Electrical and mechanical properties of Cu-Cr-Zr alloy aged under imposed direct continuous current
АґФґЖЪїЇЈєЦР№ъУРЙ«ЅрКфС§±Ё(УўОД°ж)2012ДкµЪ5ЖЪ
ВЫОДЧчХЯЈєНхЦѕЗї ЦУФЖІЁ ИДПФѕэ Нфі¬ НхЅ ХЕФц№в ИОО¬Ац ИОЦТГщ
ОДХВТіВлЈє1106 - 1111
№ШјьґКЈєCu-Cr-ZrєПЅрЈ»µјµзРФЈ»УІ¶ИЈ»µзБчК±Р§Ј»Оціц
Key wordsЈєCu-Cr-Zr alloy; electrical conductivity; hardness; current ageing; precipitation
ХЄ ТЄЈє
ФЪІ»Н¬ОВ¶ИєНµзБчГЬ¶ИПВ¶ФCu-Cr-ZrєПЅрЅшРРІ»Н¬К±јдµДК±Р§ґ¦АнЈ¬·ўПЦФЪєПККµДК±Р§ОВ¶ИПВК©јУµзБчЈ¬їЙТФН¬К±МбёЯєПЅрК±Р§єуµДµзµјВКєНУІ¶ИЎЈЛжЧЕµзБчГЬ¶ИµДФцјУЈ¬µзµјВКіКПЯРФФці¤Ј¬µ«УІ¶И±ЈіЦІ»±дЎЈПФОўЧйЦЇ№ЫІм±нГчЈ¬К©јУµзБчК±Р§єуЈ¬ЧйЦЇЦРіцПЦБЛґуБїµДО»ґнєНДЙГЧј¶ёхОціцОпЈ¬ХвКЗМбёЯµзµјВКєНУІ¶ИµДФТтЎЈµзБчЧчУГ»ъАнУлЅ№¶ъИИЎўґуБїµзЧУµДЗЁТЖј°ТтµзЧУ·зБ¦¶шМбёЯµДИЬЦКФЧУЎўїХО»єНО»ґнµДА©ЙўДЬБ¦УР№ШЎЈ
Abstract:
The Cu-Cr-Zr alloys were aged at different temperatures for different time with different current densities. The results show that both the electrical conductivity and hardness are greatly improved after being aged with current at a proper temperature. The electrical conductivity increases approximately linearly with increasing current density while the hardness remains constant. The microstructure observation reveals that a much higher density of dislocations and nanosized Cr precipitates appear after the imposition of current, which contributes to the higher electrical conductivity and hardness. The mechanism is related with three factors: 1) Joule heating due to the current, 2) migration of mass electrons, 3) solute atoms, vacancies, and dislocations promoted by electron wind force.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 1106-1111
WANG Zhi-qiang, ZHONG Yun-bo, RAO Xian-jun, WANG Chao, WANG Jiang,
ZHANG Zeng-guang, REN Wei-li, REN Zhong-ming
Department of Materials Engineering, Shanghai University, Shanghai 200072, China
Received 27 May 2011; accepted 11 November 2011
Abstract: The Cu-Cr-Zr alloys were aged at different temperatures for different time with different current densities. The results show that both the electrical conductivity and hardness are greatly improved after being aged with current at a proper temperature. The electrical conductivity increases approximately linearly with increasing current density while the hardness remains constant. The microstructure observation reveals that a much higher density of dislocations and nanosized Cr precipitates appear after the imposition of current, which contributes to the higher electrical conductivity and hardness. The mechanism is related with three factors: 1) Joule heating due to the current, 2) migration of mass electrons, 3) solute atoms, vacancies, and dislocations promoted by electron wind force.
Key words: Cu-Cr-Zr alloy; electrical conductivity; hardness; current ageing; precipitation
1 Introduction
Copper-chromium-zirconium (Cu-Cr-Zr) alloy exhibits both excellent mechanical properties and high electrical conductivity. Hence, it is one of the best choices for the materials of electronic terminal and connector. After the solution treating and quenching of an age-hardenable alloy, high resistivity remains because of the scattering of the electrons by the large number of solute atoms trapped in matrix. However, upon ageing, one would expect the resistivity to drop as the solute atoms are removed from solid solution to form the precipitates that produce the age hardening [1]. The alloy obtains a significant component of strength by age hardening. The research of addition of Zr into Cu-Cr alloy conducted by WATANABE et al [2] indicated that, the increase in strength was attributed to the decrease in inter-precipitate spacing of Cr precipitates and Cu5Zr phase. The stress relaxation resistance is improved by the nucleation and growth of Cu5Zr precipitates on dislocations, in addition to Cr precipitates.
In recent decades, the electric current has been proven to affect the solid state phase transformation in metals. It was firstly reported by KOPPENAAL and SIMCOE [3] that, a DC electric current density of about 103 A/cm2 enhanced the precipitation rate of the Al-4% Cu alloy. Because the ageing temperature is consistent, the variation in behavior observed with current during ageing is controlled principally, but not exclusively, by the current rather than by Joule heating effect. The free-electron model of conductivity of metals assumes that the conduction electrons are free to move in the metal, unconstrained by the perfect lattice of ions except for scattering interactions due to phonon vibration. The scattering does not enhance the displacement of the ion and it has no net effect on the diffusion of the ion when the electric current density is low. However, the scattering by a high current density, above 104 A/cm2, enhances the atomic displacement in the direction of electron flow. The enhanced atomic displacement and the accumulated effect of mass transport under the influence of electric field (mainly, electric current) are called electromigration [4]. CONRAD [5] considered the influence of an electric current on precipitation in metals and suggested that an electric current can either enhance or retard the precipitation rate, depending on the alloy, the current density and its frequency. Important factors are the effect of current on the quenched-in vacancies and the presence of an internal stress. The greater effects can be explained by simple electromigration theory, which suggests the cooperative motion of a large number of atoms.
The previous work [6,7] showed the influence of temperature on the ageing of Cu-Cr-Zr alloy. In order to study the influence of current density, ageing treatment and time on the electrical and mechanical properties of Cu-Cr-Zr alloy, in the present work, the direct current (DC current, with various current densities) and ageing treatment (at various temperatures for various time) were applied to the alloy, the electrical and mechanical properties were studied, the results were correlated to TEM observations and finally the mechanism was discussed.
2 Experimental
The sample alloys were supplied by Jiangyin Chuangyi Metal Material, China. The chemical composition of the alloy was 0.69% Cr, 0.33% Zr, and Cu balance. The as-received alloy was solid solution treated and cold deformed, in the form of a cylindrical rod of 4 mm in diameter. The total area reduction was more than 98%. The ageing process is schematically illustrated in Fig. 1.
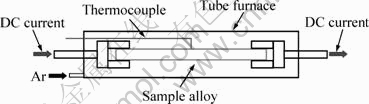
Fig. 1 Schematic illustration of ageing process
The rod was cut into 100 mm in length as a sample gripped by two steel heads to let through the current. Samples were aged in a tube furnace in the argon atmosphere and then quenched in water. A thermocouple was fitted to the surface of the sample for accurately measuring and controlling the temperature. The sample was heated to the expecting ageing temperature from room temperature in about 1 h. DC current was imposed from the beginning of heating.
In order to investigate the effect of DC current on the ageing treatment, the following three procedures were applied to the samples: 1) To investigate the influence of ageing time, the samples were aged at 400 Ўж for 1-7 h with 100 A/cm2 DC current; 2) To investigate the influence of ageing temperature, the samples were aged at 350-600Ўж for 1 h with 100 A/cm2 DC current; 3) To investigate the influence of current density, the samples were aged at 400-500 Ўж for 1 h with DC current up to 300 A/cm2.
The electrical conductivity was measured by four-probe resistivity measuring method using a Keithley 2182A nanovoltmeter and a 2400 digital sourcemeter at 20 Ўж, and presented by IACS (International annealed copper standard). Vickers hardness measurements were performed on the well-polished transverse cross-section using an Everone MH-3 microhardness tester under a load of 0.98 N for 5 s dwell time, and each hardness value was taken from the arithmetical mean of more than five indentations. Transmission electron microscopy (TEM) was performed using a JEM-2010F microscope with an acceleration voltage of 200 kV. Thin foils for TEM observations were prepared by mechanical thinning, twin-jet electropolishing in an aqueous solution of 30% HNO3 + 70% CH3OH at -40 Ўж.
3 Results and discussion
3.1 Electrical and mechanical properties
The electrical conductivity and Vickers hardness of alloy aged at 400 Ўж for 1-7 h with 100 A/cm2 current are shown in Fig. 2. In Fig. 2(a), due to more complete precipitation of solute atoms, the electrical conductivity increases with increasing ageing time. However, with imposition of current, a remarkable increase of 7% IACS is observed after 1 h, and the electrical conductivity reaches 76% IACS after 2 h aging, which is higher than that aged for 7 h without current. The additional increase in electrical conductivity with the ageing time is minor, indicating that it closes to the limit performance under 400 Ўж.
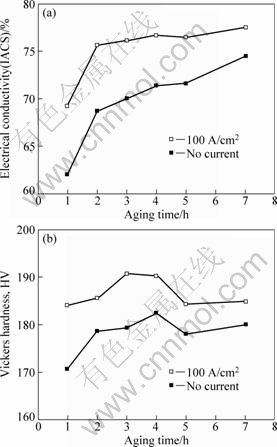
Fig. 2 Electrical conductivity (a) and Vickers hardness (b) versus ageing time for alloy aged at 400 Ўж with and without current
The microhardness of the alloy aged without current starts from HV171 at 1 h and then reaches the peak value of HV182 at 4 h, which can be attributed to the precipitation of Cr GP zones that are fully coherent with the Cu matrix and have the same FCC lattice parameter as Cu, as reported by JIN et al [8]. With the imposition of current, the hardness reaches HV184 after 1 h, which is higher than the peak value of no current ageing. Moreover, it approaches HV191 after 3 h. In general, a slight improvement occurs to the hardness after ageing with current for the investigated ageing time.
The electrical conductivity and Vickers hardness of alloy aged at 350-600 Ўж for 1 h with 100 A/cm2 current are shown in Fig.3. As shown in Fig. 3(a), due to more precipitation of solute atoms and recrystallization of deformed grains, the electrical conductivity under the both ageing conditions greatly increases with increasing temperature; however, the electrical conductivities are increased by 6% IACS by the current at the ageing temperature up to 500 Ўж.

Fig. 3 Electrical conductivity (a) and Vickers hardness (b) versus ageing temperature for alloy aged for 1 h with and without current
Precipitation occurs during ageing, which contributes to the hardening. With the increase of ageing temperature, the coherent precipitates are replaced by incoherent and coarse particulates within the alloy matrix [9]. The depletion of solutes in the solution during ageing will also decrease microhardness by removing the supersaturated solute atoms that drag dislocation motion [10]. In Fig. 3(b), both the microhardnesses come to the peak value at 450 Ўж and then decrease due to the overageing; however, the current improves the hardness by increasing the peak value by HV12 and up to H192.
Figure 4 depicts the effect of various current densities on electrical conductivity and Vickers hardness of the alloy aged at 400-500 Ўж for 1 h. At all the three ageing temperatures, the ascending current density brings on approximately linear increase of electrical conductivity, as shown in Fig. 4(a). The most prominent improvement is from the sample given ageing at 400 Ўж under 300 A/cm2, the conductivity is increased by 18% IACS and up to 80% IACS, which is equivalent to that obtained by no current ageing at 550 Ўж in Fig. 3(a). The conductivities of the alloy ageing at 450 Ўж and 500 Ўж are elevated by 13% IACS and 10% IACS after being aged with 300 A/cm2 current density, respectively. Additionally, it is clear that the slope of the electrical conductivity curve dwindles by increasing the ageing temperature, which could be attributed to the higher initial kinetics of the precipitation at a higher temperature, counteracting the enhancement effect of current ageing.
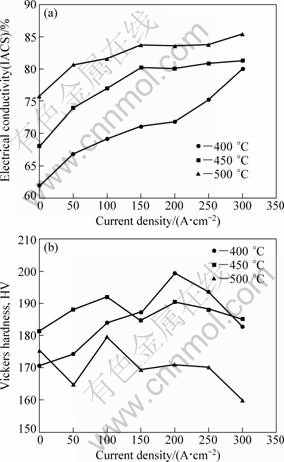
Fig. 4 Effect of various current densities on electrical conductivity (a) and Vickers hardness (b) of alloy aged at 400-500 Ўж for 1 h
In Fig. 4(b), the hardness of the alloys aged at 400 Ўж increases with increasing the current density. Microhardness climbs to HV200 after ageing at 400 Ўж with 200 A/cm2 current density, and then decreases. For the alloy aged at 450 Ўж and 500 Ўж, a slighter change of hardness can be observed. Previous results found that the high strength of the aged CuЁCCr and CuЁCCrЁCZr alloys are attributed to tiny coherent chromium-rich particles which precipitate from a supersaturated copper matrix and loose coherency with proceeding overageing. When the heat treatment is conducted at high temperatures, overageing occurs and the precipitates coarsen and lose coherency with the copper matrix, then the strength decreases [11].
3.2 Microstructure
Figure 5 illustrates the TEM images of Cu-Cr-Zr alloy aged at 400 Ўж for 1 h without current. The well defined ultra-fine grains are surrounded by clear boundaries, and some dislocations can be observed within the grains in Fig. 5(a). Moreover, scarce white precipitate particles appear in the structure in Fig. 5(b). All the above indicate that a low recovery occurs to the aged alloy, and the precipitation is at the very initial stage, which agrees with the relatively low electrical conductivity.

Fig. 5 TEM bright-field image (a) and corresponding dark-field image (b) of Cu-Cr-Zr alloy aged at 400Ўж for 1 h without current
Figure 6 shows the TEM bright-field image and the corresponding dark-field image of Cu-Cr-Zr alloy aged at 400 Ўж for 1 h with 300 A/cm2 current density. In Fig. 6(a), the center is a rather clean grain surrounded by a great amount of dislocations. A much higher dislocation density indicates that the dislocations are strongly pinned by the precipitates, which explains why the hardness is higher compared with that aged without current. According to VINOGRADOV et al [12], with such a high density of dislocations, the chaotic dislocation arrangement is energetically unfavorable, and dislocations are unevenly distributed so that the small grains in their central parts are rather free of dislocations and most dislocations are attracted to the grain boundaries, which suggests the presence of high internal stresses created by grain boundaries.
In Fig. 6(b), a tremendous number of homogenously distributed precipitates appear after imposition of 300 A/cm2 current density. The research on the precipitate reactions that occur in CuCrZr alloy suggests that small GP zones are present which exhibit a lobe-lobe appearance with a line of no contrast perpendicular to the operating (200)Cu reflection, an appearance known to occur for very small, coherent spherical particles [13]. As far as the present results are concerned, the selected area diffraction pattern (SADP) along![]() orientation suggests that these precipitates are fully coherent with the Cu matrix and FCC structure that are designated as the GP zones. The much higher electrical conductivity by ageing with 300 A/cm2 current density is attributed to the much higher precipitation rate of solute atoms, as the solute atoms in matrix act as obstacles to the movement of conduction electron and they also increase the density of electron scattering centers-lattice imperfections [14].
orientation suggests that these precipitates are fully coherent with the Cu matrix and FCC structure that are designated as the GP zones. The much higher electrical conductivity by ageing with 300 A/cm2 current density is attributed to the much higher precipitation rate of solute atoms, as the solute atoms in matrix act as obstacles to the movement of conduction electron and they also increase the density of electron scattering centers-lattice imperfections [14].

Fig. 6 TEM bright-field image (a) and corresponding dark-field image (b) of Cu-Cr-Zr alloy aged at 400 Ўж for 1 h with 300 A/cm2 current density, and SADP along ![]() zone axis
zone axis
The ageing treatment conducted at 400 Ўж for 1 h with 300 A/cm2 current density has the similar electrical conductivity to that aged at 550 Ўж without current, so the latter microstructure is provided for comparison. Figure 7 shows the TEM images of the sample given no current at 550 Ўж for 1 h ageing treatment. In Fig. 7(a), the matrix is so clean that barely precipitate particles can be found. In Fig. 7(b), a large Cr precipitate of 300 nm in diameter is confirmed by energy dispersive spectroscopy (EDS). The microstructure combines with the high electrical conductivity and low hardness, indicating that a severe overageing occurs under this ageing condition.

Fig. 7 TEM images of Cu-Cr-Zr alloy aged at 550 Ўж for 1 h without current: (a) Lower magnification; (b) Higher magnification
3.3 Mechanism
The influence of the current on the ageing treatment of Cu-Cr-Zr alloy is attributed to the interaction between Joule heating and electromigration. SILVA et al [15] investigated the reverse effects of Joule heating on the precipitation and dissolution of Co granules in Cu-Co alloys and stated that, the formation and growth of Co precipitates was produced by Joule heating. The Co granule size distribution depends on the annealing current (temperature) and the cooling rates. When the overall electron drift velocity is established, the force on the ions due to electron scattering is in the direction of the electron flow, which is known as the electron wind force. LIN et al [16] investigated the effect of pulse electric current stimulated the microstructure variation in the heat affected zone of cast hot working die steels and reported that, when the pulse electric current was passing through the bulk of metal material, some accompanying effects, such as electron wind force, thermal release and thermal stress, were induced by pulse electric current stimulation. If the induced thermal stress does not exceed the yield strength of material, the induced energies would promote the mobility of existing dislocations. Once the moving dislocations passed the barriers, such as carbide particles, dislocation loops were produced in the current stimulated specimen. The dislocation multiplication induced by pulse electric current would result in the increase of dislocation density. In addition, the distribution of nanosized carbide particles was much more dispersive after the pulse electric current simulated the fatigued specimen, which would further increase the resistance of dislocation movement and plastic deformation. NAH et al [17] studied the effect of Joule heating on electromigration-induced failure in solder joints and pointed out that, higher temperature would enhance the rate of electromigration because of the thermal effect on diffusion.
The mechanism might also be related with the interaction between the current and the mobility of solute atoms, vacancies and dislocations. The research of electroplasticity concluded that the plastic strain rate of a number of metals increases with the current density, suggesting that there exists a drift electron-dislocation interaction. The external electric field also appears to have an influence on the migration of vacancies or solute atom-vacancy complexes along grain boundaries to the charged surface [5]. The mobility of solute atoms, vacancies and dislocations is promoted by current. As a result, the formation of GP zones is highly enhanced, which finally improves the electrical and mechanical properties simultaneously.
4 Conclusions
1) The electrical conductivity reaches 76% IACS after ageing at 400 Ўж for 2 h with 100 A/cm2 current density, which is higher than that aged for 7 h without current. The hardness is slightly improved by the current.
2) The peak hardness is increased by HV12 and up to HV192 after ageing at 450 Ўж for 1 h with 100 A/cm2 current density, meanwhile, the electrical conductivity is increased by 6% IACS.
3) The sample given the 400 Ўж ageing and imposition of 300 A/cm2 current density reaches the electrical conductivity of 80% IACS, which is equivalent to that without current aged at 550 Ўж, and a peak of HV200 is reached after the alloy was aged at 400 Ўж with 200 A/cm2 current density.
4) TEM observations reveal that after ageing at 400 Ўж for 1 h with 300 A/cm2 current density, a much higher density of dislocation appears, and a tremendous number of nanosized Cr precipitates homogenously distributes in the matrix.
References
[1] ELLIS T W, KIM S T, VERHOEVEN J D. Deformation processed copper-chromium alloys: Role of age hardening [J]. J Mater Eng Perform, 1995, 4: 581-586.
[2] WATANABE C, MONZEN R, TAZAKI K. Mechanical properties of Cu-Cr system alloys with and without Zr and Ag [J]. J Mater Sci, 2008, 43: 813-819.
[3] KOPPENAAL T J, SIMCOE C R. The effect of electric current on the ageing of an Al-4 Pct Cu alloy [J]. Trans Met Soc AIME, 1963, 227: 615-618.
[4] TU K N. Recent advances on electromigration in very-large-scale- integration of interconnects [J]. J Appl Phys, 2003, 94: 5451-5473.
[5] CONRAD H. Effects of electric current on solid state phase transformations in metals [J]. Mater Sci Eng A, 2000, A287: 276-287.
[6] WANG Zhi-qiang, ZHONG Yun-bo, LEI Zuo-sheng, REN Wei-li, REN Zhong-ming, DENG Kang. Microstructure and electrical conductivity of Cu-Cr-Zr alloy aged with DC electric current [J]. J Alloy Compd, 2009, 471: 172-175.
[7] WANG Zhi-qiang, ZHONG Yun-bo, CAO Guang-hui, WANG Chao, WANG Jiang, REN Wei-li, REN Zhong-ming. Influence of dc electric current on the hardness of thermally aged Cu-Cr-Zr alloy [J]. J Alloy Compd, 2009, 479: 303-306.
[8] JIN Y, ADACHI K, TAKEUCHI T, SUZUKI H G. Ageing characteristics of Cu-Cr in-situ composite [J]. J Mater Sci, 1998, 33: 1333-1341.
[9] LIU Qiang, ZHANG Xiang, GE Yan, WANG Jiang, CUI Jian-zhong. Effect of processing and heat treatment on behavior of Cu-Cr-Zr alloys to railway contact wire [J]. Metal Mater Trans A, 2006, 37: 3233-3238.
[10] REZAEI M R, TOROGHINEJAD M R, ASHRFIZADEH F. Effects of ARB and ageing processes on mechanical properties and microstructure of 6061 aluminum alloy [J]. J Mater Process Tech, 2011, 211: 1184-1190.
[11] TU J P, QI W X, YANG Y Z, LIU F, ZHANG J T, GAN G Y, WANG N Y, ZHANG X B, LIU M S. Effect of aging treatment on the electrical sliding wear behavior of CuЁCCrЁCZr alloy [J]. Wear, 2002, 249: 1021-1027.
[12] VINOGRADOV A, PATLAN V, SUZUKI Y, KITAGAWA K, KOPYLOV V I. Structure and properties of ultra-fine grain Cu-Cr-Zr alloy produced by equal-channel angular pressing [J]. Acta Mater, 2002, 50: 1639-1651.
[13] SINGH B N, EDWARDS D J, ELDRUP M, TOFT P. Effects of heat treatments and neutron irradiation on microstructures and physical and mechanical properties of copper alloys [J]. J Nucl Mater, 1997, 249: 1-16.
[14] LIU Ping, SU Juan-hua, DONG Qi-ming, LI He-jun. Microstructure and properties of Cu-Cr-Zr alloy after rapidly solidified aging and solid solution aging [J]. J Mater Sci Technol, 2005, 21: 475-478.
[15] SILVA F C S, FERRARI E F, KNOBEL M. Precipitation and dissolution of Co granules in CuCo alloys: Reverse effects of Joule heating [J]. J Appl Phys, 1999, 86: 7170-7178.
[16] LIN H Q, ZHAO Y G, ZHAO Y, HAN L G, MA J, JIANG Q C. Effect of pulse electric current stimulation on the microstructure, mechanical properties and thermal fatigue behavior of cast-hot-working die steel [J]. ISIJ Inter, 2008, 48: 212-217.
[17] NAH J W, SUH J O, TU K N. Effect of current crowding and Joule heating on electromigration-induced failure in flip chip composite solder joints tested at room temperature [J]. J Appl Phys, 2005, 98: 1-6.
НхЦѕЗїЈ¬ЦУФЖІЁЈ¬ИДПФѕэЈ¬Нф і¬Ј¬Нх ЅЈ¬ХЕФц№вЈ¬ИОО¬АцЈ¬ИОЦТГщ
ЙПєЈґуС§ ІДБП№¤іМПµЈ¬ЙПєЈ 200072
ХЄ ТЄЈєФЪІ»Н¬ОВ¶ИєНµзБчГЬ¶ИПВ¶ФCu-Cr-ZrєПЅрЅшРРІ»Н¬К±јдµДК±Р§ґ¦АнЈ¬·ўПЦФЪєПККµДК±Р§ОВ¶ИПВК©јУµзБчЈ¬їЙТФН¬К±МбёЯєПЅрК±Р§єуµДµзµјВКєНУІ¶ИЎЈЛжЧЕµзБчГЬ¶ИµДФцјУЈ¬µзµјВКіКПЯРФФці¤Ј¬µ«УІ¶И±ЈіЦІ»±дЎЈПФОўЧйЦЇ№ЫІм±нГчЈ¬К©јУµзБчК±Р§єуЈ¬ЧйЦЇЦРіцПЦБЛґуБїµДО»ґнєНДЙГЧј¶ёхОціцОпЈ¬ХвКЗМбёЯµзµјВКєНУІ¶ИµДФТтЎЈµзБчЧчУГ»ъАнУлЅ№¶ъИИЎўґуБїµзЧУµДЗЁТЖј°ТтµзЧУ·зБ¦¶шМбёЯµДИЬЦКФЧУЎўїХО»єНО»ґнµДА©ЙўДЬБ¦УР№ШЎЈ
№ШјьґКЈєCu-Cr-ZrєПЅрЈ»µјµзРФЈ»УІ¶ИЈ»µзБчК±Р§Ј»Оціц
(Edited by LI Xiang-qun)
Foundation item: Project (2009AA03Z109) supported by the National High-tech Research and Development Program of China; Project (09zz98) supported by Key Research and Innovation Program from Shanghai Municipal Education Commission, China; Projects (09dz1206401, 09dz1206402) supported by Key Project from Science and Technology Commission of Shanghai Municipality, China
Corresponding author: ZHONG Yun-bo; Tel/Fax: +86-21-56336048; E-mail: yunboz@staff.shu.edu.cn
DOI: 10.1016/S1003-6326(11)61290-9

