
Solidification simulation of Al-Mg-Er alloys using CALPHAD method
ZHANG Li-gang(章立钢)1, LIU Li-bin(刘立斌)1, 2, HUANG Guo-xing(黄国幸)1, JIA Bing-ran(贾兵然)1
1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. Key Laboratory of Hunan Province of the Nonferrous Metal Material Science and Engineering,
Central South University, Changsha 410083, China
Received 15 July 2007; accepted 10 September 2007
Abstract: With CALPHAD(calculation of phase diagrams) method, the solidification paths of several Al-rich Al-Mg-Er alloys were calculated with Scheil―Gulliver model in the Thermo-calc software. And the amounts of several phases formed during solidification were also calculated. Synchronously, the heat-treatment temperatures of the same alloys were predicted by thermodynamic calculation. The calculated results show that by adding 0.4%Er (mass fraction), the strength effect of Er in Al-Mg alloy is better. This result is in agreement with the experimental investigations.
Key words: Al-Mg-Er alloys; solidification simulation; CALPHAD
1 Introduction
The effect of rare earths elements on the aluminum alloy is evident for their special electronic structure, because of their large atomic radius and tendency to lose two outermost level s-electrons and a 5d or 4f electron to become trivalent ion, and this has attracted attentions of many scientists. Rare earths elements are apt to react with aluminum to form refractory compounds, which are dispersed in the alloys and play the role of grain refining, and thus improve the corrosive resistance properties and the electric conductivity[1]. Recently researches have found that Er is a favorable alloying element in aluminum alloys, especially in Al-Mg and Al-Cu alloys. With increasing content of Er, the as-cast grains are refined, the thermal stability is improved, and the hardness and the strength are increased as well[2-8]. The reason of the above good properties is that the main existing form of Er in aluminum alloy is Al3Er, which is characterized as higher melting-point and good stability for its special structure. Just as Al3Sc and Al3Zr, it belongs to cubic system and Pm3m space group and coherent or semi-coherent with the Al matrix. Solidification phenomena play an important role in many processes used in fields ranging from production engineering to solid-state physics, especially in casting aluminum alloys.
The purpose of this study is to simulate the solidification processes of Al-Mg-Er alloys with CALPHAD(calculation of phase diagrams) method. And the amounts of several phases formed during solidification are also calculated.
2 Model calculation
The simplest model for describing the solidification behavior of alloys was proposed by SCHEIL[9]. The assumptions of the SCHEIL model are as follows: 1) diffusion in the solid is negligible; 2) local equilibrium maintains at the solid-liquid interface and the curvature effect at the interface is neglected; 3) uniform liquid composition; 4) equal solid and liquid densities.
Assuming linear liquidus and solidus lines, the composition of solid(cs) under solidification can be described as[10]:
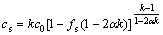 (1)
(1)
where k is the partition coefficient, c0 is the composition of original liquid alloy, fs is the fraction of solid transformed, and α is a solid-state back-diffusion parameter:
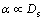 (2)
(2)
When Ds=0 (no solid-state diffusion), α=0. That is
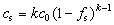 (3)
(3)
Eqn.(3) is SCHEIL’s equation.
When c parameter is replaced by T, Eqn.(3) can be rewritten as[11]
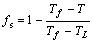 (4)
(4)
where TL and Tf are the equilibrium liquidus and solidus temperatures.
The solidification paths of some alloys are predicted with SCHEIL model in the Thermo-calc software. The α(FCC) phase will be formed from liquid at first when the Al-rich alloys solidify, then the Al3Er, τ(TAO) and Al3Mg2 phase form. The thermodynamic description of these phases is cited from the work of CACCIAMANI et al[12]. Combining Eqn.(4) with the above thermo- dynamic description, the solidification path and the phase fraction can be calculated.
3 Results and discussion
3.1 Solidification microstructure
The solidification microstructure of Al-5%Mg- 0.4%Er alloy was examined by YU et al[10], and the results are shown in Fig.1(a). The impure phase is supposed to be AlFeSi phase because the elements Fe and Si are the normal impure elements.
With the SCHEIL model in the Thermo-calc software, the solidification microstructure was calculated, as shown in Fig.1(b). Also the fraction of each phase is calculated and shown in Figs.1(c) and (d). From the calculated results, four phases (FCC, Al3Er, Al3Mg2 and τ phase) are formed in the end of solidification. The calculated results are in agreement with the experiment except the τ phase. Although the τ phase exists in our calculation, its phase fraction is much less than that of other phases. Thus it is hard to determine τ phase in the experiment. The microstructures of other alloys including Al-5%Mg-0.1%Er and Al-5%Mg-0.7%Er were also investigated by YU et al[13]. And XING et al[14] investigated the microstructures of the Al-4.5Mg-0.4Er alloy. Their results are shown in Fig.2. Similar to the Al-5Mg-0.4%Er alloys, we conducted the phase fraction calculation with SCHEIL model in the Thermo-calc software, and the results are shown in Fig.3. Comparing Fig.2 with Fig.3, it is found that there is good agreement between the calculated and experimental results.
From Figs.1(d), 2(a) and 2(b), we find that the Al-5Mg-0.4Er alloy has the higher Al3Er phase fraction among these three alloys. So the Al-5Mg-0.4Er alloy has better strength property according to our calculation. This result is in good agreement with the work of YANG [15]
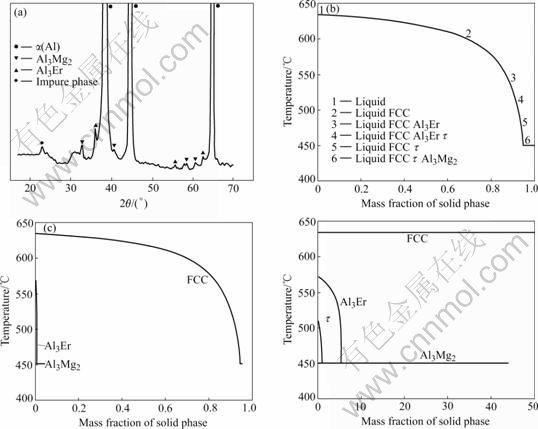
Fig.1 XRD pattern (a), calculated solidification (b) and calculated phase fraction (c, d) of Al-5%Mg-0.4%Er alloy
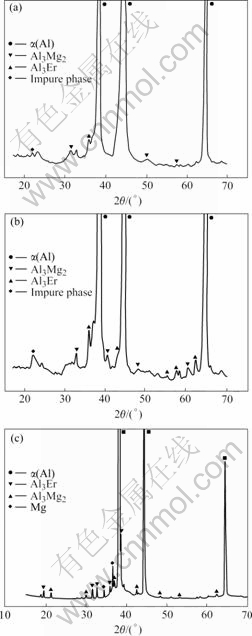
Fig.2 XRD results of some Al-Mg-Er alloys: (a) Al-5Mg-0.1Er; (b) Al-5Mg-0.7Er; (c) Al-4.5Mg-0.4Er.
3.2 Solidification path
The solidification of Al-5Mg-0.1Er alloys was simulated by SCHEIL model, and the calculated solidification path is shown in Fig.4(a). The change of mole fraction of Er and Mg in liquid during the solidification is shown in Fig.4(b).
From Fig.4(b), it is found that the mole fraction of Mg increases in the process of solidification, but the mole fraction of Er first increases and then decreases. It can be concluded that some compounds in the Al-Ersystem but no Al-Mg or Mg-Er compounds will form in the early stage of solidification, but Al3Mg2 phase forms in the end of solidification.
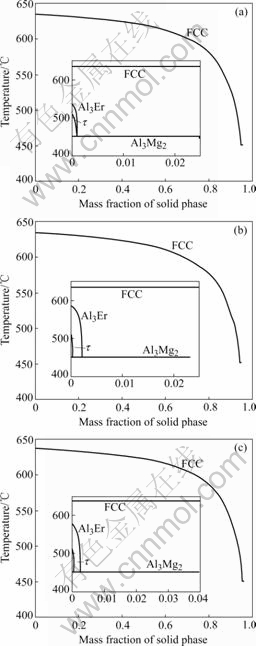
Fig.3 Calculated phase fractions of some Al-Mg-Er alloys: (a) Al-5Mg-0.1Er; (b) Al-5Mg-0.7Er; (c) Al-4.5Mg-0.4Er
The solidification paths of Al-5Mg-0.4Er, Al-5Mg- 0.7Er and Al-4.5Mg-0.4Er alloys are similar to that of Al-5Mg-0.1Er. The calculated results of these alloys are shown in Figs.5-7.
3.3 Heat-treatment temperature
In Al-Mg-Er alloys, the amount of Al3Er is very important to the properties of these alloys. So how to gain the highest amount of Al3Er from heat treatment will be a main problem to the material scientist. We use the thermodynamic database of Al-Mg-Er system to predict the heat-treatment temperatures of some Al-rich alloys.
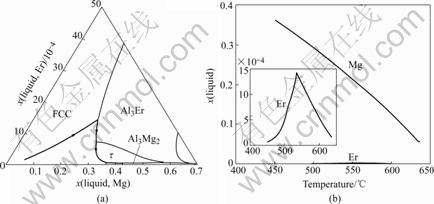
Fig.4 Solidification path of Al-5Mg-0.1Er alloy
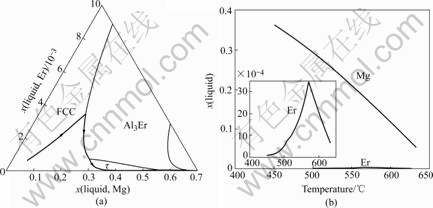
Fig.5 Solidification path of Al-5Mg-0.4Er alloy
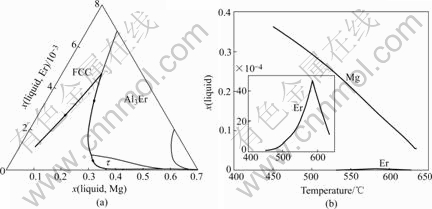
Fig.6 Solidification path of Al-5Mg-0.7Er alloy
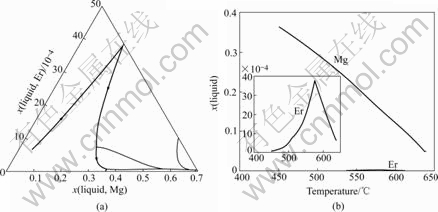
Fig.7 Solidification path of Al-4.5Mg-0.4Er alloy
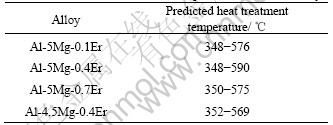
Fig.8 shows the calculated phase fraction of Al-5Mg-0.1Er alloy. In order to achieve better strength property, the phase fraction of Al3Er should be as high as possible and other compounds as little as possible. Based on this reason, the heat-treatment temperature had better to be between 348 and 576 ℃ from Fig.8. The heat treatment temperature of other alloys can be predicted as the same way. The calculated results are listed in Table 1.
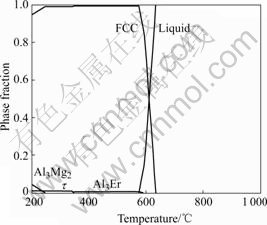
Fig.8 Calculated phase fraction of Al-5Mg-0.1Er alloy
Table 1 Predicted heat treatment temperatures of Al-rich alloys
4 Conclusions
1) The solidification path and microstructure during solidification of Al-Mg-Er alloys are calculated by CALPHAD with SCHEIL model.
2) From the calculation results, Al-5Mg-0.4Er alloy has higher Al3Er phase fraction and has better strength.
3) The heat-treatment temperatures of some alloys are predicted by thermodynamic calculation. According to this calculated results, we can choose suitable heat-treatment methods.
References
[1] NIE Z R, JIN T N, ZOU J X, FU J B, YANG J J, ZHOU T Y. Development on research of advanced rare-earth aluminum alloy [J]. Trans Nonferrous Met Soc China, 2003, 13(3): 509-514.
[2] YANG J J, NIE Z R, JIN T N, XU G F, FU J B, ZUO T Y. Effects of rare earth element Er on structure and properties of Al-4Cu [J]. Journal of the Chinese Rare Earth Society, 2002, 20(s2): 159-162.
[3] NIE Z R, JIN T N, XU G F. A kind of Al-Er alloy. CN 01134612.4 [P]. 2001-11-17.
[4] FERRO R, SACCONE A, BORZONE G. Rare earth metals in light alloys [J]. Journal of Rare Earths, 1997, 15(1): 45-61.
[5] ROSALBINO F, ANGELINI E, DE NEGRI S, SACCONE A, DELFINO S. Influence of the rare earth content on electrochemical behaviour of Al-Mg-Er alloys [J]. Intermetallics, 2003, 23: 435-441.
[6] XIAO D H, WANG J N, DING D Y. Effect of minor cerium additions on microstructure and mechanical properties of cast Al-Cu-Mg-Ag alloy [J]. Mater Sci Tech, 2004, 20(10): 1237-1240.
[7] XU G F, YANG J J, JIN T N, NIE Z R, YIN Z M. Effects of trace erbium on structure and properties of Al-5Mg alloy [J]. The Journal of Nonferrous Metals, 2006, 16(5): 768-774. (in Chinese)
[8] YANG J J, NIE Z R, JIN T N, RUAN H Q, ZHUO T Y. Form and refinement mechanism of element Er in Al-Zn-Mg alloy [J]. The Chinese Journal of Nonferrous Metals, 2004, 14(4): 620-626. (in Chinese)
[9] SCHEIL E. Retrograde saturation curves [J]. Z Metallkde, 1942, 34: 70-72.
[10] KURZ W, FISHER D J. Fundamentals of solidification [M]. Switzerland: Trams Tech Publications Ltd, 1998: 282-298.
[11] SAUNDERS N, MIODOWNIK A P. CALPHAD (calculation of phase diagrams): A comprehensive guide [M]. USA, 1998: 443-444.
[12] CACCIAMANI G, SACCONE A, NEGRI S DE, FERRO R. The Al-Er-Mg ternary system (part II): Thermodynamic modeling [J]. Journal of Phase Equilibria. 2002, 23: 38-50.
[13] YU S W, WANG W, YANG J J, ZHOU J X, NIE Z R. Thermodynamic calculation and precipitation behavior of Al3Er phase in Al-5Mg-Er alloy [J]. Journal of the Chinese Rare Earth Society, 2006, 24(3): 470-474. (in Chinese)
[14] XING Z B, NIE Z R, JI X L, WANG X D, ZHOU J X. Effect of trace erbium and manganese on microstructure and properties of Al-Mg alloys [J]. Rare Metal Materials and Engineering, 2006, 35(12): 1979-1982. (in Chinese)
[15] YANG J J. Investigations on the action mechanism of rare-earth element Er in the Al-Mg and Al-Zn-Mg alloys [D]. Beijing: Beijing University of Technology, 2005: 1-116. (in Chinese)
Foundation item: Project(50771106; 50731002) supported by the National Natural Science Foundation of China; Project(06FJ3006) supported by the Hunan Provincial Science and Technology Department, China
Corresponding author: LIU Li-bin; Tel: +86-731-8877732; E-mail: pdc@mail.csu.edu.cn
(Edited by YUAN Sai-qian)