��Ԫ�ضԼ�ѹMg-Zn-Mn�Ͻ������֯����ʴ�ԡ����������Լ��������ܵ�Ӱ��
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2021���2��
�������ߣ�����ʤ ������ ��Ծ�� ���� ����ΰ ������ ��Ӣ��
����ҳ�룺358 - 370
�ؼ��ʣ�þ�Ͻ����︯ʴ���������ԣ�����֯����ѧ����
Key words��magnesium alloy; bio-corrosion; antibacterial activities; microstructure; mechanical properties
ժ Ҫ��Ϊ��Ѱ�Һ���ֲ��Ӧ�õĿ����オ����ϣ����ð�����������ȼ�ѹ�����Ʊ�Mg-6Zn-0.3Mn-xCa(x=0��0.2��0.5����������)�Ͻ��ý���������ɨ��������������������о�Mg-6Zn-0.3Mn-xCa�Ͻ������֯����ѧ���ܡ������������CaԪ�صļ������ϸ����ѹ̬Mg-6Zn-0.3Mn�Ͻ�ľ����������ǿ�ȡ���Ca�������ֱ�Ϊ0.2%��0.5%(��������)ʱ���Ͻ�ľ����ߴ���4.8 ��m�ֱ�ϸ����4.6��4.2 ��m�����ֺϽ��У�����0.5%Ca�ĺϽ���и��õ��ۺ���ѧ���ܣ���������ǿ�Ⱥ��쳤�ʷֱ�Ϊ334 MPa��20.3%�����⣬�������о��Ͻ�ĸ�ʴ��Ϊ��ϸ�����ԺͿ������ܽ������ۡ�����Ca���������ӣ��Ͻ����ʴ�Խ��ͣ�����Ҫ������Ca2Mg6Zn3����γɵ�����Ч��������λ����������ӡ�L929ϸ����ϸ������������ʾ����Mg-6Zn-0.3Mn��Mg-6Zn-0.3Mn-0.2Ca�Ͻ���ȣ�Mg-6Zn-0.3Mn-0.5Ca�Ͻ���и��õ�ϸ�����ԡ����ڽ��ɫ�������������������ѧ���ԣ��ڹ��������У��Ͻ�Ŀ�����������Ca���������Ӷ���ǿ��
Abstract: To find suitable biodegradable materials for implant applications, Mg-6Zn-0.3Mn-xCa (x=0, 0.2 and 0.5, wt.%) alloys were prepared by semi-continuous casting followed by hot-extrusion technique. The microstructure and mechanical properties of Mg-6Zn-0.3Mn-xCa alloys were investigated using the optical microscope, scanning electron microscope and tensile testing. Results indicated that minor Ca addition can slightly refine grains of the extruded Mg-6Zn-0.3Mn alloy and improve its strength. When 0.2 wt.% and 0.5 wt.% Ca were added, the grain sizes of the as-extruded alloys were refined from 4.8 to 4.6 and 4.2 ��m, respectively. Of the three alloys studied, the alloy with 0.5 wt.% Ca exhibits better combined mechanical properties with the ultimate tensile strength and elongation of 334 MPa and 20.3%. The corrosion behaviour, cell viability and antibacterial activities of alloys studied were also evaluated. Increasing Ca content deteriorates the corrosion resistance of alloys due to the increase of amount of effective cathodic sites caused by the formation of more Ca2Mg6Zn3 phases. Cytotoxicity evaluation with L929 cells shows higher cell viability of the Mg-6Zn-0.3Mn-0.5Ca alloy compared to Mg-6Zn-0.3Mn and Mg-6Zn-0.3Mn- 0.2Ca alloys. The antibacterial activity against Staphylococcus aureus is enhanced with increasing the Ca content due to its physicochemical and biological performance in bone repairing process.
Trans. Nonferrous Met. Soc. China 31(2021) 358-370
Bao-sheng LIU1, Miao-miao CAO1, Yue-zhong ZHANG2, Yong HU1, Chang-wei GONG1, Li-feng HOU3, Ying-hui WEI1,3
1. College of Materials Science and Engineering, Taiyuan University of Science and Technology, Taiyuan 030024, China;
2. College of Chemical and Biological Engineering, Taiyuan University of Science and Technology, Taiyuan 030024, China;
3. College of Materials Science and Engineering, Taiyuan University of Technology, Taiyuan 030024, China
Received 14 March 2020; accepted 29 November 2020
Abstract: To find suitable biodegradable materials for implant applications, Mg-6Zn-0.3Mn-xCa (x=0, 0.2 and 0.5, wt.%) alloys were prepared by semi-continuous casting followed by hot-extrusion technique. The microstructure and mechanical properties of Mg-6Zn-0.3Mn-xCa alloys were investigated using the optical microscope, scanning electron microscope and tensile testing. Results indicated that minor Ca addition can slightly refine grains of the extruded Mg-6Zn-0.3Mn alloy and improve its strength. When 0.2 wt.% and 0.5 wt.% Ca were added, the grain sizes of the as-extruded alloys were refined from 4.8 to 4.6 and 4.2 ��m, respectively. Of the three alloys studied, the alloy with 0.5 wt.% Ca exhibits better combined mechanical properties with the ultimate tensile strength and elongation of 334 MPa and 20.3%. The corrosion behaviour, cell viability and antibacterial activities of alloys studied were also evaluated. Increasing Ca content deteriorates the corrosion resistance of alloys due to the increase of amount of effective cathodic sites caused by the formation of more Ca2Mg6Zn3 phases. Cytotoxicity evaluation with L929 cells shows higher cell viability of the Mg-6Zn-0.3Mn-0.5Ca alloy compared to Mg-6Zn-0.3Mn and Mg-6Zn-0.3Mn- 0.2Ca alloys. The antibacterial activity against Staphylococcus aureus is enhanced with increasing the Ca content due to its physicochemical and biological performance in bone repairing process.
Key words: magnesium alloy; bio-corrosion; antibacterial activities; microstructure; mechanical properties
1 Introduction
As temporary biodegradable metallic implant materials, magnesium (Mg) and its alloys have attracted the attention of many researchers due to their good biocompatibility and biodegradability [1]. Compared with permanent implants, temporary biodegradable Mg alloy implants can be completely degraded after the tissue recovery, resulting from the fact that the human body contains a large amount of Mg2+ ions and the degradation products of Mg can be electively metabolised. Thus, a secondary surgery for implant removal and long-term foreign-body reaction can be effectively avoided by using the temporary biodegradable Mg alloy implants, which could potentially reduce the burden on the healthcare system and benefit patients by mitigating risks and costs [2,3]. Furthermore, magnesium alloys also exhibit advantageous properties such as low density, high specific strength and low elastic modulus, which make them particularly suitable as load-bearing implants for repairing or substituting the diseased or damaged tissues [4,5]. Furthermore, Mg-based biodegradable materials have been studied as orthopaedics implants [6] and cardiovascular stent [7]. But their applications are still impeded mainly due to their poor mechanical properties [8] and corrosion resistance [9]. Besides the loss of mechanical properties in a short period , the poor corrosion resistance under physiological conditions is also related to the production of gaseous hydrogen, which may delay the healing process and cause necrosis of the tissue around [3,10]. Therefore, it is necessary and urgent to develop a Mg alloy with excellent mechanical and corrosion resistance properties to satisfy the required temporary implants.
Three main approaches have been developed to control the degradation rate of magnesium such as alloying, surface modification and coating. Recent studies have demonstrated that manipulating components and introducing the second phase by alloying is an effective way to improve the mechanical properties and corrosion resistance for Mg alloys [11], the influence of the alloying elements on the mechanical properties [12,13] and corrosion behaviour [14,15] of the Mg-based biodegradable materials has also been widely investigated. Among the as-studied alloy systems, the extruded Mg-Zn-Mn system alloy has greater application potentials since it exhibits high strength and simultaneously high ductility and biosecurity resulting from the fact that Zn and Mn are indispensable trace element in the human body [16]. Some recent studies indicated that 6 wt.% zinc is a suitable addition to magnesium for a biocompatible degradable material [17]. The addition of a small amount of Mn to Mg-Zn alloy has a significant effect in refining the grain size and enhancing the mechanical properties, and Mg-Zn-Mn alloy used as a bone implant is easily absorbed in vivo [18]. Ca is an important component of the human bone, the mechanical properties of Ca-containing biodegradable Mg alloys were investigated [19]. However, the effects of Ca on mechanical and corrosion properties of Mg-Zn-Mn alloys are still limited. What��s more, the effects of Ca on the antimicrobial activity of the Mg-Zn-Mn alloys have rarely been reported thus far.
Additionally, as biodegradable biomaterials, alloy elements in the metal implants should not only decrease the corrosion rate and improve the mechanical strength but also be biological safety and functionality [20]. Zinc (Zn) is an essential element for the human body, and it is critical to biological functions. The new developed Mg�CZn binary alloy exhibits moderate mechanical properties and good cytocompatibility [21]. Calcium (Ca) element can be metabolised and thus is considered to be biocompatible [1]. Previous investigations demonstrated that Mg-Zn-Ca alloys exhibit excellent biocompatibility both in vitro and in vivo [22]. However, Mn is a questionable element. On one hand, it can improve the corrosion and mechanical properties. On the other hand, the high content of Mn has been confirmed to cause neuro-toxicity, leading to Parkinson��s disease [23]. Consequently, it is necessary to explore the biological safety and functionality of the new Mg-Zn-Mn-xCa based biomaterials before clinical application. In vitro cell experiment, MTT, as a cheap and convenient measurement, is widely accepted to determine the cytotoxicity [20,24,25]. For the antibacterial activity, although in vitro studies have shown that Mg and its alloys possess antibacterial properties due to the high alkaline environment accompanying the degradation of the alloys [10], it is necessary to explore the biological safety for a new Mg-Zn-Mn-xCa based biomaterials.
In this study, considering that hot-extrusion is an effective approach to improve the mechanical properties and corrosion resistance of metallic materials through grain refinement, here we choose extruded Mg-6Zn-0.3Mn alloy as the target alloy. In the present work, the effects of minor Ca addition on the microstructure, mechanical properties, bio-corrosion properties, cell viability and antibacterial activity of the extruded Mg-6Zn- 0.3Mn based alloy system are systematically studied by electron microscopy, conventional tensile testing, electrochemical tests, MTT assay and plate-counting method, respectively.
2 Experimental
The Mg-6Zn-0.3Mn with different Ca additions (0, 0.2 and 0.5 wt.%) alloy ingots (ZM603, ZM60302, ZM60305) were prepared by furnace melting of pure Mg (99.8 wt.%), Zn (99.5 wt.%) and Mg-15Ca master alloys, in a mild steel crucible with protection by the RJ-2 flux and subsequently pouring into a steel mould pre-heated to 250 ��C to produce billets with 90 mm in diameter and 120 mm in length by semi-continuous casting. After homogenisation treatment at 400 ��C, the ingot was extruded directly into rods at 360 ��C with a diameter of 20 mm by a vacuum extruder (Dorst-V15, Germany).
The specimens for the microstructure characterisation were etched with a solution of 5 mL acetic acid, 5.5 g picric acid, 10 mL distilled water and 90 mL ethanol. The microstructure of the alloys was characterized by using optical microscope (OM) and field-emission scanning electron microscope (FESEM, Hitachi S-4800, Japan) with an energy dispersive spectrometer (EDS), and the average grain size was measured by line intercept method. The phase constitutions of these alloys were analyzed by using X-ray diffraction instrument (XRD, Rigaku D/MAX- 2500, Japan).
Referring to the GB/T 6394��2002 standard, the average size of grains is determined by the linear intercept method. The specific method is as follows: draw several lines with equal length every certain distance, and calculate the number of grains intersected by each straight line optical images of alloys. If there are incomplete intersecting grains at both ends of the straight line, it is calculated as a whole grain, and the average grain size is calculated by the formula (1):
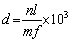 (1)
(1)
where d is the average grain diameter of the alloy (��m); n is the number of lines; l is the length of the line (mm); m is the total number of grains intersecting with all line segments; f is the magnification of the image.
As shown in Fig. 1, samples for tensile testing were machined along the longitudinal direction of alloys, and their size was 2 mm in thickness, 3.5 mm in width and 14 mm in gauge length. The tensile properties of the alloys were measured using a CMT-5504 electronic universal testing machine with a speed of 1 mm/min at room temperature.
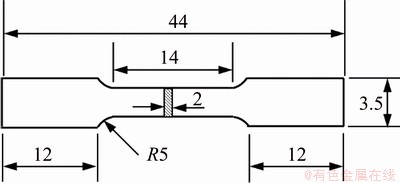
Fig. 1 Schematic diagram of tensile testing sample (unit: mm)
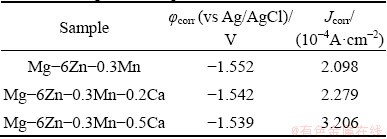
The corrosion behaviours of the alloys were investigated by the immersion tests and potentiodynamic polarization tests in Hank��s solution [26] at 37 ��C. For the immersion test, the polished samples were immersed in Hank��s solution for 10 d, then the samples were ultrasonically cleaned for 5 min in a mixture solution of AgNO3 (10 g/L) and CrO3 (200 g/L) to remove possible corrosion products and the mass loss of the samples was measured. Electrochemical measurements were conducted in a three-electrode cell using a platinum counter electrode and a Ag/AgCl reference electrode. Potentiodynamic polarization curves were measured in the Hank��s solution with a scanning rate of 0.5 mV/s.
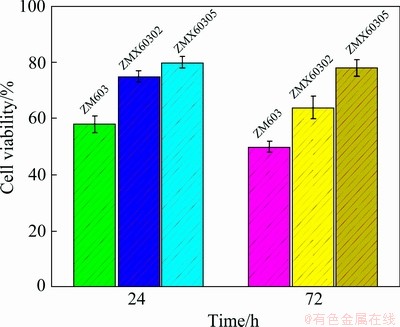
To develop the biomedical magnesium alloy, it is required to assess the cell toxicity of alloying elements. In vitro cell experiment, MTT, as a cheap and convenient measurement, is widely accepted to determine the cytotoxicity. Based on some domestic and foreign literatures [25], the L929 cells were cultured on plasma-modified PDLLA Scaffolds and incubated in a CO2 incubator at 37 ��C. A cell suspension with DMEM containing 1��105 cells was seeded on the sample which was placed in the 9 well cell culture plate and the MTT assay was conducted by incubating for 72 h at the CO2 incubator. 300 ��L of cell culture medium was added to each well. Then 30 ��L MTT solution was added followed with incubation at 37 ��C for 6 h. Then, 1 mL DMSO was also added. Viable cells were determined by the absorbance at 540 nm. All the experiments were performed in triplicate.
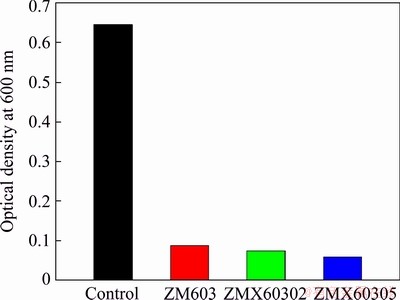
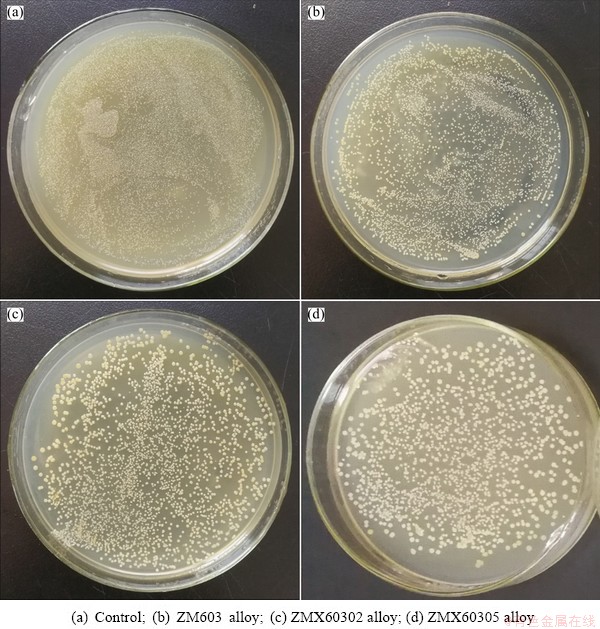
When the Mg alloys are used as the orthopedic implant, the Gram-positive bacterium Staphylococcus aureus was the main bacterium to cause infection, therefore, Staphylococcus aureus (S. aureus, ATCC 25922) was used as a model bacterium to determine the antibacterial property of the Mg-6Zn-0.3Mn-xCa alloys in vitro [27]. The antibacterial activity was investigated by the spread plate method, as described previously [28,29]. The strains were cultured at 37 ��C for 24 h on the Muller Hinton broth agar, then the cultures were diluted to 1��106 CFU/mL. 10 mL prepared bacteria suspension was added to glass tube contained samples, and incubated at 37 ��C for 8 h, then the optical density at 600 nm (OD600) was measured using a UV-visible spectrophotometer. OD600 was converted to colony-forming units (CFUs) as follows: OD600=0.1 contained ~108 CFU. After incubation, 0.1 mL of each suspension was plated onto the Muller Hinton broth and incubated at 37 ��C for 24 h. One growth control containing only the bacteria in Mueller-Hinton broth was included in the experiment. The experiment was repeated three times and the image was photographed using a digital camera. The antibacterial efficiency (��) of samples was evaluated by
 (2)
(2)
where A0 is the absorbance value of control sample, and A is the absorbance value of alloy sample.
3 Results and discussion
3.1 Microstructures and phase constitution
Figure 2 shows the optical microstructures of the as-cast Mg-6Zn-0.3Mn-xCa (x=0, 0.2, 0.5) alloys. The relatively fine particles are uniformly distributed not only at the grain boundaries but also inside the grain interiors in as-cast ZM603 alloy (Fig. 2(a)). After the addition of 0.2% Ca, the precipitates in the grains are obviously reduced and fined. The discontinuous network second phases precipitate along the grain boundary (Fig. 2(b)). After the addition of 0.5% Ca, the grains are cleaner and almost no precipitation occurs. The more continuous second phases precipitate along the network grain boundary (Fig. 2(c)). Furthermore, the grain size of the alloy decreases slightly with the increase of Ca content. This may be due to the fact that the semi-continuous/ continuous second phase hinders the grain growth during solidification [30].
For further investigation of the morphology and component of these second phase particles, SEM and EDS tests were taken out. Figure 3 shows the SEM images and EDS spectra of the as-cast Mg-6Zn-0.3Mn-xCa (x=0, 0.2, 0.5) alloys. It can be seen from Fig. 3(a) that the irregular granular second phase was randomly distributed on the substrate of as-cast ZM603 alloy. According to the results of EDS (Fig. 3(b)), the phase is rich in Zn, so it may be MgZn2 intermetallic compound [31]. After adding Ca, the hollow and lamellar second phases in the alloys are gradually connected into a continuous network (Figs. 3(c) and (e)). EDS results show that the mass ratio of Ca to Zn in the second phase is close to 2:3 (Figs. 3(d) and (f)), so it can be inferred that the phase is Ca2Mg6Zn3 [13].
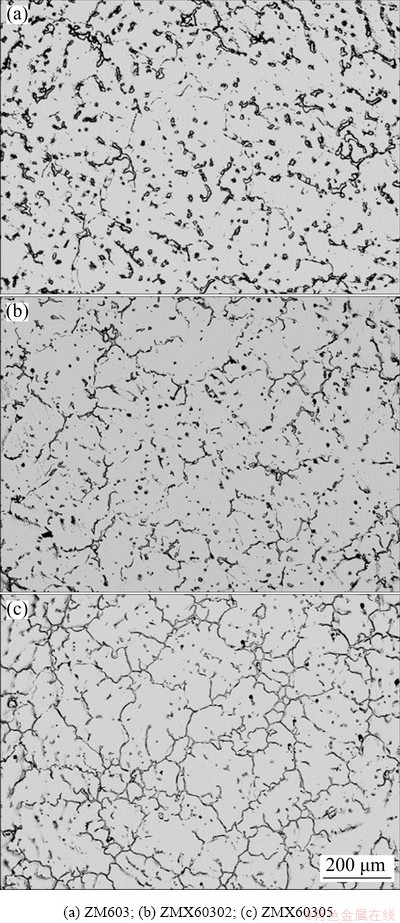
Fig. 2 Optical images of as-cast Mg-6Zn-0.3Mn-xCa alloys
Figure 4 shows the optical microstructures of the as-extruded Mg-6Zn-0.3Mn-xCa alloys perpendicular to the extrusion direction. The as-extruded ZM603 alloy has an average grain size of 4.8 ��m, indicating that a full dynamic recrystalli- zation (DXR) occurred during the extrusion (Fig. 4(a)). However, after Ca addition, the DRXed grains are found finer than that of the as-extruded ZM603 alloy (Figs. 4(b) and (c)), which implies that Ca addition refines the microstructure. The average grain sizes for the as-extruded ZMX60302 and ZMX60305 alloys are 4.6 and 4.2 ��m, respectively. The fine second phase of the as-extruded Mg-6Zn-0.3Mn-xCa alloy is distributed both in the grain interiors and at the grain boundaries. DU et al [32] have also reported that the Ca addition might hinder the grain growth during the extrusion process, which was mainly due to the undissolved second phase that retarded or postponed the dynamic recrystallization and the inhibited mobility of dislocation or grain boundaries arising from the formed fine precipitates during the extrusion.
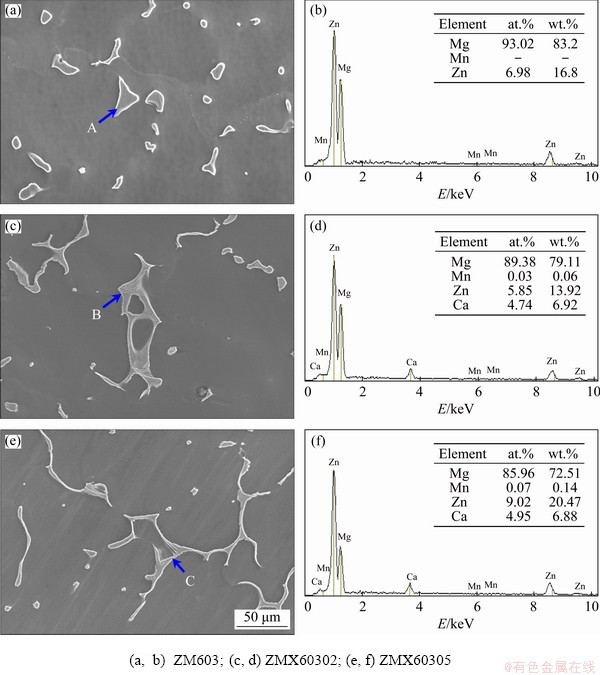
Fig. 3 SEM images (a, c, e) and EDS spectra (b, d, f) of as-cast Mg-6Zn-0.3Mn-xCa alloys

Fig. 4 Optical images of as-extruded Mg-6Zn-0.3Mn-xCa alloys
The corresponding SEM images and EDS spectra of as-extruded Mg-6Zn-0.3Mn-xCa alloys are shown in Fig. 5. As shown in Fig. 5(a), the bright second phase randomly distributed on the substrate is mainly composed of particles with a maximum diameter of 0.3 ��m. It is known that hot extrusion imparts intense plastic deformation and a severe thermal effect, which result in dispersion of the precipitate particles [33]. As a result, the fine second phase of the as-extruded Mg-6Zn-0.3Mn- xCa alloy is distributed both in the grain interiors and at the grain boundaries. EDS spectrum shows that the phase is MgZn2 phase (Fig. 5(b)). In addition to the fine particles, some flake and strip precipitates are also formed in the alloys with the addition of Ca (Figs. 5(c) and (e)). The mass ratio of Ca to Zn is close to 2:3 in strip precipitates (Figs. 5(d) and (f)), so the phase is Ca2Mg6Zn3. Additionally, based on the phase equilibrium [34], the intermetallic phase is identified to be the Ca2Mg6Zn3 ternary phase, which is also confirmed in the Mg-4.0Zn-0.2Ca alloy [24]. Further, with the increase of Ca content, the fine granular particles decreased and the flake and strip precipitates tended to increase and grow up.
The XRD patterns of the as-extruded Mg-6Zn-0.3Mn-xCa alloys are shown in Fig. 6. The ZM603 alloy is composed of ��-Mg and MgZn2 phases, while the Mn phase is undetectable in the XRD pattern as Mn content is only 0.3%. What��s more, when Ca is added, Ca2Mg6Zn3 peaks are observed and the intensity of the MgZn2 peaks decrease with increasing the Ca content. This is consistent with the results of SEM and EDS.
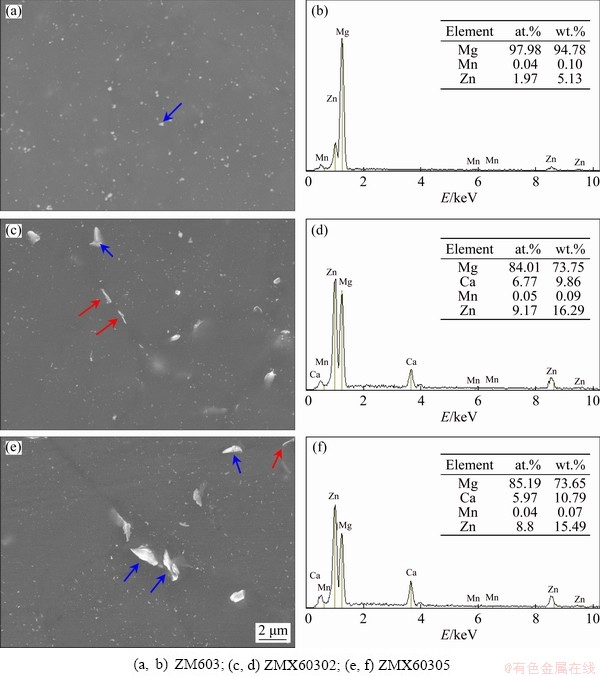
Fig. 5 SEM images (a, c, e) and EDS spectra (b, d, f) of as-extruded Mg-6Zn-0.3Mn-xCa alloys
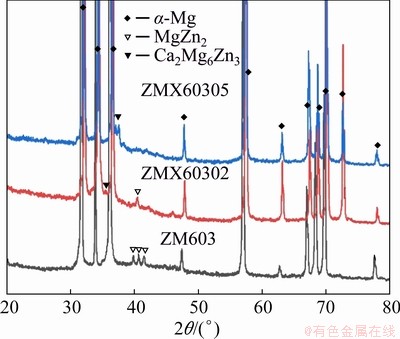
Fig. 6 XRD patterns of as-extruded alloys
3.2 Mechanical properties
The influence of Ca addition on the mechanical properties of the as-extruded ZM603 alloy is shown in Fig. 7. Overall, the yield tensile strength (YTS) and ultimate tensile strength (UTS) of the as-extruded Mg-6Zn-0.3Mn-xCa alloys are significantly increased and the elongation (EL) is slightly decreased with the increase of the Ca content. The UTSs increase to 313 and 334 MPa for the ZMX60302 and ZMX60305 alloys, respectively. The elongations decrease to 22.2% and 20.3% for the as-extruded ZMX60302 and ZMX60305 alloys, respectively.
The results show that the tensile strengths of ZM603 alloy are greatly improved with minor Ca addition. The grain size has a great influence on the yield strength of Mg alloys. According to the Hall�CPetch law [35], the grain refinement resulted in an obvious increase of TYS of the as-extruded Mg-Zn-Mn alloys by the addition of Ca. What��s more, the high amount of the second phase also contributes to the improved yield strength due to the inhibition of dislocation motion effects produced [36]. Thus, the strengthening effect caused by Ca2Mg6Zn3 phase seems to be higher in the ZMX60305 alloy than that in the ZMX60302 alloy.
To realize the fracture mechanisms of the Mg-6Zn-0.3Mn-xCa alloys, the fracture surface morphologies of the alloys after the tensile testing were examined. The results are shown in Fig. 8. A large number of tiny dimples and river patterns were observed in the fracture morphologies of the as-extruded ZM603 alloy as shown in Fig. 8(a), indicating a ductile fracture feature [37]. However, the size of dimples increases on the fracture surface of the as-extruded ZM603 alloy with increasing the Ca addition. Compared with the ZMX60302 alloy (Fig. 8(b)), the size of the dimples in the ZMX60305 alloy (Fig. 8(c)) increases significantly. As well known, the diameter and distribution of the second phase particles have a great influence on the size of the dimple. Big particles have a great dimples [38]. Due to the higher content of Ca element in the ZMX60305 alloy relative to the ZMX60302 alloy, the amount of precipitates is larger than that in the as-extruded alloy with 0.2% Ca, thus numerous large size dimples appear on the fracture surface. Thus, the elongation-to-fracture of ZM603 alloys decreases with increasing the Ca content.
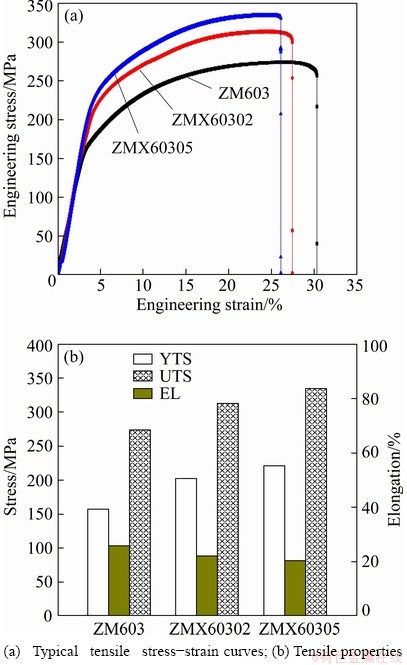
Fig. 7 Mechanical properties of as-extruded Mg-6Zn-0.3Mn-xCa alloys
3.3 Corrosion behaviors
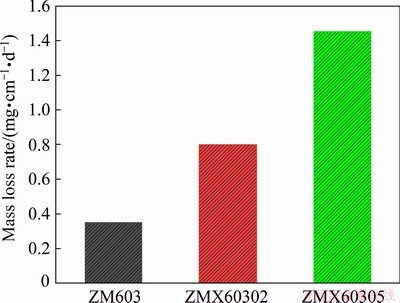
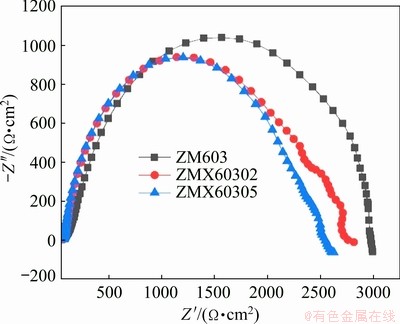
The corrosion properties of the as-extruded Mg-6Zn-0.3Mn-xCa alloys are evaluated by immersion testing and electrochemical polarization testing. The mass loss rates of the as-extruded ZM603 alloys with different Ca additions in the Hank��s solution at 37 ��C for 10 d are shown in Fig. 9. The mass loss rates in the Hank��s solution rank as: ZM603 Fig. 8 SEM images of tensile fracture for as-extruded Mg-6Zn-0.3Mn-xCa alloys Fig. 9 Mass loss rates of as-extruded Mg-6Zn-0.3Mn- xCa alloys with different Ca additions in Hank��s solution for 10 d Fig. 10 Electrochemical polarization curves of as-extruded Mg-6Zn-0.3Mn-xCa alloys The electrochemical polarization curves of the three alloys in the Hank��s solution are shown in Fig. 10. The corrosion potential (��corr) and the corrosion current density (Jcorr) are derived directly from the polarization curves by Tafel region extrapolation and summarized in Table 1. Theoretically, a lower Jcorr and higher ��corr indicate better corrosion resistance, and ��corr indicates the tendency of corrosion [40]. Compared with the as-extruded ZM603 alloy, the ��corr values of the ZMX60302 alloy (-1.542 V) and ZMX60305 alloy (-1.539 V) shift about 100 and 130 mV in the negative direction, respectively. Meanwhile, the Jcorr values of the ZMX60302 alloy and ZMX60305 alloy increase 1.1 times and 1.5 times that for ZM603, which indicates that Ca addition increases the degradation rate of the alloys, especially when Ca content reaches 0.5%. Figure 11 shows the Nyquist curves for the three alloys in the Hank��s solution. From the Nyquist curves, it can be seen that there is mainly one capacitive loop in the curves of as-extruded alloys. Generally speaking, the diameter of the capacitive loop is related to the corrosion resistance of the alloy. The larger the diameter, the better the corrosion resistance of the Mg alloy [27]. That is, the addition of Ca will reduce the corrosion resistance of ZM603 alloy. Table 1 Fitting results of polarization curves Fig. 11 Nyquist curves of as-extruded Mg-6Zn-0.3Mn- xCa alloys Increasing Ca content brings about more intermetallic phases as well as more galvanic cells, and the corrosion current is increased. The deteriorated corrosion resistance of ZM603 alloy by Ca addition may be ascribed to the formed Ca2Mg6Zn3 phase. For ZM603 alloy, a microgalvanic couple is formed between the MgZn2 phase as anode and Mg substrate as cathode since the MgZn2 phase has a higher electrochemical activity than the Mg substrate [41]. Further, the Ca2Mg6Zn3 phase is seen as the cathode and Mg substrate as the anode forming the galvanic coupling at the interface between Ca2Mg6Zn3 and Mg substrate for Ca-containing alloy [42]. As a result, the ��-Mg substrate was corroded faster compared to the MgZn2 phase and Ca2Mg6Zn3 phase. Increasing the Ca content up to 0.5 wt.% leads to more formation of Ca2Mg6Zn3 phase, as shown in Fig. 5 by SEM and Fig. 6 by XRD. Therefore, the amount of effective cathodic sites is increased and the corrosion resistance is deteriorated than that of ZMX60302 alloy. 3.4 MTT assay Figure 12 shows the cell viability of the Mg-6Zn-0.3Mn-xCa alloys tested with L929 cells using the MTT assay. The Mg-6Zn-0.3Mn alloy with different Ca additions exhibit a higher cell viability than Mg-6Zn-0.3Mn alloys without Ca addition. The increase in the cell viability at the end of 72 h confirms that ZMX60302 and ZMX60305 alloys have not released any toxic ions which could affect the cells involved in the osseointegration. In contrast, it helps in controlling the degradation rate of magnesium, thereby induces a favorable environment for cells to grow. Hence, the present study shows that ZM603 alloys with different Ca additions can promote the cell growth for superior bone healing. Fig. 12 MTT assay on as-extruded Mg-6Zn-0.3Mn-xCa alloys 3.5 Antibacterial activity In order to investigate the antibacterial activity of the Mg-6Zn-0.3Mn-xCa alloys, bacteria were incubated for 24 h and the antibacterial activities were analyzed by the optical densities and spread plate method. The optical densities of the bacteria suspension-containing alloys after 8 h incubation were measured and the corresponding number of CFUs is shown in Fig. 13. In contrast, the number of CFUs for the ZM603 alloy exhibited a reduction of about 79.7% when compared with the control group. The inhibitory effects of all the materials on the bacteria are in such an order as ZM603< ZMX60302 Fig. 13 Optical density at 600 nm (OD600) of as- extruded Mg-6Zn-0.3Mn-xCa alloys Furthermore, in order to visually evaluate the antibacterial effect, each bacteria suspension- containing alloy after 8 h incubation is plated onto the Muller Hinton broth agar plates and incubated for 24 h. S. aureus colonies after incubation are shown in Fig. 14 and strong antibacterial activity of Mg-6Zn-0.3Mn-xCa alloys is observed, which is consistent with the optical density result. The fewer the colony, the better the antibacterial efficiency. The results of antibacterial assays confirm that Mg-6Zn-0.3Mn-xCa alloys have a strong antibacterial activity and the antibacterial activity increases with the increasing Ca content. ROBINSON et al [43] reported that Mg shows similar effects on the bactericidal fluoroquinolone antibiotic (enfloxacin) against CFUs of both gram-negative and gram-positive bacteria. Their results also indicate that the Mg corrosion products [Mg(OH)2] would inhibit the growth of E. coli, P. aeruginosa and S. aureus. In addition, Zn possessing an excellent antibacterial ability has also been confirmed [44,45]. It has been reported that Zn can reduce the bacterial adhesion and growth by generating reactive oxygen species (ROS) [46], and Zn ions can restrain the bacteria growth by inhibiting multiple activities of bacteria [47]. Therefore, the released Zn ions from Mg-6Zn- 0.3Mn-xCa alloys degradation may contribute to the enhanced antibacterial ability. Furthermore, the antibacterial activity of Ca has also been confirmed. EL-RAB et al [48] found that the Ca-P apatite coat exhibited a strong antibacterial activity. Another study also indicates that Mg-Ca-Sr-Zn alloy also has antibacterial properties [49]. Fig. 14 Images of viable bacteria grown on different samples after 24 h of culture 4 Conclusions (1) The as-extruded ZM603 alloy mainly consists of ��-Mg and MgZn2 phases. While the alloys with the Ca addition of 0.2-0.5 wt.% consist of ��-Mg, MgZn2 and Ca2Mg6Zn3 phases. Minor Ca addition can slightly refine the grains of the as-extruded ZM603 alloy. (2) The addition of Ca can improve the mechanical properties of the as-extruded ZM603 alloy. The as-extruded alloy with 0.5 wt.% Ca exhibits better combined mechanical properties with the ultimate tensile strength and the elongation of 334 MPa and 20.3%, respectively. (3) The corrosion resistance of ZM603 alloy deteriorates after the addition of Ca due to the increase of amount of effective cathodic sites caused by the formation of Ca2Mg6Zn3. The cytotoxicity evaluation with L929 cells shows a higher cell viability of the ZMX60305 alloy compared with ZM603 and ZMX60302 alloys. The antibacterial activity result of Mg-6Zn-0.3Mn-xCa alloys against Staphylococcus aureus presents an enhanced antibacterial activity with increasing the Ca content. Acknowledgments The authors are grateful for the financial supports from the Natural Science Foundation of Shanxi Province, China (201901D211310), the National Natural Science Foundation of China (52071227), the Transformation of Scientific and Technological Achievements Programs of Higher Education Institutions in Shanxi, China (TSTAP), Science and Technology Major Projects of Shanxi Province, China (20191102004, 201903D111008) and the Central Special Funds Guiding the Development of Local Science and Technology, China (YDZX20181400002967). References [1] WITTE F. The history of biodegradable magnesium implants: A review [J]. Acta Biomaterialia, 2010, 6: 1680-1692. [2] WANG Shuo, ZHANG Xue-qi, LI Jin-gan, LIU Chang-sheng, GUAN Shao-kang. Investigation of Mg-Zn-Y-Nd alloy for potential application of biodegradable esophageal stent material [J]. Bioactive Materials, 2020, 5: 1-8. [3] AGARWAL S, CURTIN J, DUFFY B, JAISWAL S. Biodegradable magnesium alloys for orthopaedic applications: A review on corrosion, biocompatibility and surface modifications [J]. Materials Science and Engineering C, 2016, 68: 948-963. [4] MUNIR K, LIN Ji-xing, WEN Cui-e, WRIGHT P F A, LI Yun-cang. Mechanical, corrosion, and biocompatibility properties of Mg-Zr-Sr-Sc alloys for biodegradable implant applications [J]. Acta Biomaterialia, 2020, 102: 493-507. [5] CHENG Yuan-fen, DU Wen-bo, LIU Ke, FU Jun-jian, WANG Zhao-hui, LI Shu-bo, FU Jin-long. Mechanical properties and corrosion behaviors of Mg-4Zn-0.2Mn- 0.2Ca alloy after long term in vitro degradation [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 363-372. [6] LIN Yu-lin, LIU Jia-ning, BIAN Dong, HUANG Zhi-qiang, LIN Ze-feng, WANG Ming, CHU Xiao, LI Mei, ZHANG Yu, ZHENG Yu-feng. In vivo studies on Mg-1Sc alloy for orthopedic application: A 5-months evaluation in rabbits [J]. Materials Letters, 2020, 262: 127130-127147. [7] SHEN Zhen-quan, ZHAO Ming, ZHOU Xiao-chen, YANG Hong-tao, LIU Jia-ning, GUO Hui, ZHENG Yu-feng, YANG Jian-an. A numerical corrosion-fatigue model for biodegradable Mg alloy stents [J]. Acta Biomaterialia, 2019, 97: 671-680. [8] ZHAO Hong-liang, HUA Yun-xiao, DONG Xiang-lei, XING Hui, LU Yan-li. Influence of trace Ca addition on texture and stretch formability of AM50 magnesium alloy sheet [J]. Transactions of Nonferrous Metals Society of China, 2020, 30: 647-656. [9] LIU Yan-hui, CHENG Wei-li, ZHANG Yao, NIU Xiao-feng, WANG Hong-xia, WANG Li-fei. Microstructure, tensile properties, and corrosion resistance of extruded Mg-1Bi- 1Zn alloy: The influence of minor Ca addition [J]. Journal of Alloys and Compounds, 2020, 815: 152414-152427. [10] QIN Hui, ZHAO Yao-chao, AN Zhi-quan, CHENG Meng-qi, WANG Qi, CHENG Tao, WANG Qiao-jie, WANG Jia-xing, JIANG Yao, ZHANG Xian-long, YUAN Guang-yin. Enhanced antibacterial properties, biocompatibility, and corrosion resistance of degradable Mg-Nd-Zn-Zr alloy [J]. Biomaterials, 2015, 53: 211-220. [11] PEREZ P, ONOFRE E, CABEZA S, LLORENTE I, VALLE J A D, GARCIA-ALONSO M C, ADEVA P, ESCUDERO M L. Corrosion behaviour of Mg-Zn-Y-mischmetal alloys in phosphate buffer saline solution [J]. Corrosion Science, 2013, 69: 226-235. [12] DING Zhi-bing, ZHAO Yu-hong, LU Ruo-peng, YUAN Mei-ni, WANG Zhi-jun, LI Hui-jun, HOU Hua. Effect of Zn addition on microstructure and mechanical properties of cast Mg-Gd-Y-Zr alloys [J]. Transactions of Nonferrous Metals Society of China, 2019, 29: 722-734. [13] LIU De-xue, GUO Cheng-gong, CHAI Li-qiang, SHERMAN V R, QIN Xiao-qiong, DING Yu-tian, MEYERS M A. Mechanical properties and corrosion resistance of hot extruded Mg-2.5Zn-1Ca alloy [J]. Materials Science and Engineering B, 2015, 195: 50-58. [14] HOFSTETTER J, MARTINELLI E, POGATSCHER S, SCHMUTA P, POVODEN-KARADENIZ E., WEINBERG A M, UGGOWITZER P J, LOFFLER J F. Influence of trace impurities on the in vitro and in vivo degradation of biodegradable Mg-5Zn-0.3Ca alloys [J]. Acta Biomaterialia, 2015, 23: 347-353. [15] CHO D H, LEE B W, JIN Y P, CHO K M, PARK I M. Effect of Mn addition on corrosion properties of biodegradable Mg-4Zn-0.5Ca-xMn alloys [J]. Journal of Alloys & Compounds, 2017, 695: 1166-1174. [16] HELSEN J A, BREME H J. Metals as biomaterials [M]. New York: Wiley, 1998. [17] ZHANG Shao-xiang, ZHANG Xiao-nong, ZHAO Chang-li, LI Jia-nan, SONG Yang, XIE Chao-ying, TAO Hai-rong, ZHANG Yan, HE Yai-hua, JIANG Yao, BIAN Yu-jun. Research on an Mg-Zn alloy as a degradable biomaterial [J]. Acta Biomaterialia, 2010, 6: 626-640. [18] XU Li-ping, YU Guo-ning, ZHANG Er-lin, PAN Feng. In vivo corrosion behavior of Mg-Mn-Zn alloy for bone implant application [J]. Journal of Biomedical Materials Research (Part A), 2010, 83: 703-711. [19] YANG Jiang, PENG Jian, NYBERG E A, PAN F S. Effect of Ca addition on the corrosion behavior of Mg-Al-Mn alloy [J]. Applied Surface Science, 2016, 369: 92-100. [20] SONG Xiao-zhe, CHANG Lei, WANG Jun, ZHU Shi-jie, WANG Li-guo, FENG Kun, LUO Ya-ge, GUAN Shao-kang. Investigation on the in vitro cytocompatibility of Mg-Zn-Y-Nd-Zr alloys as degradable orthopaedic implant materials [J]. Journal of Materials Science: Materials in Medicine, 2018, 29: 44-56. [21] ZHANG Er-lin, YANG Lei, XU Jian-wei, CHEN Hai-yan. Microstructure, mechanical properties and biocorrosion properties of Mg-Si(Ca, Zn) alloy for biomedical application [J]. Acta Biomater, 2010, 6: 1756-1762. [22] HARTWIG A. Role of magnesium in genomic stability [J]. Mutat Res/Fund Mol Meeh Mutagen, 2001, 475: 113-121. [23] ASCHNER M, GUILARTE T R, SCHNEIDER J S, ZHENG W. Manganese: Recent advances in understanding its transport and neurotoxicity [J]. Toxicology and Applied Pharmacology, 2007, 221: 131-147. [24] SUN Yu, KONG Min-xiu, JIAO Xiao-hui, In-vitro evaluation of Mg-4.0Zn-0.2Ca alloy for biomedical application [J]. Transactions of Nonferrous Metals Society of China, 2011, 21: s252-s257. [25] LI Hui, PENG Qiu-ming, LI Xue-jun, LI Kun, HAN Zeng-sheng, FANG Da-qing. Microstructures, mechanical and cytocompatibility of degradable Mg-Zn based orthopedic biomaterials [J]. Materials and Design, 2014, 58: 43-51. [26] CHENG Mei-xin, CHEN Ji-hua, YAN Hong-he, SU Bin, YU Zhao-hui, XIA Wei-jun, GONG Xiao-le. Effects of minor Sr addition on microstructure, mechanical and bio-corrosion properties of the Mg-5Zn based alloy system [J]. Journal of Alloys and Compounds, 2017, 691: 95-102. [27] CHEN Jun-xiu, ZHANG Yi, IBRAHIM M, ETIM I P, TAN Li-li, YANG Ke. In vitro degradation and antibacterial property of a copper-containing micro-arc oxidation coating on Mg-2Zn-1Gd-0.5Zr alloy [J]. Colloids and Surfaces B: Biointerfaces, 2019, 179: 77-86. [28] TAN Hong-lue, PENG Zhao-xiang, LI Qing-tian, XU Xiao-fen, GUO Sheng-rong, TANG Ting-ting. The use of quaternised chitosan-loaded PMMA to inhibit biofilm formation and downregulate the virulence-associated gene expression of antibiotic-resistant staphylococcus [J]. Biomaterials, 2012, 33: 365-377. [29] WU Tian-yi, HUA Xiao-lin, HE Zhi-wei, WNAG Xin-fu, YU Xiao-wei, REN Wei-ping. The bactericidal and biocompatible characteristics of reinforced calcium phosphate cements [J]. Biomedical Materials, 2012, 7: 045003-045013. [30] SHI De-ke. Fundamentals of materials science [M]. 2nd ed. Xi��an: China Machine Press, 2015. (in Chinese) [31] YAN Yang, CAO Han-wen, KANG Yi-jun, Yu-Kun, XIAO Tao, LUO Jie, DENG You-wen, FANG Hong-jie, XIONG Han-qing, DAI Yi-long. Effects of Zn concentration and heat treatment on the microstructure, mechanical properties and corrosion behavior of as-extruded Mg-Zn alloys produced by powder metallurgy [J]. Journal of Alloys and Compounds, 2017, 693: 1277-1289. [32] DU Yu-zhou, ZHENG Ming-yi, QIAO Xiao-guang, WANG De-bao, PENG Wen-qiang, WU Kun, JIANG Bai-ling. Improving microstructure and mechanical properties in Mg�C6 mass% Zn alloys by combined addition of Ca and Ce [J]. Materials Science & Engineering A, 2016, 656: 67-74. [33] LIU Bao-sheng, YANG Yuan-xi, ZHANG Yue-zhong, DU Hua-yun, HOU Li-feng, WEI Ying-hui. Investigation of rapidly decomposable AZ91-RE-xCu (x=0, 1, 2, 3, 4) alloys for petroleum fracturing balls [J]. Journal of Physics and Chemistry of Solids, 2020, 144: 109499-109510. [34] LEVI G, AVRAHAM S, ZILBEROV A, BAMBERGER M. Solidification, solution treatment and age hardening of a Mg-1.6wt.%Ca-3.2wt.%Zn alloy [J]. Acta Materialia, 2006, 54: 523-530. [35] DU Yu-zhou, QIAO Xiao-guang, ZHENG Ming-yi, WANG D B, WU K, GOLOVIN I S. Effect of microalloying with Ca on the microstructure and mechanical properties of Mg- 6mass%Zn alloys [J]. Materials & Design, 2016, 98: 285-293. [36] SCATTERGOOD R O, BACON D J. The Orowan mechanism in anisotropic crystals [J]. Philosophical Magazine, 1975, 31: 179-198. [37] OSKUI A E, SOLTANI N. Characterization of elastic-plastic fracture toughness of AM60 Mg alloy under mixed-mode loading conditions [J]. Engineering Fracture Mechanics, 2018, 204: 388-403. [38] WEN Qi, DENG Kun-kun, SHI Ju-yan, ZHANG Bao-ping, LIANG Wei. Effect of Ca addition on the microstructure and tensile properties of Mg-4.0Zn-2.0Gd alloys [J]. Materials Science & Engineering A, 2014, 609: 1-6. [39] SANSONE V, PAGANI D, MELATO M. The effects on bone cells of metal ions released from orthopaedic implants. A review [J]. Clin Cases Mineral Bone Metabolism, 2013, 10: 34-40. [40] CUI Lan-yue, WEI Guang-bin, HAN Zhuang-zhuang, ZENG Rong-chang, WANG Lei, ZOU Yu-hong, LI Shuo-qi, XU Dao-kui,GUAN Shao-kang. In vitro corrosion resistance and antibacterial performance of novel tin dioxide-doped calcium phosphate coating on degradable Mg-1Li-1Ca alloy [J]. Journal of Materials Science & Technology, 2019, 35: 254-265. [41] BEN-HAMU G, ELIEZER D, SHIN K S. The role of MgSi on the corrosion behavior of wrought Mg-Zn-Mn alloy [J]. Intermetallics, 2008, 16: 860-867. [42] SHAHIN M, MUNIR K, WEN Cui-e, LI Yun-cang. Magnesium matrix nanocomposites for orthopedic applications: A review from mechanical, corrosion, and biological perspectives [J]. Acta Biomaterialia, 2019, 96: 1-19. [43] ROBINSON D A, GRIFFITH R W, DAN S, EVANS R B, CONZEMIUS M G. In vitro antibacterial properties of magnesium metal against Escherichia coli, Pseudomonas aeruginosa and Staphylococcus aureus [J]. Acta Biomaterialia, 2010, 6: 1869-1877. [44] HOU Kai-fu, ZHANG Xu-ming, WANG Hai-rong, ZHAO Ling-zhou, LIU Xuan-yong, CHU P K. Osteogenic activity and antibacterial effects on titanium surfaces modified with Zn-incorporated nanotube arrays [J]. Biomaterials, 2013, 34: 3467-3478. [45] JIN Guo-dong, CAO Hui-liang, QIAO Yu-qin, MENG Fan-hao, ZHU Hong-qin, LIU Xuan-yong. Osteogenic activity and antibacterial effect of zinc ion implanted titanium [J]. Colloids & Surfaces B: Biointerfaces, 2014, 117: 158-165. [46] HU H, ZHANG W, QIAO Yu-qin, JIANG Xiao-yue, LIU X, DING C. Antibacterial activity and increased bone marrow stem cell functions of Zn-incorporated TiO2 coatings on titanium [J]. Acta Biomaterialia, 2012, 8: 904-915. [47] PHAN T N, BUCKNER T, SHENG J, BALDECK J D, MARQUIS R E. Physiologic actions of zinc related to inhibition of acid and alkali production by oral streptococci in suspensions and biofilms [J]. Molecular Oral Microbiology, 2010, 19: 31-38. [48] EL-RAB S M F G, FADL-ALLAH S A, MONTSER A A. Improvement in antibacterial properties of Ti by electrodeposition of biomimetic Ca-P apatite coat on anodized titania [J]. Applied Surface Science, 2012, 261: 1-7. [49] HE Guan-ping, WU Yuan-hao, ZHANG Yu, ZHU Ye, LIU Yang, LI Nan, LI Mei, ZHENG Guan, HE Bao-hua, YIN Qing-shui, ZHENG Yu-feng, MAO Chuan-bin. Addition of Zn to the ternary Mg-Ca-Sr alloys significantly improves their antibacterial property [J]. Journal of Materials Chemistry B, 2015, 3: 6676-6689. ����ʤ1��������1����Ծ��2���� ��1������ΰ1��������3����Ӣ��1,3 1. ̫ԭ�Ƽ���ѧ ���Ͽ�ѧ�빤��ѧԺ��̫ԭ 030024�� 2. ̫ԭ�Ƽ���ѧ ��ѧ�����﹤��ѧԺ��̫ԭ 030024�� 3. ̫ԭ������ѧ ���Ͽ�ѧ�빤��ѧԺ��̫ԭ 030024 ժ Ҫ��Ϊ��Ѱ�Һ���ֲ��Ӧ�õĿ����オ����ϣ����ð�����������ȼ�ѹ�����Ʊ�Mg-6Zn-0.3Mn-xCa(x=0��0.2��0.5����������)�Ͻ��ý���������ɨ��������������������о�Mg-6Zn-0.3Mn-xCa�Ͻ������֯����ѧ���ܡ������������CaԪ�صļ������ϸ����ѹ̬Mg-6Zn-0.3Mn�Ͻ�ľ����������ǿ�ȡ���Ca�������ֱ�Ϊ0.2%��0.5%(��������)ʱ���Ͻ�ľ����ߴ���4.8 ��m�ֱ�ϸ����4.6��4.2 ��m�����ֺϽ��У�����0.5%Ca�ĺϽ���и��õ��ۺ���ѧ���ܣ���������ǿ�Ⱥ��쳤�ʷֱ�Ϊ334 MPa��20.3%�����⣬�������о��Ͻ�ĸ�ʴ��Ϊ��ϸ�����ԺͿ������ܽ������ۡ�����Ca���������ӣ��Ͻ����ʴ�Խ��ͣ�����Ҫ������Ca2Mg6Zn3����γɵ�����Ч��������λ����������ӡ�L929ϸ����ϸ������������ʾ����Mg-6Zn-0.3Mn��Mg-6Zn-0.3Mn-0.2Ca�Ͻ���ȣ�Mg-6Zn-0.3Mn-0.5Ca�Ͻ���и��õ�ϸ�����ԡ����ڽ��ɫ�������������������ѧ���ԣ��ڹ��������У��Ͻ�Ŀ�����������Ca���������Ӷ���ǿ�� �ؼ��ʣ�þ�Ͻ����︯ʴ���������ԣ�����֯����ѧ���� (Edited by Xiang-qun LI) Corresponding author: Bao-sheng LIU; Tel/Fax: +86-351-6998256, E-mail: liubaosheng@tyust.edu.cn; Yue-zhong ZHANG, E-mail: yuezhongzhang@sina.cn DOI: 10.1016/S1003-6326(21)65501-2 1003-6326/

��Ԫ�ضԼ�ѹMg-Zn-Mn�Ͻ������֯����ʴ�ԡ����������Լ��������ܵ�Ӱ��
 2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press
2021 The Nonferrous Metals Society of China. Published by Elsevier Ltd & Science Press

