
Effect of element Gd on phase constituent and mechanical property of Mg-5Sn-1Ca alloy
SHI Bin-qing(施斌卿)1, 2, CHEN Rong-shi(陈荣石)1, KE Wei(柯伟)1
1. State Key Laboratory for Corrosion and Protection, Institute of Metal Research, Shenyang 110016, China;
2. Graduate School of the Chinese Academy of Sciences, Beijing 100049, China
Received 23 September 2009; accepted 30 January 2010
Abstract: The Mg-5Sn-1Ca-xGd (x=0, 1) alloys were chosen to investigate the change in solidification paths, phase formation and mechanical properties. The microstructure of as-cast Mg-5Sn-1Ca alloy is composed of α-Mg, Mg2Sn and CaMgSn phases. With the addition of Gd, the formation of the Mg2Sn phase is impeded and the CaMgSn phase is refined, whereas the ultimate tensile strength and elongation decrease. The possible reasons for the variation in microstructure and mechanical properties were discussed.
Key words: magnesium alloy; Mg-Sn-Ca alloy; thermal analysis; mechanical property
1 Introduction
Magnesium alloys are known for lightweight, high specific stiffness and excellent cast ability and workability, and are deemed to be applied in transportation industry potentially, which requires superior heat resistance and mechanical properties[1]. Mg-Sn-Ca based alloys are being investigated, where Mg2Sn phase shows high heat resistance and Ca enhances mechanical properties at elevated temperature by forming stable CaMgSn phase in the matrix[2-5]. Recently, Mg-3Sn-2Ca alloy has been identified as one of the most promising magnesium alloys, which processes excellent thermal stability[3-7]. However, less work was conducted on Mg-Sn-Ca alloys with other compositions. Accordingly, Mg-5Sn-1Ca was chosen to investigate practical solidification paths and phase constituents.
The cooling curve method is the most widely used thermal analysis tool for investigating solidification of metals and alloys owing to its advantage in operating on casting site and controlling cooling rate compared with other techniques, such as DTA or DSC, which is designed for determining equilibrium characterization. The foremost use of cooling curves has been the determination of arrest points during solidification. Arrest points on cooling curves are related to the precipitation events, as the latent heat of formation is released and the cooling is arrested temporarily[8].
Rare earth (RE) elements, such as Gd and Y, can improve the strength of magnesium alloys at both room and elevated temperatures[9-10]. In this work, 1% Gd was added to Mg-5Sn-1Ca alloy for achieving more excellent mechanical property.
2 Experimental
Commercial pure magnesium, tin, calcium, and gadolinium were added into a heat-resistance mild steel crucible in turn to gain as-cast Mg-Sn-Ca-xGd (x=0.1) ingots. When the melt was stirred equably and held for 30 min, it was cast into a metallic mould, which was preheated previously. Samples for thermal analysis were cut from the same position of the cast ingots and machined into cylinders of 25 mm in diameter and 35 mm in length. The samples were remelted in a steel crucible by electrical resistance furnace. One shielded K-type thermocouple was placed at the center of the crucible in the melt. The temperature changes during the solidification process were recorded by using a high- speed data acquisition system linked to the computer.
The cooling rate was around 1 K/s. The thermal analysis experiment was conducted three times for each alloy composition to ensure the reliability of the results.
The microstructures of the as-cast alloys were examined by SEM (Philip XL30 ESEM-FEG/EDAX scanning electron microscope). Phase analysis was determined by a D/Max 2400 X-ray diffractometer (XRD). The chemical composition analysis of the ingots was made by the ICP-AES method and the results are listed in Table 1.
The tensile mechanical properties of experimental alloys at room temperature were determined from a complete stress―strain curve. The ultimate tensile strength (UTS) and elongation to failure were obtained based on the average of at least three tests. To examine the microstructure stability at evaluated temperatures, the samples of experimental alloys were subjected to a solution heat treatment (450 ?C, 24 h, water cooled).
Table 1 Chemical compositions of as-cast Mg-Sn-Ca alloys (mass fraction, %)
3 Results and discussion
3.1 Phase formation and solidification paths
Fig.1 and Fig.2(a) show the backscattered electron images and thermal analysis result of as-cast TX51 alloy, respectively. The first derivative of the cooling curve (dT/dt) was determined to enhance slope changes that are related to the solidification reactions for different phases, and to facilitate the determination of the critical solidification characteristics of the alloys. In the TX51 alloy, two well-defined peaks are observed at 641 ?C and 555.4 ?C, which correspond to two reactions taking place during solidification. According to the XRD pattern shown in Fig.3(a), the as-cast TX51 alloy is composed of α-Mg, Mg2Sn and CaMgSn phases, so the two actions are associated with the formation of these phases. From microstructure observation shown in Figs.1(a) and (b), one phase is inclined to be found at crossing points of ternary grains, which seems to form at the end stage of solidification. The EDAX analysis of the eutectic phase shows that the mole ratio of elements Mg to Sn is 5?1, so the eutectic phase should be composed of α-Mg and the Mg2Sn phase. In contrast to the eutectic phase, Figs.1(a) and (c) show other two phases with different morphologies: one forms uniformly as isolated needles in the grains, while the other with lamellar shape connects

Fig.1 Backscattered electron images of TX51 alloy: (a), (b), (c) In as-cast state; (d) In solution-treated state
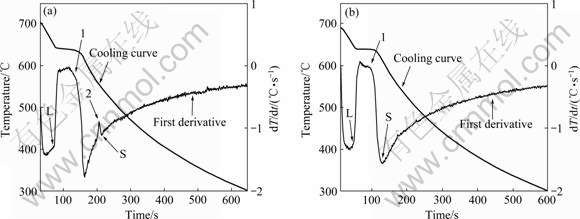
Fig.2 Thermal analysis results of as-cast TX51 alloy (a) and TXG511 alloy (b)
with α-Mg. Combined with the XRD result in Fig.3(a) and EDAX analysis, the needle-shaped and divorced- eutectic phases are determined as the same, the CaMgSn phase, which correspond to the two reactions taking place during solidification process. As demonstrated above, the details of the two reactions during solidification of TX51 alloy are listed in Table 2, which are in agreement with the calculated Mg-Sn-Ca phase diagram[7]. The microstructure of solution-treated TX51 alloy is shown in Fig.1(d). The eutectic Mg2Sn phase dissolves partly into the matrix, but the morphology of CaMgSn phase does not change, which demonstrates that the CaMgSn phase has better heat resistance than the Mg2Sn phase.

Fig.3 XRD patterns of as-cast TX51(a) and TXG511(b) alloys
When Gd is added, as shown in Fig.2(b), only one peak is observed at 640.1 ?C on the cooling curve, and the eutectic reaction related to the formation of Mg2Sn phase is hard to observe. This is in agreement with XRD results in Fig.3(b) and the microstructural observation shown in Fig.4. Fine CaMgSn phase with higher volume
Table 2 Reactions identified by thermal analysis during solidification


Fig.4 Backscattered electron images of as-cast TXG511 alloy: (a) In low magnification; (b) In high magnification
fraction distributes uniformly in the matrix as shown in Fig.4(b). The EDAX analysis shows that it possesses elements Mg, Sn, Ca and Gd with a mole ratio of 4?2?1?1. The maximum solubility of Gd in magnesium (4.53%, mole fraction) is higher than that of Sn (3.35%, mole fraction) and Ca (0.82%, mole fraction)[11], so the diffusibility of Gd is inferior to that of elements Sn and Ca. With the addition of Gd into the melted TX51 alloy, more elements Sn and Ca are ejected to the solid/liquid surface, which provides more nucleation positions and consequently consumes superfluous element Sn in Mg matrix. Maybe, that is why the CaMgSn phase is refined and eutectic Mg2Sn phase disappears. Except for the above analysis, the exact reason for the high volume fraction and refinement of CaMgSn phase in TXG511 alloy is not completely clear. Further investigations need to be carried out.
3.2 Mechanical properties and fracture behaviour
The tensile mechanical properties of experimental alloys are shown in Fig.5, including UTS and elongation. In Fig.5, ‘-C’ after alloy brand denotes the alloy in the as-cast state, and ‘-S’ means the alloy in the solution-treatment state. The tensile mechanical properties of the investigated Mg-Sn-Ca based alloys at room temperature are inferior to those of common magnesium alloys, and the as-cast TXG511 alloy exhibits worse properties than TX51 alloy. As observed in Fig.1(b) the morphology of the CaMgSn phase remains unmodified by solution heat-treatment, so the mechanical properties of TX51 alloy cannot be improved by heat-treatment. As it is known that mechanical properties are associated with microstructure and the common phase in the Mg-Sn-Ca based alloys is CaMgSn phase, so it can be concluded that the CaMgSn phase

Fig.5 Tensile properties of TX51, TXG511 and TX32 alloy (Data of as-cast TX32 alloy referred to Ref[12])
may be the origin of inferior mechanical properties.
As shown in Fig.6(a), the bright broken CaMgSn phase and residual parts distribute on the surface vertical to fracture in the sample after tensile test. The CaMgSn phase possesses different thermal expansion coefficients with Mg matrix, and the combinability between them is infirm. When being loaded, these crisp secondary phases become crack origins as stress concentration points, and then cracks propagate and the sample factures. Fig.6(b) shows the microstructure of as-cast TXG511 alloy after fracture. Compared with TX51, more broken secondary needle-shaped phases distribute uniformly on the fracture surface, which generate more cracks accordingly, and maybe that is why as-cast TXG511 alloy exhibits worse tensile properties than TX51 alloy.

Fig.6 Ruptured microstructures after tensile test: (a) As-cast TX51 alloy; (b) As-cast TXG511 alloy
Fig.7 shows SEM images of tensile-ruptured surfaces of as-cast TX51 alloy and TXG511 alloy. As observed, the fracture surface of TX51 alloy exhibits large cleavage or quasi-cleavage planes, in addition, the micro voids corresponding to fragments of secondary phases are observed. In contrast to TX51 alloy, it is seen that during tensile test the samples of TXG511 alloy suffer little tear deformation, which is the characteristic of brittle fracture.

Fig.7 SEM images of fracture surfaces: (a) As-cast TX51 alloy; (b) As-cast TXG511 alloy
4 Conclusions
1) The microstructure of as-cast TX51 alloy is composed of α-Mg, Mg2Sn and CaMgSn phases. With addition of element Gd, the formation of Mg2Sn phase is impeded and fine CaMgSn phase with higher volume fraction is present. One possible reason is that Gd benefits the ejection of elements Sn and Ca from liquid magnesium, which provides more nucleation positions for the CaMgSn phase and consequently consumes superfluous element Sn in Mg matrix
2) The tensile properties of Mg-Sn-Ca based alloys are inferior to those of common Mg alloys, which may be due to the formation of crisp CaMgSn phase.
References
[1] BAMBERGER M, DEHM G. Trends in the development of new Mg alloys [J]. Annual Review of Materials Research, 2008, 38: 505-533.
[2] BUR?IK J, BUR??KOV V, JIR?SKOV? Y, LEIL ABU T, BLAWERT C, DIETZEL W, HORT N, KAINER K U. Microstructure and micromechanical properties of as-cast Mg-Sn-Ca and Mg-Sn-Mn alloys [C]// Proceedings of the 7th ICMA and their Applications, Dresden, 2006:37-42.
[3] LEIL ABU T, HUANG Y, DIERINGA H, HORT N, KAINER K U, BUR??K J, JIR?SKOV? Y, RAO K P. Effect of heat treatment on the microstructure and creep behavior of Mg-Sn-Ca alloys [J]. Materials Science Forum, 2007, 546/549: 69-72.
[4] BOWLES A L, DIERINGA H, BLAWERT C, HORT N, KAINER K U. Investigations on the magnesium-tin system [J]. Materials Science Forum, 2005, 488/489: 135-138.
[5] HUANG Y D, HORT N, LEIL ABU T, KAINER K U, LIU Y L. Effect of microstructure inhomogeneity on creep response of Mg-Sn alloys [J]. Materials Science Forum, 2007, 345/346: 561-564.
[6] LEIL ABU T, HORT N, DIERINGA H, BLAWERT C, HUANG Y, KAINER K U, RAO K P. Proceedings of the magnesium technology in the global age [C]// The 45th Annual Conference of Metallurgists of CIM. Montreal, 2006: 739-749.
[7] KOZLOV A, OHNO M, LEIL ABU T, HORT N, KAINER K U, SCHMID-FETZER R. Phase equilibria, thermodynamics and solidification microstructures of Mg-Sn-Ca alloys. Part 1: Experimental investigation and thermodynamic modeling of the ternary Mg-Sn-Ca system [J]. Intermetallics, 2008, 16: 316-321.
[8] EMADI D, WHITING L V, NAFISI S, GHOMASHCHI R. Applications of thermal analysis in quality control of solidification processes [J]. Journal of Thermal Analysis, 2005, 81: 235-242.
[9] LIU Xi-bo, CHEN Rong-shi, HAN En-hou. Effects of ageing treatment on microstructures and properties of Mg-Gd-Y-Zr alloys with and without Zn additions [J]. Journal of Alloys and Compounds, 2008, 465: 232-238.
[10] GAO Lei, CHEN Rong-shi, HAN En-hou. Elements of rare-earth elements Gd and Y on the solid solution strengthening of Mg alloys [J]. Journal of Alloys and Compounds, 2009, 481: 379-384.
[11] Binary alloy phase diagrams [M]. OH: ASM International, Materials Park, 1990.
[12] YANG Ming-bo, PAN Fu-sheng, CHENG Liang, SHEN Jia. Effects of cerium on as-cast microstructure and mechanical properties of Mg-3Sn-2Ca magnesium alloy [J]. Mater Sci Eng A, 2009, 512: 132-138.
_______________________
Foundation item: Project(2007CB613704) supported by National Basic Research Program of China
Corresponding author: CHEN Rong-shi; Tel: +86-24-23926646; Fax: +86-24-23894149; E-mail: rschen@imr.ac.cn
(Edited by CHEN Wei-ping)