J. Cent. South Univ. Technol. (2008) 15: 49-53
DOI: 10.1007/s11771-008-0011-1
Interfacial interaction of bio-leaching of pyrite mineral
GU Guo-hua(������)1, WANG Hui(�� ��)2, SUO Jun(�� ��)1, QIU Guan-zhou(�����)1, HAO Ye(�� Ұ)2
(1. School of Resource Processing and Bioengineering, Central South University, Changsha 410083, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
Abstract: Electrokinetic and contact angle measurements were used to discuss the interfacial interaction on bio-leaching of pyrite mineral. Surface energy parameters of pyrite mineral and thiobacillus ferrooxidans were obtained by calculating according to formula of Young��s equation and contact angle measurements. The results show that surface energy of thiobacillus ferrooxidans is much higher than that of pyrite mineral, and the reaction of pyrite mineral with thiobacillus ferrooxidans causes the reduction of the pyrite surface energy. The interfacial interaction energies between pyrite mineral and thiobacillus ferrooxidans were also obtained based on polar interfacial interaction theory and electrokinetic and contact angle measurements. The thermodynamics approach only considering Lifshitz-van der Waals and Lewis acid-base interaction fails to explain the adhesion behavior of the bacteria, but the extended Derjaguin-Landan-Verwey-Overbeek theory concerning Lifshitz-van der Waals and Lewis acid-base and the electrostatic can exactly predict interfacial interaction.
Key words: extended DLVO theory; surface energy; thiobacillus ferrooxidans; pyrite; contact angle
1 Introduction
Mineral bio-processing comprises bioleaching and biobeneficiation. In bioleaching, the valuable metals are extracted from ores by the bio-oxidation of minerals. Both direct and indirect mechanisms[1] are known to be responsible for bacterial attack on sulphide ores. The indirect mechanism operates by the chemical action of acidic ferric sulphate produced by bacterial metabolism. The direct mechanism involves attack of the mineral by the bacteria, for which intimate contact is required. The mechanism of bio-oxidation is still under debate but one of the mechanisms often quoted concerns the adhesion of bacteria on mineral, thereby inducing the oxidation process[2-3]. The fundamental aspects concerning bacterial adhesion and its role in bioleaching contribute to the enhancement of leaching rates.
In general, the bacterial adhesion can be illustrated by the surface thermodynamics and by the extended Derjaguin-Landan-Verwey-Overbeek(DLVO) theory of calculating the interaction energy as a function of separation distance[4-6]. The surface interaction between bacterial and the mineral grain is affected by various factors, such as surface charge, surface hydrophobicity, etc. Any theory that attempts to explain the mechanism of bio-complicated surface interaction must incorporate all these parameters. In this paper, the change of the interfacial free energy in the bio-leaching system was analyzed in order to understand the interaction of thiobacillus ferrooxidans on pyrite minerals through surface thermodynamics and extended DLVO theory.
2 Theory foundation
2.1 Calculation of solid surface energy
Young��s equation[7] describes the relationship among the solid surface energy ��S, liquid surface energy (surface tension) ��L, solid/liquid interfacial interaction free energy ��SL, solid surface pressure ��0 and balance contact angle �� in the solid/liquid/gas triple-phase system as follows.
��S-��0-��SL=��Lcos �� (1)
For the low energy surface, surface pressure ��0 can be negligible[4, 8], Young��s equation is turned into Eqn.(2)
��S-��SL=��Lcos �� (2)
van Oss[9-10] argued that solid/liquid surface energy �� is composed of Lifshitz-van der Waals(LW) component ��LW and Lewis acid-base(AB) component ��AB, and ��AB is composed of Lewis acid component ��+ and Lewis base component ��-. Consequently, to solid/liquid, surface energy can be denoted by
 (3)
(3)
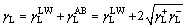 (4)
(4)
The connection between solid/liquid interfacial free energy and solid/liquid respective surface energy can be denoted by

 (5)
(5)
Substituting Eqns.(3)-(5) into Eqn.(2), we can get the connection among solid surface energy, liquid surface energy and the balance contact angle between them.

 (6)
(6)
Therefore, the solid surface energy parameters (

 ) can be obtained by mensurating the contact angle between solid surface and the three kinds of liquid with the given the values of
) can be obtained by mensurating the contact angle between solid surface and the three kinds of liquid with the given the values of  ,
, ,
, (two of the three must be polar liquid).
(two of the three must be polar liquid).
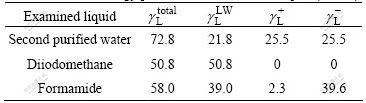
The surface tension components of the liquid (polar liquids of water, formamide and diiodomethane) are listed in Table 1[11-13].
Table 1 Surface energy parameters of examined liquid (mJ/m2)
2.2 Calculation of interfacial interaction free energy (LW-AB approach)
2.2.1 Solid/liquid and liquid/liquid interfacial interaction free energy
The interfacial interaction free energy (��GSW) between solid surface or the liquid film surface and water includes Lifshitz-van der Waals(LW) interaction free energy ( ) and Lewis acid-base(AB) interaction free energy (
) and Lewis acid-base(AB) interaction free energy ( )[14-15].
)[14-15].
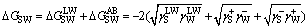 (7)
(7)
2.2.2 Interfacial interaction of component-1 and component-2 via component-3
Let water be the component-3, the interaction free energy of component-1 and component-2 via component-3 (��G1W2) includes LW interaction free energy (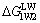 ) and AB interaction free energy (
) and AB interaction free energy ( )[16-17].
)[16-17].

 (8)
(8)
2.3 Extended EDLVO approach
EDLVO theory contains Lifshitz-van der Waals attractive forces, Lewis acid-base interactive force(AB) and attractive or repulsive electrostatic force(EL). Lifshitz-van der Waals attractive forces is the microscope-scale force of attraction between condensed-phase molecules or surfaces. Electrostatic force is caused by the double-layer interactions between two grains, acid-base interactive force is based on the interaction of the electron receptor and electron donator in aqueous solutions. The interfacial interaction of bio-leaching of sulfide mineral can be denoted as the balance between Lifshitz-van der Waals attractive forces, Lewis acid-base interactive force and attractive or repulsive electrostatic force:
 (9)
(9)
where d is the separation distance.
Because the radius of mineral particle is much longer than that of the bacteria, the mineral particle can taken as flat plate, the bacteria can be approximately be taken as the sphere. According to the EDLVO theory, we can get the connection between the relevant interaction free energy and separation distance.
2.3.1 Lifshitz-van der Waals(LW) interaction energy, ��GLW(d)[6]
 (10)
(10)
 (11)
(11)
 (12)
(12)
where A13 is the Hamaker constant of the interaction between mineral particle and the bacteria, mJ; R is the radius of mineral particle, m; d0 is the minimum separation distance between the surface of mineral particle and the surface of the bacteria, 0.657 nm; Aii is the Hamaker constant of the interaction of substance i in the vacuum, mJ; 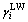 is the Lifshitz-van der Waals component of substance i surface energy, mJ/m2.
is the Lifshitz-van der Waals component of substance i surface energy, mJ/m2.
2.3.2 Lewis acid-base(AB) interaction energy, ��GAB(d)[5,18]
 (13)
(13)
where h0 is the attenuation length, 0.600 nm.
2.3.3 Electrostatic interaction energy, ��GEL(d)[19]
 (14)
(14)
where  ;
; 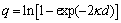 ;
;
��a is the absolute dielectric constant of separate medium, 6.95��10��10 F/m in water liquid; ��01 is the mineral particle surface potential, mV, it can be approximately replaced by electrokinetic potential ��; ��02 is the bacterial surface potential, mV, it can be approximately replaced by electrokinetic potential ��; d is the separation distance; �� is the reciprocal of Debye length, 0.104 nm-1 in standard temperature.
3 Experimental methods
3.1 Bacteria soliquid preparation
The experimental bacteria were thiobacillus ferrooxidans (cultivated by simple substance sulfur). The cultivated bacteria was put into the 50 mL centrifuge tube respectively, centrifuged for several seconds at 500 r/min. Take the upper clear solution, discard the deposition of culture medium; then centrifuge for 25 min at 4 000 r/min, discard the upper clear solution, use H2SO4 (pH��2.0) to blow the deposition to suspend, repeat the above steps three times, take the deposition and use the secondary distilled water to make the soliquid.
3.2 Contact angle measurement
The contact angle of the liquid on the solid surface was measured by the JJC-I wetting angle measurement instrument produced by the Changchun Optics Instrument Factory.
3.2.1 Mineral surfaces
By the micro sample injector(2.0 mL), the examined liquid was dropped perpendicularly at the distance of 3 mm to the solid surface to form a droplet. The droplet volume is 3-5 ��L, diameter is 1-2 mm, the measurement time can be no longer than 1 min. The average of the ten times measurement value was the contact angle of the droplet. All the measurements were operated at the room temperature (25 ��).
3.2.2 Bacteria
The ready-made bacteria soliquid was filtrated with the biomembrane. The biomembrane with the bacteria was aired at the room temperature. The detailed steps were the same as the mensuration of the mineral contact angle.
3.3 Zeta-potential measurement
0.1 g pyrite mineral powder was put into 100 mL solution with a given ion intensity, mingled for 1 h in a magnetic blender, then the DELSA440S�� Type electrokinetic instrument was used to mensurate the zeta-potential.
The ready-made bacteria soliquid was put into 2 mL solution with a given ionic strength, mingled slightly, then its the zeta-potential was mensurated. The detailed steps were the same as the mensuration of the mineral zeta-potential.
4 Results and discussion
4.1 Zeta-potential
All the measurements were made at ionic strength of 0.1 mol/L. The zeta-potentials of the pure pyrite mineral and thiobacillus ferrooxidans(cultivated by simple substance sulfur)(T.f ) as a function of pH are shown in Fig.1.
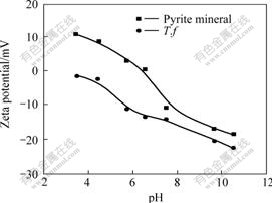
Fig.1 Zeta-potentials of pure pyrite mineral and T.f bacteria as function of pH
Fig.1 shows when the ionic strength is 0.1 mol/L, with the increase of pH, the zeta-potential of pyrite mineral has the descent trend, and the isoelectric point (IEP) is about pH 6.8 for pyrite. The cells are electronegative in the tested pH range with an IEP less than pH 2.5.
4.2 Surface free energy of pyrite mineral and thiobacillus ferrooxidans
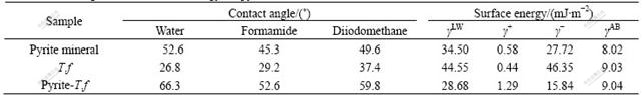
The measured contact angles of the three liquids on the surface of pyrite and T.f, and the calculated surface energy components are listed in Table 2.
From Table 2 we can see that the bacterial surfaces are more energetic than mineral surfaces, and hence are more hydrophilic, principally due to very high electron donating characteristic. After the pyrite mineral reacts with T.f bacteria, the component ��LW and electron- donating part (��-) obviously decrease, which causes the reduction of the surface energy.
Table 2 Contact angle and surface free energy for pyrite mineral and thiobacillus ferrooxidans
4.3 Interfacial interaction free energy between pyrite mineral and thiobacillus ferrooxidans
The water is defined as the 3rd phase. Using Eqns.(7) and (8), the values of the interfacial free energy between thiobacillus ferrooxidans and pyrite surfaces and its components are calculated and presented in Table 3.
Table 3 Interfacial interaction free energy between pyrite ineral and thiobacillus ferrooxidans in system pyrite-T.f

According to the thermodynamic approach (LW-AB approach), when the physicochemical interactions are considered, the adhesion is favourable only when the energy is negative. From Table 3, in the bio-leaching system, Lifshitz-van der Waals component value is negative, it is attractive to all thiobacillus ferrooxidans, but Lewis acid-base component is highly repulsive and causes the total interaction free energy between thiobacillus ferrooxidans and pyrite mineral is always positive. Therefore, by thermodynamic approach it is predicted that there will be no adhesion of thiobacillus ferrooxidans on pyrite.
Based on the extended DLVO theory, using Eqns.(10)-(14), we can build up the correlation between the separation distance and the interaction free energy of bio-leaching system. The results are shown in Fig.2.
From Fig.2, the acid-base interaction between mineral-bacteria is highly repulsive but this force operates only at close distances of 3 nm. van der Waals interaction shows very intense attraction. At pH values where the bacterial cells and mineral particles are oppositely charged, the attractive electrostatic interactions cause the formation of the total interaction while adhesion of bacterial cells is possible on mineral surface.
At pH 3.5, minerals are positively charged but bacterial cells are negatively charged and hence electrostatic interaction is highly attractive, causing second-minimum (2.9-3.7 nJ) of the total interaction. This attachment is predicted to be irreversible. From Fig.2(b), at pH 10.5, both mineral and bacteria are highly negatively charged and hence electrostatic interaction is
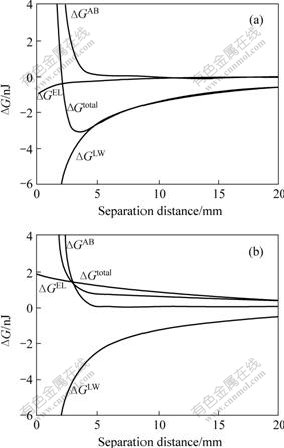
Fig.2 Curves of interfacial free energy of interacting surface vs separation distance in bio-leaching system with different pH values: (a) pH=3.5; (b) pH=10.5
repulsive. The total interaction between mineral and bacteria behaves repulsively, which means, under the condition of relatively acidic, the adhesion of bacteria on the surface of mineral cannot happen.
The above research results show that the extended DLVO theory method is effective to predict the adhesion behaviour. In acidic condition, although the acid-base interaction is repulsive, it is a short-range interaction, whereas the electrostatic interaction is attractive, also a long-range interaction, therefore the bacteria can be adsorbed on the mineral surface. In alkaline condition, however, the electrostatic interaction between bacteria and the mineral becomes repulsive force, bacteria are more liable in the medium solution, not on the surface of the pyrite mineral.
5 Conclusions
1) The bacterial surfaces are more energetic than pyrite mineral surfaces, the surface energy reduces after pyrite contacts with thiobacillus ferrooxidans.
2) The thermodynamic approach cannot describe bacterial adhesion on minerals and this discrepancy is the result of inadequate description of electrostatic interactions.
3) According to the extended DLVO theory, the adhesion of thiobacillus ferrooxidans on the surface of pyrite mineral is attributed to the electrostatic interaction that is a long-range interaction. In acidic condition, thiobacillus ferrooxidans can be attached on the surface of pyrite, as the electrostatic interaction is attractive. However, in alkaline condition, the adhesion of thiobacillus ferrooxidans on the surface of pyrite mineral cannot happen because the electrostatic interaction between bacteria and the mineral is repulsive force.
References
[1] Silverman M P, Ehrlich H L. Microbial formation and degradation of metals[J]. Adv Appl Microbiol, 1964, 6: 153-206.
[2] Tributsch H, Rojas-Chapana J A. Metal sulfide semiconductor electrochemical mechanisms induced by bacterial activity[J]. Electrochimica Acta,2000, 45(28): 4705-4716.
[3] Tributsch H. Direct versus indirect bioleaching[J]. Hydrometallurgy, 2001, 59: 177-185.
[4] Janczuk B, Bruque J M, Gonz��lez-Mart��n M L, ROM��n-GAL��N E. The contribution of double layers to the free energy of interactions in the cassiterite-SDS solution system[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 1995, 100: 93-103.
[5] Sharma P K, Hanumantha-Rao K. Adhesion of Paenibacillus polymyxa on chalcopyrite and pyrite: Surface thermodynamics and extended DLVO theory[J]. Colloids and Surfaces B: Biointerfaces, 2003, 29(1): 21-38.
[6] Hermansson M. The DLVO theory in microbial adhesion[J]. Colloids and Surfaces B: Biointerfaces, 1999, 14(1/4): 105-119.
[7] Shalel-Levanon S, Marmur A. Validity and accuracy in evaluating surface tension of solids by additive approaches[J]. Journal of Colloid and Interface Science, 2003, 262(2): 489-499.
[8] Janczuk B, Gonz��lez-Mart��n M L, Bruque J M. The influence of sodium dodecyl sulfate on the surface free energy of cassiterite[J]. Journal of Colloid and Interface Science, 1995, 170(2): 383-391.
[9] van Oss C J, Chaudhury M K, Good R J. Mechanism of partition in aqueous media[J]. Separation Science and Technology, 1987, 22(6): 1515-1526.
[10] van Oss C J, Good R J, Chaudhury M K. Estimation of the polar surface tension parameters of glycerol and formamide, for use in contact angle measurements on polar solids[J]. Journal of Dispersion Science and Technology, 1990, 11(1): 75-81.
[11] Le��n V, Tusa A, Araujo Y C. Determination of the solid surface tensions: I. The platinum case[J]. Colloids and Surfaces A: Physicochemical and Engineering Aspects, 1999, 155(2/3): 131-136.
[12] Ad?o M H V C, Saramago B J V, Fernandes A C. Estimation of the surface properties of styrene-acrylonitrile random copolymers from contact angle measurements[J]. Journal of Colloid and Interface Science, 1999, 217(1): 94-106.
[13] Park S J, Kim J S. Influence of plasma treatment on microstructures and acid�Cbase surface energetics of nanostructured carbon blacks: N2 plasma environment[J]. Journal of Colloid and Interface Science, 2001, 244(2): 336-341.
[14] Espinosa-Jim��nez M, Ontiveros-Ortega A, Gim��nez-Mart��n E. Surface energetics of the adsorption process of a cationic dye on leacril fabrics[J]. Journal of Colloid and Interface Science, 1997, 194(2): 419-426.
[15] Espinosa-Jim��nez M, Gim��nez-Mart��n E, Ontiveros- Ortega A. Effect of tannic acid on the �� potential, sorption, and surface free energy in the process of dyeing of leacril with a cationic dye[J]. Journal of Colloid and Interface Science, 1998, 207(1): 170-179.
[16] Wu W, Giese R F, van Oss C J. Stability versus flocculation of particle suspensions in water��correlation with the extended DLVO approach for aqueous systems, compared with classical DLVO theory[J]. Colloids and Surfaces B: Biointerfaces, 1999, 14(1/4): 47-55.
[17] Yang C, Dabros T, Li D q, CZARNECKI J, MASLIYAH J H. Analysis of fine bubble attachment onto a solid surface within the framework of classical DLVO theory[J]. Journal of Colloid and Interface Science, 1999, 219(1): 69-80.
[18] Poortinga A T, Bos R, Norde W, Busscher H J. Electric double layer interactions in bacterial adhesion to surfaces[J]. Surface Science Reports, 2002, 47(5): 1-32.
[19] Oliveira R. Understanding adhesion: A means for preventing fouling[J]. Thermo Fluid Sci, 1997, 14(4): 316-322.
(Edited by YANG Hua)
Foundation item: Project(2004CB619204) supported by the National Basic Research Program of China; Project(2002) supported by the Teaching and Research Award Program for Outstanding Young Teachers in Higher Education Institutions, China
Received date: 2007-06-02; Accepted date: 2007-09-18
Corresponding author: GU Guo-hua, Professor, PhD; Tel: +86-13975151469; E-mail: guguohua@126.com