Scandium recovery from ion-adsorption rare earth concentrate with HEHEHP as extractant
来源期刊:中南大学学报(英文版)2021年第3期
论文作者:宁顺艳 张伟 朱浩 刘书峰 韦悦周
文章页码:679 - 689
Key words:scandium; HEHEHP; solvent extraction; ion-adsorption rare earth
Abstract: An excellent extraction selectivity towards Sc over other REEs in 4 mol/L HCl solution was achieved with the separation factor βSc/REEs over 8000 by 2-ethylhexyl phosphoric acid mono 2-ethylhexyl ester (HEHEHP) and the extraction equilibrium can be obtained within 20 min. The extracted Sc can be stripped using 5 mol/L NaOH solution as eluent at 363 K with the stripping rate of 92.1% obtained. The extraction mechanism was clearly elucidated by slope analysis, saturation extraction, IR, and NMR analysis. It was revealed that the extraction of Sc in 4 mol/L HCl solution is still dominated by cation exchange process between P―O―H and Sc, and coordination process between P=O and Sc, with 6 molecules of extractant as dimer participating in the process. Finally, a flowsheet for the recovery of Sc from ion-adsorption rare earth elements (REEs) concentrate was proposed and proved in lab-scale experiment.
Cite this article as: ZHANG Wei, ZHU Hao, LIU Shu-feng, NING Shun-yan, WEI Yue-zhou. Scandium recovery from ion-adsorption rare earth concentrate with HEHEHP as extractant [J]. Journal of Central South University, 2021, 28(3): 679-689. DOI: https://doi.org/10.1007/s11771-021-4637-6.

J. Cent. South Univ. (2021) 28: 679-689
DOI: https://doi.org/10.1007/s11771-021-4637-6

ZHANG Wei(张伟)1, ZHU Hao(朱浩)1, LIU Shu-feng(刘书峰)1,NING Shun-yan(宁顺艳)1, WEI Yue-zhou(韦悦周)1, 2
1. Guangxi Key Laboratory of Processing for Non-ferrous Metals and Featured Materials,
MOE Key Laboratory of New Processing Technology for Non-ferrous Metals and Materials,
School of Resources, Environment and Materials, Guangxi University, Nanning 530004, China;
2. School of Nuclear Science and Engineering, Shanghai Jiao Tong University, Shanghai 200240, China
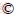 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2021
Abstract: An excellent extraction selectivity towards Sc over other REEs in 4 mol/L HCl solution was achieved with the separation factor βSc/REEs over 8000 by 2-ethylhexyl phosphoric acid mono 2-ethylhexyl ester (HEHEHP) and the extraction equilibrium can be obtained within 20 min. The extracted Sc can be stripped using 5 mol/L NaOH solution as eluent at 363 K with the stripping rate of 92.1% obtained. The extraction mechanism was clearly elucidated by slope analysis, saturation extraction, IR, and NMR analysis. It was revealed that the extraction of Sc in 4 mol/L HCl solution is still dominated by cation exchange process between P―O―H and Sc, and coordination process between P=O and Sc, with 6 molecules of extractant as dimer participating in the process. Finally, a flowsheet for the recovery of Sc from ion-adsorption rare earth elements (REEs) concentrate was proposed and proved in lab-scale experiment.
Key words: scandium; HEHEHP; solvent extraction; ion-adsorption rare earth
Cite this article as: ZHANG Wei, ZHU Hao, LIU Shu-feng, NING Shun-yan, WEI Yue-zhou. Scandium recovery from ion-adsorption rare earth concentrate with HEHEHP as extractant [J]. Journal of Central South University, 2021, 28(3): 679-689. DOI: https://doi.org/10.1007/s11771-021-4637-6.
1 Introduction
Scandium, a member of rare earth elements (REEs), plays an important role in many strategic fields, such as Al-Sc alloys, lasers, optical, solid oxide fuel cells, advanced catalyst applications, nuclear medicine [1, 2]. Although Sc is not rare, ranked the 31th in the earth’s richest elements with an average crustal abundance of 22 ppm, Sc-rich minerals such as thortveitite ((Sc, Y…)2Si2O7) are sparse [3, 4]. At present, Sc is generally recovered as a by-product from slag (bauxite residue, red mud, uranium ores, etc.) and liquid waste (such as waste acid generated during titanium dioxide production) [5-9]. The content of Sc is several orders of magnitude lower than those of impurity elements, which makes its recovery and purification process from these leaching solutions very complex.
Ion-adsorption REEs ore found in South China contains almost all rare earth elements which are especially rich in middle and heavy rare earth elements [10, 11]. Several methods for the Sc-rich concentrate are obtained from these ores containing low content of REEs. Typically, high acid solution is used to dissolving the concentrate for the later REEs purification. Nevertheless, Sc is often neglected in the extraction and separation process of other REEs. It will cause a waste of resources and decrease the purities of other rare earth products if Sc is not completely removed as an impurity. The difficulties in Sc recovery and separation from other REEs are mainly caused by their similar chemical properties. It is urgent to develop a simple and economical method for Sc recovery from ion-adsorption REEs concentrate.
Liquid-liquid extraction (LLE) has been widely applied in current industrial practice for the separation of Sc. At the moment, a variety of commercial extractants have been used for Sc separation and purification process, such as 2-ethyl-hexyl phosphonic acid mono-2-ethylhexyl ester (HEH(EHP)), bis(2,4,4-trimethylpentyl) phosphinic acid (Cyanex272), di-(2-ethyl hexyl phosphonic acid (HDEHP) and bis(2,4,4- trimethypentyl)-mono thiophosphinic acid (Cyanex302) [12-18]. The majority of these extractants are performed on a relatively low acid solution (pH≥1) via cation exchange process. To prevent the relatively large changes in acidity during the extraction process, it always needs to saponify the acidic organophosphorus extractant by alkaline reagents, typically NH3・H2O, which may result in a large undesired ammonia-nitrogen waste [19]. A new Chinese national standard is implemented which requires the concentration of ammonia-nitrogen in waste water must be lower than 15 mg/L [20]. This requirement severely limits Sc production. A green extraction method of Sc is very desirable to be developed.
Although HEHEHP is one of the most extensively applied extractants in the separation of REEs from various mediums, few researches focused on the extraction behavior of HEHEHP towards Sc. It has been studied that HEHEHP has a good selectivity towards Sc in low acidity solution containing Al, Mn, Fe, Mg, etc [13]. In addition, the extraction of Sc by HEHEHP-isooctanol from simulated red mud leach solution has also been studied [21]. However, there is less research on the separation of Sc from ion-adsorption REEs by HEHEHP.
In this work, the effects of acidity, time, temperature on the extraction of HEHEHP towards REEs would be investigated. The extraction mechanism of Sc in 4 mol/L HCl solution was elaborated by FT-IR, NMR, saturation extraction, and slope analysis. Based on these systematic theoretical and experimental researches, a flowsheet of direct separation of Sc from simulated ion adsorption rare earth enrichment was proposed and verified by lab-scale experiment.
2 Experimental
2.1 Materials and reagents
RE(Cl)3・n H2O (RE: Sc, Y, La, Ce, Pr, Nd, Pm, Sm, Eu, Gd, Tb, Dy, Ho, Er, Tm, Yb, Lu; except of Pm, n=5 or 6) with the purity of 99% were purchased from Baotou Rare Earth Research Institute, China. 2-ethylhexyl phosphoric acid mono 2-ethylhexyl ester (HEHEHP) with the purity of 99% was supplied by Meng Rong Fine Materials Baotou Co. Ltd. and used without further purification. All other reagents such as concentrated hydrochloric acid were of analytical grade and employed as received.
2.2 General solvent extraction procedure
Unless otherwise stated the extraction experiments were performed by contacting 0.5 mL organic phase consisting of HEHEHP and sulfonated kerosene (SK) (CHEHEHP: 0.3 mol/L) with 5 mL solution containing targeted metal ion in a vial. The vial was shaken mechanically at 160 r/min in a water bath with temperature control system. Then the phase separation was conducted by centrifugation for 5 min at 4000 r/min. Certain volume of aqueous phase was taken by a pipette and diluted in a volumetric flask. The concentrations of metal ions in all samples were measured by inductively coupled plasma atomic emission spectroscopy (ICP-AES: Shimadzu, ICPS-7510, Japan). The distribution coefficient (D), extraction efficiency (E) and separation factor (βA/B) can be calculated by:
 (1)
(1)
 (2)
(2)
 (3)
(3)
where C0 and C denote the metal ions concentrations in the aqueous phase before and after extraction, respectively, mg/L; Vaqu and Vorg represent the volume of aqueous phase and organic phase, respectively, mL.
The stripping tests of the metal-loaded organic phase were performed using acid solution or sodium hydroxide and the stripping rate was calculated from the ratio of the metal ion in the eluent over that in the initial loaded organic phase. Each experiment was conducted three times to confirm that the error was within 5%.
2.3 IR and NMR spectroscopy
IR spectra of pure extractant and organic-Sc complex in the range of 400 to 2500 cm-1 were collected in triplicate in pressed KBr pellets by IRTracer-100 spectrometer (Shimadzu, Japan). NMR measurements were conducted on AVANEC-600 NMR spectrometer (Bruker, Switzerland) at room temperature and the chemical shifts of 1H NMR spectra were referenced to the internal standard 10% tetramethylsilane (TMS).
3 Results and discussion
3.1 Selective extraction behavior of REEs by HEHEHP
3.1.1 Effect of acidity
The extraction behavior of HEHEHP towards REEs as a function of acidity was carried out with the results shown in Figure 1(a). HEHEHP exhibits almost no extraction towards La-Sm in 0.1-4 mol/L HCl solution, while the distribution coefficients of HEHEHP towards Tb-Lu and Y decrease in the order of Sc>Lu>Yb>Tm>Er>Y>Ho>Dy>Tb in 0.1-2 mol/L HCl solution, indicating that REEs with smaller ion radius can be extracted more easily by HEHEHP [22]. Due to the fact that the extraction drives by cation exchange mechanism, HCl concentration increases, and the deprotonation process is suppressed, which leads to a decrease in the distribution coefficients of heavy REEs. But the D value of Sc still keeps in a high level at experimental acidity and the separation factor of βSc/REEs is over 8000 in 4 mol/L HCl solution, which implies that Sc can be separated from the other REEs at high acidity.
In Figure 1(a), as the initial Sc concentration was too low that almost all Sc was extracted in 0.1-4 mol/L HCl solution, it is difficult to fully explain the effect of acidity on Sc extraction, so the effect of acidity at high Sc initial concentration was further studied solely (Figure 1(b)). A turning point of the extraction efficiency of Sc is observed when the HCl concentration is 2 mol/L. Whereas the extraction behavior is very different from those of other REEs. For example, the extraction of Lu(III) is nearly 0 at high acidity (≥3 mol/L). Generally, the appearance of a turning point indicates the possible changes of extraction mechanism from cation exchange to solvation reaction (molecular coordination) which has been mentioned in many researches but few have given other direct proofs [4, 21, 23, 24]. For the extraction of acid organophosphorus extractant towards other REEs, the turning point emerges at relatively high acidity, such as the turning point of the extraction of HDEHP towards Y, Er, Yb appearing in 8 mol/L HCl solution [25]. However, in the case of Sc, its turning point occurs at a relatively low acidity. The extraction of Sc drives by cation exchange at lower aqueous acidity (≤3 mol/L H2SO4) with Cyanex 302 as extractant and it proceeds on solvation reaction at higher aqueous acidity (≥3.5 mol/L H2SO4) [15]. A similar extraction mechanism transition can be found in the case of HBTMPP-HEHEHP and HEHEHP-isooctanol as extractant [21, 23]. Notably, the turning point of acidity in the case of Sc extraction is lower than that of other REEs extraction. The apparent distinction confused us about the extraction mechanism when the acidity is higher than turning point acidity. It will be verified in Section 3.2. In any case, the distinctive extraction behavior makes it possible to directly separate Sc with high purity from the ion adsorption rare earth concentrate.
3.1.2 Effect of initial Sc concentration on REE extraction
The effect of initial Sc concentration on REE extraction in 0.1 mol/L HCl solution was studied with the results shown in Figure 1(c). As the initial Sc concentration increases, the extraction efficiency towards Sc keeps in a high-level and meantime its extraction towards heavy REEs continuously decreases to almost zero, indicating that HEHEHP will preferentially extract Sc in REEs solution. That is to say, it provides a possible separation method for Sc from REEs solution even in 0.1 mol/L HCl, which enlarges the acidity range in the possible application.
3.1.3 Effect of time
The effect of time on Sc and REEs extraction in 4 mol/L HCl was investigated with the results shown in Figure 1(d). The extraction efficiency of Sc is as high as about 100% in the first 5 min in the case of REEs coexisting system and keeps unchanged as time increases. To further illustrate the equilibrium time needed, the effect of time on sole Sc extraction was further performed with the initial Sc concentration as high as 13 mmol/L. The results are shown in Figure 1(d). It takes about 20 min for E to reach stable, indicating that 20 min is sufficient for extraction equilibrium. All subsequent experiments were performed with the contact time of 1 h to ensure the extraction equilibrium.
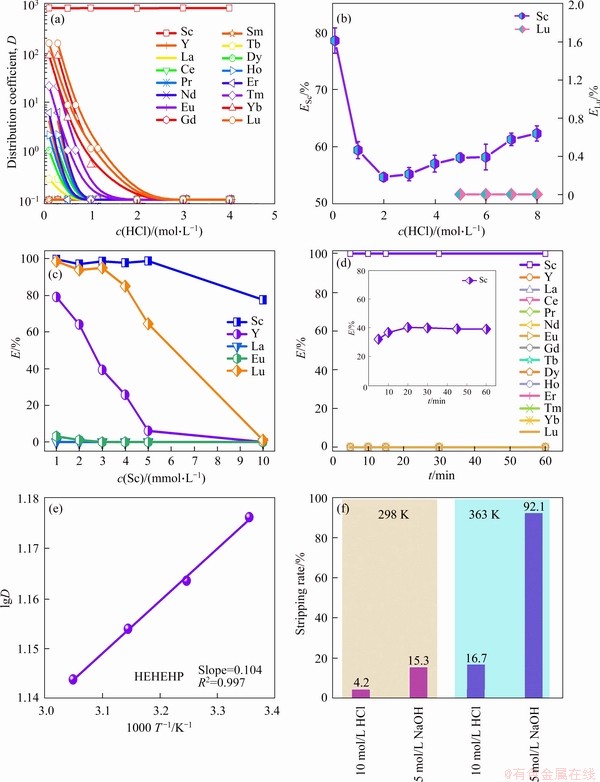
Figure 1 (a) Effect of initial HCl concentration on REEs extraction ([REEs]: 1 mmol/L); (b) Effect of initial HCl concentration on Sc or Lu extraction ([Sc]: 10 mmol/L; [Lu]: 5 mmol/L); (c) Effect of initial Sc concentration on HEHEHP extraction in REEs solution (other [REEs]: 1 mmol/L; acid: 0.1 mol/L HCl); (d) Effect of contact time on REEs extraction ([REEs]: 1 mmol/L; acid: 4 mol/L HCl), effect of contact time on Sc extraction ([Sc]:13 mmol/L; acid:4 mol/L HCl); (e) Plots of lgD versus 1000/T ([Sc]: 8 mmol/L; acid: 4 mol/L HCl); (f) Stripping of Sc from loaded HEHEHP phase with HCl and NaOH solution at different temperatures (VO/VA: 1, t=30 min)
3.1.4 Effect of temperature
The effect of temperature on Sc extraction by HEHEHP in 4 mol/L HCl solution was studied. The thermodynamic parameters can be obtained according [26]:
 (4)
(4)
where △H and R represents the changes of enthalpy, the gas constant (8.314 J/(K・mol)), respectively. The value of enthalpy was obtained from the slope of the plot of lgD versus 1000/T as shown in Figure 1(e). The negative △H (-2 kJ/mol) indicates that the extraction process of HEHEHP towards Sc is exothermal. High temperature is unfavorable for Sc(III) extraction by HEHEHP in 4 mol/L HCl solution. But on the contrary, it may promote the stripping process which is proved, in the following stripping test.
3.1.5 Stripping test
Commonly, the stripping of REEs-loaded HEHEHP can be realized using high concentrated HCl solution because there is a large number of hydrogen ions competing with the extracted metal ions for effective sites in HEHEHP, making the REEs strip back to the eluent [27]. Considering that the extraction efficiency of Sc(III) keeps at relatively high value in the experimental acidity (0.1-8 mol/L HCl) and the extraction process is exothermal, 10 mol/L HCl solution was firstly tried as eluent at different temperatures to strip Sc but relatively low stripping rate is obtained with the results shown in Figure 1(f), which is mainly due to the strong coordination interaction between Sc and HEHEHP. But it has a remarkable improvement by elevating the temperature, i.e., 363 K, revealing that increasing temperature is an effective method to obtain a higher stripping rate. As it was reported in some literature, Sc is prone to be stripped from HEHEHP by NaOH solution when the extraction of Sc is performed in a relatively low acidity [28]. Herein, 5 mol/L NaOH is adopted as the stripping solution via precipitation process. The stripping rate of Sc(III) significantly increases from 15.3% to 92.1% with temperature increasing from 298 K to 363 K, suggesting that 5 mol/L NaOH solution can effectively strip Sc from loaded HEHEHP phase at elevated temperature in a single step.
3.2 Mechanism of selective extraction of scandium
Generally, there are two kinds of mechanisms in the extraction process of HEHEHP or other acid organophosphorus extractants towards metal ions, i.e., cation exchange and solvation reaction. Usually, it is widely thought that a turning point means the transition from cation exchange to solvation reaction, but little research has given direct proofs. Herein, slope analysis, saturation extraction, IR and 1H NMR analysis are performed to give direct proofs to elucidate the extraction mechanism of HEHEHP towards Sc, especially focusing on the extraction mechanism in 4 mol/L HCl solution.
3.2.1 Saturation extraction
To verify the stoichiometry of the extracted species in 4 mol/L HCl solution, the saturation extraction method as an auxiliary means was employed (Figure 2(a)). 0.3 mol/L extractant was repeatedly contacted with fresh aqueous solution at a volume ratio (O/A) of 10 mL/20 mL until the Sc concentration in the aqueous phase unchanged. After 7 times of extraction, the organic phase reached saturation and the extracted Sc was calculated to be 0.504 mmol. Considering that the amount of HEHEHP in the organic phase is 3.09 mmol, the value obtained by dividing the amount of extractant by the amount of extracted Sc is 6.1, indicating that in 4 mol/L HCl solution 6 molecules of HEHEHP are needed to extract one molecule of Sc.
3.2.2 Slope analysis
In general, HEHEHP presents as dimer combined by an inter hydrogen bond in non-polar diluent such as SK. The extraction of Sc from chloride by HEHEHP in low acidity proceeds through cation exchange process and the extraction reaction can be expressed as [20]:
 (5)
(5)
where (HL)2 denotes HEHEHP dimer and the apparent equilibrium constant K1 can be defined as:
 (6)
(6)

Figure 2 (a) Binding stoichiometry estimation of HEHEHP with Sc(III) ([Sc]:7 mmol/L; VO/VA: 0.5; acid: 4 mol/L HCl);(b) Effect of HEHEHP concentration at different acidies on extraction of Sc (initial [Sc] in 0.1 mol/L HCl: 13.8 mmol/L and in 4 mol/L; HCl: 9.4 mmol/L); (c) IR spectra of fresh and Sc-loaded organic phase; (d) 1H NMR spectra of fresh HEHEHP and Sc-loaded organic phase (600 MHz, CDCl3)
The extraction of Sc by HEHEHP in high acidity drives by solvation reaction and the combination of n molecules of HEHEHP dimer bonded as monomer with Sc3+ can be expressed as:
 (7)
(7)
where HL represents HEHEHP monomer and the equilibrium constant K2 can be described as:
 (8)
(8)
The effect of HEHEHP concentration on the lgD value of Sc in 0.1 mol/L HCl and 4 mol/L HCl solution was performed with the results shown in Figure 2(b). A linear correlation with a slope of around 3 is obtained from slope analysis of the extraction of Sc both from 0.1 mol/L HCl and 4 mol/L HCl solution, indicating that around 6 mol HEHEHP as dimer participates in the extraction reaction, consistent with the result of saturation extraction. But according to the slope analysis, it is still hard to tell whether the extraction is cation exchange or solvation reaction.
3.2.3 IR analysis
To further elucidate the extraction mechanism of HEHEHP towards Sc at different acidity, IR spectra were collected on the samples of both the fresh organic phase and Sc-loaded organic phase with the results shown in Figure 2(c). The weakening of dimeric peak of hydrogen bonds at 1670 cm-1 after extraction indicates that intermolecular hydrogen bonds in HEHEHP are not absolutely eliminated [29]. The peak of P=O stretching vibration observed at 1198 cm-1 in the spectra of fresh organic phase shifts to 1167 cm-1 in Sc-loaded spectra, revealing the strong interaction of Sc with P=O group [30, 31]. The peak of P―O―C stretching vibration at 1035 cm-1 shifts to 1039 cm-1, attributed to the extraction of Sc [32]. It is worth to note that the weakening of the peaks, e.g., 2320 cm-1 assigned to stretching vibration and 983 cm-1 contributed by the bending vibration of P―O―H group, suggesting that the extraction of Sc by HEHEHP is still dominated by cation exchange [33]. Furthermore, the peak of P―O―H at 983 cm-1 gradually shifts with the aqueous acidity increasing in the range of 0.1-2 mol/L HCl solution, then stabilizes at 975 cm-1, indicating that the force between HEHEHP and Sc is not exactly the same in Sc-loaded organic phase at different acidity. All the remained peaks contributed by hydrocarbon moieties in SK keep the same before and after Sc-loaded extractant. The IR spectra confirm that the extraction of Sc from 4 mol/L HCl solution is predominantly driven by cation exchange.
3.2.4 1H NMR analysis
1H NMR spectra of fresh and Sc-loaded oil phase were collected to identify the types of hydrogen atoms present in the organic phase and to help elucidate the extraction mechanism with the results shown in Figure 2(d). To clearly illustrate the spectra discrepancy in cation exchange and solvation reaction, the extraction samples were prepared in the range of 0.1-8 mol/L HCl solution. The peak at 11.549 ppm is observed in the 1H NMR spectra of fresh organic phase,which is attributed to the hydrogen of P―O―H group [29, 33]. After extracting Sc from 0.1 mol/L HCl solution, the peak of P―O―H disappears, suggesting that the hydrogen in this group is replaced by Sc through cation exchange. When the extraction of Sc is performed in 4 mol/L or 6 mol/L HCl solution, the peak of P―O―H is still not obvious, indicating that cation exchange still dominates. As the aqueous acidity further increases, this peak gradually appears again due to the fact that the solvation reaction occurs in the extraction process. In the spectra of Sc-loaded organic phase in the case of extraction experiment performed in 8 mol/L HCl solution, this peak area becomes very obvious and even larger compared with that in the fresh organic phase, indicating existing the large amount of P―O―H groups, which means that the extraction mechanism changes from cation exchange to solvation reaction. The large peak may be caused by the extraction towards HCl, which can enhance the intensity of the hydroxyl hydrogen [34]. Notably, the tiny shift of the peak of P―O―H may be caused by the HEHEHP concentration changes in 1H NMR analysis [35].
Based on the above analysis, it can be summarized that the extraction of HEHEHP towards Sc in 4 mol/L HCl solution is still predominated by cation exchange process between P―O―H and Sc3+, and HEHEHP in SK extracts Sc in the form of dimer that it needs 6 mol of extractant to extract 1 mol Sc. The possible structure of the Sc-loaded HEHEHP in 4 mol/L HCl solution is shown in Figure 3.
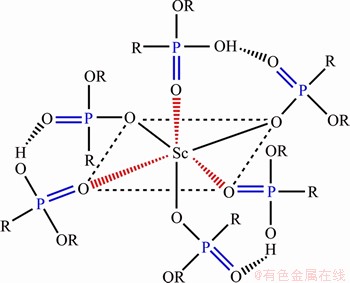
Figure 3 Structure of Sc-loaded HEHEHP in 4 mol/L HCl solution
3.3 Flowsheet of recovery of Sc from ion- adsorption REEs concentrate
A process for recovery of Sc from ion-adsorption REEs concentrate is proposed and shown in Figure 4. The major chemical composition of simulated ion-adsorption REEs concentrate is prepared according to the reference shown in Table 1 [36]. Firstly, to obtain the optimal extraction efficiency in Sc recovery, the effect of phase ratio on Sc extraction was performed with the results shown in Figure 5. The extraction efficiency of Sc is almost close to 100% when the A/O increases from 0.5 to 1, and then it pronouncedly decreases with the A/O further increasing, mainly due to the saturation extraction. Hence, the phase ratio of 1 is selected in the next experiment.
Furthermore, a lab-scale experiment was performed to verify the feasibility of the process. The extraction efficiency of Sc by HEHEHP from simulated REEs feed solution reaches 100% and it exhibits almost no extraction towards other REEs. Furthermore, the single stripping rate by 5 mol/L NaOH solution at 363 K is 89%. Compared with other recovery processes of Sc as a byproduct, this process is much simpler and greener that only single solvent extraction-back extraction is needed without extraction circuit stage and no ammonia-nitrogen waste is generated. Further study for the Sc recovery in low content from ion-adsorption ore liquor will be expected to testify the various application of the flow-sheet in our next works.
Figure 4 Flowsheet for recovery of Sc from ion-adsorption REEs concentrate
Table 1 Main composition of simulated ion-adsorption REEs concentrate


Figure 5 Effect of phase ratio on extraction of Sc from simulated ion-adsorption REEs concentrate ([HEHEHP]: 0.3 mol/L; acid: 4 mol/L HCl; t=1 h, T=298 K)
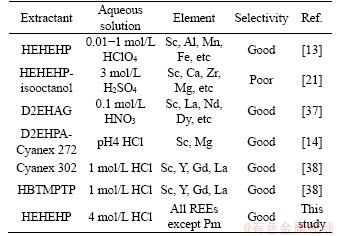
In addition, a comparison of this Sc recovery method with reported Sc extraction is made with results shown in Table 2. It is clearly found that this method proposed in this study can be used for the effective extraction of Sc from 4 mol/L HCl solution, which the acid of aqueous solution is higher than the listed studies. Although HEHEHP-isooctanol can be used to extracted Sc in 3 mol/L H2SO4 solution, the aqueous solution is the simluated red mud leach solution, which composed of Sc, Ca, Mg etc. The HEHEHP-isooctanol extracted the Sc by antagonistic effect and facilitates efficient Sc stripping, but the extraction amount is also decreased. Furthermore, the selectivity of HEHEHP-isooctanol towards Sc from ion-adsorption concentrate is unkown. Thus the advantage of this study is the effective extraction of Sc from high acid (4 mol/L HCl) and the excellent selectivity towards Sc among other REEs.
Table 2 Comparison of extraction methods of Sc
4 Conclusions
To efficiently recover Sc from ion-adsorption REEs concentrate, the extraction behavior and mechanism of HEHEHP in sulfonated kerosene as extractant towards Sc were firstly studied. An excellent extraction selectivity towards Sc over other REEs in 0.1-4 mol/L HCl solution is observed. The equilibrium time is obtained within 20 min and the extraction process is exothermal in 4 mol/L HCl solution. The stripping of Sc from loaded HEHEHP can be successfully achieved in a single step with 5 mol/L NaOH solution at 363 K. The interaction mechanism between HEHEHP and Sc in 4 mol/L HCl solution has been investigated based on slope analysis, saturation extraction, IR, and NMR analysis, which confirms the extracted complex formed by cation exchange between P―O―H and Sc and coordination process between P=O and Sc. Furthermore, a flowsheet for Sc recovery from ion-adsorption REEs concentrate is proposed and testified. The single extraction efficiency close to 100% and the stripping rate under optimal conditions is 89%. These results above indicate that it’s a promising flowsheet for the recovery of Sc from ion-adsorption REEs concentrate.
Contributors
ZHANG Wei conducted the study and wrote the manuscript. ZHU Hao and LIU Shu-feng was involved in spectrum analysis. NING Shun-yan reviewed and edited the manuscript. WEI Yue-zhou planned and executed the research activity.
Conflict of interest
ZHANG Wei, ZHU Hao, LIU Shu-feng, NING Shun-yan and WEI Yue-zhou declare that they have no conflict of interest.
References
[1] CUI Hong-min, CHEN Ji, LI Hai-lian, ZOU Dan, LIU Yu, DENG Yue-feng. High-performance polymer-supported extractants with phosphonate ligands for scandium(III) separation [J]. AlChE J, 2016, 62(7): 2479-2489. DOI: 10.1002/aic.15236.
[2] HERNANDEZ R, VALDOVINOS H F, YANG Yu-nan, CHAKRAVARTY R, HONG Hao, BARNHART T E, CAI Wei-bo. 44Sc: An attractive isotope for peptide-based PET imaging [J]. Mol Pharm, 2014, 11(8): 2954-2961. DOI: 10.1021/mp500343j.
[3] ZHANG Wei, NING Shun-yan, ZHANG Shi-chang, WANG Si-yi, ZHOU Jie, WANG Xin-peng, WEI Yue-zhou. Synthesis of functional silica composite resin for the selective separation of zirconium from scandium [J]. Microporous Mesoporous Mater, 2019, 288: 109602. DOI: 10.1016/j.micromeso.2019.109602.
[4] ZHANG Wei, YU Shu-qi, ZHANG Shi-chang, ZHOU Jie, NING Shun-yan, WANG Xin-peng, WEI Yue-zhou. Separation of scandium from the other rare earth elements with a novel macro-porous silica-polymer based adsorbent HDEHP/SiO2-P [J]. Hydrometallurgy, 2019, 185: 117-124. DOI: 10.1016/j.hydromet.2019.01.012.
[5] YU Qing, NING Shun-yan, ZHANG Wei, WANG Xin-peng, WEI Yue-zhou. Recovery of scandium from sulfuric acid solution with a macro porous TRPO/SiO2-P adsorbent [J]. Hydrometallurgy, 2018, 181: 74-81. DOI: 10.1016/ j.hydromet.2018.07.025.
[6] GIRET S, HU Yi-mu, MASOUMIFARD N, BOULANGER J F, ESTELLE J, KLEITZ F, LARIVIERE D. Selective separation and preconcentration of scandium with mesoporous silica [J]. ACS Appl Mater Interfaces, 2018, 10(1): 448-457. DOI: 10.1021/acsami.7b13336.
[7] ZHANG Na, LI Hong-xu, LIU Xiao-ming. Recovery of scandium from bauxite residue-red mud: A review [J]. Rare Met, 2016, 35(12): 887-900. DOI: 10.1007/s12598-016- 0805-5.
[8] ZHANG Xue-kai, ZHOU Kang-gen, CHEN Wei, LEI Qing-yuan, HUANG Ying, PENG Chang-hong. Recovery of iron and rare earth elements from red mud through an acid leaching-stepwise extraction approach [J]. J Cent South Univ, 2019, 26(2): 458-466. DOI: 10.1007/s11771-019-4018-6.
[9] PENG Zhen, LI Qing-gang, LI Zhao-yang, ZHANG Gui-qing, CAO Zuo-ying, GUAN Wen-juan. Removal of impurities from scandium solutions by ion exchange [J]. J Cent South Univ, 2018, 25(12): 2953-2961. DOI: 10.1007/s11771-018-3965-7.
[10] YANG X-jin, LIN Ai-jun, LI Xiao-liang, WU Yi-ding, ZHOU Wen-bin, CHEN Zhan-heng. China’s ion-adsorption rare earth resources, mining consequences and preservation [J]. Environ Dev, 2013, 8: 131-136. DOI: 10.1016/ j.envdev.2013.03.006.
[11] WANG Yan-liang, GUO Xiang-guang, BI Yan-feng, SU Jia, KONG Wei-chang, SUN Xiao-qi. Enrichment of trace rare earth elements from the leaching liquor of ion-absorption minerals using a solid complex centrifugal separation process [J]. Green Chem, 2018, 20(9): 1998-2006. DOI: 10.1039/c7gc03674d.
[12] WU Dong-bei, NIU Chun-ji, LI De-qian, BAI Yan. Solvent extraction of scandium(III), yttrium(III), lanthanum(III) and gadolinium(III) using Cyanex 302 in heptane from hydrochloric acid solutions [J]. J Alloys Compd, 2004, 374(1, 2): 442-446. DOI: 10.1016/j.jallcom.2003.11.058.
[13] WANG Wei-wei, CHENG Chu-yong. Separation and purification of scandium by solvent extraction and related technologies: A review [J]. J Chem Technol Biotechnol, 2011, 86(10): 1237-1246. DOI: 10.1002/jctb.2655.
[14] DAS S, BEHERA S S, MURMU B M, MOHAPATRA R K, MANDAL D, SAMANTRAY R, PARHI P K, SENANAYAKE G. Extraction of scandium(III) from acidic solutions using organo-phosphoric acid reagents: A comparative study [J]. Sep Purif Technol, 2018, 202: 248-258. DOI: 10.1016/j.seppur.2018.03.023.
[15] WANG Chun, LI De-qian. Solvent extraction of Sc(III), Zr(lV), Th(IV), Fe(lll), and Lu(lll) with thiosubstituted organophosphinic acid extractants [J]. Solvent Extr Ion Exch, 1995, 13(3): 503-523. DOI: 10.1080/07366299508918288.
[16] WANG Wei-wei, PRANOLO Y, CHENG Chu-yong. Recovery of scandium from synthetic red mud leach solutions by solvent extraction with D2EHPA [J]. Sep Purif Technol, 2013, 108: 96-102. DOI: 10.1016/j.seppur.2013. 02.001.
[17] ZHOU Jie, YU Qing, HUANG Yu, MENG Jie-jie, CHEN Ye-dan, NING Shun-yan, WANG Xin-peng, WEI Yue-zhou, YIN Xiang-biao, LIANG Jie. Recovery of scandium from white waste acid generated from the titanium sulphate process using solvent extraction with TRPO [J]. Hydrometallurgy, 2020, 195: 105398. DOI: 10.1016/ j.hydromet.2020.105398.
[18] ZHOU Jie, NING Shun-yan, MENG Jie-jie, ZHANG Shi-chang, ZHANG Wei, WANG Si-yi, CHEN Ye-dan, WANG Xin-peng, WEI Yue-zhou. Purification of scandium from concentrate generated from titanium pigments production waste [J]. J Rare Earths, 2021, 39(2): 194-200. DOI: 10.1016/j.jre.2020.02.008
[19] LIU Ying-hui, CHEN Ji, LI De-qian. Application and perspective of ionic liquids on rare earths green separation [J]. Sep Sci Technol, 2012, 47(2): 223-232. DOI: 10.1080/01496395.2011.635171.
[20] WANG Liang-shi, HUANG Xiao-wei, YU Ying, XIAO Yan-fei, LONG Zhi-qi, CUI Da-li. Eliminating ammonia emissions during rare earth separation through control of equilibrium acidity in a HEH(EHP)-Cl system [J]. Green Chem, 2013, 15(7): 1889-1894. DOI: 10.1039/c3gc40470f.
[21] LIU Chuan-ying, CHEN Li, CHEN Ji, ZOU Dan, DENG Yue-feng, LI De-qian. Application of P507 and isooctanol extraction system in recovery of scandium from simulated red mud leach solution [J]. J Rare Earths, 2019, 37(9): 1002-1008. DOI: 10.1016/j.jre.2018.12.004.
[22] HUGHES I D, DANE M, ERNST A, HERGERT W, LUDERS M, POULTER J, STAUNTON J B, SVANE A, SZOTEK Z, TEMMERMAN W M. Lanthanide contraction and magnetism in the heavy rare earth elements [J]. Nature, 2007, 446(7136): 650-653. DOI: 10.1038/nature05668.
[23] WANG Chun, LI De-qian. Extraction Mechanism of Sc(III) and separation from Th(IV), Fe(III) and Lu(III) with bis(2,4,4-trimethylpentyl)phos- phinic acid in N-hexane from sulphuric acid solutions [J]. Solvent Extr Ion Exch, 1994, 12(3): 615-631. DOI: 10.1080/07366299 408918228.
[24] JAYACHANDRAN J, DHADKE P M. Solvent extraction of titanium (IV) with 2-ethyl hexyl phosphonic acid mono 2-ethyl hexyl ester (HEHEHP) [J]. J Chem Eng Jpn, 1998, 31(3): 465-468. DOI: 10.1252/jcej.31.465.
[25] MICHELSEN O B, SMUTZ M. Separation of yttrium, holmium, and erbium with di-(2-ethylhexyl) phosphoric acid in chloride and nitrate systems [J]. J Inorg Nucl Chem, 1971, 33(1): 265-278. DOI: 10.1016/0022-1902(71)80028-3.
[26] ZHAO Jun-mei, HUO Fang, PAN Feng, LI De-qian, LIU Hui-zhou. Distribution behaviors of light rare earths by di-(2-ethylhexyl) 2-ethylhexyl phosphonate in kerosene under the action of a self-salting-out effect [J]. Ind Eng Chem Res, 2014, 53(4): 1598-1605. DOI: 10.1021/ ie403414j.
[27] HUANG Xiao-wei, DONG Jin-shi, WANG Liang-shi, FENG Zong-yu, XUE Qian-nan, MENG Xiang-long. Selective recovery of rare earth elements from ion-adsorption rare earth element ores by stepwise extraction with HEH(EHP) and HDEHP [J]. Green Chem, 2017, 19(5): 1345-1352. DOI: 10.1039/c6gc03388a.
[28] ZHU Xiao-bo, LI Wang, TANG Sen, ZENG Ma-jian, BAI Peng-yuan, CHEN Lun-jian. Selective recovery of vanadium and scandium by ion exchange with D201 and solvent extraction using P507 from hydrochloric acid leaching solution of red mud [J]. Chemosphere, 2017, 175: 365-372. DOI: 10.1016/j.chemosphere.2017.02.083.
[29] ZHANG Feng-yun, DAI Jing-jie, WANG A-min, WU Wen-yuan. Investigation of the synergistic extraction behavior between cerium (III) and two acidic organophosphorus extractants using FT-IR, NMR and mass spectrometry [J]. Inorg Chim Acta, 2017, 466: 333-342. DOI: 10.1016/j.ica.2017.06.016.
[30] DONG Jin-shi, XU Yang, WANG Liang-shi, HUANG Xiao-wei, LONG Zhi-qi, WU Sheng-xi. Thermodynamics and kinetics of lutetium extraction with HEH(EHP) in hydrochloric acid medium [J]. J Rare Earths, 2016, 34(3): 300-307. DOI: 10.1016/s1002-0721(16)60029-7.
[31] TKAC P, VANDEGRIFT G F, LUMETTA G J, GELIS A V. Study of the interaction between HDEHP and CMPO and its effect on the extraction of selected lanthanides [J]. Ind Eng Chem Res, 2012, 51(31): 10433-10444. DOI: 10.1021/ ie300326d.
[32] ZHAO Qi, LI Yan-ling, KUANG Sheng-ting, ZHANG Zhi-feng, BIAN Xue, LIAO Wu-ping. Synergistic extraction of heavy rare earths by mixture of α-aminophosphonic acid HEHAMP and HEHEHP [J]. J Rare Earths, 2019, 37(4): 422-428. DOI: 10.1016/j.jre.2018.07.013.
[33] CHEN Kai-hua, HE Yuan, SRINIVASAKANNAN C, LI Shi-wei, YIN Shao-hua, PENG Jin-hui, GUO Sheng-hui, ZHANG Li-bo. Characterization of the interaction of rare earth elements with P507 in a microfluidic extraction system using spectroscopic analysis [J]. Chem Eng J, 2019, 356: 453-460. DOI: 10.1016/j.cej.2018.09.039.
[34] LI De-qian, ZHANG Jie, XU Min. Studies of extraction mechanism of rare earth compounds with mono (2-ethyl hexyl) 2-ethyl hexyl phosphonate (HEH (EHP)) [J]. Chin J Appl Chem, 1985, 2(2): 17-23. (in Chinese)
[35] FERRIS T D, ZEIDLER M D, FARRAR T C. The concentration dependence of the proton chemical shift and the deuterium quadrupole coupling parameter for binary solutions of ethanol [J]. Mol Phys, 2000, 98(11): 737-744. DOI: 10.1080/00268970009483343.
[36] LIAO Chun-sheng, JIA Jiang-tao, ZHANG Yi, XU Gang, YAN Chun-hua, LI Biao-guo, XU Guang-xian. Extraction of scandium from ion-adsorptive rare earth deposit by naphthenic acid [J]. J Alloys Compd, 2001, 323-324: 833-837. DOI: 10.1016/S0925-8388(01)01155-0.
[37] BABA Y, FUKAMI A, KUBOTA F, KAMIYA N, GOTO M. Selective extraction of scandium from yttrium and lanthanides with amic acid-type extractant containing alkylamide and glycine moieties [J]. RSC Adv, 2014, 4(92): 50726-50730. DOI: 10.1039/c4ra08897b.
[38] WU Dong-bei, XIONG Ying, LI De-qian. Studies on the roles of different components in cyanex 302 for rare earth ions extraction and separation [J]. Sep Sci Technol, 2006, 41(8): 1725-1739. DOI: 10.1080/014963906 00674943.
(Edited by ZHENG Yu-tong)
中文导读
HEHEHP从离子吸附型稀土精矿中回收钪
摘要:2-乙基己基磷酸单(2-乙基己基)酯(HEHEHP)对4 mol/L HCl稀土溶液中的钪具有极高的选择性,其分离系数βSc/REEs超过了8000,且在20 min内达到萃取平衡。萃取后的钪在363 K下,利用5 mol/L NaOH作为洗脱剂,洗脱率达到92.1%。利用斜率法、饱和萃取法、红外光谱分析和核磁共振分析阐明了萃取机理。结果表明,HEHEHP在4 mol/L HCl溶液中萃取Sc主要是以P―O―H中的H与Sc的离子交换过程,以及P=O与Sc的配位过程为主导,且6个萃取剂分子作为二聚体参与萃取。最后,提出了从离子吸附型稀土精矿中回收钪的工艺流程,并在实验室进行了验证。
关键词:钪;HEHEHP;溶剂萃取;离子吸附型稀土
Foundation item: Projects(11705032, 11975082, U1967218) supported by the National Natural Science Foundation of China; Project(2017GXNSFBA198175) supported by the Natural Science Foundation of Guangxi Province, China; Project(AA17204100) supported by the Science and Technology Major Project of Guangxi Province, China
Received date: 2020-05-08; Accepted date: 2020-11-23
Corresponding author: NING Shun-yan, PhD, Associate Professor; Tel: +86-771-3392507; E-mail: ningshunyan@gxu.edu.cn; ORCID: https://orcid.org/0000-0002-9573-5960

