Ar/CO�������������������Ʊ�SiC/SiO2������������������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2020���11��
�������ߣ�л��F ���ܰ� �ƶ� ��� ���Ƿ� ������ ������
����ҳ�룺3058 - 3066
�ؼ��ʣ��ϳɣ�����������SiC/SiO2������������������һ����̼�����·�������
Key words��synthesis; growth mechanism; SiC/SiO2 nanochains; thermal evaporation method; carbon monoxide; photoluminescence properties
ժ Ҫ����Ar/CO�����в�����������������̿��ά��������Ʊ�SiC/SiO2��������XRD��FT-IR��SEM��TEM������������Ʊ���SiC/SiO2�������ɵ����ṹSiC�����ߺ����νṹSiO2����ɡ�CO�������ܴٽ��γ�SiO2���Ӷ�����ȴ�������γ�SiC/SiO2�����������⣬���·�����������������SiC/SiO2��������Լ 350 nm����ʾ��һ���Ͽ��ķ���壬��Ҫ������SiO2���е���ƫ���Լ�SiC/SiO2�Ľ���ЧӦ�����о���Ϊһά̼��������������������о��ṩָ����
Abstract: SiC/SiO2 nanochains were synthesized on a carbon fiber substrate by a catalyst-free thermal evaporation method in the Ar/CO atmosphere. X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR), scanning electron microscopy (SEM) and transmission electron microscopy (TEM) revealed that the as-synthesized SiC/SiO2 nanochains are composed of single-crystalline SiC nanowires and amorphous SiO2 beads. The introduction of CO can promote the formation of SiO2, so that the SiC/SiO2 nanochains are subsequently formed during cooling. In addition, the photoluminescence spectrum of SiC/SiO2 nanochains showed a broad emission peak at around 350 nm, which is ascribed to the oxygen discrepancy in the SiO2 beads as well as the SiC/SiO2 interfacial effect. These findings can provide guidance for further study of the vapor growth of 1D SiC-based materials.
Trans. Nonferrous Met. Soc. China 30(2020) 3058-3066
Xiang-min XIE1, Zhe-an SU1, Dong HUANG2, Cheng YANG1,Ya-feng WANG1, Ding-yu JIANG1, Qi-zhong HUANG1
1. National Key Laboratory of Science and Technology for National Defence on High-strength Structural Materials, Central South University, Changsha 410083, China;
2. Hunan Provincial Engineering Research Center for High Performance Pitch Based Carbon Materials, Hunan Toyi Carbon Material Technology Co., Ltd., Changsha 410205, China
Received 10 January 2020; accepted 10 July 2020
Abstract: SiC/SiO2 nanochains were synthesized on a carbon fiber substrate by a catalyst-free thermal evaporation method in the Ar/CO atmosphere. X-ray diffraction (XRD), Fourier-transform infrared spectroscopy (FT-IR), scanning electron microscopy (SEM) and transmission electron microscopy (TEM) revealed that the as-synthesized SiC/SiO2 nanochains are composed of single-crystalline SiC nanowires and amorphous SiO2 beads. The introduction of CO can promote the formation of SiO2, so that the SiC/SiO2 nanochains are subsequently formed during cooling. In addition, the photoluminescence spectrum of SiC/SiO2 nanochains showed a broad emission peak at around 350 nm, which is ascribed to the oxygen discrepancy in the SiO2 beads as well as the SiC/SiO2 interfacial effect. These findings can provide guidance for further study of the vapor growth of 1D SiC-based materials.
Key words: synthesis; growth mechanism; SiC/SiO2 nanochains; thermal evaporation method; carbon monoxide; photoluminescence properties
1 Introduction
Silicon carbide (SiC) is an important material which is widely applied in nuclear industry, aerospace, and semiconductor [1-4]. The SiC nanowires have, in particular, demonstrated that they are good candidates for making functional optoelectronic devices thanks to their large band gap, as well as the quantum confinement effect when their crystallite size is reduced to nanoscale [5]. Various technologies such as pyrolysis of precursor [6], chemical vapor deposition [7,8], microwave heating [9] and carbothermal reduction [10,11] enable us to precisely fabricate SiC nanowires. However, most of these methods involve metal catalyst which is difficult to be removed and may affect properties of nanowires [6,12], so the catalyst-free thermal evaporation method is now widely accepted for the fabrication of SiC nanowires [13,14].
The SiC nanowires which are coated with SiO2 beads, are called SiC/SiO2 nanochains [15-18]. These SiC/SiO2 nanochains exhibit the enhanced electronic properties due to the SiC/SiO2 interfacial effect, which is a great advantage in the application for functional materials [19]. LI et al [20] synthesized SiC/SiO2 nanochains via molten salt-mediated chemical vapor reaction technique, and suggested that the SiO2 beads can effectively enhance the photoluminescence properties of the SiC/SiO2 nanochains. It was proposed that the controllable and predictable fabrication of SiO2 can broaden the application of SiC/SiO2 nanochains greatly [21,22]. As a result, many researchers have investigated the growth mechanisms of SiC/SiO2 nanochains. For instance, HOU et al [15] synthesized SiC/SiOx nanochains via polymer- pyrolysis process, and found that the reactant concentration played a crucial role in controlling the final morphology of SiO2. CHU et al [23] synthesized SiC/SiO2 nanochains with different morphologies at different temperatures, and they ascribed this kind of results to the differences in both the SiO partial pressure and the Plateau- Rayleigh instability. Carbon monoxide (CO) is a common reductant, which is closely related to the carbothermal reduction synthesis of metal oxide nanostructures [24-26]. Similarly, it is assumed that CO can also affect the formation mechanism of SiO2 nanostructures during the vapor growth of SiC nanowires, but no effort has been made so far to verify this assumption experimentally.
In this work, SiC/SiO2 nanochains were synthesized by a catalyst-free thermal evaporation method in the Ar/CO atmosphere at 1400 ��C. The comparative experiments conducted in the Ar atmosphere enable us to understand the effect of CO on the vapor growth of SiO2, and determine the formation mechanism of the SiC/SiO2 nanochains. In addition, the photoluminescence properties of the SiC/SiO2 nanochains were also characterized, which may be served as a reference for the application in the field of optoelectronic device.
2 Experimental
2.1 Synthesis of SiC/SiO2 nanochains
Figure 1 illustrates the experimental setup for the preparation of SiC/SiO2 nanochains. The silicon powder purchased from Tianjin Kermel Chemical Reagent Co., Ltd., was used as the silicon source, and the carbon fiber purchased from Toray Industries, Inc. was used as both the carbon source and the substrate. Firstly, 5 g of silicon powder was added into a graphite crucible, and a carbon fiber with a length of 5 cm was fixed on the top of the silicon powder. Secondly, the graphite crucible was placed into an electric resistance furnace. After being evacuated to about 100 Pa by a vacuum pump, the furnace was filled with Ar (99.99%) until total pressure reached 0.11 MPa. Thirdly, the furnace was heated until the temperature reached 1400 ��C with CO (99.99%) being introduced into the furnace for a dwell time of 5 h. The flow rates of Ar and CO were 1 and 0.3 L/min, respectively. Finally, the furnace was cooled down naturally in the Ar atmosphere, and the white cotton-like nanostructures were obtained on the carbon fiber substrate. A comparative experiment was also performed by the same procedure but without introducing CO.

Fig. 1 Experimental setup for fabricating SiC/SiO2 nanochains
2.2 Characterization
The morphologies of the products were investigated using scanning electron microscopy (SEM, FEI Quanta 250 FEG). The crystalline phase measurements were carried out using X-ray diffraction (XRD, Rigaku Dmax/2550 18 kW) from a Cu K�� source with a wavelength �� of 0.1542 nm. The surface functional groups were identified using Fourier-transform infrared spectroscopy (FT-IR, Thermo Fisher Scientific). The microstructures were characterized by transmission electron microscopy (TEM, FEI, Titan G2 60-300) including high-resolution transmission electron microscopy (HRTEM). The room temperature photoluminescence (PL) emissions were recorded using a fluorescence spectrophotometer (Hitachi F-7000, Xe lamp).
3 Results and discussion
3.1 Morphologies and microstructures of SiC nanowires and SiC/SiO2 nanochains
Figure 2 shows the SEM images of the products synthesized in different atmospheres. Figures 2(a) and (b) display that a large number of entangled and uniform nanowires are formed after vapor growth in the Ar atmosphere. Their diameters are in a range of 30-60 nm and their lengths are hundreds of micrometers. Other nanostructures such as beads have not been detected. By contrast, the nanochains are formed after vapor growth in the Ar/CO atmosphere (Fig. 2(c)). The lengths of the nanochains are about several tens of micrometers. Figure 2(d) further reveals that the nanochains mainly consist of beads and nanowires. The periodic beads display a smooth surface with a diameter range of 300-600 nm, and they are strung together by the nanowires with a diameter in the range of 30-60 nm. It is inferred that the introduction of CO can induce the formation of periodic beads on nanowires during vapor growth and thus affect the final morphology of the 1D nanostructures.

Fig. 2 SEM images of products synthesized under different atmospheres
To determine the crystalline phase and surface functional groups of the 1D nanostructures, X-ray diffraction and FT-IR spectroscopy analyses were performed. Figure 3 displays the XRD patterns of the products obtained in the Ar atmosphere (Fig. 3(a)) and the Ar/CO atmosphere (Fig. 3(b)), respectively. A strong diffraction peak at 2�� value of 25.8�� is associated with the (002) crystal plane of graphite (JCPDS No. 75-1621), which is derived from the carbon phase of the fiber substrate. In addition, three diffraction peaks at 2�� values of 35.8��, 60.3�� and 72.1�� match the (111), (220) and (311) planes of the cubic SiC (JCPDS No. 29-1129), respectively. Interestingly, a sharp peak at 2�� value of 21.7�� appears in Fig. 3(b), which is indexed to the crystal structure of the SiO2 (JCPDS No. 39-1425). Figure 4 presents the FT-IR spectra of the products obtained in the Ar atmosphere (Fig. 4(a)) and the Ar/CO atmosphere (Fig. 4(b)). A sharp peak at around 797 cm-1 is observed (Fig. 4(a)), corresponding to the Si��C stretching vibration band [27]. Besides, two other absorption peaks can be seen at around 1101 and 478 cm-1 (Fig. 4(b)), which can be ascribed to the Si��O��Si stretching band [28]. Combined with the XRD results, it can be inferred that the main product synthesized in the Ar atmosphere is SiC, whereas the main products synthesized in the Ar/CO atmosphere are SiC and SiO2, suggesting that the introduction of CO can induce the formation of SiO2.

Fig. 3 XRD patterns of products synthesized in different atmospheres

Fig. 4 FT-IR spectra of products synthesized in different atmospheres

Fig. 5 TEM (a) and HRTEM (b) images of nanowire synthesized in Ar atmosphere

Fig. 6 TEM images of SiC/SiO2 nanochains synthesized in Ar/CO atmosphere
TEM examinations including the HRTEM were conducted to acquire the microstructural information of the 1D nanostructures. Figure 5(a) illustrates the TEM image of a typical SiC nanowire synthesized in the Ar atmosphere, which has a diameter of 35 nm and grows along the axial direction. The spacing between two lattice planes along the growth direction is 0.25 nm (Fig. 5(b)), indicating that the nanowire has a cubic SiC structure, but no other crystalline or amorphous structures can be observed. The growth mechanism of high purity SiC nanowires formed in the Ar atmosphere can be explained as follows. Firstly, both the silicon powder and the graphite crucible can react with O2 to generate SiO and CO gases at the synthesis temperature, as described in Eqs. (1) and (2) (the residual O2 is believed to come from the residual air at 100 Pa in the furnace and the impurity of the Ar [29,30]). Subsequently, the formed SiO will react with C and CO on the carbon fiber according to Eqs. (3) and (4), and thus generates the SiC nanowires. This oxide-assisted mechanism for the fabrication of SiC nanowires has also been reported by some other researchers [31,32].
2Si(s)+O2(g)=2SiO(g) (1)
2C(s)+O2(g)=2CO(g) (2)
SiO(g)+2C(s)=SiC(s)+CO(g) (3)
SiO(g)+3CO(g)=SiC(s)+2CO2(g) (4)
Figure 6 shows the TEM images of SiC/SiO2 nanochain synthesized in the Ar/CO atmosphere. To better observe and study the microstructure of SiC/SiO2 nanochain, we divide it into two parts: a bead and a string. The bead (Fig. 6(a)) possesses a smooth surface and its diameter is about 350 nm. HRTEM image (Fig. 6(b)) clearly shows the lattice fringes in inner part of the bead, and the interplanar spacing (0.25 nm) can be indexed to the (111) plane of SiC, indicating that the inner part of the bead is the single-crystalline SiC. Moreover, no lattice fringes but irregular structures in outer part of the bead are observed so that the outer part of the bead should be amorphous SiO2. The string (Fig. 6(c)) shows that a core-shell nanowire with a diameter of about 40 nm has a crystalline cubic SiC core, as confirmed by HRTEM image (Fig. 6(d)) which also shows an amorphous SiO2 shell with a thickness of 3 nm. This kind of SiC/SiO2 core-shell nanowire has been studied for two decades, and it is believed that the SiO2 is formed by the reaction between SiO and CO gases, as described in Eq. (5) [33]:
3SiO(g)+CO(g)=SiC(s)+2SiO2(s) (5)
3.2 Growth mechanism of SiC/SiO2 nanochains
Figure 7(a) shows the standard Gibbs free energy change (��G��) of Reactions (1)-(5). It can be noticed that the ��G�� values of Reactions (1) and (2) are relatively low, which indicates that both the silicon and carbon are easy to react with oxygen at high temperature to form the SiO and CO gases, in spite of low oxygen content in the system. In addition, the ��G�� values of Reactions (3) and (5) are also negative. It seems to suggest that these two reactions may occur easily, leading to the formation of SiC and/or SiO2. However, no SiO2 has been detected after reaction in the Ar atmosphere (Figs. 3(a) and 4(a)) in our experiment, presumably because Reaction (5) is not thermodynamically feasible. In order to investigate the influence of vapor pressure of CO gas on the occurrence of Reaction (5), the phase diagrams as the function of p(CO) and temperature are illustrated in Figs. 7(b) and (c). As the SiO and CO gases are both generated in the Ar atmosphere, the vapor pressures of both SiO and CO gases are supposed to be similar (i.e. p(CO)��p(SiO)). When the reaction occurs in the Ar atmosphere, the SiO and CO gases from trace oxygen are insufficient to allow the formation of SiO2 (Fig. 7(b)). By contrast, when the reaction occurs in the Ar/CO atmosphere (about 23 kPa CO in this work), the external CO gas can effectively promote the vapor reaction (Fig. 7(c)), forming a large amount of SiO2. Consequently, the formation of SiO2 during vapor growth process is believed to depend on the vapor pressure of CO.

Fig. 7 Standard Gibbs free energy change for Reactions (1)-(5) (a), phase diagrams as functions of temperature and p(CO) in Ar (b) and Ar/CO (c) atmosphere
On the basis of comparative experiments and systematic characterization, the catalyst-free growth mechanism of the SiC/SiO2 nanochains in the Ar/CO atmosphere can be proposed as follows. Firstly, SiC nanowires are generated from SiO and CO gases, as described in Figs. 8(a-c). Secondly, a high CO partial pressure can make Reaction (5) happen, resulting in the formation of both the SiC and SiO2 phases as illustrated in Fig. 8(d). The deposited SiC diffuses to the tips of the nanowires and assists further growth of nanowires along the [111] direction. The SiO2 phase is absorbed by the SiC nanowires and becomes a molten layer due to good chemical compatibility and wettability between SiO2 and SiC at a high temperature of 1400 ��C. Finally, the SiO2 melt condenses into the periodic SiO2 beads on the SiC nanowires during cooling (Figs. 8(e) and (f)). The driving force of wetting is believed to come from the competition between the surface energy of SiO2 melt and the interfacial energy of SiO2/SiC. At the synthesis temperature of 1400 ��C, the wetting behavior is dominated by the interfacial energy of SiO2/SiC, and consequently the SiO2 melt with low viscosity covers the SiC nanowires. As the temperature decreases during cooling, the SiO2 melt migrates and forms the SiO2 droplets periodically based on the lowest surface energy principle. These SiO2 droplets would eventually condense into solid beads on the SiC nanowires (Figs. 6(a) and (b)). At the same time, some SiC nanowires are still coated with SiO2 film and finally form the SiC/SiO2 core-shell nanowires (Figs. 6(c) and (d)), because the high viscosity can suppress the migration of SiO2 on the SiC nanowires during cooling.
3.3 Photoluminescence properties of SiC nanowires and SiC/SiO2 nanochains
The SiC nanowires are regarded as a good candidate for the application in violet-blue optoelectronic devices, and the SiO2 is believed to affect the PL properties of SiC nanowires owing to the special electronic structure of the SiC/SiO2 interface [5,19]. Figure 9(a) shows the photo- luminescence (PL) spectra of SiC/SiO2 nanochains at the excitation wavelength of 250 nm. Four emission peaks at 350, 398, 451 and 467 nm are observed, which are blue-shifted compared to the original 3C��SiC [34]. Similar emission peaks were also reported by WEI et al [9]. Figure 9(b) shows the normalized emission intensity of SiC/SiO2 nanochains and SiC nanowires. The emission peak of SiC/SiO2 nanochains shows an obvious blue-shift to 350 nm compared with that of SiC nanowires, which can be ascribed to the influence of SiC/SiO2 interface interaction and the oxygen discrepancy in the SiO2 beads [35-37]. This result suggests that the presence of SiO2 beads can tailor the photoluminescence properties of SiC nanowires.
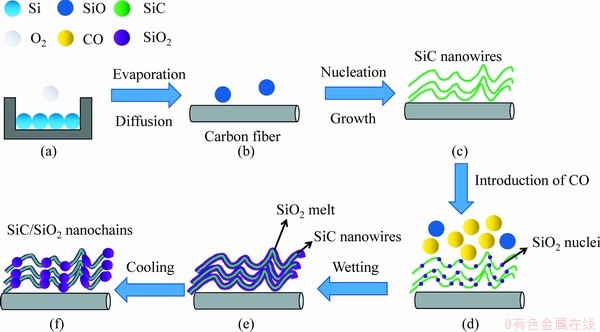
Fig. 8 Growth process of SiC/SiO2 nanochains

Fig. 9 Photoluminescence spectra of SiC/SiO2 nanochains (a) and normalized SiC/SiO2 nanochains and SiC nano- wires (b)
4 Conclusions
(1) SiC/SiO2 nanochains were synthesized by a catalyst-free thermal evaporation method in the Ar/CO atmosphere. The obtained SiC/SiO2 nano- chains consist of crystalline SiC nanowires and amorphous SiO2 beads.
(2) The comparative experiments revealed that a high CO partial pressure can thermodynamically promote the formation of SiO2 on SiC nanowires at synthesis temperature, thus leading to the formation of SiC/SiO2 nanochains during cooling.
(3) The SiC/SiO2 nanochains showed an obvious blue-shift to 350 nm compared with SiC nanowires, which can provide a way to tune the optoelectronic behavior of SiC-based functional device.
References
[1] ZATKO B, HRUBCIN L, SAGATOVA A, OSVALD J, BOHACEK P, ZAPRAZNY Z, SEDLACKOVA K, SEKACOVA M, DUBECKY F, SKURATOV V A, KORYTAR D, NECAS V. Schottky barrier detectors based on high quality 4H-SiC semiconductor: Electrical and detection properties [J]. Applied Surface Science, 2018, 461: 276-280.
[2] SAVINO R, CRISCUOLO L, DI MARTINO G D, MUNGIGUERRA S. Aero-thermo-chemical characterization of ultra-high-temperature ceramics for aerospace applications [J]. Journal of the European Ceramic Society, 2018, 38: 2937-2953.
[3] ZENG K H, MA Q S, GU X Y. Effects of SiC interfacial coating on mechanical properties of carbon fiber needled felt reinforced sol-derived Al2O3 composites [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 463-471.
[4] RAN L P, RAO F, PENG K, YIN H, YI M Z. Preparation and properties of C/C-ZrB2-SiC composites by high-solid- loading slurry impregnation and polymer infiltration and pyrolysis (PIP) [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 2141-2150.
[5] FAN J Y, WU X L, CHU P K. Low-dimensional SiC nanostructures: Fabrication, luminescence, and electrical properties [J]. Progress in Materials Science, 2006, 51: 983-1031.
[6] FENG W, MA J T, YANG W Y. Precise control on the growth of SiC nanowires [J]. Cryst Eng Comm, 2012, 14: 1210-1212.
[7] LIU S L, LIU H T, HUANG Z H, FANG M H, LIU Y G, WU X W. Synthesis of beta-SiC nanowires via a facile CVD method and their photoluminescence properties [J]. RSC Advances, 2016, 6: 24267-24272.
[8] GE Y C, LIU Y Q, WU S, HUANG W Q, MAO P L, YI M Z. Characterization of SiC nanowires prepared on C/C composite without catalyst by CVD [J]. Transactions of Nonferrous Metals Society of China, 2015, 25: 3258-3264.
[9] WEI G D, QIN W P, ZHENG K Z, ZHANG D S, SUN J B, LIN J J, KIM R J, WANG G F, ZHU P F, WANG L L. Synthesis and properties of SiC/SiO2 nanochain heterojunctions by microwave method [J]. Crystal Growth & Design, 2009, 9: 1431-1435.
[10] KUDRENKO E, RODDATIS V, ZHOKHOV A, ZVERKOVA I, KHODOS I, EMELCHENKO G. Morphology of SiC nanowires grown on the surface of carbon fibers [J]. RSC Advances, 2012, 2: 4913-4919.
[11] XIN L P, SHI Q, CHEN J J, TANG W H, WANG N Y, LIU Y, LIN Y X. Morphological evolution of one-dimensional SiC nanomaterials controlled by sol-gel carbothermal reduction [J]. Materials Characterization, 2012, 65: 55-61.
[12] LI Z J, GAO W D, MENG A, GENG Z D, WAN L B. Effects of Fe and Ni on the yield and morphology of the 1D SiC nanostructures prepared by chemical vapor reaction [J]. Journal of Crystal Growth, 2008, 310: 4401-4406.
[13] DAI J X, SHA J J, ZHANG Z Q, SHAO J Q, ZU Y F, LEI M K. Catalyst-free growth of multi-shaped SiC nanofibers on carbon fibers at elevated temperatures [J]. Ceramics International, 2017, 43: 17057-17063.
[14] DAI J X, SHA J J, ZU Y F, SHAO J Q, LEI M K, FLAUDER S, LANGHOF N, KRENKEL W. Synthesis and growth mechanism of SiC nanofibres on carbon fabrics [J]. Cryst Eng Comm, 2017, 19: 1279-1285.
[15] HOU H L, WANG L, GAO F M, WEI G D, ZHENG J J, CHENG X M, TANG B, YANG W Y. Mass production of SiC/SiOx nanochain heterojunctions with high purities [J]. Cryst Eng Comm, 2013, 15: 2986-2991.
[16] ZHONG B, KONG L Q, ZHANG B, YU Y L, XIA L. Fabrication of novel hydrophobic SiC/SiO2 bead-string like core-shell nanochains via a facile catalyst/template-free thermal chemical vapor deposition process [J]. Materials Chemistry and Physics, 2018, 217: 111-116.
[17] LI W, JIA Q L, LIU X H, ZHANG J. Large scale synthesis and photoluminescence properties of necklace-like SiC/SiOx heterojunctions via a molten salt mediated vapor reaction technique [J]. Ceramics International, 2017, 43: 2950-2955.
[18] DAI J X, SHA J J, ZHANG Z F, SHAO J Q, ZU Y F, LEI M K. Synthesis of novel hierarchical SiC-SiO2 heterostructures via a catalyst free method [J]. Cryst Eng Comm, 2017, 19: 6540-6546.
[19] FAN J Y, CHU P K. Silicon carbide nanostructures: Fabrication, structure, and properties [M]. Heidelberg: Springer, 2014.
[20] LI W, JIA Q L, YANG D Y, LIU X H. Tunable synthesis of SiC/SiO2 heterojunctions via temperature modulation [J]. Materials, 2018, 11: 766-776.
[21] LI Z J, ZHAO J, ZHANG M, XIA J Y, MENG A. SiC nanowires with thickness-controlled SiO2 shells: Fabrication, mechanism, reaction kinetics and photoluminescence properties [J]. Nano Research, 2015, 7: 462-472.
[22] RYU Y, TAK Y, YONG K. Direct growth of core-shell SiC-SiO2 nanowires and field emission characteristics [J]. Nanotechnology, 2005, 16: 370-374.
[23] CHU Y H, JING S Y, YU X, ZHAO Y L. High-temperature plateau-rayleigh growth of beaded SiC/SiO2 nanochain heterostructures [J]. Crystal Growth & Design, 2018, 18: 2941-2947.
[24] WU F, MYUNG Y, BANERJEE P. Rayleigh instability driven nodular Cu2O nanowires via carbothermal reduction of CuO nanowires [J]. Crystal Growth & Design, 2014, 15: 1588-1595.
[25] GORUP L, SOUZA NETO F, KUBO A, SANTOS SOUZA J A, FERNANDES SCAPPATICCI R, LOPES G, MONTEIRO D, BARROS BARBOSA D, CAMARGO E. Recent advances in complex functional materials [M]. Heidelberg: Springer, 2017.
[26] SUBANNAJUI K, RAMGIR N S, GRIMM R, MICHIELS R, YANG Y, MULLER S, ZACHARIAS M. ZnO Nanowire growth: A deeper understanding based on simulations and controlled oxygen experiments [J]. Crystal Growth & Design, 2010, 10: 1585-1589.
[27] RAY S, DAS D, BARUA A K. Infrared vibrational spectra of hydrogenated amorphous silicon carbide thin films prepared by glow discharge [J]. Solar Energy Materials, 1987, 15: 45-57.
[28] MACDONALD S A, SCHARDT C R, MASIELLO D J, SIMMONS J H. Dispersion analysis of FTIR reflection measurements in silicate glasses [J]. Journal of Non- Crystalline Solids, 2000, 275: 72-82.
[29] CHU Y H, JING S Y, CHEN J K. In situ synthesis of homogeneously dispersed SiC nanowires in reaction sintered silicon-based ceramic powders [J]. Ceramics International, 2018, 44: 6681-6685.
[30] KHOLMANOV I N, KHARLAMOV A, BARBORINI E, LENARDI C, LI BASSI A, BOTTANI C E, DUCATI C, MAFFI S, KIRILLOVA N V, MILANI P. A simple method for the synthesis of silicon carbide nanorods [J]. Journal of Nanoscience and Nanotechnology, 2002, 2: 453-456.
[31] DAI J J, SHA J J, ZHANG Z F, WANG Y, KRENKEL W. Synthesis of high crystalline beta SiC nanowires on a large scale without catalyst [J]. Ceramics International, 2015, 41: 9637-9641.
[32] HUANG Z H, LIU H T, CHEN K, FANG M H, HUANG J T, LIU S Y, HUANG S F, LIU Y G, WU X W. Synthesis and formation mechanism of twinned SiC nanowires made by a catalyst-free thermal chemical vapour deposition method [J]. RSC Advances, 2014, 4: 18360-18364.
[33] LI B S, WU R B, PAN Y, WU L L, YANG G Y, CHEN J J, ZHU Q M. Simultaneous growth of SiC nanowires, SiC nanotubes, and SiC/SiO2 core�Cshell nanocables [J]. Journal of Alloys and Compounds, 2008, 462: 446-451.
[34] CHIU S C, LI Y Y. SiC nanowires in large quantities: Synthesis, band gap characterization, and photoluminescence properties [J]. Journal of Crystal Growth, 2009, 311: 1036-1041.
[35] WEI G D, QIN W P, ZHU P F, KIM R J, WANG G F, ZHANG D S, ZHENG K Z, WANG L L. Large-scale synthesis and photoluminescence properties of aligned multicore SiC-SiO2 nanocables [J]. Journal of Nanoscience and Nanotechnology, 2010, 10: 1964-1968.
[36] LIANG C, MENG G, ZHANG L, WU Y, CUI Z. Large-scale synthesis of ��-SiC nanowires by using mesoporous silica embedded with Fe nanoparticles [J]. Chemical Physics Letters, 2000, 329: 323-328.
[37] CAI K F, ZHANG A X, YIN J L, WANG H F, YUAN X H. Preparation, characterization and photoluminescence properties of ultra long SiC/SiOx nanocables [J]. Applied Physics A, 2008, 91: 579-584.
л��F1�����ܰ�1���� ��2���� ��1�����Ƿ�1��������1��������1
1. ���ϴ�ѧ ���ʸ�ǿ�ṹ���Ϲ����Ƽ��ص�ʵ���ң���ɳ 410083��
2. ���϶�ӳ̼���ϿƼ�����˾ ����ʡ�����������̼���Ϲ����о����ģ���ɳ 410205
ժ Ҫ����Ar/CO�����в�����������������̿��ά��������Ʊ�SiC/SiO2��������XRD��FT-IR��SEM��TEM������������Ʊ���SiC/SiO2�������ɵ����ṹSiC�����ߺ����νṹSiO2����ɡ�CO�������ܴٽ��γ�SiO2���Ӷ�����ȴ�������γ�SiC/SiO2�����������⣬���·�����������������SiC/SiO2��������Լ 350 nm����ʾ��һ���Ͽ��ķ���壬��Ҫ������SiO2���е���ƫ���Լ�SiC/SiO2�Ľ���ЧӦ�����о���Ϊһά̼��������������������о��ṩָ����
�ؼ��ʣ��ϳɣ�����������SiC/SiO2������������������һ����̼�����·�������
(Edited by Wei-ping CHEN)
Foundation item: Project (U19A2088) supported by the National Natural Science Foundation of China; Project (2019RS2058) supported by the Special Fund for Innovative Construction of Hunan Province, China
Corresponding author: Zhe-an SU; Tel: +86-13873188002; E-mail: suza@csu.edu.cn
DOI: 10.1016/S1003-6326(20)65442-5

