Trans. Nonferrous Met. Soc. China 26(2016) 2502-2508
Temperature-programmed pyrolysis of magnesium ammonium phosphate and removal of ammonia-nitrogen by its pyrolysate
Jian-jun TANG1, Yi-qing CHEN1, Zhen-hui ZHONG2, Wen-long LI1, Juan YIN1
1. Shenzhen Key Laboratory of Industrial Water Conservation & Municipal Wastewater Resources Technology, Shenzhen Polytechnic, Shenzhen 518055, China;
2. School of Civil Engineering, Guangzhou University, Guangzhou 510006, China
Received 11 November 2015; accepted 21 April 2016
Abstract: In order to achieve the dual goals of complete deamination of magnesium ammonium phosphate (MAP) and ensure the pyrolysate��s good removal properties towards ammonia-nitrogen, a temperature-programmed method for the pyrolysis of MAP was studied, as well as the thermodynamic and kinetic processes involved in the removal reaction system between MAP pyrolysate and aqueous ammonium. It was found that the pyrolysis method and pyrolysis final temperature had significant effects on the MAP pyrolysate��s removal properties towards aqueous ammonium, and the following conditions were deemed to be more appropriate: pyrolysis final temperature and heating rate at 180 ��C and 5 ��C/min, respectively, and a pH level of 9.5 for the removal reaction system. The resultant ammonium removal capacity by the MAP pyrolysate was 95.62 mg/g. After 120 min, the removal rate with an initial concentration of 1000 mg/L was 82%. The kinetic and thermodynamic results indicated that the removal of aqueous ammonium by MAP pyrolysate was the exchange process between H+ and NH4+ via MAP precipitation. The kinetics complied with the Lagergren quasi second-order model with an equilibrium time of 120 min, while the isothermal curves complied with the Freundlich model.
Key words: magnesium ammonium phosphate; temperature-programmed heating; pyrolysate; ammonia-nitrogen
1 Introduction
The issue of eutrophication caused by ammonia- nitrogen pollution has become increasingly prominent, burdening environmental workers with the task of finding economical but effective ways to treat ammonia- nitrogen wastewater. The main treatment methods used currently include chemical precipitation [1-3], physical stripping [4,5], and biological method [6]. Among these, the first method is the most popular due to its various advantages, such as the simplicity of its process, being economical and effective, and high treatment efficiency. It has already been applied to actual engineering projects, with many examples available.
The biggest constraint affecting the practical application of the chemical precipitation method is the price of the precipitation agents (soluble phosphate and magnesium salts), which increases the processing costs. Various researchers sought ways to reduce processing costs during the actual application of this method. AHMAD and IDRIS [7] studied the regeneration of MgNH4PO4��6H2O (MAP) under acidic conditions: MgNH4PO4��6H2O(s)+H+ MgHPO4��3H2O+NH4++ 3H2O. HE et al [8] studied the regeneration of MAP under alkaline conditions: MgNH4PO4��6H2O(s)+NaOH��MgNaPO4(s)+NH3+7H2O and MgNaPO4(s)+ NH4+ + 6H2O �� MgNH4PO4��6H2O (s) + Na+, thereby recycling MAP and solving the cost issue of using precipitating agents. Since acidic regeneration involves the use of ammonium salts to recover ammonia-nitrogen in the wastewater, the recovered liquor inevitably contains small amounts of Mg2+ and
MgHPO4��3H2O+NH4++ 3H2O. HE et al [8] studied the regeneration of MAP under alkaline conditions: MgNH4PO4��6H2O(s)+NaOH��MgNaPO4(s)+NH3+7H2O and MgNaPO4(s)+ NH4+ + 6H2O �� MgNH4PO4��6H2O (s) + Na+, thereby recycling MAP and solving the cost issue of using precipitating agents. Since acidic regeneration involves the use of ammonium salts to recover ammonia-nitrogen in the wastewater, the recovered liquor inevitably contains small amounts of Mg2+ and  , making the discharging of the liquor problematic. On the other hand, although regeneration methods using alkaline conditions can recover ammonia-nitrogen in the form of aqueous ammonia, an equivalent amount of NaOH must be consumed, which is not cost effectively.
, making the discharging of the liquor problematic. On the other hand, although regeneration methods using alkaline conditions can recover ammonia-nitrogen in the form of aqueous ammonia, an equivalent amount of NaOH must be consumed, which is not cost effectively.
The study by BHUIYAN et al [9] showed that after pyrolysis, MAP became a mixture comprising MgHPO4, Mg3(PO4)2, and Mg2P2O7. SUGIYAMA and YOKOYAMA [10] found that MgHPO4, followed by Mg3(PO4)2, had good removal properties towards NH4+, but Mg2P2O7 showed no removal effect. RINAUDO et al [11] and ZHAO et al [12] indicated that at a suitable pH value, the exchange reaction between MgHPO4 and NH4+ would create MAP. Theoretical calculations further indicated that upon complete pyrolysis of MAP, aqueous ammonia at 13.6% could be obtained. Hence, the controlled pyrolysis of MAP under specific conditions could lead to the recovery of highly-concentrated aqueous ammonia. Furthermore, the pyrolysate could be used as an agent for the treatment of ammonia-nitrogen wastewater. The potential for recycling would also help to address the cost issue of using agents to treat ammonia-nitrogen wastewater.
During the preliminary work of this research [13], the pyrolytic behavior of MAP and the removal properties of its pyrolysate towards ammonia-nitrogen were studied. It was found that ammonia was released when MAP was pyrolyzed under specific conditions, and the pyrolysate could be recycled as an agent for the treatment of ammonia-nitrogen wastewater. However, the results also showed that when the pyrolysis temperature was low, the deamination of MAP was incomplete. Although complete deamination was achieved at high temperatures, the pyrolytic process of MAP produced Mg2P2O4, which had no removal effect towards ammonia-nitrogen. This resulted in the deterioration of removal properties of the pyrolysate. As such, we studied a temperature-programmed method for the pyrolysis of MAP, as well as the thermodynamic and kinetic processes involved in the removal reaction system between pyrolysate and aqueous ammonium, thereby achieving the dual goals of implementing the complete deamination of MAP and ensuring that the pyrolysate has good removal properties towards ammonia-nitrogen.
2 Experimental
2.1 Preparation of MAP
NH4Cl(AR) was used to prepare 1000 mg/L simulated aqueous ammonium wastewater. Na2HPO4��12H2O(AR) and MgCl2��6H2O(AR), the precipitating agents, were then added before the MAP was prepared in a magnetic stirring device under constant temperature. The reaction conditions were as follows: the molar ratio of N:P:Mg based on the precipitation agents added was 1:1:1, pH value was 9.5, reaction temperature was room temperature (25 ��C), and reaction time was 40 min. After the reaction was completed, the MAP precipitate was filtered, rinsed, and dried in a drying oven at a constant temperature of 40 ��C for 48 h. The dried MAP precipitation was placed in a dryer for subsequent use.
2.2 Pyrolysis of MAP
An appropriate amount of MAP was placed inside a tubular resistance furnace and subjected to controlled pyrolysis through a temperature-programmed method. The heating rate was 2, 3, 5, 7, 10, or 20 ��C/min from room temperature (25 ��C) to the pyrolysis final temperature at 180-240 ��C. Unless otherwise specified, the heating rate was 5 ��C/min, final pyrolysis temperature was 180 ��C, and the holding time at the final pyrolysis temperature was 3 h. The pyrolysate was sealed and stored for subsequent use.
2.3 Acid dissolution experiment
0.1 g of solidified powders (MAP or its pyrolysate) were placed in 200 mL of deionized water. The pH value was adjusted with the addition of 2 mol/L hydrochloric acid solution until the solidified powder was completely dissolved. The concentrations of ammonia-nitrogen, total phosphorus, and magnesium were then analyzed, and the deamination rate of the pyrolysate was calculated.
2.4 Removal experiment
200 mL (1000 mg/L) of aqueous ammonium was prepared inside a conical flask before NaOH and HCl solutions were added to adjust it to a particular pH. Unless otherwise stated, the pH value was controlled at 9.5. Next, 2 g of MAP pyrolysate was added before stirring was done in a constant-temperature magnetic chamber, with the temperature controlled at room temperature (25 ��C). During the removal process, tests were done at regular intervals to maintain the reaction system at a constant pH level. Samples were extracted for analysis of ammonia-nitrogen concentration, as well as calculation of the removal capacity of the pyrolysate and the removal rate. Unless otherwise specified, the reaction duration was 120 min.
2.5 Analysis and characterization
Analysis of aqueous ammonium and total phosphorus concentrations was made using the spectrophotometry method. Analysis of Mg2+ concentration was done using flame atomic absorption spectrometry. The STA-449-F3 thermal analyzer was used for thermogravimetric and differential thermal analyses, changes in the heating rate were controlled at 2, 3, 5, 7, 10, or 20 ��C/min, with the final temperature being 350 ��C and air being the reaction atmosphere (20 mL/min). The S-4800 scanning electron microscope (SEM) was used to analyze the microscopic crystal structures. For X-ray diffraction (XRD) analysis, continuous scans were done at room temperature with a Cu K source and at 40 kV and 40 mA.
3 Results and discussion
3.1 Characterization of materials
The SEM images of MAP before and after pyrolysis are shown in Fig. 1. MAP before pyrolysis was strip-type with originally rhomboid crystals of uniform size, its length was 30-50 ��m. After pyrolysis, there were significant changes to its form: particle size became smaller, with a length of 10-20 ��m, its degree of crystallinity was greatly reduced.
The XRD patterns of MAP, MAP��s pyrolysate and the pyrolysate after reaction with ammonia-nitrogen are shown in Fig. 2. MAP is a crystalline material of high purity, with its XRD pattern mainly exhibiting the characteristic diffraction peak of MgNH4PO4��6H2O. In contrast, the crystallinity of the MAP pyrolysate was poor, with amorphous structure and no characteristic diffraction peak of MgNH4PO4��6H2O. However, the characteristic diffraction peak of MgNH4PO4��6H2O reappeared in its XRD pattern after reaction with ammonia-nitrogen.

Fig. 1 SEM images of MAP before (a) and after (b) pyrolysis
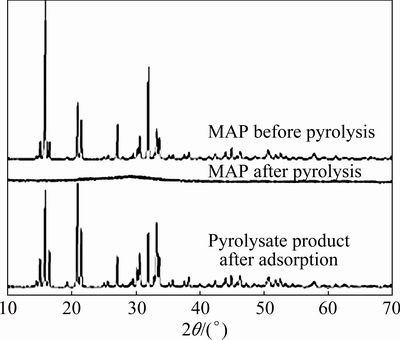
Fig. 2 XRD patterns of MAP and its pyrolysis product
3.2 Effect of pyrolysis process
The pyrolysis of MAP follows by this equation: MgNH4PO4��6H2O �� MgHPO4 + 6H2O�� + NH3��. There was a mass loss of 51.03% after complete pyrolysis, indicating that mass loss of MAP after complete deamination was approximately 50%. The results of the TG�CDSC curves (Fig. 3) indicate that when the heating rates were controlled at 2, 3, 5, 7, 10, and 20 ��C/min, the corresponding pyrolysis temperatures at end point with mass loss of approximately 50% were 155, 180, 200, 215, 235, and 255 ��C, respectively. This indicates that the temperature-programmed method has an impact on the pyrolysis of MAP. Specifically, a rapidly-rising heating rate would limit the deamination and dehydration of MAP. When the rate reached 10 ��C/min or above, with a mass loss of 50%, the pyrolysis temperature exceeded 200 ��C.
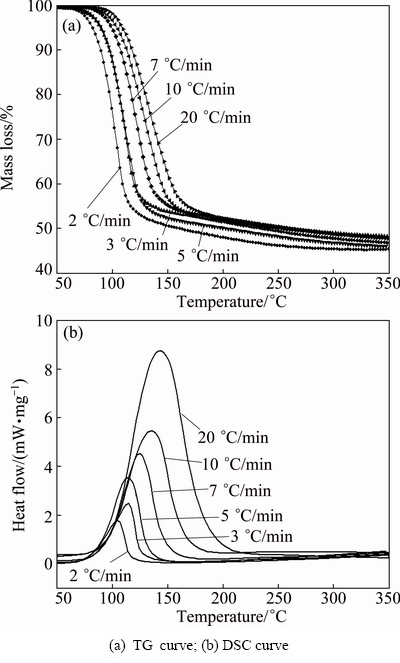
Fig. 3 TG�CDSC curves with different heating rates
The experimental results in Fig. 4 further show that the heating rate affected not only the pyrolysis process of MAP, but also the removal properties of its pyrolysate. When the rate was increased from 2 to 5 ��C/min and the mass loss was 50%, the final pyrolysis temperature increased from 155 to 200 ��C. However, the aqueous ammonium removal capacity of the pyrolysate increased, reaching an amount greater than 95 mg/g. When the heating rate was increased to above 5 ��C/min, not only the pyrolysis final temperature increased, but the removal capacity of the pyrolysate deteriorated. Combining the experimental results in Figs. 3 and 4, controlling the temperature-programmed heating rate of MAP at 5 ��C/min was more appropriate.
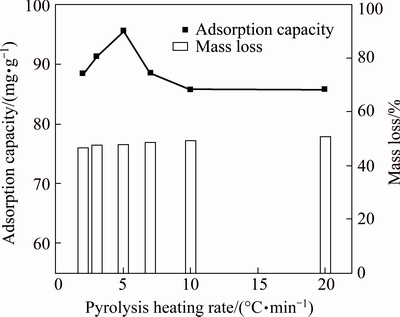
Fig. 4 Effect of pyrolysis heating rates on adsorption capacity and mass loss
The experimental results in Fig. 5 show that when the heating rate for temperature-programmed pyrolysis was controlled at 5 ��C/min, the deamination rate increased with increase in the final pyrolysis temperature. When the latter temperature was 180 ��C, the ammonia-nitrogen removal capacity of the pyrolysate reached 95 mg/g. Further increasing the final pyrolysis temperature would cause the capacity to decline. The reason was that although there would be complete deamination during MAP pyrolysis at high temperatures, the pyrolysate would begin to generate by-products, such as Mg2P2O7 and Mg3(PO4)2, with poor ammonia-nitrogen removal effects.
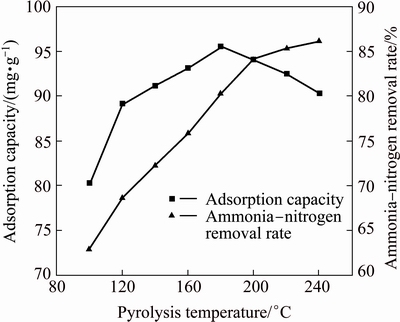
Fig. 5 Effect of final pyrolysis temperatures on adsorption capacity and ammonia-nitrogen removal rate
3.3 Removal mechanism
To better understand the mechanism by which the MAP pyrolysate removes aqueous ammonium, the initial pH value of the reaction system was controlled at 9.5. During the experiment, a fixed amount of MAP pyrolysate (2 g) was added. Changes in pH value of the reaction system and ammonia-nitrogen concentration were then measured at various reaction time. Separately, the amount of pyrolysate added was also varied, pH value and ammonia-nitrogen concentration were measured after reaction. The results are shown in Fig. 6.
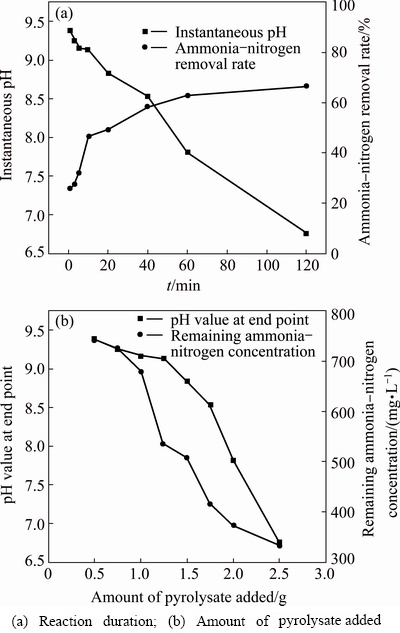
Fig. 6 Mechanism of ammonia-nitrogen adsorption by MAP pyrolysis product
It can be seen that the removal rate of ammonia-nitrogen increased with increase in reaction time, although pH value of the reaction system decreased continuously. This trend became even more apparent with increase in the amount of pyrolysate added. This illustrates that the removal mechanism of aqueous ammonium by MAP pyrolysate was through an exchange reaction between NH4+ and H+ via MAP precipitation: MgHPO4 + NH4+ + 6H2O �� MgNH4PO4��6H2O + H+.
Figure 6 shows that pH value was one of the main factors affecting the removal of ammonia-nitrogen by MAP pyrolysate. Hence, the impact of pH value on ammonia-nitrogen removal rate was further studied, as shown in Fig. 7. When pH value was initially low and then gradually increased to 9.5, the removal capacity of MAP pyrolysate increased, as did the ammonia-nitrogen removal rate of the reaction system. However, further increase of pH value resulted in sharp decline in both removal capacity and removal rate.
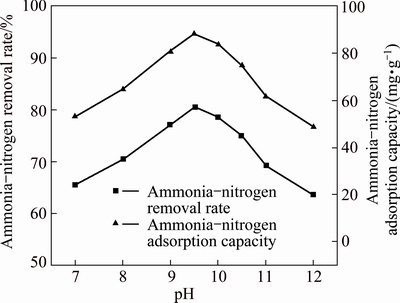
Fig. 7 Effect of pH value on removal capacity of pyrolysis product
Since the removal mechanism of aqueous ammonium by MAP pyrolysate was the exchange reaction between  and H+, this exchange reaction was an equimolar reaction. When
and H+, this exchange reaction was an equimolar reaction. When  combined with the pyrolysate to generate MgNH4PO4��6H2O, an equimolar of H+ would enter into the solution. When the pH value was low, the generated MgNH4PO4��6H2O would dissolve and in turn enter the solution as ions, causing a removal decline. When pH value was high, the pyrolysate could be converted to Mg(OH)2 or Mg3(PO4)2, leading to a deterioration in removal effect. From the experimental results shown in Fig. 7, controlling pH value of the reaction system at 9.5 was more appropriate.
combined with the pyrolysate to generate MgNH4PO4��6H2O, an equimolar of H+ would enter into the solution. When the pH value was low, the generated MgNH4PO4��6H2O would dissolve and in turn enter the solution as ions, causing a removal decline. When pH value was high, the pyrolysate could be converted to Mg(OH)2 or Mg3(PO4)2, leading to a deterioration in removal effect. From the experimental results shown in Fig. 7, controlling pH value of the reaction system at 9.5 was more appropriate.
3.4 Kinetics and thermodynamics of removal
Figure 8 shows the removal capacity of MAP pyrolysate at 15, 25, and 35 ��C. When the removal duration was 0-50 min, the removal capacity of ammonia-nitrogen increased rapidly with increase in removal duration. When the removal duration reached 120 min and more, removal capacity tended towards equilibrium. At this stage, the reaction point tended towards saturation. The removal equilibrium time was 120 min.
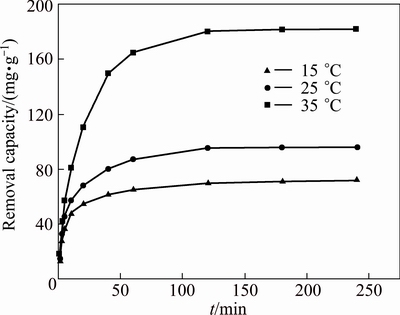
Fig. 8 Effect of duration on ammonia-nitrogen removal by MAP pyrolysate
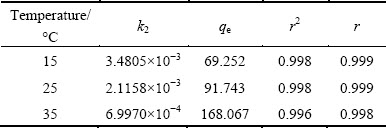
The kinetic model of ammonia-nitrogen removal by MAP pyrolysate is shown in Table 1, and the results indicate that the model complies with the Lagergren quasi second-order model as shown in the following equation:
 (1)
(1)
where qt represents the capacity at duration t; qe represents the equilibrium capacity; and k2 is the quasi second-order reaction rate constant.
Table 1 Parameters for quasi second-order kinetic model
The reaction isotherms of MAP pyrolysate at 15, 25, and 35 ��C and an removal duration of 120 min are shown in Fig. 9. When the equilibrium concentration was less than 300 mg/L, the equilibrium removal capacity increased sharply with increase in equilibrium concentration. However, further increase in equilibrium concentration did not result in any obvious trend for increase in equilibrium removal capacity. The reason was that the removal capacity of MAP pyrolysate did not reach saturation when the equilibrium concentration was low, and when the equilibrium concentration became higher, the reaction point would tend towards saturation, and so further increase of the equilibrium concentration would have no impact on the removal capacity.
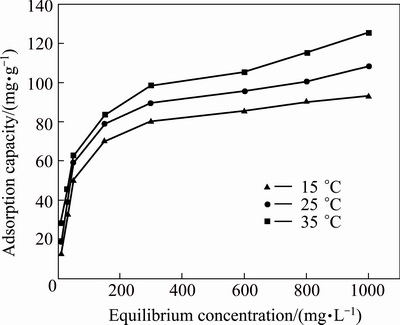
Fig. 9 Effect of equilibrium concentrations on ammonia- nitrogen removal by MAP pyrolysate
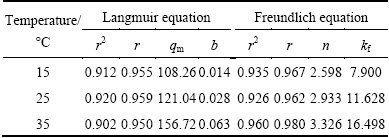
Based on the experimental results in Fig. 9, the Langmuir model and Freundlich model qe=kfC1/n were used to analyze the isothermal reaction between MAP pyrolysate and aqueous ammonium, where qm is the removal amount at saturation, qe is the equilibrium removal amount; Ce is the equilibrium concentration of ammonia-nitrogen; b, kf, and n are parameters of the equation.
and Freundlich model qe=kfC1/n were used to analyze the isothermal reaction between MAP pyrolysate and aqueous ammonium, where qm is the removal amount at saturation, qe is the equilibrium removal amount; Ce is the equilibrium concentration of ammonia-nitrogen; b, kf, and n are parameters of the equation.
The analysis results are shown in Table 2, which indicate that the reaction isotherms between MAP pyrolysate and ammonia-nitrogen were closer to the Freundlich model. The reason was that the material for aqueous ammonium removal in this study was prepared using heating method, resulting in it having an amorphous micro-structure and poor uniformity. Consequently, it was closer to the Freundlich model, which was derived from multilayer adsorption, with the assumption that the adsorbent has an uneven surface [14].
Table 2 Fitting parameters for Langmuir and Freundlich equations
4 Conclusions
1) A temperature-programmed heating method was used for direct pyrolysis of MAP to prepare an amorphous material for the removal of aqueous ammonium. During actual operation of this method, controlling the final pyrolysis temperature, heating rate, and holding time at the final pyrolysis temperature at 180 ��C, 5 ��C/min and 3 h, respectively, was appropriate.
2) The specific surface area of the MAP pyrolysate was 201.2 m2/g, while the total pore volume was 0.2798 cm3/g. Its equilibrium removal capacity reached 95.62 mg/g. The basic removal mechanism was an exchange reaction between NH4+ and H+ via MAP precipitation: MgHPO4 + NH4+ + 6H2O �� MgNH4PO4�� 6H2O+H+.
3) The MAP pyrolysate has poor uniformity, with an equilibrium time of 120 min, which complied with the Lagergren quasi second-order model. Isothermal removal reaction complied with the Freundlich model.
References
[1] RAHMAN M M, SALLEH M A M, RASHID U. Production of slow release crystal fertilizer from wastewaters through struvite crystallization��A review [J]. Arabian Journal of Chemistry, 2014, 7(1): 139-155.
[2] ZHONG Zhen-hui, CHEN Yi-qing, RONG Hong-wei, TANG Jian-jun, YIN Juan. Study of map precipitation improvement for ammoniacal nitrogen wastewater treatment [J]. Technology of Water Treatment, 2014, 40(10): 53-57. (in Chinese)
[3] PRABHU M, MUTNURI S. Cow urine as a potential source for struvite production [J]. International Journal of Recycling of Organic Waste in Agriculture, 2014, 3(1): 1-12.
[4] LIU Hua, LI Jing, SUN Li-na, CHEN Xi-jian. Treatment of high concentration ammonia nitrogen wastewater by distillation and stripping [J]. CHina Water & Wastewater, 2013, 29(20): 96-99. (in Chinese)
[5] JIN Ya-wen, LIU Zhen-hong, XUE Gang, GAO Pin, LI Shan-shan. Study on the pretreatment of dyeing wastewater with high- concentration ammonia by air-stripping [J]. Environment Science and Technology, 2013(S1): 147-149. (in Chinese)
[6] IACONI C D, ROSSETTI S, LOPEZ A. Effective treatment of stabilized municipal landfill leachates [J]. Chemical Engineering Journal, 2011, 168(3):1085-1092.
[7] AHMAD A A, IDRIS A. Release and recovery of phosphorus from wastewater treatment sludge via struvite precipitation [J]. Desalination and Water Treatment, 2014, 52(28-30): 1-8.
[8] HE S L, ZHANG Y, YANG M. Repeated use of MAP decomposition residues for the removal of high ammonium concentration from landfill leachate [J]. Chemosphere, 2007, 66(11): 2233-2238.
[9] BHUIYAN M I H, MAVINIC D S, KOCH F A. Thermal decomposition of struvite and its phase transition [J]. Chemosphere, 2008, 70(8): 1347-1356.
[10] SUGIYAMA S, YOKOYAMA M. Removal of aqueous ammonium with magnesium phosphates obtained from ammonium-elimination of magnesium ammonium phosphate [J]. Journal of colloid and interface science, 2005, 292(1): 133-138.
[11] RINAUDO C, ABBONA F, BOISTELLE R. Heterogeneous nucleation and epitaxy of newberyite and yuric acid crystals [J]. Journal of Crystal Growth, 1984, 66(3): 607-615.
[12] ZHAO Ting, ZHOU Kang-gen, WANG Hao, LIU Rong-yi. Study on optimal conditions of absorbing ammonia-nitrogen in wastewater with MHP [J]. Technology of water treatment, 2008, 34(4): 69-72. (in Chinese)
[13] CHEN Yi-qing, TANG Jian-jun, LI Wen-long, ZHONG Zhen-hui, YIN Juan. Thermal decomposition of magnesium phosphate and adsorption properties of its pyrolysis products toward ammonia nitrogen [J]. Transactions of Nonferrous Metals Society of China, 2015, 25(2): 497-503.
[14] LI Xu-qiang, ZHU Chun-shui, XU Guo-xiang, HAN Xi. Removal of Pb(II) with amorphous titanium (IV) hydrogen phosphate loaded on SiO2 [J]. Chinese Journal of Environmental Engineering, 2014, 8(4): 1510-1514. (in Chinese).
���������Ƚ����ᰱþ���Ƚ����İ����ѳ�����
�ƽ���1��������1�������2��������1���� ��1
1. ����ְҵ����ѧԺ �����й�ҵ��ˮ�������ˮ��Դ�������ص�ʵ���ң����� 518055��
2. ���ݴ�ѧ ��ľ����ѧԺ������ 510006
ժ Ҫ��Ϊʹ���ᰱþ(MAP)�Ѱ���ȫ�������Ƚ����İ���ȥ�����ܣ��о���һ�ֳ������µ��Ƚⷽʽ����̽�����Ƚ����ȥ������������ѧ������ѧ���̡�����������Ƚⷽʽ���Ƚ��յ��¶�����Ӱ��MAP�Ƚ����İ���ȥ�����ܣ������Ƚ��յ��¶�Ϊ180 ��C��5 ��C/min�ij������·�ʽ���Լ�������Ӧ��ϵ��pHֵΪ9.5��Ϊ���ʣ��ڴ������£�MAP�Ƚ���������ȥ������95.62 mg/g����120 min����ʼŨ��Ϊ1000 mg/L������ȥ���ʴ�82%������ѧ������ѧ���������MAP�Ƚ����ȥ��������H+�� �����ӽ������̣��䶯��ѧ����Lagergren����ģ�ͣ�ƽ��ʱ��Ϊ120 min���������߷���Freundlichģ�͡�
�����ӽ������̣��䶯��ѧ����Lagergren����ģ�ͣ�ƽ��ʱ��Ϊ120 min���������߷���Freundlichģ�͡�
�ؼ��ʣ����ᰱþ���������£��Ƚ�������
(Edited by Xiang-qun LI)
Foundation item: Project (ZDSY20120619093952884) supported by Shenzhen Strategic New Industry Development, China
Corresponding author: Jian-jun TANG; Tel: +86-755-26731157; E-mail: tangjj7384@szpt.edu.cn
DOI: 10.1016/S1003-6326(16)64372-8