Extraction of copper and zinc from naturally contaminated copper mine soils: Chemical fractionation analysis and risk assessment
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2018���6��
�������ߣ����� Ф�Ż� �ܿ� ��˼�� ��ΰ�� ������ ��ҫ�� Ф��ѩ ¦���t ������
����ҳ�룺1274 - 1284
Key words��waste dump soil; heavy metal; leaching; chemical fractions
Abstract: The leaching behaviour of Cu and Zn from contaminated soils at a copper mine was investigated using four extractants: citric acid (CA), oxalic acid (OA), ethylenediaminetetraacetic acid (EDTA) and nitrilotriacetic acid (NTA). Six soil samples were characterized for the distribution of four chemical fractions of Cu or Zn (acid-soluble, reducible, oxidizable and residual). For the extraction of Cu, EDTA is more effective than other extractants when the concentration is less than 0.02 mol/L. The leaching efficiency for Cu was at least 15% higher for EDTA than for the other extractants at the same concentrations. Similar leaching behaviour was observed in the extraction of Zn. After extraction by CA, OA, EDTA or NTA, the acid-soluble fractions and the reducible fractions of Cu were the main fractions extracted. The potential environmental risks related to speciation were evaluated, and after extraction these risks were reduced.
Cite this article as: XIAO Jiu-hua, ZHOU Jun, WANG Zhao-hui, LI Si-yue, ZHANG Wei-chao, FANG Chang-ling, GUO Yao-guang, XIAO Dong-xue, LOU Xiao-yi, LIU Jian-she. Extraction of copper and zinc from naturally contaminated copper mine soils: chemical fractionation analysis and risk assessment [J]. Journal of Central South University, 2018, 25(6): 1274�C1284. DOI: https://doi.org/10.1007/s11771-018-3824-6.

J. Cent. South Univ. (2018) 25: 1274-1284
DOI: https://doi.org/10.1007/s11771-018-3824-6
XIAO Jiu-hua(Ф�Ż�)1, ZHOU Jun(�ܿ�)1, WANG Zhao-hui(����)1, 2, LI Si-yue(��˼��)3,
ZHANG Wei-chao(��ΰ��)1, FANG Chang-ling(������)1, GUO Yao-guang(��ҫ��)1,
XIAO Dong-xue(Ф��ѩ)1, LOU Xiao-yi(¦���t)1, LIU Jian-she(������)1
1. State Environmental Protection Engineering Center for Pollution Treatment and Control in Textile Industry (College of Environmental Science and Engineering, Donghua University),Shanghai 201620, China;
2. International Center for Balanced Land Use (ICBLU), The University of Newcastle,Callaghan, NSW 2308, Australia;
3. Key Laboratory of Reservoir Aquatic Environment, Chongqing Institute of Green and Intelligent Technology, Chinese Academy of Sciences, Chongqing 400714, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2018
Abstract: The leaching behaviour of Cu and Zn from contaminated soils at a copper mine was investigated using four extractants: citric acid (CA), oxalic acid (OA), ethylenediaminetetraacetic acid (EDTA) and nitrilotriacetic acid (NTA). Six soil samples were characterized for the distribution of four chemical fractions of Cu or Zn (acid-soluble, reducible, oxidizable and residual). For the extraction of Cu, EDTA is more effective than other extractants when the concentration is less than 0.02 mol/L. The leaching efficiency for Cu was at least 15% higher for EDTA than for the other extractants at the same concentrations. Similar leaching behaviour was observed in the extraction of Zn. After extraction by CA, OA, EDTA or NTA, the acid-soluble fractions and the reducible fractions of Cu were the main fractions extracted. The potential environmental risks related to speciation were evaluated, and after extraction these risks were reduced.
Key words: waste dump soil; heavy metal; leaching; chemical fractions
Cite this article as: XIAO Jiu-hua, ZHOU Jun, WANG Zhao-hui, LI Si-yue, ZHANG Wei-chao, FANG Chang-ling, GUO Yao-guang, XIAO Dong-xue, LOU Xiao-yi, LIU Jian-she. Extraction of copper and zinc from naturally contaminated copper mine soils: chemical fractionation analysis and risk assessment [J]. Journal of Central South University, 2018, 25(6): 1274�C1284. DOI: https://doi.org/10.1007/s11771-018-3824-6.
1 Introduction
The copper mine located in Jiangxi province is the second largest open pit copper mine in China. A large quantity of waste was abandoned without proper treatment, resulting in a number of environmental problems, including heavy metal pollution from the oxidation of sulfide minerals [1]. The heavy metals leached from waste disposal sites may end up in groundwater, rivers, and soils around the deposit, which poses a threat to human health and the ecosystem [2]. Hence, it is imperative to develop suitable technologies to remediate the contaminated waste materials associated with this copper mine.Various remediation techniques have been developed in recent years. In situ remediation methods (i.e. soil flushing, electrokinetic remediation, and phytoremediation) seem very promising for decontamination as they are less disruptive than ex-situ treatment and have low handling costs [3, 4]. However, the remediation efficiency of these methods depends on the binding strength of heavy metals to soil sorption sites [4]. Extractants such as acids, bases, chelating agents, and surfactants can be used in such techniques to enhance metal mobilization [5]. Low molecular weight organic acids (LWMOAs) (e.g. citric acid (CA), oxalic acid (OA), ethylenediaminetetraacetic acid (EDTA), and nitrilotriacetic acid (NTA)), are among the most commonly used extractants, due to their ability to form strong water-soluble complexes with heavy metals. The leaching behavior of heavy metals is closely related to soil composition and properties. Many researchers have investigated the effect of CA, OA, EDTA and NTA on the leaching of heavy metals from paddy soils, urban soils, agricultural soils, cultivated soil and artificially contaminated soils [6�C9]. Sources of contamination can affect the extraction efficiency with the same extracting agents. Extraction of metals from artificially spiked soils is more efficient than from naturally contaminated soils because the added metals are in a much more accessible form, having not undergone aging [10, 11]. Therefore, data obtained in cultivated or artificially contaminated soils cannot be extrapolated to field-scale remediation of contaminated sites when chelating agents are evaluated for field use. Although the application of LWMOAs for metal extraction has been investigated, there are limited data available on the leaching behavior of heavy metals associated with naturally contaminated copper mine waste soils. An understanding of how, and to what extent, these extractants affect leaching of metals from copper mine waste soils will provide guidance for their remediation.
The leaching behavior of heavy metals is strongly correlated with metal fractionation in the soil environment. The content of different chemical fractions also determines the bioavailability of heavy metals [12]. It is, therefore, important to investigate the chemical speciation of heavy metals before and after extraction. Most studies overemphasize the extraction efficiency of total heavy metals. Minimal attention has been paid to the distribution of chemical speciation of heavy metals after extraction with LWMOAs in copper mine deposit soils.
The aim of the study is to investigate the influence of CA, OA, EDTA and NTA on the leaching behaviors of heavy metals from copper mine waste soils by analyzing Cu and Zn speciation. An evaluation of the potential environmental risk of heavy metals from copper mine waste soils is also proposed.
2 Methods and materials
2.1 Sites
The sample sites are located in the copper mine deposit located in the Jiangxi province of China. Approximately 10300 t of open pit mining wastes are deposited each day. Soil samples were collected from six areas (DE1, DE2, DE3, DE4, DW1 and DW2). DE1 and DE3 were collected from different sites of a waste dump to the east where fines have been carried by runoff. DE2 and DE4 were collected from the slopes of a waste dump to the east, abandoned 20 years ago. DW1 was collected from a recently abandoned waste dump to the west. DW2 was selected from a flat area of the waste dump to the west.
2.2 Soil characterization
Soil samples were collected from the surface layer (within 30 cm). The soil samples were prepared for analysis by air drying and passing through a 2 mm sieve. The pH was measured with a calibrated pH-meter (pHS-3C, Shanghai Leici Instrument Factory, China) in distilled water (1:2.5) and 1 mol/L KCl solution (1:2.5). Organic matter content was determined by potassium dichromate digestion [13]. Soil texture was determined by the pipette method [14]. Cation exchange capacity (CEC) was determined by the ammonium acetate method [14]. Total Cu and Zn concentrations were determined after dissolution by microwave-assisted acid digestion with aqua regia (ISO 11466-1995). Concentrations in the extracts were determined by atomic absorption spectrophotometry (AAS, Z-2000, Hitachi High-Technologies Corporation, Japan).
2.3 Extraction of Cu and Zn
Soil samples (0.1 g) were equilibrated in a shaker (25��5 ��C) for 24 h (200 r/min) with 20 mL of CA, OA, EDTA or NTA solutions. Previous studies indicated that 24 h was sufficient to reach equilibrium [15]. The pH was adjusted to 5.0��0.1 with either 1 mol/L NaOH or 1 mol/L HNO3 after the addition of chelating agent. Metal concentrations in the extracted solutions were measured by inductively coupled plasma mass spectrometer (ICP-MS, iCAP Q, Thermo Fisher Scientific, USA).
2.4 BCR sequential extraction
The mobility and bioavailability of heavy metals are closely related with chemical speciation. To estimate the mobility and bioavailability of heavy metals, a modified BCR sequential extraction procedure was used. All the soil samples were extracted according to the procedure described by CUONG et al [18], as shown in Table 1.
Three heavily contaminated soil samples (DE1, DE4 and DW1) were selected to examine the changes in chemical fractionation and to elucidate the specific metal leaching behavior by CA, OA, EDTA, and NTA. After extracting the soil samples with 0.2 mol/L CA, OA, EDTA or NTA, the residues were rinsed with deionized water and submitted to the modified BCR sequential extraction. Cu and Zn concentrations in all BCR extractions were determined by AAS. All plastic and glassware were soaked in 5% HNO3 overnight and rinsed with deionized water before use. All soil analyses were conducted in triplicate.
3 Results and discussion
3.1 Soil characteristics
The basic soil properties from the copper mine deposits are listed in Table 2. The pH values of DE1, DE2 and DE3 range from 6.7 to 7.2 and the pH values of DE4, DW1 and DW2 are 4.5, 5.6 and 3.0, respectively. The lower pH of the soil samples to the west is probably due to oxidation of sulfide minerals in the waste dump. The CEC of all the samples is less than 0.1 mol/kg, except for DE4 and DW1. All soils have low organic matter content of less than 0.5%, except for DE4 and DW1 at approximately 1%. According to soil texture categories [16], DE1, DE2 and DW1 are sandy loam while DE3, DE4 and DW2 are sandy clay loam, silty clay loam and loamy sand, respectively.
Metal removal efficiency is dependent on the speciation of metals in soils and the physicochemical properties. An increase in soil organic matter content, CEC, or clay content decreases chemical extraction efficiency [17]. Since DE3 and DE4 contain high clay and organic matter contents, the metal extractability is expected to be low at these sites. Elevated concentrations of Cu and Zn are observed in all soil samples from the mine waste dump (Table 2). The total concentrations of Cu and Zn exceed the China Environmental Quality Standard for soils (Grade III, Cu: 400 mg/kg, Zn: 500 mg/kg. GB15618-1995 of China). Of all the soil samples, DW1 has the highest Cu and Zn concentrations: 11326 mg/kg and 6012 mg/kg, respectively. This is attributed to environmental contamination from the nearby electrode copper plant.
3.2 Distribution of heavy metal speciation
In order to justify BCR sequential extraction, an internal check was performed [18]. The sum of heavy metals extracted in the four steps shown in Table 1 was compared with the heavy metal pseudo-total. The sums of the four fractions were in agreement with the total metal content, ranging from 95% to 125% for Cu, and from 78% to 109% for Zn. In addition, the concentration of metal impurities in the BCR sequential extraction reagents was below detection limits.
Metal partitioning among the different chemical fractions is shown in Figure 1. For Cu, the distribution of the chemical fractions in the different soil samples varies. In the DE1 and DE4 samples, 41% and 55% of the total Cu is bound to Fe�CMn oxides, respectively. For the DE2, DE3 and DW2 samples, the residual fraction dominates, ranging from 34% to 65%. Exchangeable, water-soluble and acid-soluble concentrations are the highest in the soil samples, making up 46% of the total Cu concentration. The role of the oxidizable fraction bound to organic matter and sulfide with Cu is less significant and is as low as 12%, except for DW1. It is worth noting that, except for DW2, all samples are abundant in potential mobile phases (e.g. the acid-soluble fraction makes up 46% of total Cu in DW1 and the mobile fraction 68% in DE1). These experimental results are consistent with previous studies [19].
Table 1 BCR sequential extraction scheme [18]

Table 2 Properties of copper mine waste dump soils
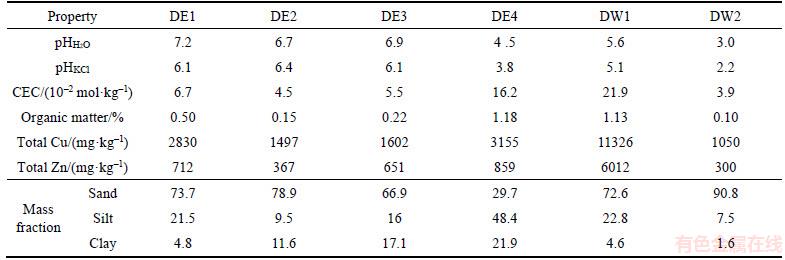

Figure 1 Speciation distribution of Cu and Zn in waste dump soil as determined by BCR sequential extraction
Distribution of Zn in the residual fraction ranges from 57% to 79% in all soil samples (Figure 1) implying that Zn is not readily released from the soils. Previous studies also concluded that the residual fractions accounted for the highest proportions of heavy metal in the soils from mine tailings or soils from copper mine discharge [20�C22]. Variation in the distribution of the residual fraction is small compared with the variation of the other three fractions. The acid-soluble fraction of Zn is less than 15% except for DE1 (28%). The reducible fractions of DE2 and DE3 comprise 21% and 16%, respectively; the contents of DE1, DE4, DW1 and DW2 samples are 7%, 11%, 6% and 6%, respectively. Other than DW1 (average 16% of pseudo-total Zn), the metal content in the organic matter and sulfide form (oxidizable fraction) only accounts for 9%, 10%, 4%, 8% and 10% in DE1, DE2, DE3, DE4 and DW2 soils.
The distribution of metals between the BCR fractions mainly depends on the aging time, the topography of the waste dump, the composition and properties of the soil, and the nature of the heavy metal itself [5, 15, 21, 23]. The higher percentage of reducible and residual Cu identified in the DE1, DE2, DE3 and DE4 samples may be partly due to the transformation of mobile to less reactive fractions over time or high levels of oxidizable materials (e.g. Fe and Mn oxides) [21, 22, 24]. The relatively low content of acid-soluble and oxidizable fractions may result from the topography and low organic matter contents.
3.3 Leaching of heavy metal
The extracted Cu concentrations from the copper mine waste dump form an inverted L-shape when plotted against extractant concentration (Figure 2(a)). They rapidly increase with extractant strength up to 0.05 mol/L, then level off until a plateau is formed. The leaching of Cu from all samples is optimal for 0.05 to 0.1 mol/L NTA, 0.02 to 0.1 mol/L EDTA, and 0.05 to 0.2 mol/L CA or OA, which is in agreement with previous studies [5, 25].
However, the patterns of Cu and Zn removal efficiencies vary with the soil samples. The leaching efficiencies for Cu in 0.2 mol/L CA, OA, EDTA, and NTA are 65%, 61%, 81% and 63%, respectively. The high leaching efficiency of DW1 is attributed to the dominance of the mobile fraction (acid-soluble fraction) (Figure 1). However, lower efficiencies for the other soils are due to a variety of reasons including: (i) high clay content, organic matter, and high CEC of the soil samples from DW1, which improves metal extraction, (ii) a small proportion bound to exchangeable, carbonate and reducible oxide fractions after leaching, and (iii) the readsorption of extracted metal by organic ligands [5, 26, 27].

Figure 2 Extracted Cu (a) and Zn (b) concentrations from DE1, DE2, DE3, DE4, DW1 and DW2 soil samples with CA, OA, EDTA and NTA
The extraction efficiency for Zn for all samples ranges from 18% to 29% with 0.2 mol/L CA, from 16% to 31% with 0.2 mol/L OA, from 10% to 28% with 0.2 mol/L EDTA, and from 6% to 18% with 0.2 mol/L NTA. These extraction efficiencies are low because the residual fraction dominates Zn speciation in these samples. The leaching efficiency of the four extractants is so low (less than 35%) that increasing extractant concentration has little effect. Aging may play a role in Zn binding and real waste soils are not expected to contain zinc in loosely bound forms [5].
When extractant concentrations were lt;0.02 mol/L, the amount of extracted Cu followed the order: EDTA>NTA��CA>OA, for all samples except DW1. However, at extractant concentrations higher than 0.02 mol/L, OA was the most effective Cu extractant and NTA was the least effective. The results are in agreement with Ref. [28]. PETERS et al [29] found that CA was more effective in extracting Cu and Zn from soils than EDTA at high concentrations. The leaching behavior for Zn is similar to that for Cu (Figure 2(b)). However, the leaching curve is irregular, which is attributed to the mutual competition for binding sites by different heavy metals [30]. In general, when extractant concentrations are low, EDTA is more effective than either NTA, OA or CA. However, OA outperforms the other chemicals when extractant concentrations are high. This phenomenon is attributed to: i) bonding strength and chemical form [31], and ii) a change in pH over the course of the experiment [8]. When the extractant concentrations are low, extractants first attack Cu and Zn weakly bound to exchangeable and carbonate fractions. The dosage of organic ligands has the greatest impact, with the maximum extraction efficiency obtained by treatment with low doses of EDTA with four organic ligands. Increasing the extractant concentration not only chelates weakly bound metals but also extracts metals strongly bound to the fractions associated with soil (e.g., reducible fraction). The pH also plays an important role in extracting metals bound to Fe and Mn oxides [8, 23, 26]. For OA and CA, the low pH in the post- extraction solution suggests dissolution of Fe and Mn oxides and this is reflected in the mobilization of associated metals [9, 19, 32]. Fe�CMn oxides are highly soluble in the OA extractant. Furthermore, surface charge, organic matter, and the distribution of particle size in the soils can influence the release and readsorption of heavy metals [2, 26, 33]. Therefore, further study is required to improve the leaching efficiency of CA, OA, EDTA and NTA for heavy metals.
3.4 Heavy metal speciation change before and after extraction
Speciation distribution after extraction with CA, OA, EDTA or NTA was studied in DE1, DE4, and DW1, chosen because of the high Cu and Zn concentrations in these samples, and the results are shown in Figure 3.
Cu and Zn in the exchangeable/carbonate (acid-soluble) phases and the Fe�CMn oxide (reducible) phases are the main species extracted by the extractants (Figure 3), while in the oxidizable and residual fractions, they are stationary. The acid-soluble fraction of Cu is reduced by around 80%, 40% and 90% in DE1, DE4 and DW1 soils, respectively. The reducible fraction of Cu decreases by 42%�C73%, 59%�C78% and 73%�C82%, in DE1, DE4 and DW1 soils, respectively. For Zn, a slight decrease is observed for each fraction, except for the oxidizable fraction in DW1. The relative decrease in acid-soluble Zn for DE1, DE4 and DW1 is 5%�C87%, 10%�C71% and 81%�C94%, respectively. The reducible and oxidizable fractions decreased by 11%�C67%, except for the oxidizable fraction in DW1. However, the oxidizable Cu fractions increased by around 17% to 29% in DE1 soil. The residual fraction of DE1 increased by 14% to 22%. The residual fraction of DE4 increased by 14% to 40%. PEREZ-ESTEBAN et al [32] found similar results, with organic matter and sulfide fractions increasing after CA extraction. Similar results were also reported for the extractant ethylenediaminedisuccinic (EDDS) acid [15]. The results suggest that reducing conditions during extraction lead to the formation of insoluble metal sulfides.
In addition, the order of extractability of Cu and Zn with 0.2 mol/L CA, OA, EDTA or NTA corresponds well with the order of leaching efficiencies observed for Cu and Zn. Generally,EDTA has much better extractability for the acid-soluble phase of Cu and Zn extraction, whereas liberation of Cu and Zn from the Fe�CMn oxide bound phase requires more intense chemical attack. This phenomenon explains why the optimal extraction efficiency of Cu was found in this case for the OA extractant.

Figure 3 Sequential extraction of Cu and Zn in DE1, DE4, and DW1 soil before and after extraction with 0.2 mol/L CA, OA, EDTA or NTA (E-Cu: extracted Cu with CA, OA, EDTA, or NTA; E-Zn: extracted Zn with CA, OA, EDTA or NTA)
Although significant uncertainty exists in the BCR sequential extraction process due to mass transfer and loss of soil in the operation and redistribution of metals [9, 34], the speciation of metals gives an indication of the potential environmental risk and the relative extraction efficiency of each extractant from different phases.
3.5 Assessment of potential environmental risk
In order to understand the bioavailability and mobility of heavy metals, two approaches are used to evaluate the potential environmental risk from copper mine waste dump soils. A risk assessment code (RAC) was introduced by several authors to evaluate the potential environmental risk of heavy metals in soils and sediments [22, 35]. The risk levels of heavy metals are evaluated by this method based on the percentage of metals bound in exchangeable and carbonate forms. These forms represent the loosely and weakly bound phases which are readily released into the environment, causing potential ecotoxicological and human risks [31].
The classification of soil samples before and after extraction with CA, OA, EDTA or NTA, according to RAC guidelines, is presented in Table 3. Before extraction, in the DE1, DE4 and DW1 soils, the content of Cu bound with the exchangeable and carbonate fractions is categorized as medium risk (MR), low risk (LR) and high risk (HR), respectively, while the content of Zn is LR, LR and MR, respectively. After extraction with CA, OA, EDTA or NTA, the contents for Cu and Zn are all NR or LR. This implies that the potential environmental risk is reduced after extraction; however, RACs are only based on the acid-soluble fraction. To investigate the change in reducible and oxidizable fractions (Cu and Zn bound with Fe�CMn oxides, organic matter, and sulfides), an assessment method developed by YOO et al [36] is used.
YOO et al [36] use the bioavailability index (BIM) and potential mobility index of metals (PMIM) (Eqs. (1) and (2)) to evaluate the risk to human health and the environment.
 (1)
(1)
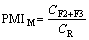 (2)
(2)
where CF1, CF2 and CF3 are the concentrations of metals in soil samples in the acid soluble (F1), reducible (F2), and oxidizable (F3) fractions, and CR is the reference value of metals according to the China Environmental Quality Standard (Grade III).
A BIM value of <1 indicates that the metals have a low potential mobility and bioavailability, whereas a BIM value of >1 indicates high potential mobility and bioavailability of metals. PMIM is a similar indicator to the BIM and has the same threshold values. At PMIM >1, the metal fraction could be changed to acid-soluble if surrounding conditions change, and vice versa. The BIM and PMIM values for Cu and Zn in the soils before and after BCR sequential extraction with CA, OA, EDTA or NTA are presented in Figure 4. The BIM values of Cu in DE1 and DW1 are >1 before extraction, but they are <1 after extraction with CA, OA, EDTA or NTA. On the other hand, the PMIM values of Cu, both before and after extraction with CA, OA, EDTA or NTA, are >1, although a decrease did occur after extraction. For Zn, the BIM and PMIM values are <1 in DE1 and DE4 before and after extraction, while the BIM and PMIM values are >1 in the original DW1 soil. It should be pointed out that the PMIM values of Cu in DE1, DE4 and DW1 soils are reduced but still >1. The PMIM value of Zn in DW1 soil is higher after extraction with OA due to an increase in the oxidizable fraction after extraction. The potential environment risk is reduced after extraction; however, if ambient environmental conditions are to change, the risk would increase.
Table 3 Risk assessment of Cu and Zn using RAC for DE1, DE4 and DW1 soils before and after extraction with CA, OA, EDTA, or NTA

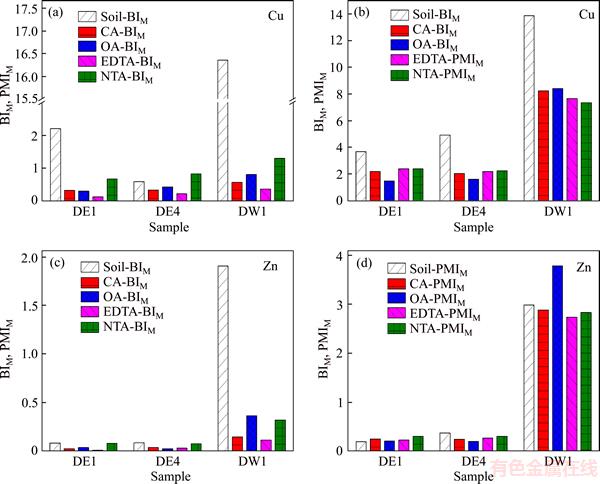
Figure 4 BIM and PMIM values of metals in DE1, DE4 and DW1 soil samples by BCR sequential extraction before and after extraction using CA, OA, EDTA or NTA
The two assessment methods used in this work suggest that the influence of heavy metals on environmental and human health would be reduced due to the release of metals bound in exchangeable and carbonate forms after extraction with CA, OA, EDTA or NTA. However, many of the PMIM values remain higher than the threshold, potentially leading to high mobility and bioavailability if alterations in environmental conditions caused the heavy metals to move to the acid-soluble fractions.
4 Conclusions
The leaching behavior and changes in Cu and Zn chemical fractions from waste dump soils of the copper mine before and after extracting with EDTA, NTA, CA or OA were investigated. Except for DW1, the extraction efficiency of Cu and Zn in respective soil samples was less than 60%. When the extractant concentration was less than 0.2 mol/L,
EDTA was more effective for extracting Cu and Zn. When the extractant concentration was more than 0.2 mol/L, OA was more effective for extracting Cu. The dominant chemical phases of Cu in soil were the reducible fraction for DE1 and DE4, the residual fraction for DE2 and DE3, and the acid-soluble fraction for DW2 and DW1. The dominant chemical phase of Zn was the residual fraction. After extracting with EDTA, NTA, CA or OA, the acid-soluble and reducible fractions of Cu and Zn were decreased, but there was a slight increase in oxidizable and residual fractions for some soil samples. Assessment of the potential environmental risk indicates that metals bound with exchangeable and carbonate phases are reduced after extraction, but a potential environment risk still remains.
Although the results provide some references for soil remediation, further research is needed to explore the applicability and cost effectiveness of these extractants at a large scale for the remediation of contaminated soil from the copper mine.
Acknowledgements
The authors would like to thank Dr. Nicholas Ward and Dr. Nadia Toppler of Southern Cross University for their editing assistance.
References
[1] GUO Yao-guang, HUANG Peng, ZHANG Wu-gang, YUAN Xue-wu, FAN Feng-xia, WANG Huan-li, LIU Jian-she, WANG Zhao-hui. Leaching of heavy metals from Dexing copper mine tailings pond [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(10): 3068�C3075.
[2] HANSEN H K, YIANATOS J B, OTTOSEN L M. Speciation and leachability of copper in mine tailings from porphyry copper mining: Influence of particle size [J]. Chemosphere, 2005, 60(10): 1497�C1503.
[3] DERMONT G, BERGERON M, MERCIER G, RICHER- LAFLECHE M. Metal-contaminated soils: Remediation practices and treatment technologies [J]. Practice Periodical of Hazardous, Toxic, and Radioactive Waste Management, 2008, 12(3): 188�C209.
[4] MULLIGAN C N, YONG R N, GIBBS B F. Remediation technologies for metal-contaminated soils and groundwater: An evaluation [J]. Engineering Geology, 2001, 60(1�C4): 193�C207.
[5] PETERS R W. Chelate extraction of heavy metals from contaminated soils [J]. Journal of Hazardous Materials, 1999, 66(1, 2): 151�C210.
[6] WU L H, LUO Y M, CHRISTIE P, WONG M H. Effects of EDTA and low molecular weight organic acids on soil solution properties of a heavy metal polluted soil [J]. Chemosphere, 2003, 50(6): 819�C822.
[7] QIN Fei, SHAN Xiao-quan, WEI Bei. Effects of low-molecular-weight organic acids and residence time on desorption of Cu, Cd, and Pb from soils [J]. Chemosphere, 2004, 57(4): 253�C263.
[8] YUAN Song-hu, XI Zhi-min, JIANG Yi, WAN Jin-zhong, WU Chan, ZHENG Zhong-hua, LU Xiao-hua. Desorption of copper and cadmium from soils enhanced by organic acids [J]. Chemosphere, 2007, 68(7): 1�C9.
[9] LI Li-jun, HOLM P E, MARCUSSEN H, BRUUN H C. Release of cadmium, copper and lead from urban soils of Copenhagen [J]. Environmental Pollution, 2014, 187: 90�C97.
[10] GAO Yan-zheng, HE Ji-zheng, LING Wan-ting, HU Hong-qing, LIU Fan. Effects of organic acids on copper and cadmium desorption from contaminated soils [J]. Environment International, 2003, 29(5): 613�C618.
[11] JIANG Hong, LI Ting-qiang, HAN Xuan, YANG Xiao-e, HE Zhen-li. Effects of pH and low molecular weight organic acids on competitive adsorption and desorption of cadmium and lead in paddy soils [J]. Environmental Monitoring and Assessment, 2012, 184(10): 6325�C6335.
[12] MADRID F, REINOSO R, FLORIDO M C, DIAZ- BARRIENTOS E, AJMONE-MARSEN F, DAVIDSON C M, MADRID L. Estimating the extractability of potentially toxic metals in urban soils: A comparison of several extracting solutions [J]. Environmental Pollution, 2007, 147(3): 713�C722.
[13] CHEN Su-lan, HU Guan-jiu. A preliminary discussion on the analysis methods for inorganic elements used in the national investigation program of soil pollution [J]. Environ Monit China, 2007, 23(5): 6�C10. (in Chinese)
[14] LU R K. Analytical methods for soils and agricultural chemistry [M]. Beijing: China Agricultural Science and Technology Press, 1999. (in Chinese)
[15] TANDY S, BOSSART K, MUELLER R, RITSCHEL J, HAUSER L, SCHULIN R, NOWACK B. Extraction of heavy metals from soils using biodegradable chelating agents [J]. Environmental Science & Technology, 2004, 38(3): 937�C944.
[16] ZHAI Yu-shun, THOMASSON J A, BOGGESS III J E, SUI Rui-xiu. Soil texture classification with artificial neural networks operating on remote sensing data [J]. Computers and Electronics in Agriculture, 2006, 54(2): 53�C68.
[17] DERMONT G, BERGERON M, MERCIER G, RICHER- LAFLECHE M. Soil washing for metal removal: A review of physical/chemical technologies and field applications [J]. Journal of Hazardous Materials, 2008, 152(1): 1�C31.
[18] CUONG D T, OBBARD J P. Metal speciation in coastal marine sediments from Singapore using a modified BCR- sequential extraction procedure [J]. Applied Geochemistry, 2006, 21(8): 1335�C1346.
[19] ARENAS-LAGO D, ANDRADE M L, LAGO-VILO M, RODRIGUEZ-SEIJO A, VEGA F A. Sequential extraction of heavy metals in soils from a copper mine: Distribution in geochemical fractions [J]. Geoderma, 2014, 230�C231: 108�C118.
[20] YANG J S, LEE J Y, BAEK K, KWON T S, CHOI J. Extraction behavior of As, Pb, and Zn from mine tailings with acid and base solutions [J]. Journal of Hazardous Materials, 2009, 171(1�C3): 443�C451.
[21] REMIREZ M, MASSOLO S, FRACHE R, CORREA J A. Metal speciation and environmental impact on sandy beaches due to El Salvador copper mine, Chile [J]. Marine Pollution Bulletin, 2005, 50(1): 62�C72.
[22] RODRIGUEZ L, RUIZ E, ALONSO-AZCARATE J, RINCON J. Heavy metal distribution and chemical speciation in tailings and soils around a Pb�CZn mine in Spain [J]. J Environ Manage, 2009, 90(2): 1106�C1116.
[23] ZALECKAS E, PAULAUSKAS V, SENDZIKIENE E. Fractionation of heavy metals in sewage sludge and their removal using low-molecular-weight organic acids [J]. Journal of Environmental Management, 2013, 21(3): 189�C198.
[24] BURT R, WILON M A, KECK T J, DOUGHERTY B D, STROM D E, LINDAHL J A. Trace element speciation in selected smelter-contaminated soils in Anaconda and Deer Lodge Valley Montana, USA [J]. Advances in Environmental Research, 2003, 8(1): 51�C67.
[25] GIANNIS A, NIKOLAOU A, PENTARI D, GIDARAKOS E. Chelating agent-assisted electrokinetic removal of cadmium, lead and copper from contaminated soils [J]. Environmental Pollution, 2009, 157(12): 3379�C3386.
[26] KRISHNAMURTI G S R, CIESLINSKI G, HUANG P M, VAN-REES K C J. Kinetics of cadmium release from soils as influenced by organic acids: Implication in cadmium availability [J]. Journal of Environmental Quality, 1997, 26(1): 271�C277.
[27] CHAIRIDCHAI P, RITCHIE G S P. Zinc adsorption by a lateritic soil in the presence of organic ligands [J]. Soil Science Society of America Journal, 1990, 54(5): 1242�C1248.
[28] XIA Wen-bin, LI Xin, GAO Hui, HUANG Bao-rong, ZHANG Hui-zhi, LIU Yun-guo, ZENG Guang-ming, FAN Ting. Influence factors analysis of removing heavy metals from multiple metal-contaminated soils with different extractants [J]. Journal of Central South University of Technology, 2009, 16(1): 108-111.
[29] PETERS R W. Feasibility treatability studies for removal of heavy metals from training range soils at the Grafenwoehr training area, Germany [M]. Argonne, IL: ANL/ESD/TM-81, Argonne National Laboratory, 1995
[30] VEGA F A, COVELO E F, ANDRADE M L. Competitive sorption and desorption of heavy metals in mine soils: Influence of mine soil characteristics [J]. Journal of Colloid and Interface Science, 2006, 298(2): 582�C592.
[31] CHENG Zhong-qi, LEE L, DAYAN S, GRINSHTEIN M, SHAW R. Speciation of heavy metals in garden soils: evidences from selective and sequential chemical leaching [J]. Journal of Soils and Sediments, 2011, 11(4): 628�C638.
[32] PEREZ-ESTEBAN J, ESCOLASTICO C, MOLINER A, MASAGUER A. Chemical speciation and mobilization of copper and zinc in naturally contaminated mine soils with citric and tartaric acids [J]. Chemosphere, 2013, 90(2): 276�C 283.
[33] SHAN Xiao-quan, LIAN Jun, WEN Bei. Effect of organic acids on adsorption and desorption of rare earth elements [J]. Chemosphere, 2002, 47(7): 701�C710.
[34] BACON J R, DAVIDSON C M. Is there a future for sequential chemical extraction? [J]. Analyst, 2008, 133(1): 25�C46.
[35] SINGH K P, MOHAN D, SINGH V K, MALIK A. Studies on distribution and fractionation of heavy metals in Gomti river sediments-a tributary of the Ganges, India [J]. Journal of Hydrology, 2005, 312(1): 14�C27.
[36] YOO J C, LEE C D, YANG J S, BAEK K. Extraction characteristics of heavy metals from marine sediments [J]. Chemical Engineering Journal, 2013, 228(28): 688�C699.
(Edited by FANG Jing-hua)
���ĵ���
���ò�ͬ��ȡ�������������������ؽ�����ȡ
ժҪ����ͭ������Ⱦ����Ϊ�����о������ᣨCA�������ᣨOA�����Ҷ��������ᣨEDTA�����������ᣨNTA��������ȡ����ͭ��п�Ľ���Ч������������������Ʒ������̬����ԭ̬������̬������̬��ʽ����ͭ��п��ռ�ȡ��о��������������ȡ�����ܶ�С�� 0.02 mol/Lʱ�����������������ȡ����EDTA ����ͭ����ȡЧ����ã�����ͬ��ȡ��Ũ���£�EDTA��������ȡ������ȡЧ�����ٸ�15%������ȡ�����ܶ�С�� 0.02 mol/Lʱ��EDTA ��п����ȡЧ����á��ֱ�ⶨ������ȡ����ȡǰ����ͭ�Ļ�ѧ������̬������������̬�ͻ�ԭ̬��ʽ���ڵ�ͭ��������ȡ�������������١������������շ��֣���ȡ֮�����������½���
�ؼ��ʣ����������ؽ��������ˣ���ѧ��̬
Foundation item: Project(2232013A3-08) supported by the Fundamental Research Funds for the Central Universities, China; Project supported by Shanghai Pujiang Program and DHU Distinguished Young Professor Program, China
Received date: 2016-12-17; Accepted date: 2017-01-23
Corresponding author: WANG Zhao-hui, PhD, Professor; Tel: +86�C21�C67792557; Fax: +86�C21�C67792522; E-mail: zhaohuiwang@ dhu.edu.cn; ORCID: 0000-0003-2101-093X

