
Preparation of high-purity tantalum ethoxide by vacuum distillation
YANG Sheng-hai(杨声海)1, CHEN Yong-ming(陈永明)1, YANG Hai-ping(杨海平)1,
LIU Yin-yuan(刘银元)1, TANG Mo-tang(唐谟堂)1, QIU Guan-zhou(邱冠周)2
1.School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China;
2. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 6 February 2007; accepted 9 May 2007
Abstract: Effects of reflux ratio, water addition and content of water in ethanol on the purity and yield of tantalum ethoxide during vacuum distillation were investigated under the operational conditions of pressure of 1 kPa, oil bath temperature of 210-230 ℃, and outlet temperature of 190 ℃. The condensate sample was characterized by FTIR, 1H-NMR spectroscopy and Raman spectra, respectively. The contents of tantalum, carbon and hydrogen in the sample were also determined with elemental analysis instrument. The obtained results consistently demonstrate that the condensate is tantalum ethoxide. The content of impurity, such as Al, As, Ca, Co, Cr, Cu, Fe, K, Mg, Mn, Mo, Na, Ni, Pb, Sn, Ti, V and Zn, in tantalum ethoxide is less than 0.000 05%, while Nb content is less than 0.000 5%. The content of impurities in tantalum ethoxide sample excels that of Epichem Group’s requirement for Ta(OC2H5)5 of 99.999%.
Key words: tantalum ethoxide; distillation; FTIR
1 Introduction
Tantalum ethoxide is mainly used for the production of thin oxide films, adopted in optical, semiconductor and optoelectronics applications, by sol-gol[1] or metalorganic chemical vapor deposition(MOCVD)[2] methods. Tantalum oxide thin films with high dielectric constant, large refractive index and remarkable chemical stability are usually selected as key materials for dynamic random-access memory(DRAM), anti-refection coating, gas sensor and capacitor[3]. Especially in the fields of ultra-large-scale integrated(ULSI)[4] and metal-insulator-semiconductor(MIS) switch, higher dielectric constant of tantalum oxide (25-35) than that of SiO2(3.8) is the main reason for larger capacitance with the same thickness[5]. In addition, tantalum oxide has been widely applied in synthesizing tantalum ceramics [6], ferroelectric tantalates, dielectric films[7], and high-purity nano-sized tantalum oxide powders[8].
Tantalum pentachloride is usually employed as the raw material in present technique for synthesis of tantalum ethoxide which is the product of the reaction between tantalum pentachloride and appropriate ethanol in diluted solvent of benzene or toluene[9]. As metal halide reacts with alcohol only at partial replacement, it is necessary to add some reagents like ammonia or pyridine to impel the reaction to complete. In order to remove the trace halide solved in ethanol and tantalum ethoxide, alkali metals are added for the formation of metal halides[10]. Thus, this preparation method has some disadvantages, such as restrict requirements on equipments, bad work condition, low recovery ratio and toxic diluted solvent pollution. Significant progress was made in the electrochemical synthesis of metalorganic complexes in last several decades. Many similar complexes of reactive metals, such as iron, cobalt, nickel and copper, have been produced by this technique[11]. In contrast to conventional methods, the electrochemical synthetic process is more productive and inexpensive with regards to apparatus and raw materials. Furthermore, it is friendly to the environment.
Because some impurity metals have greatly negative influences on the properties of tantalum oxide thin films[12], the high-purity of tantalum ethoxide is necessary absolutely. On the basis of previous electrochemical synthesis of crude tantalum ethoxide [13], studies on the preparation of high purity tantalum ethoxide by distillation were carried out in this work. 2 Experimental
2.1 Raw materials
Anhydrous ethanol (AR grade) was purchased from Changsha Chemical Reagent Corporation, China and tetramethylammonium chloride (TMAC) (AR grade) was purchased from Jintan Huadong Chemical Research Institute of Jiangsu Province, China. All reagents were used without further purification.
A stainless steel plate was used as cathode, and its working area was 19.0 cm×12.0 cm. Tantalum plate (1.0 kg), purchased from Zhuzhou Cement Carbide Group Limited Corporation, was employed as anode, and its working area was 18.0 cm×10.0 cm. The tantalum plates were made from tantalum powders of metallurgical grade with self-resistance sintering, electron bombardment and rolling treatment. The chemical composition of tantalum plate is listed in Table 1.
Table 1 Chemical composition of tantalum plate (mass fraction, %)

2.2 Tantalum ethoxide synthesis and purification
As referred elsewhere[13], electrochemical synthesis of tantalum ethoxide was performed in a cell with current of 4 A at boiling temperature (about 78 ℃) for 72 h. In the cell, a tantalum plate and stainless steel plate were adopted as anode and cathode, respectively, with distance of 2.3 cm between them. The electrolyte consisted of 2.5 L anhydrous ethanol and 0.04 mol/L TMAC, used as conductive agent. Distillation was performed at ambient pressure to separate redundant ethanol from mixed solution of ethanol and tantalum ethoxide at temperature above 78 ℃. Then the distillation temperature was raised to 150 ℃, and a little amount of ester was removed. According to tantalum ethoxide temperature―vapor pressure curve and the maximum vacuum of distillation tower or vacuum pump, vacuum distillation of 200 mL crude tantalum ethoxide solution was conducted at pressure of 1 kPa and oil bath temperature of 210-230 ℃. The outlet temperature and tail gas outlet temperature were maintained at 190 ℃ and 150 ℃, respectively. The different vacuum distillation experiments were performed under following conditions: 1) Reflux ratio was 9?1, and vacuum distillation tower was not filled with packing (No.1); 2) Vacuum distillation tower was filled with packing, and reflux ratio was varied as 0, 1?1 and 9?1, respectively (No.2-4); 3) Vacuum distillation tower was filled with packing. Amounts of water and anhydrous ethanol were mixed and added into distillation tank before vacuum distillation. The mass ratio of water addition to tantalum ethoxide was 0.1% and 0.5% (No.5, No.6), respectively. The volume of anhydrous ethanol solution was 100 times of water addition; 4) Vacuum distillation tower was filled with packing, and alcohol solution recovered from previous distillation process substituted for anhydrous ethanol during tantalum ethoxide electrochemical synthesis (No.7). The distillation product obtained was saved in Duran bacteria culture bottle produced in Germany with Ar atmosphere protection.
2.3 Characterization
FTIR spectroscopy of condensate and distilled residue was measured with a Perkin Elmer 2000 IR spectrometer. Raman spectra were recorded using a Labram I Micro-Raman Spectroscopy System of Dilor with 632.8 nm He-Ne laser. The 1H-NMR spectrum was recorded by an INOVA-400 (Varian) Nuclear Magnetic Resonance spectroscopy, and chloroform-d was adopted as dilute reagent. The elemental analyses of carbon and hydrogen were determined with a Perkin Elmer PE 2400 elemental analysis instrument. In order to determine impurity contents of tantalum ethoxide, some amount of water was added into tantalum ethoxide solution for hydrolysis reaction. After desiccation in desiccator at 100 ℃ for 12 h, the sample was calcined in muffle furnace at 900 ℃ for 1 h. The silicon content of tantalum oxide obtained was determined with spectroscopic analyser, and other impurity contents were obtained by ICP-Mass Agilent 7500a analyzer. And then the impurity content of tantalum oxide was converted into that of tantalum ethoxide. The impurity elements were classified as Al, Nb, main group metals (such as, As, Pb, Sn), mobile metals (such as Ba, Ca, Li, Mg, K, Na, Sr) and transition metals (such as Cr, Co, Cu, Fe, Mn, Mo, Ni, Ti, V, Zn).
2.4 Experimental equipment
The sketched vacuum distillation system is illustrated in Fig.1. The equipment consists of constant temperature oil bath, three-mouth flask with volume of 1.0 L, vacuum distillation section, product collecting bottle and water cooling pump. The heights of refining distillation section, glass spring packing and water cooling pump are 500, 400 and 300 mm, respectively, with the diameter of 25 mm. The HH-9A constant temperature oil bath with temperature range of 0-300 ℃was purchased from Jintan Yitong Electron Limited Corporation, Jiangsu Province, China.

Fig.1 Sketch map of distillation equipment: 1 Constant temperature oil bath; 2 Transformer; 3 Glass spring packing; 4 Heat preservation area; 5 Product collecting bottle; 6 Soft connection; 7 Magnetic valve; 8 Vacuum balance pump; 9 Vacuum buffer bottle; 10 Circulated cooling water; 11 Buffer bottle for pressure measurement; 12 Automation controller; T1 Thermometer at kettle; T2 Thermometer at tower body; T3 Thermometer at tower peak; T4 Thermometer for tail gas
3 Experimental principle
According to Refs.[14-15], the vapor pressure of organic compound can be expressed as
lg p=A-B/(t+C) (1)
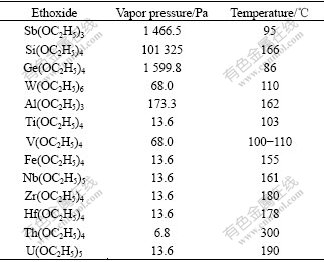
where A, B, and C are constants, t represents temperature. The temperature―vapor pressure curves of C2H5OH, C2H3O2C2H5, Al(OC2H5)3, Nb(OC2H5)5, Ti(OC2H5)4, Ta(OC2H5)5, Si(OC2H5)4 and Zr(OC2H5)4, are presented in Fig.2, and the vapor pressures of the other metal ethoxides are listed in Table 2. The vapor pressure of Ta(OC2H5)5 at 146 ℃ (20.0 Pa) and 202 ℃ (1 333 Pa) were reported in Ref.[15], and Ta(OC2H5)5 begins to decompose when temperature is elevated to above 275 ℃[16]. We measured the vapor pressure values of Ta(OC2H5)5 in order to obtain the temperature―vapor pressure curve in temperature range from 110 ℃ to 230 ℃. According to vapor pressure values obtained, the vapor pressure of Ta(OC2H5)5 can be expressed as
 (2)
(2)

Fig.2 Temperature―vapor pressure curves of metal ethoxides: 1 C2H5OH; 2 C2H3O2H5C2; 3 Si(OC2H5)4; 4 Ti(OC2H5)4; 5 Al(OC2H5)3; 6 Ta(OC2H5)5; 7 Zr(OEt)4; 8 Nb(OC2H5)5
Table 2 Vapor pressure data of metal ethoxides
Consequently, the experiment of vacuum distillation was conducted at 200 ℃ and 1 kPa.
As shown in Fig.2, the boiling point of C2H5OH and C2H3O2H5C2 is about 80 ℃, and they can be removed with atmospheric distillation method. According to Ref.[16], ethoxides of alkalis (except Li) and alkaline-earth metals are generally insoluble, non-volatile compounds and decomposable at higher temperature. Therefore, they remain in residue during vacuum distillation. Ethoxides of group ⅢA, ⅣA, ⅤA elements, such as Sb(OC2H5)3, Si(OC2H5)4, Ge(OC2H5)4 and As(OC2H5)3, have higher vapor pressure value than Ta(OC2H5)5 at desired temperature, so they can be separated from Ta(OC2H5)5 easily by vacuum distillation. Al(OC2H5)3 and Ta(OC2H5)5 have similar vapor pressure values. For ethoxides of metal elements in the same group, such as Ge(OC2H5)4, Sn(OC2H5)4 and Pb(OC2H5)2, their molecular complexity degree increases with atomic number, whereas the volatility decreases. For ethoxides of transition elements, the vapor pressure of Ti(OC2H5)4, W(OC2H5)6 and V(OC2H5)4, is higher than that of Ta(OC2H5)5, and the others have greatly lower vapor pressure values at the same temperature. These non-volatile ethoxides have constant vapor pressure values at lower temperature, and then decompose when temperature is elevated to certain value. For example, uranium pentaethoxide has been found to be thermally stable at 170 ℃ during vacuum distillation, but it decomposes when vacuum distillation temperature is elevated to 180-200 ℃.
In conclusion, the metal ethoxides with lower boiling point can be volatilized into off-gas and removed from tantalum ethoxide by vacuum distillation when distillation temperature and vapor pressure are maintained at about 200 ℃ and 1 kPa, respectively. At the same time, the metal ethoxides with higher boiling point, such as alkali metals, alkaline metals, main group metals, and transition metals, are left in distillation residue, and completely separated from tantalum ethoxide.
4 Results and discussion
The vacuum distillation conditions and tantalum ethoxide yield are presented in Table 3. The contents of impurities in tantalum ethoxide are converted from those in tantalum oxide. The impurity content of samples and Epichem’s requirement for content of impurities are listed in Table 4.
Table 3 Vacuum distillation conditions and tantalum ethoxide yield

Table 4 Impurity contents in tantalum ethoxide and Epichem’s requirement for contents of impurities (%)
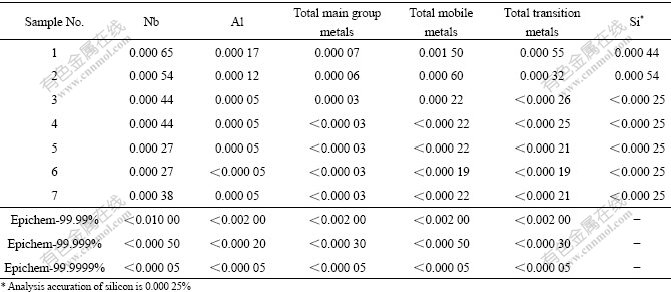
As shown in Table 3 and Table 4, distillation packing and reflux ratio have slight influences on the tantalum ethoxide yield, but they have favorable effects on the purity of tantalum ethoxide. Impurity content of transition metals and mobile metals (mainly as alkali metals) in sample greatly decreases (No.1-4). The possible reason for this phenomena may be as follows: the volatile rate of vacuum distillation without glass packing is larger, and impurity metal ethoxides, Me(OC2H5)n, are taken out by Ta(OC2H5).5 gas. Single ethoxide of alkali or alkaline-earth metals is non-volatile and decomposable during vacuum distillation at high temperature, but double-metal ethoxide (MeTa(OC2H5)6 or Me[Ta(OC2H5)6]2) with high boiling point form in presence of tantalum ethoxide[17]. Consequently, little amount of double-metal ethoxide is volatilized into tantalum ethoxide condensate, and larger amount is left in distillation residue. The ethoxide of transition metals, composed mainly of W(OC2H5)6 and Nb(OC2H5)5, have similar vapor pressure with Ta(OC2H5)5 at distillation temperature. Thereby, it is necessary to rectify tantalum ethoxide sample in order to remove these impurities.
Higher separating efficiency was obtained with larger reflux ratio and more tray number. The preferential hydrolyzation of some impurities occurs with water addition and the purity of tantalum ethoxide increases further, but the tantalum ethoxide yield decreases remarkably (No.5 and No.6). When ethanol recovered from vacuum distillation substitutes for AR grade of anhydrous ethanol during the electrochemical synthesis of tantalum ethoxide, the product has the similar purity with a yield higher than 90% (No.7). The impurity content of tantalum ethoxide obtained under normal distillation conditions (No.4-7), including Al, As, Ca, Co, Cr, Cu, Fe, K, Mg, Mn, Mo, Na, Ni, Pb, Sn, Ti, V and Zn, is less than 0.000 05%. Si and Nb contents are less than 0.000 25% and 0.000 5%, respectively. The content of impurities in tantalum ethoxide sample excels that of Epichem Group’s requirement for Ta(OC2H5)5 of 99.999%. The contents of carbon, hydrogen and tantalum in tantalum ethoxide are 29.5%, 6.1% and 44.6% (the last value, 44.6%, is obtained by the conversion of content of tantalum oxide), respectively. It has the good consistence with the theoretical contents of carbon (29.56), hydrogen (6.16%) and tantalum (44.57%) in tantalum ethoxide compound.
The content of tantalum in No.4 distillation residue is 66.79%. The FTIR spectra of No.4 distillation residue and condensate sample are shown in Fig.3 and Fig.4, respectively. The 1H-NMR spectra of condensate sample and Epichem sample are shown in Fig.5.
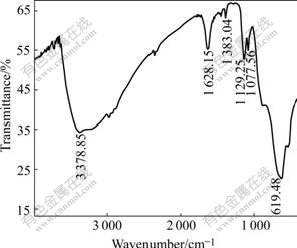
Fig.3 FTIR spectrum of distillation residue
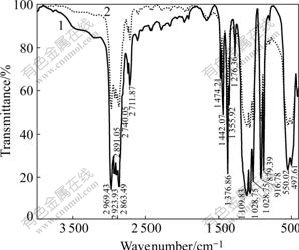
Fig.4 FTIR spectrum of Ta(OC2H5)5 samples: 1 Condensate; 2 Epichem
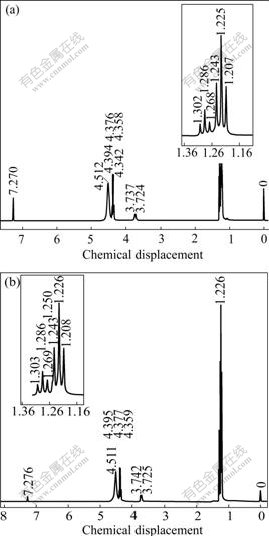
Fig.5 1H-NMR spectra of Ta(OC2H5)5 samples: (a) Epichem; (b) Condensate
As shown in Fig.3, the broad band at 3 379 cm-1 due to Ta―O―H vibration and the sharp band at 619 cm-1 due to Ta―O stretching mode are observed in the FTIR spectrum of distillation residue. This indictes that the hydrolyzation of partial tantalum ethoxide occurs. The band around 1 129―1 077 cm-1 due to Ta―O―R vibration testifies the existence of Ta-OC2H5. Consequently, tantalum presents in the residue in the form of Ta(OC2H5)5-n-2m(OH)nOm according to the tantalum content of distillation residue (66.79%). In order to improve tantalum ethoxide yield and reduce the distillation reside amount, it is necessary to avoid the hydrolyzation of tantalum ethoxide. The main reason for tantalum ethoxide hydrolyzation is the addition of AR grade of ethanol containing about 0.3% water. During electrochemical synthesis, 2-3 kg of anhydrous ethanol is added when 1 kg of tantalum ethoxide is produced, and therefore about 15% of tantalum is transformed to Ta(OC2H5)5-n-2m(OH)nOm (No.2-4). The boiling temperature of Ta(OC2H5)5-n-2m(OH)nOm is much higher than that of tantalum ethoxide, and it will preserve in the residue during vacuum distillation. As a result, the tantalum ethoxide yield can be improved greatly when the ethanol recovered from previous distillation process substitutes for AR grade ethanol during tantalum ethoxide electrochemical synthesis (No.7).
The FTIR spectrum of condensate sample exhibits sharp bands around 2 970, 2 924 and 2 864 cm-1 due to symmetric stretch of the ethoxy ligands and sharp bands around 1 473-1 356 cm-1 due to the δ(CH2) vibrations. Bands at 1 113-879 cm-1 correspond to the Ta―O―C vibrations of ethoxy groups bound to Ta. The presence of two individual bands, instead of one broad band, is indicative of the dimeric nature of ethoxide. The broad envelope of bands below 700 cm-1 is due to the Ta―O stretching modes occurring along with bending and torsional modes of the ligand.
As shown in Fig.5, the 1H-NMR spectrum of condensate sample is identical with that of high purity tantalum ethoxide (99.999%) purchased from Epichem Group with same analysis methods. Raman spectrum of condensate sample is consistent with Ref.[18]. In conclusion, the condensate of vacuum distillation can be clearly determined as tantalum ethoxide.
5 Conclusions
1) The condensate obtained from vacuum distillation was characterized by FTIR, Raman spectra and 1H-NMR spectroscopy. The contents of tantalum, carbon and hydrogen in the sample were also determined by elemental analysis instrument. The results consistently demonstrate that the condensate is tantalum ethoxide.
2) Distillation packing and reflux ratio have favorable effects on the purity of tantalum ethoxide. The preferential hydrolyzation of some impurities occurs with water addition and the purity of tantalum ethoxide increases further, but the tantalum ethoxide yield decreases remarkably. When ethanol recovered from distillation substitutes for AR grade of anhydrous ethanol during the electrochemical synthesis of tantalum ethoxide, the product has the similar purity with a yield higher than 90%.
3) The tantalum content of product is 44.6%. The impurity content of tantalum ethoxide, including Al, As, Ca, Co, Cr, Cu, Fe, K, Mg, Mn, Mo, Na, Ni, Pb, Sn, Ti, V, Zn, is less than 0.000 05%. Si and Nb contents are less than 0.000 25% and 0.000 5%, respectively. The content of impurities in tantalum ethoxide sample excels that of Epichem Group’s requirement for Ta(OC2H5)5 of 99.999%.
References
[1] OZER N, LAMPERT C M. Structural and optical properties of sol-gel deposited proton conducting Ta2O5 films [J]. Journal of Sol-Gel Technology, 1997, 8(1/3): 703-709.
[2] MIKKO R, KAUPO K, ANTTI R, RAISANEN P I, LESKELA M, SAJAVAARA T, KEINONEN J. Atomic layer deposition of oxide thin films with metal alkoxides as oxygen sources [J]. Science, 2000, 288(5464): 319-321.
[3] DONGJOON M, SUNGHO P, BUM-SEOK S, CHOI S, LEE N, LEE J H. Low temperature crystallization of high permittivity Ta oxide using an Nb oxide thin film for metal/insulator/metal capacitors in dynamic random access memory applications [J]. Journal of Vacuum Science and Technology B, 2005, 23(1): 80-83.
[4] KERN W, CHEN A, SANDLER N. MOCVD of tantalum pentoxide for large-area ULSI circuit wafers [J]. Journal de Physique IV, 1991, C2(Suppl): 311-317.
[5] CAVA R F, PECK W F, KRAJEWSKI J J. Enhancement of the dielectric constant of Ta2O5 through substitution with TiO2 [J]. Nature, 1995, 377(6546): 215-217.
[6] WANG Wen, JIA De-chang, ZHOU Yu, YE Feng. Synthesis and characterization of nanosized SrBi2Ta2O9 powder by a novel sol-gel process [J]. Materials Research Bulletin, 2002, 37(15): 2517-2524.
[7] CHENG Z X, KIMURA H, OZAWA K, MIYAZAKI A, KANNA C V. Ferroelectric lithium tantalate thin film derived from peroxide [J]. Journal of Alloys and Compounds, 2005, 402(1/2): 208-212.
[8] YANG Sheng-hai, WANG Yi-nan, HE Jing, TANG Mo-tang, QIU Guan-zhou. Preparation of high-purity tantalum oxide nanoparticles [J]. Rare Metal Materials and Engineering, 2007, 36(2): 282-286. (in Chinese)
[9] MEHROTRA R C. Synthesis and reactions of metal alkoxides [J]. Journal of Non-crystalline Solids, 1988(100): 1-15.
[10] FRIEDRICH Z. Process for preparing tantalum alkoxides and niobium alkoxides [P]. US 6548685, 2003-4-15.
[11] VECCHIO-SADUS A M. The electrochemical synthesis of inorganic and organometallic complexes in non-aqueous media [J]. Journal of Applied Electrochemistry, 1993, 23(5): 401-416.
[12] SHINDO Y, YAMAGUCHI S. High purity tantalum for thin film formation and its productions [P]. JP 2000212678, 2000-8-2.
[13] YANG Sheng-hai, PAN Ze-qiang, LI Zhao-hui, TANG Mo-tang, QIU Guan-zhou. Electrochemical synthesis of tantalum ethoxide using sacrificing anode [J]. Rare Metal Materials and Engineering, 2006, 35(4): 625-628. (in Chinese)
[14] LIDE D R, MILNE A G W. Handbook of data on common organic compounds(Ⅱ) [M]. Florida: CRC Press, 1995: 2131.
[15] BRANDLEY D C, MEHROTRA R C, GAUR D P. Metal alkoxide [M]. New York: Academic Press, 1978: 42-74.
[16] KUKLI K, RITALA M, LESKEL? M. Atomic layer epitaxy growth of tantalum oxide thin films from Ta(OC2H5)5 and H2O [J]. Journal of the Electrochemical Society, 1995, 142(5): 1670-1674.
[17] REN Xu. The challenge of precursor compounds in the MOCVD of oxides [J]. JOM, 1997, 49(10): 1-9.
[18] NAKAMOTO N. Infrared and Raman spectra of inorganic and coordination compounds [M]. New York: John Wily, 1978: 613.
Foundation item: Project(50404011) supported by the National Natural Science Foundation of China; Project(20040350187) supported by the Postdoctoral Science Foundation of China; Project(2007AA03Z425) supported by the Hi-Tech Research and Development Program of China
Corresponding author: YANG Sheng-hai; Tel: +86-731-8830470; E-mail: 75894838@163.com
(Edited by YANG Bing)