
Morphology modification of Mg2Si by Sr addition in Mg-4%Si alloy
TANG Shou-qiu1, ZHOU Ji-xue1, 2, TIAN Chang-wen1, YANG Yuan-sheng1, 2
1. New Materials Research Institute, Shandong Academy of Sciences, Ji’nan 250014, China;
2. Institute of Metal Research, Chinese Academy of Sciences, Shenyang 110016, China
Received 17 February 2011; accepted 25 April 2011
Abstract: A modification of Mg2Si in the hypereutectic Mg-4%Si alloy (mass fraction) with Sr was investigated. Two types of Mg2Si in the alloys were found: polygonal primary Mg2Si and Chinese script type eutectic Mg2Si. Adding Al-10% Sr master alloy to the Mg-4%Si alloy clearly reduced the average size of primary Mg2Si and changed the morphology of eutectic Mg2Si from Chinese script type to polyhedral or fine fibre shape. The refinement of primary Mg2Si is mainly attributed to the heterogeneous nucleation mechanism induced by the Sr-rich particles. The modification of eutectic Mg2Si results from the dissolved Sr, which alters the preferred growth manner of the eutectic.
Key words: Mg-Si alloy; Al-Sr alloy; Mg2Si; modification; refinement
1 Introduction
Growing demands from the automobile and aerospace industries for lightweight components that reduce energy consumption and air pollution lead to an increasing interest in magnesium alloys and their composites because of their low density, high strength-to-mass ratio and good machinability [1-3].
Among magnesium alloys, the Si-containing Mg alloys offer significant potential as structural materials that can be subjected to comparatively high temperature [4-7]. This is because they are reinforced with Mg2Si, which exhibit a high melting temperature of 1 085 °C, a low density of 1.99 g/cm3, a high hardness of 4.5?109 N/m2, a low thermal expansion coefficient of 7.5?10-6 K-1, and a reasonably high elastic modulus of 120 GPa [8-9].
On the other hand, Si-containing Mg alloys exhibit a low strength and very low ductility due to the large Mg2Si particles and the brittle eutectic Mg2Si [10-11]. For this reason, refinement and modification of Mg2Si will be needed to improve the mechanical properties. It was reported that the additions of KBF4, Ca, P, Y or RE produced positive effects on the morphology of Mg2Si in magnesium alloys containing Si [10-13]. As a beneficial element to the grain refinement of Mg-Al alloys [14-15], Sr was also proven effective in modifying Mg2Si in Mg-Al-Si alloys by changing their shape from network shape to fine fibrous shape [16-20]. YANG et al [17-18] reported that Sr could refine Mg2Si in AZ61-0.7Si alloy because Al4Sr can theoretically act as the heterogeneous nucleus for Mg2Si according to the calculation of the disregistry by the two-dimensional lattice misfit mathematical model, and Sr may lower the onset crystallizing temperature and increase the undercooling level. SRINIVASAN et al [19] found that Sr may be incorporated in the Mg2Si precipitates during solidification of AZ91-Si alloy.
To date, less work has been carried out with regard to the modification effect of Sr on morphology of primary Mg2Si in hypereutectic Mg-Si alloys. The further research is needed to understand the mechanism for the refinement in structure. The present paper studies the modification effect of Sr on Mg2Si (both eutectic and primary Mg2Si crystals) by adding the Al-10%Sr (mass fraction) alloy to Mg-4%Si (mass fraction) alloy. The modification effect of Al on the Mg2Si phases is also investigated to clarify the modification mechanism of Sr in Al-10%Sr master alloys.
2 Experimental
Commercial pure Mg ingots (99.9% purity) and Si (99.9% purity) were used as raw materials to prepare Mg-4%Si alloy. The Mg-4%Si alloy was melted at 750 °C in an electrical resistance furnace under a protective atmosphere (0.5% SF6+99.5%CO2, volume fraction), and 2.7% Al or 3% Al-10%Sr master alloy was then added to the Mg-Si alloy melt. The melt was stirred for 5 min, held at 750 °C for 30 min, and poured into a steel mold preheated at 200 °C to cast bar samples of d40 mm?100 mm. The microstructure was investigated by optical microscopy. Characteristic and qualitative analysis of the microstructure were conducted using SEM equipped with an energy-dispersive spectrometer (EDS). The size of the primary Mg2Si particles was measured and calculated with SISCIAS8.0 software.
Table 1 lists the actual chemical compositions of the experimental alloys, which were analyzed by inductively coupled plasma spectroscopy.
Table 1 Chemical compositions of experimental alloys
3 Results and discussion
3.1 Microstructure of Mg-4%Si alloy
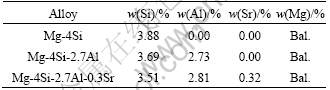
The microstructure and EDS analysis of Mg-4%Si alloy are shown in Fig. 1 and Table 2. According to the Mg-Si binary phase diagram, Mg-4%Si alloy is a hypereutectic alloy with a solidified microstructure of Mg2Si primary and Mg2Si-Mg eutectic. The dark area (A) is primary Mg2Si, the gray area (B) is Mg, and the area C is a rod-like shaped eutectic Mg2Si. The primary Mg2Si phases are surrounded by Mg halos, around which the Mg-Mg2Si eutectic is formed, which is similar to the microstructure of the Mg-5%Si alloy [12]. Due to the relatively high cooling rate, its solidification deviates from the equilibrium route. During solidification, the Mg2Si precipitates primarily, resulting in an enrichment of magnesium around the primary Mg2Si, which is attributed to the rejection of magnesium solute. Mg is thus able to nucleate and grow surrounding the primary Mg2Si. As the temperature decreases to the eutectic point, the eutectic structures of Mg2Si-Mg form around the Mg halos.
Table 2 Chemical compositions of zones A, B and C analyzed by EDS in Fig. 1(a)

3.2 Effects of Sr addition on Mg2Si
The microstructures of Mg-4%Si alloys unmodified and modified by 2.7%Al or 3% Al-10%Sr are compared in Fig. 2. The magnified microstructures of Mg-4%Si alloys modified by 3% Al-10%Sr master alloy is shown in Fig. 3. By addition of 3% Al-10%Sr to Mg-4%Si alloy, the shape of the primary Mg2Si does not change obviously while the morphology of eutectic Mg2Si phase changes from Chinese script type to a polyhedral or fine fibers shape, as shown in Figs. 2(a), 2(b) and Fig. 3. Furthermore, the average size of primary Mg2Si decreases from about 46 μm to 16 μm. In contrast, when only 2.7% is added to the alloy, the morphology of primary Mg2Si or eutectic Mg2Si does not change obviously and the average size of primary Mg2Si slightly reduces from about 46 μm to 36 μm while the amount of eutectic Mg2Si decreases, as shown in Fig. 2(c).
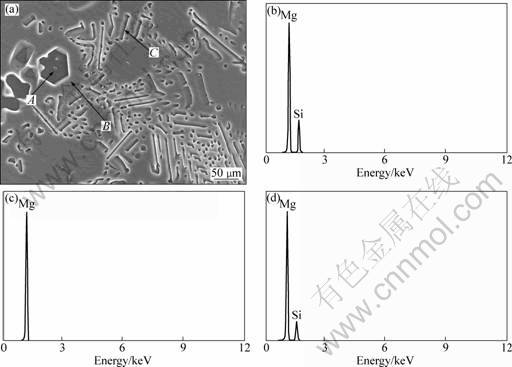
Fig. 1 SEM image of Mg-4%Si alloy (a) and EDS spectra for primary Mg2Si marked with ‘A’ (b), Mg marked with ‘B’ (c) and eutectic Mg2Si marked with ‘C’ (d)
SEM image of the primary Mg2Si phase, and the concentration profiles of elements Mg, Al, Si and Sr in Mg-4%Si alloy with 3% Al-10%Sr, are shown in Fig. 4. Small particles are found to exist inside the primary Mg2Si phases, which presumably act as nucleation sites for the primary Mg2Si. The concentration profiles of elements show that the primary Mg2Si, especially the core particle inside, is enriched in Sr element. As shown in Fig. 4, the separate Al addition could not induce an obvious refinement of primary Mg2Si, nor is there a core particle introduced inside the primary Mg2Si in Mg-4%Si alloy by the addition of 2.7% Al. It is thus presumed that the refinement of primary Mg2Si mainly depends on the mechanism of heterogeneous nucleation by the core particles that are rich in Sr. According to YANG et al [17-18], Al4Sr can theoretically act as the heterogeneous nucleus for the Mg2Si particles according to the calculation of the disregistry by the two- dimensional lattice misfit mathematical model. In addition, the Sr dissolved within primary Mg2Si is able to modify the primary Mg2Si crystals.

Fig. 2 Microstructures of Mg-4%Si alloys unmodified (a) and modified by 3% Al-10%Sr master alloy (b) and 2.7% Al (c)

Fig. 3 Magnified microstructure of Mg-4%Si alloy modified by 3% Al-10%Sr master alloy
A SEM image of eutectic Mg2Si in Mg-4%Si alloy with 3% Al-10%Sr alloy is shown in Fig. 5. The average contents of Mg, Al, Si, Sr within areas A and B in Fig. 5, measured by EDS analysis, are listed in Table 3. As shown in Fig. 5, there is eutectic Mg2Si in area A but none in area B. It is also noted that the average Sr content in area A is much higher than that in area B, as shown in Table 3, indicating that the eutectic Mg2Si is enriched in Sr either inside the eutectic Mg2Si or at the interfaces of eutectic Mg2Si and Mg. However, no nucleation core was observed in the eutectic Mg2Si. It is concluded that the dissolved Sr is responsible for the modification of eutectic Mg2Si.
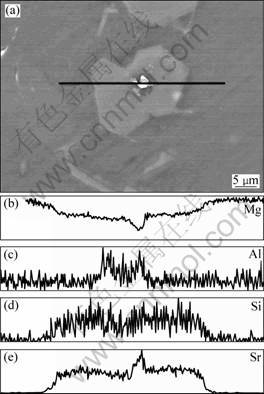
Fig. 4 SEM image (a) and line scanning maps of element Mg (b), Al (c), Si (d) and Sr (e) of primary phase Mg2Si in Mg-4%Si alloy modified by 3% Al-10%Sr master alloy

Fig. 5 SEM image of Mg-4%Si alloy modified by 3%Al- 10%Sr master alloy
Table 3 Chemical compositions of zones A and B analyzed by EDS in Fig. 5

In the unmodified alloys, the coarse eutectic Mg2Si crystals are formed by the preferred growth that occurs at the tips of the branches [18]. When the alloy is modified with 3% Al-10%Sr during the solidification, in addition to the segregation of Sr at the liquid-solid interface, some of the alloys may be adsorbed on the Mg2Si crystal plane boundaries [19], causing changes of the surface energy of the Mg2Si crystals from lattice distortion. The preferred growth manner of the eutectic Mg2Si is depressed, and another type of growth, the isotropic growth manner, is present during the solidification of the modified alloys. Thus, the eutectic Mg2Si crystals are modified from Chinese script shape to polyhedral or fine fibers shape.
4 Conclusions
1) The solidified microstructure of Mg-4%Si alloy with 3% Al-10%Sr master alloy consists of Mg2Si primary and Mg2Si- Mg eutectic. The primary Mg2Si crystals are surrounded by a layer of divorced eutectic Mg halos, which are surrounded by the eutectic Mg2Si-Mg structures.
2) The addition of 3% Al-10%Sr master alloy to Mg-4%Si alloys modifies the eutectic Mg2Si from Chinese script shape to polyhedral or fine fibers shape, and clearly decreases the average size of primary Mg2Si phase.
3) The modification of primary Mg2Si phase depends on the heterogeneous nucleation mechanism induced by Sr-rich particles. The modification of eutectic Mg2Si phase mainly depends on the dissolved Sr, in which the preferred growth manner of the Mg2Si phase is depressed, while the isotropic growth is enhanced in the modified alloys.
References
[1] KIM J M, SHIN K, KIM K T, JUNG W J. Microstructure and mechanical properties of a thixocast Mg-Cu-Y alloy [J]. Scripta Mater, 2003, 49: 687-691.
[2] HUMBLE P. Towards a cheap resistant magnesium alloy [J]. Mater Forum, 1997, 21: 45-56.
[3] BRONFIN B, KATSIR M, AGHION E. Preparation and solidification features of AS21 magnesium alloy [J]. Mater Sci Eng A, 2001, 302: 46-50.
[4] LU L, LAI M O, HOE M L. Formation of nanocrystalline Mg2Si and Mg2Si dispersion strengthened Mg-Al alloy by mechanical alloying [J]. Nanostruct Mater, 1998, 10: 551-563.
[5] MABUCHI M, HIGASHI K. Strengthening mechanisms of Mg-Si alloys [J]. Acta Mater, 1996, 44(11): 4611-4618.
[6] ZHANG J, FAN Z, WANG Y Q, ZHOU B L. Microstructural development of Al-15wt.%Mg2Si in situ composite with mischmetal addition [J]. Mater Sci Eng A, 2000, 281: 104-112.
[7] WANG H Y, WANG W, ZHA M, ZHENG N, GU Z H, LI D, JIANG Q C. Influence of the amount of KBF4 on the morphology of Mg2Si in Mg-5Si alloys [J]. Mater Chem Phys, 2008, 108: 353-358.
[8] PAN Y C, LIU X F, YANG H. Microstructural formation in a hypereutectic Mg-Si alloy [J]. Mater Charact, 2005, 55: 241-247.
[9] BLUM W, ZHANG P, WATZINGER B, GROSSMANN B V, HALDENWANGER H G. Comparative study of creep of the die-cast Mg-alloys AZ91, AS21, AS41, AM60 and AE42 [J]. Mater Sci Eng A, 2001, 319-321: 735-740.
[10] WANG H Y, JIANG Q C, MA B X, WANG Y, WANG J G, LI J B. Modification of Mg2Si in Mg-Si alloys with K2TiF6, KBF4 and KBF4+K2TiF6 [J]. J Alloys Compd, 2005, 387: 105-108.
[11] KIM J J, KIM D H, SHIN K S, KIM N J. Modification of Mg2Si morphology in squeeze cast Mg-Al-Zn-Si alloys by Ca or P addition [J]. Scripta Mater, 1999, 41: 333-340.
[12] JIANG Q C, WANG H Y, WANG Y, MA B X, WANG J G. Modification of Mg2Si in Mg-Si alloys with yttrium [J]. Mater Sci Eng A, 2005, 392: 130-135.
[13] SPIGARELLI S, CICCARELLI D, EVANGELISTA E. Compressive deformation of an Mg-Al-Si-RE alloy between 120 and 180 °C [J]. Mater Lett, 2004, 58: 460-464.
[14] LEE Y C, DAHLE A K, StJOHN D H. The role of solute in grain refinement of magnesium [J]. Metall Mater Trans A, 2000, 31: 2895-2905.
[15] ZHOU Ji-xue, WANG Bin, TONG Wen-hui, YANG Yuan-sheng. Effect of strontium addition on dendrite growth and phase precipitation in AZ91D magnesium alloy [J]. Acta Metallurgica Sinica, 2007, 43(11): 1171-1175. (in Chinese)
[16] NAM K Y, SONG D H, LEE C W, LEE S W, PARK Y H, CHO K M, PARK I M. Modification of Mg2Si morphology in as-cast Mg-Al-Si alloys with strontium and antimony [J]. Mater Sci Forum, 2006, 510-511: 238-241.
[17] YANG Ming-bo, PAN Fu-sheng, SHEN Jia, BAI Liang. Comparison of Sb and Sr on modification and refinement of Mg2Si phase in AZ61-0.7Si magnesium alloy [J]. Transactions of Nonferrous Metals Society of China, 2009, 19: 287-292.
[18] YANG Ming-bo, PAN Fu-sheng, SHEN Jia. Modification and refinement mechanism of Mg2Si phase in Sr-containing AZ61-0.7Si magnesium alloy [J]. China Foundry, 2009, 6(1): 37-42. (in Chinese)
[19] SRINIVASAN A, PILLAI U T S, SWAMINATHAN J, DAS S K, PAI B C. Observations of microstructural refinement in Mg-Al-Si alloys containing strontium [J]. J Mater Sci, 2006, 41: 6087-6089.
[20] WANG Zhi-ming, GENG Hao-ran, TENG Xin-ying, ZHOU Guo-rong. Influence of Sr on microstructure and mechanical properties of AS41 alloy [J]. Foundry Technology, 2009, 30(10): 1299-1302.
Sr对Mg-4%Si合金中Mg2Si的变质作用
唐守秋1,周吉学1, 2,田长文1,杨院生1, 2
1. 山东省科学院 新材料研究所,济南 250014;
2. 中国科学院 金属研究所,沈阳 110016
摘 要:研究Sr对过共晶Mg-4%Si(质量分数)合金中Mg2Si相的变质作用与机理。Mg-4%Si合金中存在多面体形初生Mg2Si相与汉字状共晶Mg2Si相。添加Al-10%Sr可以明显细化初生Mg2Si相,同时可以将共晶Mg2Si相由汉字状变质为多面体状或者纤维状。对初生Mg2Si相的细化作用主要是由凝固过程中含Sr颗粒的异质形核作用引起的,而对共晶Mg2Si相的变质作用是由在凝固过程中熔体中的Sr原子在Mg2Si晶体生长表面富集,从而改变了其生长优势所致的。
关键词:Mg-Si合金;Al-Sr合金;Mg2Si;变质;细化
(Edited by YANG Hua)
Foundation item: Project (2011BAE22B00) supported by the National Key Technology R&D Program of China; Projects (ZR2010EQ021, ZR2010EL013) supported by the Natural Science Foundation of Shandong Province, China
Corresponding author: ZHOU Ji-xue; Tel/Fax: +86-531-88728308; E-mail: jxzhou_1982@163.com
DOI: 10.1016/S1003-6326(11)60952-7