Zr55Cu30Ni5Al10�����������ĵ�ʱ�͵��¾���
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2012���3��
�������ߣ�J. C. QIAO J. M. PELLETIER
����ҳ�룺577 - 584
�ؼ��ʣ�Zr55Cu30Ni5Al10�������������ȷ�������������ѧ��Avrami ָ��
Key words��Zr55Cu30Ni5Al10; metallic glass; thermal analysis; crystallization kinetics; Avrami exponent

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 577-584
J. C. QIAO, J. M. PELLETIER
Universit�� de Lyon, MATEIS, UMR CNRS5510, Bat. B. Pascal, INSA-Lyon,F-69621 Villeurbanne cedex, France
Received 26 August 2011; accepted 10 December 2011
Abstract: Non-isothermal crystallization transformation kinetics and isothermal crystallization kinetics in super-cooled liquid region (SLR) in Zr55Cu30Ni5Al10 bulk metallic glasses were studied by differential scanning calorimetry (DSC) and X-ray diffraction (XRD). In isochronal mode, the average values of activation energy in Zr55Cu30Ni5Al10 bulk metallic glass determined by different models (Kissinger method, Flynn-Wall-Ozawa method and Augis-Bennett method) are in good agreement with each other. In addition, the isothermal transformation kinetics in Zr55Cu30Ni5Al10 bulk metallic glasses was described by the Johnson-Mehl-Avrami (JMA) model. For Zr55Cu30Ni5Al10 bulk metallic glass, the Avrami exponent n ranges from 2.2 to 2.9, indicating that crystallization mechanism in the bulk metallic glass was mainly diffusion-controlled; crystal growth is controlled by long range ordering diffusion in three-dimensional growth during isothermal crystallization process. The average value of activation energy in Zr55Cu30Ni5Al10 bulk metallic glass is 469 kJ/mol in isothermal transformation process.
Key words: Zr55Cu30Ni5Al10; metallic glass; thermal analysis; crystallization kinetics; Avrami exponent
1 Introduction
Unlike the crystallized counterparts, bulk metallic glasses lack of long-range order in the atomic assembly. Therefore, metallic glasses possess unique combination of structural and functional properties, such as high strength, large elasticity, low elastic modulus and excellent corrosion resistance [1-11]. Thus, bulk metallic glasses are potential materials as applications in various engineering fields. However, amorphous alloys, which are in a metastable state, can change to crystalline state in the super-cooled liquid region. The crystallization kinetics for bulk metallic glasses is an important property, which is always linked with physical properties and mechanical properties.
There are many publications concerning the crystallization kinetics either in isochronal or isothermal modes in amorphous materials (polymer, metallic glass and glass). In bulk metallic glasses, many researchers performed differential scanning calorimetry (DSC) or differential thermal analysis (DTA) and used Johnson-Mehl-Avrami (JMA) model to analyze their results, especially the kinetics of this evolution. It is well known that the kinetics of crystallization in amorphous materials is sensitive to the kinetic parameters, which mainly include the activation energy of crystallization, mechanisms of nucleation and growth process for bulk metallic glasses, nature of crystalline phases, crystallization time and temperature. ROY and RAGHUVANSHI [12] analyzed the isothermal and non-isothermal crystallization kinetics in Al65Cu20Ti15 bulk metallic glass. In fact, the Avarami exponent indicated that crystallization was governed by three-dimensional diffusion-controlled growth in isothermal mode. In our previous investigation [13], the crystallization transformation kinetics in Cu46Zr45Al7Y2 bulk metallic glass in isothermal and non-isothermal modes were analyzed by Kissinger model and JMA model. It is interesting to note that the activation energy corresponding to isothermal conditions calculated by Arrhenius equation is larger than the value calculated by the Kissinger method under isochronal conditions. The reason lies in an energy barrier higher in isothermal annealing mode than in isochronal mode. More recently, KONG et al [14] have investigated the non-isothermal kinetics in Fe78Zr7B15 amorphous alloy with JMA model, and they have shown that the crystallization mechanisms are different in two crystallization stages. In particular, ABU EL-OYOUN [15-19] reported the crystallization kinetic parameters in different amorphous glasses by isoconversional methods: Kissinger-Akahira-Sunose (KAS) method, Tang method, Starink method, Flynn-Wall-Ozawa (FWO) and Vyazokin method. A new analysis method (various heating rates, VHR) was also used to discuss the crystallization kinetic parameters (apparent activation energy Ea and kinetic exponent n) in amorphous materials in continuous heating mode [20,21]. AL-HENITI [22] reported that under non- isothermal crystallization conditions in Fe78Si9B13 metallic glass, the Avrami exponent (n) for the two stages of crystallization were n1=4 and n2=5. Based on the JMA model, PRASHANTH et al [23] demonstrated that the crystallization process in Zr65Ag5Cu12.5Ni10Al7.5 glassy powder was diffusion-controlled with a three- dimensional growth. OKAI et al [24] investigated the Ca50Mg22.5Cu27.5 bulk metallic glass in super-cooled liquid region (isothermal annealing), and showed that the crystal growth was determined by a long-range diffusion processes. Finally, crystallization kinetics in metallic glass-matrix composites (TiC/Ti-based metallic glass matrix composite) was also in agreement with JMA model [25].
However, Zr55Cu30Ni5Al10 bulk metallic glasses have received considerable attention because of their possible application as structural and functional materials. For instance, Zr55Cu30Ni5Al10 [26-28] bulk metallic glasses show excellent glass forming ability, wide super-cooled liquid region and high mechanical properties. In the Zr55Cu30Ni5Al10 bulk metallic glass, many features have been investigated, such as crystallization [26, 29-32], structural relaxation [33-35], glass forming ability [36]. However, there is only little work on the non-isothermal crystallization properties and some discrepancies exist between the different reported results and some questions are open: 1) Cu46Zr45Al7Y2 [37-39] and Zr55Cu30Ni5Al10 [26-28] bulk metallic glasses show excellent glass forming ability, wide super-cooled liquid region and high mechanical properties. The thermal properties of the two bulk metallic glasses: glass transition temperature (Tg), temperature of the onset of crystallization (Tx) and crystallization peak temperature (Tp), are very similar in the two alloys. These two alloys have similar characteristic temperature (Tg, Tx). Do they exhibit similar crystallization features? 2) Experiments could be performed under isothermal or non-isothermal conditions. Does this difference lead to different behavior? Therefore, the aim of the present work is to give answers to these questions and to try to describe the kinetics using theoretical models.
2 Theoretical background
2.1 Isothermal annealing
During an isothermal annealing, transformation kinetics from the amorphous state to the crystalline state can occur, and the phase transformation is characterized by the crystallized volume fraction, ��. This evolution can be described by the JMA model and the following equation [30, 36, 40-42]:
![]() (1)
(1)
where n is the kinetics exponent (Avrami exponent), which reflects both the crystallization mechanism and the dimensionality of the crystallization process; t is the annealing time; K is the reaction rate constant (K depends on the annealing temperature), with an Arrhenius temperature dependence:
![]() (2)
(2)
where K0 is the frequency factor; Ea is the effective activation energy during the overall crystallization process; R is the mole gas constant; T is the thermodynamic temperature.
2.2 Non-isothermal annealing
During the isochronal phase transaction process, the phase transaction rate d��/dt [36, 42] is usually written as:
![]() (3)
(3)
where f(��) is a transformation function of the crystallized volume fraction �� [36, 42]. This function f(��) is usually defined as:
![]() (4)
(4)
During the crystallization process in amorphous materials, the activation energy of the phase transformation can be evaluated using various methods: Kissinger method [43-45], Flynn-Wall-Ozawa method [43,45] or Augis-Bennett method [46]. The Kissinger method is the most common one.
2.2.1 Kissinger method
According to this model, the activation energy Ea of phase transformation occurring in bulk metallic glass can be determined using DSC experiments carried out with various heating rates [43-45] and using the following equation:
![]() (5)
(5)
where Rh is the heating rate; Ta is the temperature at a fixed stage of the relaxation in amorphous materials, such as Tg, Tx and Tp in this investigation; C is a constant. It can be found that Ea could be obtained from a plot of ![]() vs 1000/Ta.
vs 1000/Ta.
2.2.2 Flynn-Wall-Ozawa method
During the non-isothermal crystallization process, the Flynn-Wall-Ozawa method is another effective method to calculate the activation energy for bulk metallic glasses [43,45]. The formula could be expressed as:
![]() (6)
(6)
In a similar way, a plot of ln Rh vs 1000/Ta gives a value of the activation energy Ea.
2.2.3 Augis-Bennett method
The activation energy of crystallization process for the samples could also be calculated by Augis-Bennett method [46]. In this model,
![]() (7)
(7)
where T0 is a characteristic (and adjustable) temperature.
Let us mention that these three methods are very similar and require similar plots.
3 Experimental
In the present work, master ingots of alloys with composition of Zr55Cu30Ni5Al10 were kindly provided by Prof. Y. Yokoyama, Institute of Materials Research, Tohoku University, Sendai, Japan. Plate samples were mechanically cut to prepare DSC samples. In order to remove surface oxidation prior to experiments, these samples were carefully polished using diamond paste and finally washed in ethanol in an ultrasonic cleaning machine. Structure of the specimens in the bulk metallic glasses was examined by X-ray diffraction using Cu K�� radiation produced by a commercial device (D8, Bruker AXS Gmbh, Germany), either at room temperature or during continuous heating up to 873 K. The working conditions were 40 kV and 40 mA for the X-ray tube and a scanning rate of 0.025 (��)/step.
The thermal properties and phase transformations were investigated by a standard commercial instrument (Pekin Elmer, DSC-7) under high purity dry nitrogen at a flow rate of 20 mL/min. Aluminium pans were used as sample holders. In order to ensure the reliability of experimental data, temperature and enthalpy were calibrated by indium standard specimen (Tm=429.7 K, ��Hc=28.48 J/g) and zinc standard with 3.283 mg (Tm=692.6 K, ��Hc=108.37 J/g), with an accuracy of ��0.2 K and ��0.02 mW, respectively.
The crystallization kinetics of the samples was characterized by DSC, at different constant heating rate ranges. In the case of isothermal crystallization investigation, isothermal crystallization of the samples in super-cooled liquid region (SLR) was tested at temperature above Tg and below Tx. The samples were heated to the isothermal tested temperature at a heating rate of 20 K/min. The temperature was held for various periods to induce the crystallization behaviour until completion. Finally, the samples were cooled down to room temperature.
4 Results and discussion
4.1 Non-isothermal crystallization
Figure 1 displays the XRD pattern of the as-cast Zr55Cu30Ni5Al10 bulk metallic glass. A typical broad diffraction peak appears, and the diffraction peak corresponding to a crystalline phase is not observed. Therefore, Zr55Cu30Ni5Al10 bulk metallic glass exhibits typical characteristics of an amorphous structure.

Fig. 1 XRD pattern of as-cast Zr55Cu30Ni5Al10 bulk metallic glass
The DSC curves of Zr55Cu30Ni5Al10 bulk metallic glass obtained using continuous heating with different heating rates are shown in Fig. 2. The crystallization peak temperature (Tp) shifts to higher temperature as heating rate is increased. This result is similar to what is classically observed. In the Zr55Cu30Ni5Al10 bulk metallic glass, the peak is a little more complicated, since a kind of double peak is observed. However, the question is: Are these two phases due to same phase or not? When the heating rate is 20 K/min, peaks occur at 761 K (the first peak) and 765 K (the second peak), respectively. Therefore, two apparent peaks during the course of heating with 20 K/min up to 761 K (the first peak) and 765 K (the second peak) occur and then XRD patterns indicate that the corresponding crystalline phases are identical (Fig. 3).
In the literature, the crystallization behaviour in Zr55Cu30Ni5Al10 bulk metallic glass was investigated by in-situ synchrotron radiation. YAVARI et al [47,48] found that crystallization in Zr55Cu30Ni5Al10 bulk metallic glass occurs via initial nucleation and growth of a metastable phase that transforms to equilibrium tetragonal Zr2Cu. To confirm this mechanism, X-ray diffraction experiments were performed during a continuous heating. the average heating rate was 1.5 K/min and diffractograms were recorded each 5 K (Fig. 4).

Fig. 2 DSC curves of Zr55Cu30Ni5Al10 bulk metallic glasses with different heating rates
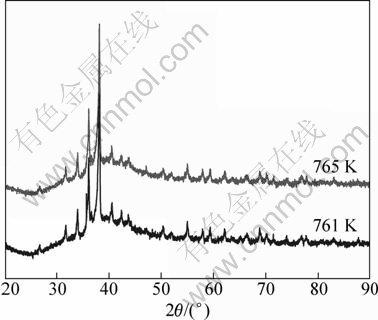
Fig. 3 XRD patterns after heating at 761 K or 765 K with heating rate of 20 K/min for Zr55Cu30Ni5Al10 bulk metallic glass

Fig. 4 XRD spectrum (3D representation) during continuous heating at 1.5 K/min from 573 K to 873 K for Zr55Cu30Ni5Al10 bulk metallic glass
The formation of crystalline particles is clearly revealed by the apparition of diffraction peaks. The nature of the crystalline phases change when the temperature increases. For instance, the peak in the range 37.5��-38.0�� disappears and a new peak appears close to 37��. This new peak can be attributed to a body-centered tetragonal phase similar to Zr2Cu. Figure 5 illustrates this evolution. But it is not possible on this figure to detect a two-steps evolution in the low temperature crystallization process (in the range of 700-750 K). TEM observation should also give precious information. However, since the present work is focused mainly on the kinetics aspects rather than on the nature of the crystalline phases, and no detailed investigation of the phenomenon was conducted.

Fig. 5 Diffraction peak area versus time during continuous heating at 1.5 K/min for Zr55Cu30Ni5Al10 bulk metallic glass
The relationship between the crystallization volume fraction �� and temperature during non-isothermal crystallization process of Zr55Cu30Ni5Al10 bulk metallic glass is shown in Fig. 6. All the curves present a sigmoid dependence with temperature. The same evolution is observed in all the amorphous materials (bulk metallic glasses and polymers) in the non-isothermal crystallization mode.

Fig. 6 Plots of crystallization volume fraction �� of Zr55Cu30Ni5Al10 bulk metallic glass as function of temperature at different heating rates
The activation energy Ea can be determined by Kissinger��s method, Flynn-Wall-Ozawa (FWO) method and Augis-Bennett (AB) method. The relevant fitting results by FWO method at different crystalline volume fraction �� (ranging from 0.1 to 0.9) are shown in Fig. 7. The results are in good agreement with the Kissinger��s equation, FWO equation and AB equation since straight lines are clearly observed.

Fig. 7 FWO plots of Zr55Cu30Ni5Al10 in non-isothermal heating mode at any fixed crystallization volume fraction �� (range from 0.1 to 0.9)
Figure 8 presents the calculated results of activation energy for Zr55Cu30Ni5Al10 bulk metallic glass. The activation energy for crystallization in the Zr55Cu30Ni5Al10 bulk metallic glass is not constant, fluctuating slightly with crystallization volume fraction �� in non-isothermal heating mode. The average values of activation energy for Cu46Zr45Al7Y2 and Zr55Cu30Ni5Al10 bulk metallic glasses in non-isothermal heating mode are very similar [13]. Meanwhile, the average values of activation energy for Zr55Cu30Ni5Al10 bulk metallic glass determined by different models (Kissinger method, FWO method and AB method) are in good agreement with each other.

Fig. 8 Activation energy Ea as a function of crystallization volume fraction �� in Zr55Cu30Ni5Al10 bulk metallic glasses (�� range from 0.1 to 0.9) in non-isothermal heating process
4.2 Isothermal behaviours
In order to well understand the crystallization kinetics of the Zr55Cu30Ni5Al10 bulk metallic glass, the crystallization kinetic of the samples was investigated in isothermal situation at different annealing temperatures. The DSC curves obtained for Zr55Cu30Ni5Al10 bulk metallic glass at different annealing temperatures are shown in Fig. 9. They look like those recorded under non-isothermal conditions. It can be found that every DSC curve exhibits a single exothermic peak after a certain incubation time (��). Additionally, by increasing annealing temperature, it is obvious that the incubation time slumps. The reason is that atoms in the samples have a higher mobility at higher annealing temperature, which leads to concentration fluctuations necessary for large-scale crystallization to set in [49]. From these curves, the evolution of crystalline volume fraction versus the annealing time during isothermal annealing is shown in Fig. 10. The higher the annealing temperature is, the steeper the curves are. Meanwhile, all the curves exhibit a typical sigmoid evolution.
As mentioned above, the transformation kinetics can be described using the JMA equation [36, 50]:
![]() (8)
(8)
where �� is the incubation time, which in fact is a fitting parameter. Taking the double logarithm of Eq. (8), the following expression is deduced [36]:
![]() (9)
(9)
By plotting ln[-ln(1-��)] versus ln(t-��) at various annealing temperatures with 0.2�ܦ���0.8 (Fig. 11), the kinetic exponent n and the reaction rate constant K can be calculated and the values are given in Table 1.

Fig. 9 Isothermal DSC curves of Zr55Cu30Ni5Al10 bulk metallic glass at various annealing temperatures
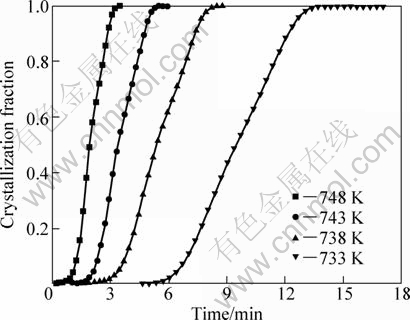
Fig. 10 Plots of crystallization volume fraction �� of Zr55Cu30Ni5Al10 bulk metallic glass as function of time at different annealing temperatures

Fig. 11 Avrami plots of ln[-ln(1-��)] vs ln(t-��) at various annealing temperature with 0.2�ܦ���0.8 from isothermal DSC curves for Zr55Cu30Ni5Al10 bulk metallic glass
Table 1 Kinetic exponent n, reaction rate constant K and incubation time �� at different annealing temperatures with 0.2�ܦ���0.8 in Zr55Cu30Ni5Al10 bulk metallic glasses

The kinetic exponent n varies with annealing temperatures from 2.2 to 2.9 in the Zr55Cu30Ni5Al10 bulk metallic glass. Diffusion-controlled growth theory [25,51,52] predicts that: 1
The average kinetic exponent n for Zr55Cu30Ni5Al10 bulk metallic glass is about 2.5, which implies that crystallization mechanism in the Zr55Cu30Ni5Al10 bulk metallic glass is mainly diffusion-controlled [30], and crystal growth is controlled by long range ordering (LRO) diffusion in three-dimensional growth during isothermal crystallization process [53].
Let us now turn to the influence of the nucleation and growth behaviour during crystallization on the activation energy. The activation energy for the crystallization process in isothermal mode can be determined using the Arrhenius equation [30]:
![]() (10)
(10)
where t0 is a constant. The plots of ln t(a) vs 1/Ta in Zr55Cu30Ni5Al10 bulk metallic glass during isothermal process are shown in Fig. 12. All the fitting lines are almost straight lines. The average value of activation energy for Zr55Cu30Ni5Al10 bulk metallic glass is 469 kJ/mol. However, this activation energy decreases when the crystalline fraction increases (Fig. 13). This decrease could be due to the fact that this energy is the sum of two components: one term corresponding to the nucleation and the other to the growth (connected to the diffusion). When crystallization progresses, the energy required for nucleation can progressively disappear and then Ea can decrease.

Fig. 12 Plots for calculation of activation energy in Zr55Cu30Ni5Al10 bulk metallic glass
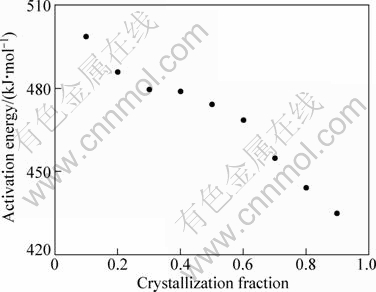
Fig. 13 Activation energy Ea as function of crystallization volume fraction �� in Zr55Cu30Ni5Al10 (�� ranging from 0.1 to 0.9) in isothermal mode
5 Conclusions
1) In non-isothermal mode, the average values of activation energy for Zr55Cu30Ni5Al10 bulk metallic glass determined by different models (Kissinger method, Flynn-Wall-Ozawa method and Augis-Bennett method) are good agreement with each other.
2) Under isothermal annealing conditions, the Avrami exponent n ranges from 2.2 to 2.9 in the Zr55Cu30Ni5Al10 bulk metallic glass. These values, close to 2.5, indicate that the crystallization mechanism is mainly diffusion-controlled. Crystal growth occurs by a three-dimensional long range ordering.
3) In isothermal transformation process, the average value of activation energy for Zr55Cu30Ni5Al10 bulk metallic glass is 469 kJ/mol. The classical models developed in the literature are really appropriated to describe the crystallization process in Zr55Cu30Ni5Al10 bulk metallic glass.
Acknowledgements
One of the author, J. C. QIAO, wants to express his thanks to Dr. J. M. CHENAL and Dr. S. CARDINAL, for valuable discussion and assistance in XRD and DSC experiments during the course of the work, and Prof. Y. Yokoyama, Institute of Materials Research, Tohoku University, Sendai, Japan for providing samples. QIAO thanks China Scholarship Council (CSC) for providing the scholarship.
References
[1] GREER A L. Metallic glasses [J]. Science, 1995, 267: 1947-1953.
[2] BYRNE C J, ELDRUP M. Materials science��Bulk metallic glasses [J]. Science, 2008, 321: 502-503.
[3] INOUE A. Stabilization of metallic supercooled liquid and bulk amorphous alloys[J]. Acta Materialia, 2000, 48: 279-306.
[4] WANG W H, DONG C, SHEK C H. Bulk metallic glasses [J]. Materials Science and Engineering R, 2004, 44: 45-89.
[5] INOUE A, ZHANG W, ZHANG T, KURODAKA K. High-strength Cu-based bulk glassy alloys in Cu-Zr-Ti and Cu-Hf-Ti ternary systems [J]. Acta Materialia, 2001, 49: 2645-2652.
[6] WANG W H. Bulk metallic glasses with functional physical properties [J]. Advanced Materials, 2009, 21: 4524-4544.
[7] WANG W H. Roles of minor additions in formation and properties of bulk metallic glasses [J]. Progress in Materials Science, 2007, 52: 540-596.
[8] LUO Q, WANG W H. Rare earth based bulk metallic glasses[J]. Journal of Non-Crystalline Solids, 2009, 355: 759-775.
[9] CHENG Y Q, MA E. Atomic-level structure and structure�Cproperty relationship in metallic glasses [J]. Progress in Materials Science, 2011, 56: 379-473.
[10] JOHNSON W L. Thermodynamic and kinetic aspects of the crystal to glass transformation in metallic materials[J]. Progress in Materials Science, 1986, 30: 81-134.
[11] INOUE A, TAKEUCHI A. Recent development and application products of bulk glassy alloys [J]. Acta Materialia, 2011, 59: 2243-2267.
[12] ROY D, RAGHUVANSHI H. Study on crystallization kinetics of Al65Cu20Ti15 amorphous alloy [J]. Journal of Non-Crystalline Solids, 2011, 357: 1701-1704.
[13] QIAO J C, PELLETIER J M. Crystallization kinetics in Cu46Zr45Al7Y2 bulk metallic glass by differential scanning calorimetry (DSC) [J]. Journal of Non-Crystalline Solids, 2011, 357: 2590-2594.
[14] KONG L H, GAO Y L, SONG T T, WANG G, ZHAI Q J. Non-isothermal crystallization kinetics of FeZrB amorphous alloy[J]. Thermochimica Acta, 2011, 522: 166-172.
[15] ABU EL-OYOUN M. An investigation of the kinetic transformation mechanism of Ge12.5Te87.5 chalcogenide glass under non-isothermal regime [J]. Journal of Non-Crystalline Solids, 2011, 357: 1729-1735.
[16] ABU EL-OYOUN M. Determination of the crystallization kinetic parameters of Ge22.5Te77.5 glass using model-free and model-fitting methods[J]. Journal of Alloys and Compounds, 2009, 486: 1-8.
[17] ABU EL-OYOUN M. Evaluation of the transformation kinetics of Ga7.5Se92.5 chalcogenide glass using the theoretical method developed and isoconversional analyses [J]. Journal of Alloys and Compounds,2010, 507: 6-15.
[18] ABU EL-OYOUN M. Effect of wide range of heating rate on the crystallization kinetic parameters of Se77Te20Sb3 glass [J]. Thermochimica Acta, 2009, 494: 129-135.
[19] ABU EL-OYOUN M. The effect of addition of gallium on the thermal stability and crystallization kinetic parameters of GaxSe100-x glass system[J].Physica B, 2011, 406: 125-133.
[20] ABD EL-RAHEEM M M, ALI HM. Crystallization kinetics determination of Pb15Ge27Se58 chalcogenide glass by using the various heating rates (VHR) method [J]. Journal of Non-Crystalline Solids, 2010, 356: 77-82.
[21] RASHEEDY M R, SOLTAN A S, ABD-ELMAGEED A A I. Various heating rates (VHR) as a method for crystallization kinetics determination and its application to Se75.5Te20Sb4.5 chalcogenide glass [J]. Journal of Alloys and Compounds, 2009, 472: 581-586.
[22] AL-HENITI S H. Kinetic study of non-isothermal crystallization in Fe78Si9B13 metallic glass[J]. Journal of Alloys and Compounds, 2009, 48: 177-184.
[23] PRASHANTH K G, SCUDINO S, SURREDDI K B, SAKALIYSKA M, MURTY B S, ECKERT J. Crystallization kinetics of Zr65Ag5Cu12.5Ni10Al7.5 glassy powders produced by ball milling of pre-alloyed ingots [J]. Materials Science and Engineering A, 2009, 513-514: 279-285.
[24] OKAI D, SHIMIZU Y, HIRANO N, FUKAMI T, YAMASAKIT, INOUE A. Isothermal crystallization in supercooled liquid state for Ca50Mg22.5Cu27.5 metallic glass [J]. Journal of Alloys and Compounds, 2010, 504: s247-s250.
[25] ZHANG L C, XU J, ECKERT J. Thermal stability and crystallization kinetics of mechanically alloyed TiC/Ti-based metallic glass matrix composite [J]. Journal of Applied Physics, 2006, 100: 033514.
[26] LIU L, WU Z F, ZHANG J. Crystallization kinetics of Zr55Cu30Al10Ni5 bulk amorphous alloy[J]. Journal of Alloys and Compounds, 2002, 339: 90-95.
[27] KERYVIN N, VAILLANTM L, ROUXEL T, HUGER M, GLORIANT T, KAWAMURA Y. Thermal stability and crystallisation of a Zr55Cu30Al10Ni5 bulk metallic glass studied by in situ ultrasonic echography[J]. Intermetallics, 2002, 10: 1289-1296.
[28] CASTELLERO A, BOSSUYT S, STOICA M, DELEDDA S, ECKERT J, CHEN G Z, FRAY D J, GREER A L. Improvement of the glass-forming ability of Zr55Cu30Al10Ni5 and Cu47Ti34Zr11Ni8 alloys by electro-deoxidation of the melts[J]. Scripta Materialia, 2006, 55: 87-90.
[29] GAO Y L, SHEN J, SUN J F, WANG G, XING D W, XIAN H F, ZHOU B D. Crystallization behavior of ZrAlNiCu bulk metallic glass with wide supercooled liquid region[J]. Materials Letters, 2003, 57:1894-1898.
[30] YANG Y J, XING D W, SHEN J, SUN J F, WEI S D, HE L J, MCCARTNEY D G. Crystallization kinetics of a bulk amorphous Cu-Ti-Zr-Ni alloy investigated by differential scanning calorimetry[J]. Journal of Alloys and Compounds, 2006, 415: 106-110.
[31] HU Z Q, ZHANG Q S, ZHANG H F, DING B Z, RAO Z M. Isothermal crystallization of Zr55Al10Ni5Cu30 bulk amorphous alloy near the glass transition temperature [J]. Materials Science Forum, 2004, 449-452: 933-936.
[32] HARUYAMA O, NAKATAMAY, WADA R, TOKUNAGA H, OKADA J, ISHIKAWA T, YOKOYAMA Y. Volume and enthalpy relaxation in Zr55Cu30Ni5Al10 bulk metallic glass[J]. Acta Materialia, 2010, 58: 1829-1836.
[33] SLIPENYUK A, ECKERT J. Correlation between enthalpy change and free volume reduction during structural relaxation of Zr55Cu30Al10Ni5 metallic glass [J]. Scripta Materialia, 2004, 50: 39-44.
[34] INOUE A, ZHANG T. Fabrication of bulky Zr-based glassy alloys by suction casting into copper mold [J]. Materials Transactions JIM, 1995, 36: 1184-1187.
[35] QIAO J C, PELLETIER JM. Enthalpy relaxation in Cu46Zr45Al7Y2 and Zr55Cu30Ni5Al10 bulk metallic glasses by differential scanning calorimetry (DSC)[J]. Intermetallics, 2011, 19: 9-18.
[36] XU D H, DUANG G, JOHNSON W L. Unusual glass-forming ability of bulk amorphous alloys based on ordinary metal copper [J]. Physical Review Letters, 2004, 92: 245504.
[37] YUAN Z Z, BAO S L, LU Y, ZHANG D P, YAO L. A new criterion for evaluating the glass-forming ability of bulk glass forming alloys [J]. Journal of Alloys and Compounds, 2008, 459: 251-260.
[38] CHEN Q J, SHEN J, ZHANG D L, FAN H B, SUN J F, MCCARTNEY D G. A new criterion for evaluating the glass-forming ability of bulk metallic glasses[J]. Materials Science and Engineering A, 2006, 433: 155-160.
[39] LIN L Z, WEI H Q, DING Y H, ZHANG P, XIE G Q, INOUE A. A new criterion for predicting the glass-forming ability of bulk metallic glasses[J]. Journal of Alloys and Compounds, 2009, 475: 207-219.
[40] LI J F, HUANG Z H, ZHOU Y H. Crystallization of amorphous Zr60Al15Ni25 alloy [J]. Intermetallics, 2007, 15: 1013-1019.
[41] YUAN Z Z, CHEN X D, WANG B X, WANG Y J. Kinetics study on non-isothermal crystallization of the metallic Co43Fe20Ta5.5B31.5 glass [J]. Journal of Alloys and Compounds, 2006, 407: 163-169.
[42] LIU L, ZHAO X J, MA C L, ZHANG T. Kinetics of crystallization process for Pd-based bulk metallic glasses[J]. Intermetallics, 2009, 17: 241-245.
[43] BAYRI N, IZGI T, GENCER H, SOVAK P, GUNES M, ATALAY S. Crystallization kinetics of Fe73.5-xMnxCu1Nb3Si13.5B9 (x=0, 1, 3, 5, 7) amorphous alloys [J]. Journal of Non-Crystalline Solids, 2009, 355: 12-16.
[44] LOPEZ-ALEMANY P L, VAZQUEZ J, VILLARES P, JIMENEZ-GARAY R. A kinetic study on non-isothermal crystallization of the glassy alloy Sb0.16As0.43Se0.41 [J]. Journal of Non-Crystalline Solids, 2000, 274: 249-256.
[45] PARK E S, LEE J Y, KIM D H, GEBERT A, SCHULTZ L. Correlation between plasticity and fragility in Mg-based bulk metallic glasses with modulated heterogeneity [J]. Journal of Applied Physics, 2008, 104: 023520.
[46] OZAWA T. Estimation of activation energy by isoconversion methods [J]. Thermochimica Acta, 1992, 203: 159-165.
[47] YAVARI A R, LE MOULEC A, BOTTA W J, INOUE A, REJMANKOVA P, KVICK A. In situ crystallization of Zr55Cu30Al10Ni5 bulk glass forming from the glassy and undercooled liquid states using synchrotron radiation[J]. Journal of Non-Crystalline Solids, 1999, 247: 31-34.
[48] YAVARI A R, Le MOULEC A, INOUE A, BOTTA W J, VAUGHAN G, KVICK A. Metastable phases in Zr-based bulk glass-forming alloys detected using a synchrotron beam in transmission [J]. Materials Science and Engineering A, 2001, 304-306: 34-38.
[49] VENKATARAMAN S, ROZHVOKA E, ECKERT J, SCHULTA L, SORDELET D J. Thermal stability and crystallization kinetics of Cu-reinforced Cu47Ti33Zr11Ni8Si1 metallic glass composite powders synthesized by ball milling: the effect of particulate reinforcement[J]. Intermetallics, 2005, 13: 833-840.
[50] XU F, JIANG J Z, CAO Q P, DU Y W. Isothermal crystallization kinetics analysis of melt-spun Pd42.5Cu30Ni7.5P20 amorphous ribbons[J]. Journal of Alloys and Compounds, 2005, 392: 173-176.
[51] OKAI D, SHIMIZU Y, HIRANO N, FUKAMI T, YAMASAKI T, INOUE A. Isothermal crystallization in supercooled liquid state for Ca50Mg22.5Cu27.5 metallic glass [J]. Journal of Alloys and Compounds, 2010, 504: s247-s250.
[52] WEI S W, DING B Z, LI T Q, HU Z Q. Crystallization of amorphous Zr65Cu27Al8 alloy with wide supercooled liquid region[J]. Materials Letters, 1998, 37: 263-267.
[53] WU X F, MENG L K, ZHAO W, SUO Z Y, SI Y, QIU K Q. Crystallization kinetics of misch metal based bulk metallic glasses [J]. Journal of Rare Earths, 2007, 25: 189-193.
J. C. QIAO, J. M. PELLETIER
Universit�� de Lyon, MATEIS, UMR CNRS5510, Bat. B. Pascal, INSA-Lyon, F-69621 Villeurbanne cedex, France
ժ Ҫ�����ò�ʾɨ�������ǣ�DSC����X����������(XRD) �о�Zr55Cu30Ni5Al10�����������ķǵ��¾���ת�䶯��ѧ���ڹ���Һ�����ĵ��¾�������ѧ��Ϊ���ڷǵ��¹����У����ò�ͬ����(Kissinger, Flynn-Wall-Ozawa��Augis-Bennett)�õ���Zr55Cu30Ni5Al10����������ƽ�������ܱ˴�֮���ǺϺܺá����⣬����Johnson-Mehl- Avrami(JMA)ģ������Zr55Cu30Ni5Al10�����������ĵ���ת�䶯��ѧ���о����������Zr55Cu30Ni5Al10������������Avrami ָ��n����2.2��2.9֮�䣬�����侧��������Ҫ����ɢ���ƹ��̡��ڵ��¾����Ĺ����У����˳�����Ҫ����ά�ij���������ɢ���ƵĹ��̣�ƽ��������Ϊ469 kJ/mol��
�ؼ��ʣ�Zr55Cu30Ni5Al10�������������ȷ�������������ѧ��Avrami ָ��
(Edited by YANG Hua)
Corresponding author: J. M. PELLETIER; Tel: +33 4 72 43 83 18; Fax: +33 4 72 43 85 28; E-mail: jean-marc.pelletier@insa-lyon.fr
DOI: 10.1016/S1003-6326(11)61216-8

