
Preparation and properties of K2NiF4-type perovskite oxides La2NiO4 catalysts for steam reforming of ethanol
Zhang Li-feng(张利峰), Wang Yi-ping(王一平), Huang Qun-wu(黄群武)
School of Chemical Engineering and Technology, Tianjin University, Tianjin 300072, China
Received 10 August 2009; accepted 15 September 2009
Abstract: The performance of La2NiO4 perovskite catalysts, prepared using a citric acid complexation method, for the steam reforming of ethanol was studied. The catalysts were characterized by X-ray diffractometry (XRD), specific surface area measurements (BET), thermogravimetric analysis (TGA) and scanning electron microscopy (SEM). The experimental results show that the calcination temperature and the amount of citric acid (CA) have a significant influence on the characteristics of the catalysts and their catalytic activity. Among the catalysts tested, the La2NiO4 catalyst calcined at 700 ℃ with n(La)?n(Ni)?n(CA) of 2?1?3 exhibits the best activity and excellent stability as well as very low coke formation.
Key words: hydrogen; ethanol; perovskite-like oxides; La2NiO4
1 Introduction
In the wake of the growing demand for carbon- neutral and zero-emission alternative fuels, considerable attention has been paid to transform the biomass-derived compounds into hydrogen-rich gas. Of special interest among such compounds is bioethanol obtained by biomass fermentation, in view of the increasing availability of raw material, ease of handling in the liquid state, high hydrogen content and low toxicity[1-3]. The biomass-derived ethanol, an aqueous solution containing 12%-15% ethanol, requires the distillation for direct use. Hydrogen manufacture by the catalytic steam reforming of ethanol without the distillation can thus be considered to be one of the most effective ways for the utilization of the biomass-derived ethanol. Catalysts using a wide variety of transition metals and supports have previously been reported for the steam reforming of ethanol[4-8]. Among the catalysts reported so far, rhodium, cobalt and nickel are considered to be the most promising metals[9]. Although rhodium seems to be most active, the use of noble metal is considered to be economically unfavorable. On the other hand, more inexpensive nickel catalysts often suffer from catalyst deactivation due to coke formation and sintering. One promising method for the inhibition of carbon deposition over Ni catalysts is using perovskite-type oxides. CHOUDHARY et al[10] reported that complex oxides with a perovskite structure, like LaNiO3, La0.8Ca(or Sr)0.2NiO3 and LaNi1-xCoxO3 (x=0.2-1.0), were resistant to coking for the partial oxidation of methane to synthesis gas. To inhibit carbon deposition, one must keep the size of the metal clusters smaller than the critical size needed for coke formation. In this work, the Ni catalysts from K2NiF4-type perovskite oxides La2NiO4 exhibit high activity and a rather high stability. The effects of calcination temperature and citric acid concentration on the structure and the catalytic activity for the steam reforming of ethanol are studied.
2 Experimental
2.1 Catalyst preparation
The La2NiO4 catalysts were prepared by a citric acid complexation method. Stoichiometric amounts of Ni(NO3)2・6H2O and La(NO3)2・6H2O were dissolved completely in deionized water, and a stoichiometric amount of citric acid was mixed into the solution. The solution was then heated to 80 ℃ with constant stirring until a gel was formed. The gel was then dried at 110 ℃ for 12 h, and subsequently calcined at different temperatures for 5 h. The calcined samples were pressed, crushed and sieved to obtain a particle size of 0.40- 0.50 mm.
2.2 Catalyst characterization
The powder X-ray diffraction (XRD) experiments were carried out on Philips PANalytical apparatus with Co Kα radiation, at 40 kV and 40 mA. The diffraction angle (2θ) was scanned from 10? to 90?. Specific areas were calculated using the BET method from the nitrogen adsorption isotherms, recorded at the temperature of liquid nitrogen on a NOVA-2000 instrument. The thermogravimetric analysis (TGA) was carried out under an oxidative atmosphere with a Pyris Diamond Analyzer. About 10 mg of sample was heated from room temperature to 850 ℃ at 10 ℃/min. Scanning electron microscopy (SEM) was performed with a PHILIPS XL30 scanning electron microscope operating at 20 kV.
2.3 Catalytic tests
Steam reforming tests were performed in a fixed-bed flow quartz tubular reactor (12 mm of inner diameter) operating at atmospheric pressure. The catalyst (0.10 g) was first subjected to in situ reduction under pure hydrogen flow (3 mL/min) at 500 ℃ for 1 h. Following reduction, hydrogen was replaced by argon (20 mL/h) and temperature was adjusted to reaction temperature. The liquid ethanol-water solution was fed by means of a syringe pum into a heating chamber (150 ℃) and vaporized completely in the stream of argon. The ethanol feed rate was 5.8×10-5 mol/min in all tests and the ethanol-to-water-to-argon molar ratio of 1?9?14.3 was used. The gas products were analyzed on-line by gas chromatography using both flame ionization and thermal conductivity detector. Results were expressed in terms of molar composition in the gas phase for H2, CO2, CO, and CH4.
3 Results and discussion
3.1 Influence of calcination temperature
The XRD patterns of La2NiO4 samples calcined at different temperatures are shown in Fig.1. After the amorphous precursor was calcined at 500 ℃ for 4 h, intense peaks for La2O3 were observed. When the temperature was increased to 700 ℃, typical diffraction peaks for the perovskite-like oxide La2NiO4, which was identified as single phase with a tetragonal K2NiF4 structure, as well as a small amount of La2O3 were observed. When the calcination temperature was increased to 850 ℃, the diffraction peaks for the spinel structure significantly intensified and became sharper, which suggests that the crystalline phase of La2NiO4 became more perfect. This result indicates that La2NiO4 with pure spinel structure can be formed by complexing citric acid with a mixture of La3+ and Ni2+ and calcining the obtained gel above 700 ℃. The specific surface areas and crystal sizes of the La2NiO4, calculated from the half-width of a diffraction peak using Scherrer formula, are listed in Table 1. Upon increasing calcination temperature, the particle sizes increased, and the specific surface area decreased dramatically. The drop in specific surface area may originate from the aggregation of the perovskite phase, which indicates the structure properties are significantly dependent on the calcination temperature.
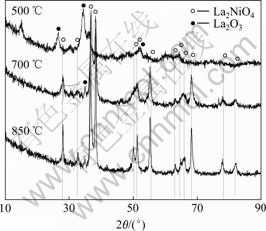
Fig.1 XRD patterns of La2NiO4 calcined at different temperatures
Table 1 Specific surface area and crystal size of La2NiO4 calcined at different temperatures

Steam reforming tests were performed at 500 ℃ and 650 ℃ over La2NiO4 catalysts calcined at different temperatures. Total ethanol conversion took place all along the 3 h runs at both reaction temperatures. The typical gaseous products H2, CO2, CO and CH4 were obtained in all cases while other common intermediates or by-products like ethylene, diethylether and acetaldehyde were detected only in negligible amounts (<0.10%). Table 2 shows the typical molar composition of the gaseous mixture obtained with the La2NiO4 catalysts calcined at different temperatures. For every catalyst, the H2 content was found to increase with temperature. At 650 ℃, it was found to be 70.3%- 71.6%, approaching the theoretical reaction stoichiometry (H2: 75%). Also in agreement with stoichiometry, the H2-to-CO2 molar ratio was found to approach 3.0 for the catalysts tested at 650 ℃. In contrast, the CH4 molar fraction showed a decreasing trend, which is consistent with a higher reaction rate of the reforming reaction(CH4+2H2O→CO2+4H2), which is favored both thermodynamically and kinetically by the temperature increment. The CO and CO2 contents were found to have opposite trends with increasing temperature, which is consistent with the temperature dependence of the thermodynamic equilibrium constant of the exothermic water gas shift reaction (WGSR) (CO+H2O→CO2+H2). Ni is known as a not very active catalyst for the WGSR[11]. Thus, under the experimental conditions here the CO-to-CO2 molar ratios used could be far from the equilibrium values and more dependent on kinetics parameters and textural properties of La2NiO4 catalysts. Thus, these La2NiO4 catalysts could be explained by differences in the influence of textural properties on the rate of the direct and reverse WGSR.
Table 2 C2H5OH reforming using La2NiO4 catalysts calcined at different temperatures

By comparing the performance of catalysts at the same reforming temperature, the catalyst La2NiO4 (700 ℃) led to a higher H2 content (71.6%) and a lower CH4 content (0.10%) in the gaseous mixture than the catalysts La2NiO4 (500 ℃) and La2NiO4 (85 ℃) at 650 ℃. This is ascribed not only to a higher rate of the CH4 reforming reaction being linked to a higher surface specific area, but also to that being closely linked to the smaller La2NiO4 crystal size in the case of La2NiO4 (700 ℃).
3.2 Influence of amount of citric acid
Citric acid (CA) is used as a chelating reagent in the synthesis of the La2NiO4 powders. The amount of citric acid plays an important role in the morphology and activity of the catalysts. If the amount of citric acid is too low, some of the ions may not chelate with the citric acid and resulting gel may not be homogeneous. On the other hand, too much citric acid causes waste and precipitation from unchelated citrate. The SEM micrographs of the catalysts with different molar ratios of La to Ni to CA are shown in Fig.2. An homogeneous phase is visible when molar ratios are 2?1?1 and 2?1?3. Thus, we propose that this technique is a good and convenient method to disperse the active metal. While the catalyst powders with n(La)?n(Ni)?n(CA) of 2?1?4 have the relatively large average granularity due to significant agglomeration of fine powders.

Fig.2 SEM images of La2NiO4 catalysts prepared with different n(La)?n(Ni)?n(CA): (a) 2?1?1; (b) 2?1?3; (c) 2?1?4
Fig.3 shows the XRD patterns of the La2NiO4 catalysts calcined at 700 ℃ with different molar ratios of La to Ni to CA. All these catalysts exhibited the characteristic diffraction peaks for the La2NiO4 spinel structure. Weak diffraction lines of NiO were also observed in the catalysts with n(La)?n(Ni)?n(CA)=2?1?1 and n(La)?n(Ni)?n(CA)=2?1?4. This phenomenon indicates that the optimal amount of citric acid needed to obtain a pure perovskite structure of La2NiO4 is n(La)?n(Ni)?n(CA)=2?1?3.
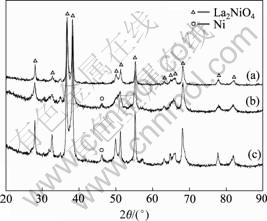
Fig.3 XRD patterns of La2NiO4 prepared with different n(La)?n(Ni)?n(CA): (a) 2?1?3; (b) 2?1?1; (c) 2?1?4
The surface areas of fresh and used La2NiO4 catalysts prepared with different amounts of citric acid are listed in Table 3. As the amount of citric acid increased from n(La)?n(Ni)?n(CA)=2?1?1 to 2?1?4, the specific surface areas of the fresh catalysts decreased from 7.78 to 6.48 m2/g. After the ethanol steam reforming reaction, the specific surface area for n(La)?n(Ni)?n(CA)=2?1?1 decreased dramatically, and that for n(La)?n(Ni)?n(CA)=2?1?4 decreased slightly, but the specific surface area for n(La)?n(Ni)?n(CA)=2?1?3 was almost the same. This suggests that the La2NiO4 catalyst with n(La)?n(Ni)?n(CA)=2?1?3 has the most stable structure.
Table 3 Specific surface area of La2NiO4 prepared with different n(La)?n(Ni)?n(CA)

The conversion of C2H5OH and the content of products at 500 ℃ and 650 ℃ over La2NiO4 with different molar radios of La to Ni to CA were studied and the results are shown in Table 4. Total ethanol conversion took place all along the 3 h runs at both reaction temperatures. The typical gaseous products H2, CO2, CO and CH4 were obtained in all cases while other common intermediates or by-products like ethylene, diethylether and acetaldehyde were detected only in negligible amounts (<0.10%). For every catalyst, the H2 content was found to increase with temperature. By comparing the performance of catalysts at the same reforming temperature, the catalyst La2NiO4 with n(La)?n(Ni)?n(CA)=2?1?3 led to a higher H2 content (71.6%) and a lower CH4 content (0.10%) in the gaseous mixture than the catalysts La2NiO4 with n(La)?n(Ni)?n(CA)=2?1?1 and 2?1?4 at 650℃. This is ascribed to a higher rate of the CH4 reforming reaction linked to a higher active metal dispersion degree seen from Fig.2.
Table 4 C2H5OH reforming using La2NiO4 catalysts prepared with different n(La)?n(Ni)?n(CA)

3.3 Stability of La2NiO4 catalyst
Fig.4 shows the conversion of CH4, and the content of H2, CO, CH4 and CO2 as a function of time on stream at 500 ℃ with the ethanol feed rate of 5.8×10-5 mol/min and the ethanol-to-water-to-argon molar ratio of 1?9?14.3 over a La2NiO4 catalyst calcined at 700 ℃ with n(La)?n(Ni)?n(CA)=2?1?3. It is clear that during the first 55 h reaction ethanol was completely converted. When the operation time was longer than 56 h, the ethanol conversion started to decrease slightly and arrived about 97.5% at 80 h, and the selectivities of H2, CO, CH4 and CO2 were stable at around 67.5%, 1.44%, 2.73% and 28.3%. Meanwhile, other common intermediates or by-products like ethylene, diethylether and acetaldehyde were detected only in negligible amounts (<0.10%) in the whole 80 h, which suggests that the La2NiO4 catalysts are quite stable and catalytically highly active. In agreement with these results, Ni-La catalysts also showed constant activity under the same reforming conditions[12].
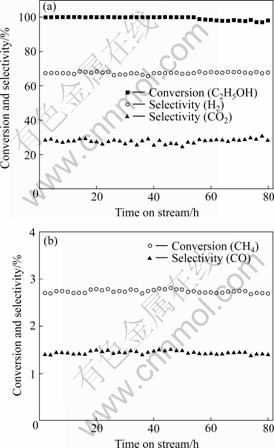
Fig.4 Stability of La2NiO4 catalyst for ethanol steam reforming reaction at 500 ℃
The X-ray diffraction patterns of the fresh, reduced and used La2NiO4 catalysts are shown in Fig.5. For the fresh La2NiO4 catalyst, typical diffraction peaks for the La2NiO4 spinel structure were observed. After the catalyst was reduced by hydrogen at 500 ℃ for 1 h, most of the diffraction peaks attributed to La2NiO4 disappeared, whereas those belonging to La2O3 appeared. And nickel existed chiefly as Ni0, and the average crystal size of Ni0 (21 nm) for La2NiO4 (after reduction), obtained from the XRD line broadening, was found to be much smaller than that (125 nm) for Ni-La2O3[13]. This suggests that the La2NiO4 has been reduced and decomposed by hydrogen to form nanoscale Ni particles, which are segregated by La2O3. LIU and AU[14] suggested that La2O3 can prevent transition metals from agglomeration and promote the dispersion of nanoscale Ni0 particles, resulting in an enhancement of catalytic activity and stability. After reaction, the catalysts showed similar patterns with the results of ZHANG and VERYKIOS[15]. The La2O3 phase that existed in the reduced catalysts disappeared, and the La2O2CO3 phase was formed due to the adsorption of CO2 on La2O3 (CO2+La2O3→La2O2CO3). The La2O2CO3 phase that existed in the used La2NiO4 catalyst was mainly hexagonal. Ni0 particles were not observed, thus, Ni0 particles might be present in an amorphous form or they were highly dispersed. ZHANG and VERYKIOS[15] have noted that the carbon species formed on the Ni sites were easily removed by the oxygen species originating from La2O2CO3 (La2O2CO3+C*→La2O3+2CO+*), thus producing an active and stable catalyst due to the existence of synergetic sites which consist of Ni and La elements. As indicated in this experiment, La2O2CO3 may play a crucial role in the ethanol steam reforming reaction using La containing catalyst prepared from perovskite precursors.

Fig.5 X-ray diffraction patterns of La2NiO4: (a) After calcination at 850 ℃; (b) After 1 h reduction at 500 ℃; (c) After reaction
The TG/DTG curves for the La2NiO4 catalyst after ethanol steam reforming reaction at 500 ℃ for 80 h are shown in Fig.6. The DTG curve of the used catalyst distinctly indicated a mass loss due to the removal of carbon. Three DTG features were observed at 363, 513 and 700 ℃. This indicated that at least three kinds of carbon depositions were formed on the La2NiO4. One of the carbon depositions was likely attributed to La2O2CO3,

Fig.6 TG/DTG profiles of La2NiO4 catalysts after ethanol steam reforming reaction at 500 ℃ for 80 h
which had been verified by XRD. La2O2CO3, formed by the interaction of La2O3 with CO2, may decompose into CO and produce oxygen species, which react with the surface carbon species on the Ni sites, thus giving active and stable catalytic performance for the ethanol steam reforming reaction, indicating La2NiO4 catalyst has higher résistance to coke formation, as well as higher catalytic stability as shown in Fig.4 and Fig.6.
4 Conclusions
1) Better catalytic performances can be achieved using the perovskite La2NiO4 as catalyst precursor. Among the catalysts tested, the catalyst La2NiO4 prepared with n(La)?n(Ni)?n(CA)=2?1?3 calcined at 700 ℃ exhibits the best activity with excellent stability.
2) The XRD results confirm that La2NiO4 exhibits a typical spinel structure.
3) During the ethanol steam reforming reaction, the active Ni0 particles do not aggregate and sinter, and the catalyst shows good stability as well as higher resistance to coke formation within an on-stream time of 80 h.
References
[1] FRENI S, CAVALLARO S, MONDELLO N, SPADARO L, FRUSTERI F. Steam reforming of ethanol on Ni/MgO catalysts: H2 production for MCFC [J]. Journal of Power Sources, 2002, 108(1/2): 53-57.
[2] BREEN J P, BURCH R, COLEMAN H M. Metal-catalysed steam reforming of ethanol in the production of hydrogen for fuel cell applications [J]. Applied Catalysis B: Environmental, 2002, 39(1): 65-74.
[3] VIZCA?NO A J, CARRERO A, CALLES J A. Hydrogen production by ethanol steam reforming over Cu-Ni supported catalysts [J]. International Journal of Hydrogen Energy, 2007, 32(10/11): 1450-1461.
[4] LIGURAS D K, KONDARIDES D I, VERYKIOS X E. Production of hydrogen for fuel cells by steam reforming of ethanol over supported noble metal catalysts [J]. Applied Catalysis B: Environmental, 2003, 43(4): 345-354.
[5] KUGAI J, SUBRAMANI V, SONG C, ENGELHARD M H, CHIN Y H. Effects of nanocrystalline CeO2 supports on the properties and performance of Ni-Rh bimetallic catalyst for oxidative steam reforming of ethanol [J]. Journal of Catalysis, 2006, 238(2): 430-440.
[6] BATISTA M S, SANTOS R K S, ASSAF E M, ASSAF J M, TICIANELLI E A. Characterization of the activity and stability of supported cobalt catalysts for the steam reforming of ethanol [J]. Journal of Power Sources, 2003, 124(1): 99-103.
[7] FATSIKOSTAS A N, VERYKIOS X E. Reaction network of steam reforming of ethanol over Ni-based catalysts [J]. Journal of Catalysis, 2004, 225(2): 439-452.
[8] SHENG P Y, YEE A, BOWMAKER G A, IDRIS H. H2 production from ethanol over Rh-Pt/CeO2 catalysts: The role of Rh for the efficient dissociation of the carbon-carbon bond [J]. Journal of Catalysis, 2002, 208(2): 393-403.
[9] HARYANTO A, FERNANDO S, MURALI N, ADHIKARI S. Current status of hydrogen production techniques by steam reforming of ethanol: A review [J]. Energy Fuels, 2005, 19(5): 2098-2106.
[10] CHOUDHARY V R, UPHADE B S, BELHEKAR A A. Oxidative conversion of methane to syngas over LaNiO3 perovskite with or without simultaneous steam and CO2 reforming reactions: Influence of partial substitution of La and Ni [J]. Journal of Catalysis, 1996, 163(2): 312-318.
[11] AUPRETRE F, DESCORME C, DUPREZ D, CASANAVE D, UZIO D. Ethanol steam reforming over MgxNi1-xAl2O3 spinel oxide- supported Rh catalysts [J]. Journal of Catalysis, 2005, 233(2): 464-477.
[12] SUN Jie, QIU Xin-ping, WU Feng, ZHU Wen-tao. H2 from steam reforming of ethanol at low temperature over Ni/Y2O3, Ni/La2O3 and Ni/Al2O3 catalysts for fuel-cell application [J]. International Journal of Hydrogen Energy, 2005, 30(4): 437-445.
[13] CHOUDHARY V R, RANE V H, RAJPUT A M. Selective oxidation of methane to CO and H2 over unreduced NiO-rare earth oxide catalysts [J]. Catalysis Letter, 1993, 22(4): 289-297.
[14] LIU B S, AU C T. Sol-gel-generated La2NiO4 for CH4/CO2 reforming [J]. Catalysis Letters, 2003, 85(3/4): 165-170.
[15] ZHANG Z, VERYKIOS X E. Carbon dioxide reforming of methane to synthesis gas over Ni/La2O3 catalysts [J]. Applied Catalysis A: General, 1996, 138(1): 109-133.
Corresponding author: Huang Qun-wu; Tel: +86-22-27404771; E-mail: huangqw@tju.edu.cn
DOI: 10.1016/S1003-6326(09)60048-0
(Edited by YANG Bing)