
Thermodynamic principle and properties of electroplated RE-Ni-W-P-B4C composite coatings
ZHU Cheng-yi(�����), LI Guang-qiang(���ǿ), QIN Qing-wei(����ΰ), MA Guo-jun(������)
Key Laboratory for Ferrous Metallurgy and Resources Utilization of Ministry of Education, Wuhan University of Science and Technology, Wuhan 430081, China
Received 15 July 2007; accepted 10 September 2007
Abstract��The �գ�pH diagram of Ni-W-P-H2O system at 298.15 K, 10 1325 Pa was plotted by thermodynamic calculation software FactSageTM 5.1. The results show that tungsten and phosphorus can be co-deposited with nickel in type of Ni4W, Ni3P, Ni5P2, respectively. XRD analysis shows that the main phase of RE-Ni-W-P-B4C coatings is amorphous as deposited. After heated at 673 K for 3 h, part phases change to crystalline which are Ni, Ni3P, P, W, Ni3C, Ni3B, CeO2. SEM shows the micrograph of the coatings is even and the solid particles scatter well. The thickness of the coating is 2 219 ?m after electroplating for 96 h. The micro-hardness of the coatings is HV 825-HV 1 097 as-deposited and increases to HV 1 236 after heat treated. The wear resistance of the coatings is good and the friction coefficient changes from 0.10 to 0.33 during the abrasion process. The resistance to oxidation of the composite coatings is better than Ni-W-P alloy coatings and worse than that of RE-Ni-W-P-SiC coatings.
Key words: RE-Ni-W-P-B4C coatings; microstructure; micro-hardness; wear resistance; high temperature oxidation resistance
1 Introduction
The copperplate of the mold used in liquid steel casting must have high hardness, good wear resistance, good heat conductivity and high temperature heat corrosion resistance to adapting to high efficiency continuous casting. At present, Ni[1], Ni-Cr[2], Ni-Co[2-3] and Ni-Fe[4-6] coatings prepared by electrodepositing are widely used domestic and foreign. The newly developed coatings are Ni-W-Co[7-8], Ni-P[9] alloy and solid particles co-depositing with the above alloys[10]. There are no reports that new coatings are used in the steel plant and the preparation technologies need to perfect.
Boron carbide has good qualities such as extreme hardness, which is only lower than diamond and cubic boron nitride, excellent chemical resistance, high melting point and low density, which make it extremely useful for applications at both room and high temperature[11]. Very little work has been reported on composite coatings containing B4C particles used in the continuous mold. MONIR VAGHEFI et al[12] studied abrasion- resistant electroless Ni-P-B4C composite coatings and pointed out that these composite coatings have different applications in automotive industry, mining, aerospace, a good protective coating used in nuclear system due to high neutron absorption ability and etc. SHRESTHA et al[13] reported electroplated Ni/B4C composition coatings properties. The present author had prepared electroplated Ni-W-P-B4C composite coatings as the first phase of preparations for using on the mold[14]. RE has been applied in the surface technologies such as plating chromium, chemical heat treatment and transforming films[15-18], meanwhile, a few applications of RE in the composite plate is helpful to fine crystal particles and increase deposition velocity[19-20].
The present authors plotted the �գ�pH diagram of Ni-W-P-H2O system at 298.15 K under the standard pressure condition using thermodynamic calculation software FactSageTM 5.1 so that the thermodynamic principle of co-electrodeposition of Ni-W-P was analyzed. Otherwise, based on the previous work of Ni-W-P-B4C coatings, the present authors studied the influence of the heat treatment temperature on the microstructure, morphology, hardness, wear resistance and high temperature oxidation resistance of the RE-Ni-W-P-B4C composite coatings.
2 Experimental 2.1 Materials and technology conditions
The compositions of the solution in the bath and the conditions of the technology are listed in Table 1. In the experiments, copperplate containing beryllium (0.3%, mass fraction) and chromium (2.5%) was used as cathode, and the stainless steel plate was used as anode.
Table 1 Compositions of solution and conditions of technology

2.2 Technological process and analysis methods of coatings properties
The technological process for preparation the RE-Ni-W-P-B4C composite coatings could be illustrated as follows. Before being electroplated, the copperplate was degreased in the alkaline solutions, and then was activated after washing. Further more, a little corrosion should be applied before electroplating. The coatings were heat treated for some time from 473 K to 1 113 K in the air and the oxidation mass changing with the treatment time was recorded by electronic thermal balance. After cooling to room temperature, the samples were analyzed by X-ray diffraction.
The principle of co-electrodeposition of Ni-W-P was analyzed thermodynamically by using the �գ�pH diagram of Ni-W-P-H2O system at 298.15 K under the standard pressure condition plotted by thermodynamic calculation soft FactSageTM 5.1. The Philip type XRD was used to analyze the phase of the composite coatings. XL30W/TMP type SEM was used to analyze the section micrograph of the composite coatings. The micro-hardness of the coatings was analyzed with HM-114 hardness meter. The WIM-IE mini-type friction abrasion testing machine was used to analyze the wear resistance of the composite coatings.
3 Results and discussion 3.1 Thermodynamic principle analysis of co- electrodeposition of Ni-W-P alloy
It can be seen from the �գ�pH diagram of Ni-P-H2O system[21-22] that P can deposit at cathode from the bath under the conditions similar to Ni in the form of Ni3P. Since the potential of Ni3P is much more positive than that of P, Ni3P deposits at cathode more easyly than P. Therefore, the co-deposition of Ni, P in the form of Ni3P can be illustrated as the following reactions.
3Ni2++H2PO4-+6H++11e=Ni3P+4H2O
3Ni+H2PO4-+6H++5e=Ni3P+4H2O
However, W can not deposit from the WO42- (Na2WO4��2H2O) burdening solutions seen from the E-pH diagram of W-H2O system[23]. It has been reported that W can deposit at cathode induced by Ni or other Fe system metals[24]. The present author has plotted E-pH diagram of Ni-W-P-H2O system at 298.15 K under the standard pressure condition by thermodynamic calculation software FactSageTM 5.1, which is shown in Fig.1. It can be seen that W can deposit at cathode in the form of Ni4W induced by Ni, while P can deposit in the form of Ni3P or Ni5P2. Meanwhile, the solid particles CeO2 and B4C can deposit on the cathode with the co-action of fluid force field, electric field, concentration field and the deposition of the other metals in the bath.
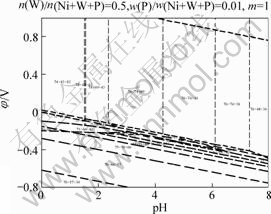

Fig.1 ��-pH diagram of Ni-W-P-H2O system at 298.15 K
3.2 Phase structure of composite coatings
In order to study the microstructure of solid particles and alloys in the composite coatings, XRD patterns of B4C powder and RE-Ni-W-P-B4C composite coatings as-deposited and after heat treatment are shown in Fig.2.
X-ray diffraction pattern of B4C powder in Fig.2(a) shows there is a little B crystal besides B4C crystal. In Fig.2 (b), the diffraction peaks of Ni, Ni3P and CeO2 appear and it is clear that there is a ��bread�� peak at 2�� =
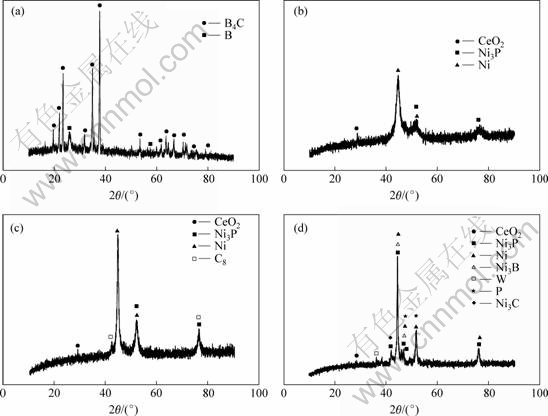
Fig.2 XRD patterns of RE-Ni-W-P-B4C composite coatings under different conditions: (a) B4C; (b) As-deposited; (c) Heat treated at 573 K for 3 h; (d) Heat treated at 673 K for 3 h
45? in the diffraction pattern of composite coatings as-deposited, which indicates that the coating as-deposited includes amorphous phase. There are no sharp diffraction peaks of B4C in the pattern, indicating no crystal B4C particles in the coatings. In this experiment, we chose 573 K and 673 K as the heat treatment temperature for the coatings since the working temperature of the coatings is often at the range from 623 K to 673 K. However, the microstructure of the coatings treated at 573 K has no apparent difference comparing with the as-deposited coatings, which is shown in Fig.2(c). Compared with Fig.2(c), it can be also seen from Fig.2(d) the range of the amorphous state is obviously reduced and Ni, Ni3P and CeO2 peaks are more remarkable. In addition, there appears the diffraction peaks of P, W, Ni3C, and these are typical characteristic of crystal state. The most apparent peaks are N3P in the coatings. This indicates that the crystallization of the composite coating is strengthened after being heat treated at 673 K for 3 h. Besides, the appearance of Ni3P, Ni3B, W, Ni3C phases indicate that nickel atoms and W, P atoms in the coating diffuse mutually, and form transition phase Ni3P, Ni3B, W, Ni3C phases. Because the amorphous state particles are in unstable state, and it has much free energy difference compared with the equilibrium crystal state particles at the room temperature. However, when the heat treated temperature exceeds 673 K, the movement ability of the atom enhances and then amorphous state transit to relatively stable crystal state. Since the electroplating temperature is 323 K, B4C particles decompose perhaps at the heat treated temperature. B atoms either mixed in B4C particles or decomposed by B4C particles combine with Ni or W atoms to become a new phase in the coatings. In this way, these new phases will be good in improving the properties of the composite coatings.
3.3 Micro-hardness analysis of composite coatings
The micro-hardness of the electrodeposited RE-Ni-W-P-B4C composite coatings was analyzed by the HM-114 hardometer. The micro-hardness of the as-deposited coatings is about HV 825-HV 1 097. The micro-hardness of the heat treated coatings changing with the heat treatment temperature is shown in Fig.3. It is clear that the micro-hardness of coatings increases with the heat treatment temperature. However, when the heat treatment temperature exceeds 673 K, the hardness of the coatings increases a little. The decrease of the hardness is owing to new dispersion strengthened phase such as Ni3P, P, W, Ni3C, Ni3B and too high temperature has no advance for improving the hardness of the coatings since there is no other new dispersion phase to separate out. When the coatings are heat treated for 3 h from 573 K to 773 K, the hardness of the coatings changes from HV 1 100 to HV 1 236.
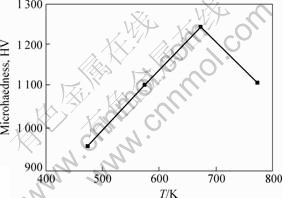
Fig.3 Influence of heat treatment temperature on hardness of composite coatings
Meanwhile, the influence of heat treatment time on the hardness at 673 K is shown in Fig.4. It suggests that the micro-hardness of coatings increases with the heat treatment time. However, the hardness of the coatings decreases when the heat treatment time exceeds 3 h. When the heat treatment temperature is unchanged, the dispersion strengthened phases increase with the increase of the heat treatment time. This phenomenon does not exist since new other dispersion phase increases. Unfortunately, too long heat treated time leads to the conglomeration of the new dispersion phase grain to grow up. Therefore, the coatings become loosen and the hardness of the coatings decreases.
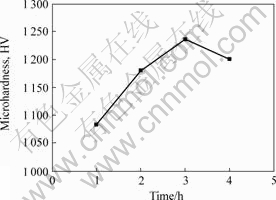
Fig.4 Influence of heat treatment time on hardness of composite coatings at 673 K
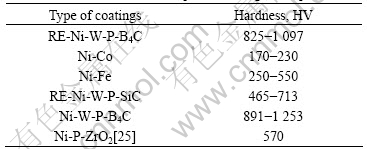
The micro-hardness of the present RE-Ni-W-P-SiC composite coatings as-deposited compared with the other coatings is listed in Table 2. The results show that the micro-hardness of the present coatings is about HV 825- HV 1 097, which is much higher than Ni-Co alloy coatings(HV 170-HV 230) and Ni-Fe alloy coatings(HV 250-HV 550) and also higher RE-Ni-W-P-SiC composite coatings(HV 465-HV 713) that we have studied. The micro-hardness of the present coatings is slightly less than that of Ni-W-P-B4C coatings with the value HV 891-HV 1 253, which is the highest in the following samples.
Table 2 Micro-hardness of composite coatings as-deposited
3.4 Wear resistance analysis of composite coatings
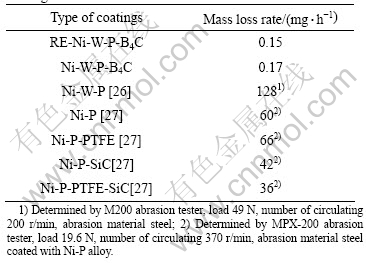
The wear resistance of the electrodeposited RE-Ni-W-P-B4C composite coatings was studied and compared with the other coatings. And the results are listed in Table 3. The wear resistance of the coatings is tested by WIM-IE mini-type friction abrasion testing machine. In this study, we used the mass loss rate of the coatings weighed by analytic balance (regulated within ��0.1 mg) after the coatings abrading to steel for 3 h with 1.47 g in load. The results show that the abrasions of the coatings are very low. They are much lower than Ni-W-P alloy coating [26] we had researched in our previous work and Ni-P, Ni-P-PTFE, Ni-P-SiC, Ni-P-PTFE-SiC coatings reported in Ref.[27].
Table 3 Abrasion weight loss rate of the different as-deposited coatings
The relationships of friction coefficient and time of the composite coatings are showed in Fig.5. The friction coefficient of Ni-W-P-B4C coatings changes little with the value 0.12 before 90 min and after 90 min increases suddenly to 0.35 as shown in Fig.5(a). The friction coefficient of the present composite coatings is very low with the value 0.10 before 90 min and after 90 min increases near to 0.33 as shown in Fig.5(b). It is explained that the particles adhering on the surface falling off to become affixation abrasion particles to prick up abrasion to increase friction coefficient. The results show that the addition of CeO2 in the bath leads to low friction coefficient, which can be explained that the addition of CeO2 in the coatings improves the surface quality of the coatings and fines the deposition grain[6]. The friction coefficient of Ni-W-P-B4C composite coatings is lower than Ni-P coating which is 0.60-0.63 and Ni-P-PTFE��25%-30%, volume fraction��which is 0.16 reported in Ref.[28]. Therefore, the wear resistance of RE-Ni-W-P-B4C is the best among the above coatings.
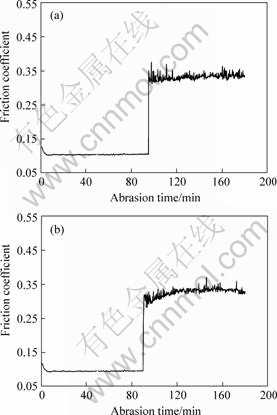
Fig.5 Relationship of friction coefficient and time of composite coatings: (a) Ni-W-P-B4C; (b) RE-Ni-W-P-B4C
A few elements such as W, P, B, C can strengthen the properties of the coatings. Ni3P in the coatings is a diffusion phase with lower friction coefficient. W is a horniness element and C has lubricating property. So the micro-hardness and wear resistance of RE-Ni-W-P-B4C composite coatings will be good compared with the other reported coatings used in the mold.
3.5 Morphologies of composite coatings
Morphologies of composite coatings obtained in the present work are shown in Fig.6. It suggests that the microstructure of the composite coatings is uniformity and particles disperse in the matrix evenly. Seen from Fig.6(b) that there is a boundary between the composite coatings and the matrix and it is a mechanical combination. To improve their binding force, special treatment must be taken. The thickness of the coatings is 124 ?m and the coatings 2 219 ?m in thickness can be prepared by prolong electroplating time to 96 h. The surface scan morphologies of the elements of RE-Ni-W-P-B4C composite coatings are shown in Fig.7 which indicates the elements in the coatings disperse evenly.

Fig.6 SEM images of RE-Ni-W-P-B4C composite coatings: (a) Surface morphology; (b) Section morphology
3.6 High temperature oxidation resistance of RE- Ni-W-P-B4C composition coatings
The high temperature-oxidation resistance of RE-Ni-W-P-B4C composite coatings as well as the matrix copper, Ni-W-P alloy coatings and RE-Ni-W-P-SiC composite coatings was determined by means of mass gain method, the specimen mass changing with the treatment time was recorded by electronic thermal balance. A vertical type of electric resistant furnace with a quartz reaction tube was employed. The oxidation experiments were carried out at the temperatures from 673 K to 1 113 K. The temperature was measured and controlled by two S type thermocouples, respectively. The experimental temperature accuracy was controlled within ��2 K by a PID controller. The oxidation rate was the mass gain per unit area of the samples (mg/cm2).
 Fig.7 Surface morphologies of and surface scanning elements for RE-Ni-W-P-B4C composite coatings: (a) SE image; (b) BK image; (c) C; (d) W; (e) P; (f) Ce; (g) Fe; (h) Ni
Fig.7 Surface morphologies of and surface scanning elements for RE-Ni-W-P-B4C composite coatings: (a) SE image; (b) BK image; (c) C; (d) W; (e) P; (f) Ce; (g) Fe; (h) Ni
The measurement results of the high temperature oxidation kinetics of Cu matrix electrodeposited Ni-W-P, RE-Ni-W-P-B4C and RE-Ni-W-P-SiC coatings are shown in Fig.8. The results indicate that the relationship between the oxidized film weight and the oxidation time is nearly a parabola curve, and the following regression equations can be obtained by regression analysis of the data:
For Cu matrix:
mCu=3.494 7[1-exp(-0.007 65t)], R2=0.99 (1)
For Ni-W-P coating:
mNi-W-P=1.1716[1-exp(-0.007 65t)], R2=0.99 (2)
For RE-Ni-W-P-B4C coating:
mRE-Ni-W-P-B4C=0.864 5[1-exp(-0.009 01t) ], R2=0.99 (3)
For RE-Ni-W-P-SiC coating:
mRE-Ni-W-P-SiC=0.578 4[1-exp(-0.011 8t)], R2=0.98 (4)
where m is mass gain, mg/cm2; t is oxidation time, min.
The correlation coefficients of above regression equations are all equal to or greater than 0.98, which means there is a certain relationship between the oxidized film mass of coating and the oxidation time during high temperature oxidation. When the oxidation time is over 60 min, the growth regularity of oxidized film can be expressed as power function Eqns.(1) to (4), and the reaction rate is controlled by the mass transfer rate. The oxidized film mass of Cu matrix, Ni-W-P, Ni-W-P-SiC and RE-Ni-W-P-B4C coatings is approximately a linear function of the oxidation time when oxidation time is less than 60 min. When the oxidation time is less than 60 min the oxidized film is very thin, the diffusion rate is relatively fast and the
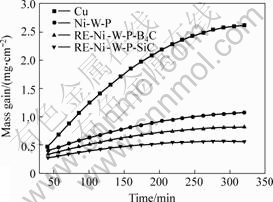
Fig.8 Relationship of mass gain of coatings and oxidation time
surface reaction resistance is not ignorable, the surface reaction is the controlling factor and the oxidation rate is constant.
The oxidation results of four coatings at different temperatures for 3 h are represented as the curves in Fig.9 and the following regressions can be obtained by regression analysis of the data.
For Cu matrix:
mCu=5.4��10-4exp(T/121.06)+0.411 6, R2=0.995 (5)
For Ni-W-P coating:
mni-W-P=5.0��10-5exp(T/105.51)+0.403 2, R2=0.999 (6)
For RE-Ni-W-P-B4C coating:
mRE-Ni-W-P-B4C=1.0��10-5exp(T/97.84)+0.425 3, R2=0.998 (7)
For RE-Ni-W-P-SiC coating:
mRE-Ni-W-P-SiC=1.89��10-7exp(T/74.90)+0.441 3, R2=0.980 (8)
where m is mass gain, mg/cm2, T is oxidation temperature, K.
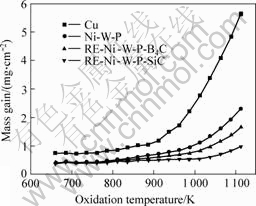
Fig.9 Relationships between mass gain and oxidation temperature (oxidation time: 3h )
It can be seen from Fig.9 that RE-Ni-W-P-SiC coating is superior to Ni-W-P coating and RE-Ni-W-P-B4C coating in the oxidation resistance when heat treated temperature is above 873 K. From 673 K to 873 K, the difference of the oxidation resistance of coatings is small. When the temperature is over 1 073 K, the oxidation resistance of RE-Ni-W-P-SiC coating is evidently superior to those of Ni-W-P and RE-Ni-W-P-B4C coatings. And when the temperature is higher than 1 073 K the oxidized film weight increases sharply, which indicates that the oxidation resistances of these coatings decrease sharply when the temperature is over 1 073 K.
4 Conclusions
1) RE-Ni-W-P-B4C composite coatings were with suitable quantity particle were prepared. The coatings have good wear resistance and oxidation resistance properties.
2) The phase of the composite coatings is main amorphous as deposited. After heated at 673 K for 3 h, the composite coatings change to crystal partly, the main phases are Ni, Ni3P, P, W, Ni3C, Ni3B and CeO2.
3) The micrograph of composite coatings is in uniformity and the solid particles scatter well in the coatings. The surface scan morphologies of the elements of RE-Ni-W-P-B4C composite coatings indicate the elements in the coatings disperse evenly.
4) The micro-hardness of the coatings is high and the value is about HV 825 -HV 1 097, and it is HV 1 236 after heat treated at 673 K for 3 h.
5) The friction resistance of the composite coatings is good and the mass loss rate is 0.15 mg/h. The friction coefficient of the coatings is below 0.33 and the minim value is 0.10.
6) The increased mass of oxidized film of Cu matrix, Ni-W-P, RE-Ni-W-P-SiC and RE-Ni-W-P-B4C coatings is approximately a linear function of the oxidation time when oxidation time is less than 60 min, while the former is a power function of the latter when the heat treatment time is over 60 min. The oxidation rate order of the four coatings from high to low is Cu matrix, Ni-W-P, RE-Ni-W-P-B4C, RE-Ni-W-P-SiC.
7) The oxidized film mass of Cu matrix, Ni-W-P, Ni-W-P-SiC and RE-Ni-W-P-B4C coating is an exponential function of the oxidation temperature during high temperature oxidation.
References
[1] WANG Long-shou. Long life technology of mould copper plates in Baosteel[J]. Continuous Casting, 2002 (5): 18-21. (in Chinese)
[2] KUBOTA A, TAKATA M, HORI K, UMEYAMA Y, YAMAMOTO K, CSAKO S. Extending CC mold life by thermal spray coating[J]. Continuous Casting, 2001 (4): 13-14, 46. (in Chinese)
[3] YANG Ming-duo, ZHANG Tie-jun. Technology of electroplate Ni-Co alloy for sheet copper of continuous caster crystallizer[J]. Angang Technology, 2003 (1): 18-20. (in Chinese)
[4] HE Yue-lin. Functional thick nickel-iron alloy plating[J]. Material Protection, 1997, 30(5): 22-23. (in Chinese)
[5] HE Yue-lin. Study on functional thick nickel-iron alloy plating[J]. Electroplating and Finishing, 2002, 21(2): 1-13. (in Chinese)
[6] WANG Bao-yu, LIU Shu-lan. The effect of RE on Ni-Fe alloy electrodeposition[J]. Material Protection, 1998, 31(1): 12-14. (in Chinese)
[7] MATSUYAMA T, OTA T. Continuous casting mold for steel and it��s manufacturing method: JP, 2000-218346[P]. 2000-08-08.
[8] DONG Yun, LIN Xiao-ping. Study on liquor of brush plating Ni-W-Co alloy and brush plating process[J]. Journal of Heibei University of Technology, 1997, 26(3): 68-72. (in Chinese)
[9] YUAN Qing-long, HOU Wen-yi. Study on strengthening of crystallizer with Ni-P alloy by brush-plating [J]. Journal of Taiwan University of Technology, 2001, 32(2): 162-164. (in Chinese)
[10] FU Han-guang, XING Jian-dong, ZHANG Li, HONG Tao-sheng. Ceramic-lined compound copper pipe preparation by self-propagating high-temperature synthesis[J]. Nonferrous Metals, 2002, 54(3): 15-18.
[11] REIGADA D C, PRIOLI R, JACOBSOHN L G, FREIRE JR. F L. Boron carbide films deposited by a magnetron sputter�Cion plating process: film composition and tribological properties[J]. Diamond and Related Materials, 2000, 9: 489-493.
[12] MONIR VAGHEFI S M, SAATCHI A, EBRAHIMIAN-HOSEINABADI M. Deposition and properties of electroless Ni-P-B4C composite coatings[J]. Surface & Coatings Technology, 2003, 168: 259-262.
[13] SHRESTHA N K, KAWAI M, SAJI T. Co-deposition of B4C particles and nickel under the influence of a redox-active surfactant and anti-wear property of the coatings[J]. Surface & Coatings Technology, 2005, 200: 2414-2419.
[14] ZHU Cheng-yi, LI Guang-qiang, PENG Qi-chun, LI Yuan-yuan. Study on the properties of electroplated Ni-W-P-B4C composite coatings[C]// LIU Guang-lin, KONG Jian-yi NI Hong-wei, et al. Proceedings of the International Workshop on Modern Science and Technology in 2006. Beijing: China Science and Technology Press, 2006: 26-31.
[15] FANG Guo-xin , YU Zhi-gang. Preparation of organic RE additives for Cr plating [J ]. Materials Protection, 1993, 26(5): 26-27. (in Chinese)
[16] QIAN Da-ren. Application of RE in electroplating[J]. Plating & Finishing, 1992 , 14(3) : 19-21. (in Chinese)
[17] GUO Zhong-cheng. Effects of rare earth process and properties of composite plating[J]. Acta Metallurgica Sinica, 1996, 32(5): 516-520. (in Chinese)
[18] GUO Zhong-cheng, LIU Hong-kang, WANG Zhi-yin, WANG Min. Process and properties of electroless RE-Ni-B-SiC composite coatings[J]. Acta Metallurgica Sinica, 1995 , 8(2): 118-121.
[19] ZHU Cheng-yi, GUO Zhong-cheng. Effects of addition of RE on the properties and structure of Ni-W-B-SiC composite coatings[J]. Engineering Chemistry & Metallurgy, 1999, 20(3): 225-228. (in Chinese)
[20] GUO Zhong-cheng, ZHU Cheng-yi, ZHAI Da-cheng, YANG Xian-wan. Microstructure of electrodeposited RE-Ni-W-P-SiC composite coating[J]. Tran Nonferrous Met Soc China, 2000, 10(1): 15-17.
[21] LIANG Ying-jiao, CHE Ying-chang. Handbook of inorganic thermodynamics[M]. Shenyang: Northeastern University Press, 1993. (in Chinese)
[22] WEN Ming-fen. Study on new type of solar energy selective heat absorption coatings[D]. Kunming: Kunming University of Science and Technology, 1998: 23-25. (in Chinese)
[23] YANG Xi-zhen, YANG Wu. Electrochemical thermo-dynamics of metallic corrosion potential-pH diagram and it��s application[M]. Beijing: Chemistry Industry Press, 1991. (in Chinese)
[24] TU Zhen-mi. The principle and technology of electroplated alloys[M]. Beijing: National Defence Press, 1993. (in Chinese)
[25] JIANG Wen-hui. Process and properties of electroless plating Ni-P-ZrO2 alloy[J]. Journal of Central South University of Technology, 2001, 32(1): 46-49. (in Chinese)
[26] ZHU Cheng-yi, YAO Hua-xin, NI Hong-wei. Effect of concentration of sodium tungsten in electroplating bath on properties of nickel-tungsten-phosphorus ternary alloy[J]. Electroplating and Finishing, 2003, 22(2): 1-3. (in Chinese)
[27] CHI Yi, FAN Hui-yu, GAO Jin-bo. A study on electroless composite Ni-P-PTFE-SiC plating[J]. Mater Sci Technol, 2003, 11(1): 77-80. (in Chinese)
[28] YU Sheng-xue, SHAO Guang-jie, SONG Wei-ming. Technologies and properties of electroless Ni-P-PTFE composite coating[J]. Physics Examination and Testing, 2001(1): 14-17. (in Chinese)
(Edited by YANG Hua)
Foundation item: Project (2005XZ1) supported by the Scientific Research Key Foundation of Wuhan University of Science and Technology, China
Corresponding author: ZHU Cheng-yi; Tel: +86-27-63166470; E-mail: zhchyhsy-2002@163.com