
ARTICLE
J. Cent. South Univ. (2019) 26: 1573-1581
DOI: https://doi.org/10.1007/s11771-019-4113-8
Corrosion behavior of dissimilar copper/brass joints welded by friction stir lap welding in alkaline solution
Kamran AMINI1, 2, Farhad GHARAVI3
1. Department of Mechanical Engineering, Tiran Branch, Islamic Azad University, Isfahan, Iran;
2. Center for Advanced Engineering Research, Majlesi Branch, Islamic Azad University, Isfahan, Iran;
3. Department of Materials Engineering, Sirjan Branch, Islamic Azad University, Sirjan, Iran
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: This study was done to evaluate the nugget zone (NZ) corrosion behavior of dissimilar copper/brass joints welded by friction stir lap welding (FSLW) in a solution of 0.015 mol/L borax (pH 9.3). To this end, dissimilar copper/ brass plates were welded with two dissimilar heat inputs (low and high) during the welding procedure. The high and low heat inputs were conducted with 710 r/min, 16 mm/min and 450 r/min, 25 mm/min, respectively. Using open circuit potential (OCP) measurements, electrochemical impedance spectroscopy (EIS) and Tafel polarization tests, the electrochemical behavior of the specimens in borate buffer solution was assessed. With the help of scanning electron microscope (SEM), the morphology of welded specimen surfaces was examined after immersion in the test solution. According to the results, the NZ grain size and resistance improvement reduced due to the nugget zone corrosion with a decreased heat input. The results obtained from Tafel polarization and EIS indicated the improved corrosion behavior of the welded specimen NZ with a decrease in the heat input during the welding process unlike the copper and brass metals. Furthermore, an increased heat input during the welding process shows a reduction in the conditions for forming the passive films with higher protection behavior.
Key words: friction stir welding; copper; brass alloy; corrosion behavior; alkaline solution; dissimilar joint
Cite this article as: Kamran AMINI, Farhad GHARAVI. Corrosion behavior of dissimilar copper/brass joints welded by friction stir lap welding in alkaline solution [J]. Journal of Central South University, 2019, 26(6): 1573-1581. DOI: https://doi.org/10.1007/s11771-019-4113-8.
1 Introduction
Due to the high thermal conductivity, good mechanical workability and good corrosion resistance, copper and its alloys have been widely used as condenser and heat exchanger tubing materials in power plants and other manufacturing facilities [1]. In general, it is difficult to join copper and brass by conventional fusion welding approaches due to formation of cracks and porosities, sever deformation, and high residual stress as well as alloying element evaporation during the fusion welding [2, 3]. Friction stir welding (FSW), as a new solid state joining process, is nowadays used for joining copper and its alloys to prevent the evaporation of alloying element and to provide extremely high heat inputs required for fast heat radiation as well as to diminish the strength loss in the welding zone [2-4]. As one of the most significant copper alloys, brass is extensively used for its proper corrosion properties. However, brass has a comparatively high corrosion resistance rate in diverse aggressive media. Thus, it is used in shipboard condensers and heat exchangers [5, 6]. In spite of good corrosion resistance, brass seems to be vulnerable to corrosion in chloride and sulphate media [5, 7]. Many studies have proven a suitable corrosion resistance for copper and its alloys because of the passive film stability in alkaline solutions. In general, copper and its alloys appear to be the widely used multi-component construction materials for chemical and petrochemical industries [8-12]. In fact, numerous factors such as pH, aerating conditions, aggressive anions and immersion time affect the composition of the passive films formed on copper and its alloys in aqueous solutions [9]. Obviously, this passive film forms an effective shield against the metal dissolution. The electrochemical behavior of copper and zinc in alkaline solutions has been studied [13, 14].
There are also reports on the electrochemical behavior of Cu-Zn alloys [15, 16]. Many studies have focused on the FSW of low melting point metals such as Al and Mg alloys, whereas the impact of this welding technique on copper and its alloys has been rarely studied. Indeed, the electrochemical and corrosion properties can be significantly influenced by diverse factors such as residual stress and grain size [13]. Corrosion resistance of friction stir welded materials formed during welding at various zones is not similar, due to the obvious microstructure gradient caused by the storage heating effect and plastic deformation during FSW. Therefore, FSW can affect the corrosion performance of copper and its alloys welds. Yet, little research has been done on the corrosion behavior of the friction stir welded copper/brass metals. Therefore, this study aims to investigate the heat input effect on the electrochemical behavior of the welded joints nugget zone (NZ) in 0.015 mol/L borax solution (pH 9.3) during the welding process using Tafel polarization, and electrochemical impedance spectroscopy (EIS) analysis. The scanning electron microscope (SEM) was used to examine the surface corrosion morphology of welded joints NZ.
2 Experimental procedure
In this research, pure copper and brass plates with 220 mm in length, 140 mm in width, and 5 mm in thickness were welded by friction stir lap welding (FSLW) approach. Tables 1 and 2 show the chemical composition of pure copper and brass plates.
Pure copper plate was produced by cold rolling under DIN EN1652 standard condition, and the brass plates with DIN 17660 standard condition were produced by hot rolling. Before the welding process, based on annealing temperature of copper and its alloys and thickness of plates, all the copper and brass components were annealed at 500 ��C, holding for 30 min, and then cooled in the furnace. A tool made of DIN 1.2344 hot working tool steel was used with a geometry given in Figure 1. The welding tool was subjected to oil quenching and three-stage tempering. An automatic CNC machine performed the welding experiments, while the joint design was selected to produce lap welds. Figure 2 shows the schematic representation of the welding process and the joint design. The brass and copper plates were respectively put in advancing and retreating sides in the joint design. The welding tool key dimensions are summarized in Table 3. The welding direction was the normal of the plates rolling direction. A tilt angle of 3��and a plunge depth of 0.2 mm were applied to the FSLW tool through the FSLW. All welded samples were welded at two welding heat inputs, denoted as FSLW1 with a low heat input (450 r/min, 25 mm/min); FSLW2 with a high heat input (710 r/min, 16 mm/min).
Table 1 Copper plate chemical composition (mass fraction, %)
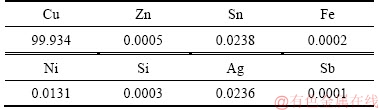
Table 2 Brass plate chemical composition (mass fraction, %)
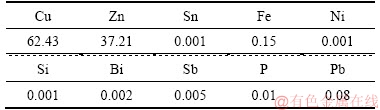

Figure 1 Shape of welding tool
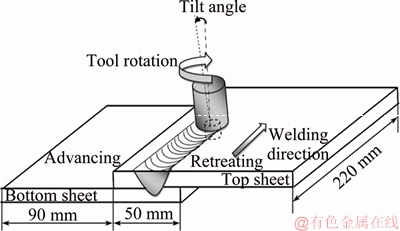
Figure 2 Schematic of friction stir lap welding process and joint design
Table 3 Welding tool key dimensions
Using a conventional three-electrode electrochemical glass cell, the electrochemical measurements were made by a computer to control the Ivium System (model: compactstas. h standard). The cell was opened to the air and the measurements were made at the ambient temperature. Each set of working electrodes, including the NZ and base metals (BM) of copper and brass specimens, were connected to a copper wire, and sealed with epoxy resin with a 1 cm2 exposure area for all specimens. A graphite rode and the saturated calomel electrode (SCE) were used as the counter rode and the reference rode, respectively.
During the measurements, the solution was not stirred. Using abrasive SiC papers from 600 to 1200 grades, the exposed surface of each specimen was ground. Then, it was mechanically polished with 1 ��m diamond paste, rinsed with double distilled water, and finally, degreased with ethanol. The degreased electrodes were dipped in the concentrated HNO3 for 30 s. Following the procedure, they were rinsed with deionized water and then inserted into the alkaline solution of 0.015 mol/L borax solution (pH 9.3). To form a steady state passive film prior to all electrochemical tests, the specimens were immersed in open circuit potential (OCP) for 3600 s in the test solution. In the Tafel polarization tests, the potential was scanned from -0.25 V to 1.10 V regarding the OCP at scan rate of 1 mV/s. The EIS test was performed at OCP and AC potential with a 10 mV amplitude and a frequency range of 100 kHz to 10 MHz to investigate the specimens' passivation in the alkaline solution. The Zsimp 3.2 impedance software was used for the EIS modeling and curve-fitting method.
The average microhardness of pure copper and brass and the NZ of welded joints were calculated using the Buehler microhardness measurement set up (model: Wilson, VH1150). To this end, the specimens were grounded to 2000 grit, and then cleaned with deionized water. Vickers hardness was measured with load of 50 g for 10 s. Using the Clemex software, average grain size measurement was done across the welded joints nugget zone according to ASTM E-112. Furthermore, the specimens' corroded surface morphology was examined under a JEOLJSM- 840A scanning electron microscope.
3 Results and discussion
3.1 Grain size and microhardness
Figure 3 illustrates the average Vickers hardness distribution and the grain size throughout the nugget zone of welded joints. As stated in Figure 3, the hardness of the welded joints is higher than that of copper and brass metals.
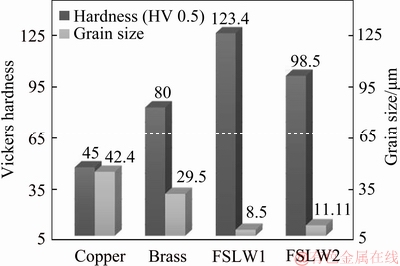
Figure 3 Average Vickers hardness distribution and grain size across nugget zone of welded joints
In addition, the highest level of hardness can be provided in the welded specimen nugget zone with using a low heat input during the welding process. Thus, enhanced heat input increases the average grain size and decreases the hardness at the nugget zone of welded joints. It should be noted that the hardness value is closely related to the grain size. With a decreasing heat input, the grain refined meaningfully in the welded joints NZ. While the welding heat input enhances, the hardness decreases in the NZ of the FSLW2 sample and the grain size increases due to the low cooling rate after the welding process. The cooling rate leads to increment of grain size by grain growth mechanism [17-19].
In this regard, the studies suggest that corrosion resistance is improved and grain size decreases, and it is generally demonstrated that fine grain microstructure indicates decrease of corrosion susceptibility because of enhanced passivation kinetics and a more stable passive film motion [20, 21]. Therefore, the highest hardness is found in the NZ at welding condition with a low heat input.
3.2 Open circuit potential
Figure 4 presents Eocp data stated for pure copper, brass alloy and the two nugget zones of welded copper/brass in the as-prepared borate buffer solution for 3600 s. Upon specimen, immersion prior to the steady state rate, the Eocp curves of Cu and the welded specimens NZ showed reduction. At first, a sharp decrease is seen in the Eocp curve of welded specimens, and subsequently, there is an increase in the Eocp to a value near to that for pure copper. The decrease of Eocp is an outcome of oxide film breakdown. Yet, the Eocp turns out to be more positive with time after around 200 s for all the specimens. This indicates the passive film formation and its role in increasing the productivity over time [7, 22]. In addition, the brass specimen's Eocp goes clearly toward positive values at the start of immersion. This will result in enabling the potential as the copper content enrichment on the surface of brass for its dezincification. Moreover,according to Figure 4, the welded specimen's Eocp tends to shift to positive values with a decrease in the heat input during the welding process. Consequently, the welded specimens show bigger and lesser tendencies to corrode thermodynamically in comparison with pure copper and brass alloy, respectively. Furthermore, the change of microstructure during the welding process strongly affects the stability and nature of passive film formed on the welded specimens.
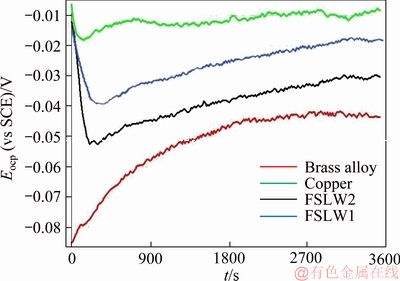
Figure 4 Eocp data of Cu, brass alloy and welded copper/brass nugget zones in 0.015 mol/L borax (pH 9.3) for a time period of 3600 s experiment
3.3 Polarization behavior
Figure 5 indicates the polarization behavior of pure copper and brass, as the base materials and of the welded specimen NZ at different heat inputs in the borate buffer solution. Obviously, the behavior at active region seems to be unaffected by buffer agent, suggesting that the apparent Tafel region has a similar slope. This implies that the pH remained almost unchanged during the Cu dissolution, as expected. As E moves toward positive rates, the film breakdown becomes apparent initially. But then, the passive film is formed once again and remained stable at about 1.0 V.
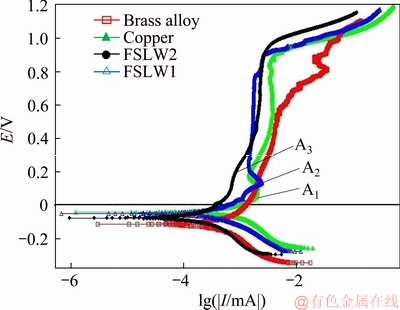
Figure 5 Polarization behavior of Cu, brass alloy and welded copper/brass nugget zones in 0.015 mol/L borax (pH 9.3)
Three peaks (A1, A2, and A3) were observed at pure copper and the welded specimens NZ (Figure 5), which are related to the Cu (I)-oxide formation [23]. Cu2O is the prime oxidation product in the alkaline solution, which was confirmed by the following equation:
2Cu+2OH-��Cu2O+H2O+2e
The corrosion potential (Ecorr), passive current density (Jpass), zero current potential (EI=0), and the corrosion current density (Jcorr) are shown in Table 4. As seen in the tabulated values, the zero current potential is more positive than the measured corrosion potentials. To be precise, the surface layer reformed after cathodic polarization appears to be operative for surface protection [7]. Again, according to Table 4, the densities of passive current were found to be further increased in the welded specimens than the pure copper and brass by analyzing the polarization curves. The density of corrosion current increased without significant changes in the potential throughout the initial stage of the passivation with an increased heat input during the welding process. Moreover, there was no obvious current peak. Furthermore, the pure copper and the welded specimen NZ with a low heat input showed similar features with a passive potential range ranging from the corrosion potential to the trans-passivity onset.
Nevertheless, while the passive region is practically the same, the passive film of the welded specimen NZ with a low heat input is more stable than that of pure copper. Instead, similar features are seen in brass alloy and the welded specimen NZ with a high heat input, but the passive film of welded specimen is more stable than that of brass alloy. Therefore, during the friction stir welding process, especially the heat input, induces a more prominent impact on the rate of anodic reaction compared with that of the cathodic reaction. Although the heat input increase could not decrease the cathodic reaction, the anodic current densities significantly decreased in the welded specimens. Yet, no significant shift to positive potential was seen by the corrosion potentials and they were near to the pure copper corrosion potential. Finally, it is concluded that when the welding heat input decreases, the rate of the NZ corrosion in the welded specimen was observed to decrease in borax solution due to both decreased anodic and cathodic reaction kinetics [21]. The improved passive film stability is attributed to decreasing grain size and higher grain boundary density, which is posited to aid in the rapid formation of a passive layer, through diffusion and more active electrons near grain boundaries. Additionally, increased grain boundaries density may promote mechanical development and other researchers [20, 21] presented adhesion of a passive film via a pegging mechanism.
3.4 Electrochemical behavior
Figure 6 shows the Nyquist and Bode plots of pure copper, brass alloy and the welded specimens NZ at OCP after 3600 s immersion in the borate buffer solution. The difference in real impedance between lower and higher frequencies is usually seen as a charge transfer resistance throughout the assessment of Nyquist plots.
Table 4 Electrochemical data for Cu, brass alloy and welded copper/brass nugget zones in 0.015 mol/L borax (pH 9.3)

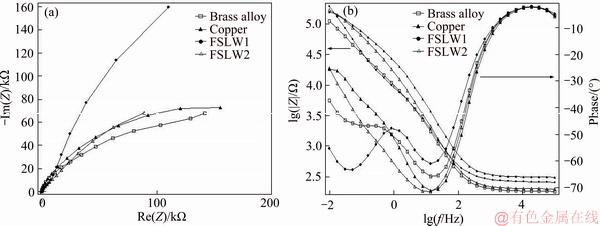
Figure 6 Nyquist (a) and Bode plots (b) of Cu brass alloy and welded copper/brass nugget zones in 0.015 mol/L borax (pH 9.3)
The charge transfer resistance should be consistent with the resistance between metal and outer helmholtz plane (OHP).
The influence of all resistances is related to the metal/solution interface, suggesting the need to consider the charge transfer resistance, accumulation resistance, diffuse layer resistance, film resistance, etc. Therefore, the difference in real impedance between lower and higher frequencies was considered as the polarization resistance (Rpol) in this study [16]. Based on Figure 6(a), Nyquist plots for pure copper and brass have similar behaviors as well as evident two time constants. The first time constant is seen in high frequencies in an incomplete semicircle form, and the second time constant is observed in middle to low frequencies range. However, as seen in Figure 6, the heat input during the welding process results in a change in the impedance diagrams for both shape and size. Thus, with an increased heat input, the passive film feature varies in both welded specimens, and the corrosion mechanism could not be controlled likewise. The equivalent circuit shown in Figure 7 was used to simulate the measured impedance data on copper and brass metals in the borate buffer solution.
In this equivalent model, Qp is the constant phase element of passive film; Rp is the passive film resistance; Rct is the charge-transfer resistance; Qdl is the constant phase element of the double layer; and Rs is the solution resistance. As mentioned in Table 4, the passive film resistance and the charge-transfer resistance of copper specimen are higher than those of the brass sample. Clearly, the polarization resistance rate (Rpol=Rp+Rct) of copper specimen showed increase as compared to the brass specimen. There was more reduction in the corrosion current densities of copper specimen than the other one. These results seem to be consistent with the potentiodynamic polarization results. The equivalent circuit indicated in Figure 7(b) was applied to simulate the measured impedance data on the welded specimen NZ with a high heat input in the borate buffer solution. However, the equivalent circuit applied to simulate the measured impedance data on the welded specimen NZ with a low heat input was consistent with the equivalent circuit offered in Figure 7(a).
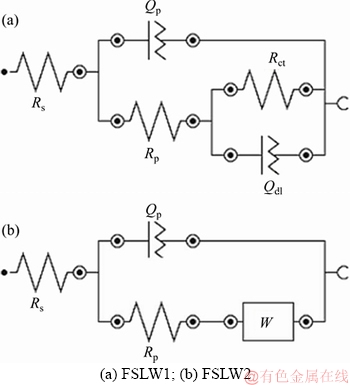
Figure 7 Equivalent circuit used for modeling experimental EIS data of:
In this regard, Table 4 shows the calculated equivalent circuit parameters of the welded specimen NZ with a low heat input. In Figure 7(b), the equivalent circuit consists of the constant phase element of passive film Qp, the passive film resistance Rp, in series with the Warburg impedance (W). The diffusion process may imply the control of corrosion mechanism by charge-transfer step and the diffusion process. The calculated corresponding circuit parameters of the welded specimen NZ with a low heat input are also demonstrated in Table 4. As shown in Table 4, the higher heat input results in less polarization resistance rate in the welded specimen NZ. This suggests the higher resistance of the welded specimen polarization with a low heat input may be related to the superior nature of passive film formed on the specimen due to its smaller grain size. Finally, the EIS plots present that the decrease in welding heat input increases the impedance value, due to the improved passive film stability by decreasing grain size, which is consistent with the result of the polarization behavior.
3.5 SEM micrograph
Defining a precise composition for copper and its alloy surface in the alkaline solution appears to be difficult. In addition, the composition of formed surface layer rests on potential. Some researches described a layer on the brass surface in the alkaline solution, which is mainly ZnO at lower potentials [23]. Thus, ZnO and Zn(OH)2 were mostly formed at negative potentials, whereas Cu2O, CuO and Cu(OH)2 formation occurred at higher potentials [23]. Instead, the FSW process is believed to help obtain fine grain structures with a high internal energy and a larger fraction of grain boundaries. In addition, the dislocation density and residual stress are high on the grain boundaries side and inside some grains. Therefore, high residual stress provides the fine grain copper and its alloys with more nuclei to form heavier passive films, which will improve the corrosion resistance rate. Furthermore, the formation of grain structures results in the rapid passive film growth [24-26].
Figures 8 and 9 specify the SEM micrographs of copper, brass and the welded specimens NZ surface in the borate buffer solution after the electrochemical tests. According to Figure 8(a), copper surface is covered with a porous coarse layer of corrosion products, which expected a cuprite Cu2O or Cu(OH)2. The porous layer is corresponded to pit corrosion as seen. However, Figure 8(b) shows that the brass specimen surface is protected with a very porous passive layer, formed by the occurrence of Zn dealloying phenomenon. As well, this film can be defensive, which leads to a diffusion-control corrosion process. As shown in Figures 9(a) and (b), the welded specimens NZ surfaces are protected with almost different dense porous passive films. Thus, the passive film is more porous and thinner than the one formed on the welded specimen surface with a high heat input. According to Figure 9(b), the welded specimen with a low heat input has a denser porous passive layer with the lower pit corrosion area across the surface. Finally, the SEM assessment concluded that the corrosion mechanism for welded specimen with a high heat input occurs in two steps, including a charge-transfer step and a diffusion process step. However, the corrosion mechanism for welded specimen with a low heat input happened in a single charge- transfer step.

Figure 8 SEM micrographs of Cu and brass surfaces formed in 0.015 mol/L borax solution:
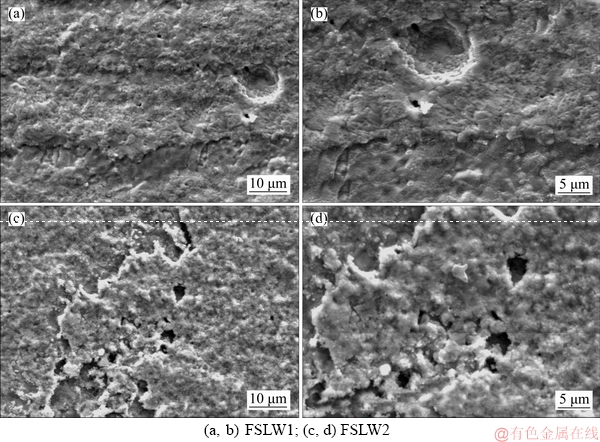
Figure 9 SEM micrographs of welded specimens NZ surfaces formed in 0.015 mol/L borax solution:
4 Conclusions
The corrosion behavior of dissimilar copper/ brass welded joints of the nugget zone in a 0.015 mol/L borax solution (pH 9.3) was investigated using the electrochemical impedance spectroscopy, potentiodynamic polarization curves, open circuit potential, and surface morphology studies. The conclusions are as follows:
Figure 9 The WNZ surfaces SEM micrographs formed in 0.015 M Borax solution, (a, b) FSLW1; (c, d) FSLW2
1) The open circuit potential measurements indicated that the E
ocp of welded specimen nugget zone is shifted to positive values with a decrease in heat input during welding process.
2) Thermodynamically, the welded specimens have a bigger and lesser tendency to corrode against copper and brass metals, respectively.
3) Polarization plots showed that the increased heat input leads to current densities. Reduction and significant shift to positive values for corrosion potentials.
4) EIS results specified that the higher heat input leads to lesser polarization resistance in the welded nugget zone.
5) The FSW process with a low heat input produced better conditions for the passive films formation with higher protection behavior.
References
[1] KUNDIG K J A, COWIE J G. Copper and copper alloys [M]// MYER K. Mechanical Engineers�� Handbook. New Jersey: Wiley Interscience, 2006: 117-220.
[2] ZHOU L, HUANG Y X, DONG S, ZHOU W L, FENG J C, HE W X. Effect of rotation speed on the microstructure and mechanical properties of dissimilar friction stir-welded copper/brass metals [J]. International Journal of Advanced Manufacturing Technology, 2016, 84(5-8): 1335-1343. DOI: 10.1007/s00170-015- 7792-9.
[3] ERDEM M. Study the structure and mechanical properties of copper-brass plates joined by friction stir welding [J]. International Journal of Advanced Manufacturing Technology, 2015, 76: 1583-1592.
[4] THOMAS W M, NICHOLAS E D, TEMPLESMITH P, DAWES C J, NEEDHAM J C, MURCH M G. Friction stir welding: International Patent Application. No. PCT/ GB92102203 and Great Britain Patent Application. No. 9125978.8 [P]. 1991.
[5] RADOVANOVI M B, MILI
M B, MILI S M, SIMONOVI
S M, SIMONOVI A T, PETROVI
A T, PETROVI MB, ANTONIJEVI
MB, ANTONIJEVI M. The impact of purine on brass behavior in neutral and alkaline sulphate solutions [J]. International Journal of Electrochemistry Society, 2012, 7: 11796-11810.
M. The impact of purine on brass behavior in neutral and alkaline sulphate solutions [J]. International Journal of Electrochemistry Society, 2012, 7: 11796-11810.
[6] MERAN C. The joint properties of brass plates by friction stir welding [J]. Materials & Design, 2006, 27: 719-726.
[7] ALFANTAZI A M, AHMED T M, TROMANS D. The copper alloys corrosion behavior of in chloride media [J]. Materials & Design, 2009, 30: 2425-2430.
[8] PROCACCINI R, V ZQUEZ M, CER
ZQUEZ M, CER S. Copper and brass aged at open circuit potential in slightly alkaline solutions [J]. Electrochimica Acta, 2009, 54: 7324-7329.
S. Copper and brass aged at open circuit potential in slightly alkaline solutions [J]. Electrochimica Acta, 2009, 54: 7324-7329.
[9] PROCACCINI R, VAZQUEZ M, SCHREINER W H, CER S. The study of films' surface formed on copper and brass at open circuit potential [J]. Applied Surface Science, 2013, 268: 171-178.
S. The study of films' surface formed on copper and brass at open circuit potential [J]. Applied Surface Science, 2013, 268: 171-178.
[10] BITON M, SALITRA G, ILZYCER D, AURBACH D, MISHKOV P. The electrochemical behavior and passivation of copper and brass (Cu70/Zn30) electrodes in the concentrated aqueous KOH solutions [J]. Journal of Electrochimica Society, 2006, 153: B555-B565.
[11] KUNZE J, MAURICE V, STREHBLOW H, KLEIN L H, MARCUS P. In situ STM study of the duplex passive films formed on Cu (111) and Cu (001) in 0.1 M NaOH [J]. Corrosion Science, 2004, 46: 245-264.
[12] MIKIC T K, MILOSEV I, PIHLAR B. Passivity and corrosion of Cu�CXZn (X=10-40 wt.%) alloys in borate buffer containing chloride ions [J]. Journal of Applied Electrochemistry, 2005, 35: 975-984.
[13] FATTAH-ALHOSSEINI A, KESHAVARZ M K, TAHERI A H. Friction stir welding effects on electrochemical behavior of pure copper [J]. Transactions of Indian Institute of Metals. 2016, 69(7): 1423-1434.
[14] FENG Y, TAN K L, SIOW K S, TEO W K, HSIEH A K. The corrosion mechanisms and products of copper in aqueous solutions at different pH values [J]. Corrosion, 1997, 53(5): 389-398.
[15] MILOSEV I, MIKI'C T K, GABER��S��CEK M. The effect of Cu-rich sub-layer on the increased corrosion resistance rate of Cu�CxZn alloys in chloride solution containing borate buffer [J]. Electrochimica Acta, 2006, 52: 415-426.
[16] KILINCCEKER G, ERBIL M. The effects of phosphate ions on the electrochemical behavior of brass in sulphate solutions [J]. Materials Chemistry & Physics, 2010, 119: 30-39.
[17] GHARAVI F, FADAEIFARD F, MATORI KA, YUNUS R, OTHMAN N K. Mechanical and structural evaluation of friction stir welded 6061 aluminum alloy lap joints at different welding speeds [J]. Kovove Materially, 2016, 54: 1-11.
[18] AMINI K, GHARAVI F. Influence of welding speed on corrosion behavior of friction stir welded AA5086 aluminum alloy [J]. Journal of Central South University, 2016, 23: 1301-1311.
[19] GHARAVI F, EBRAHIMZADEH I, AMINI K, DARYA P. Evaluation of the microstructure and mechanical properties of friction stir-welded copper/brass dissimilar joints [J]. Materials Research Express, 2018, 5(7): 076517. https://doi.org/10.1088/2053-1591/aacf20.
[20] TAO S, LI D Y. Tribological, mechanical and electrochemical properties of monocrystalline copper deposits produced by pulse electrodeposition [J]. Nanotechnology, 2006, 17: 65-78.
[21] RALSTON K D, BIRBILLIS N. Effect of grain size on corrosion: A review [J]. Corrosion, 2010, 66(7): 075005.
[22] BADAWY W A, AL-KHARAFI F M. The brass alloys corrosion behavior in aqueous solutions with different pHs [J]. Corrosion, 1999, 55: 268-274.
[23] ANTONIJEVIC M, PETROVIC M B, BOGDANOVIC G D, RADOVANOVIC M B, STAMENKOVIC A T. The effects of pH and chloride ions on electrochemical behavior of brass in alkaline solution [J]. International Journal of Electrochemical Society, 2009, 4: 654-661.
[24] SARLAK H, ATAPOUR M, ESMAILZADEH M. The corrosion behavior of friction stir welded lean duplex stainless steel [J]. Materials & Design, 2015, 66: 209-215.
[25] FATTAH-ALHOSSEINI A, IMANTALAB O. The accumulative roll bonding process effect on the electrochemical behavior of pure copper [J]. Journal of Alloys & Compounds, 2015, 632: 48-54.
[26] IMANTALAB O, FATTAH-ALHOSSEINI A. Electrochemical and passive behaviors of pure copper fabricated by accumulative roll bonding (ARB) process [J]. Journal of Materials Engineering and Performance, 2015, 24: 2579-2585.
(Edited by ZHENG Yu-tong)
���ĵ���
ͭ/��ͭ���ֽ���Ħ������ͷ�ڼ�����Һ�и�ʴ��Ϊ
ժҪ�����о�ּ���о�0.015 mol/L��ɰ��Һ(pH 9.3)��ͭ/��ͭ���ֽ���Ħ����(FSLW)��ͷ�ĵ㺸�ۺ���(NZ)�ĸ�ʴ��Ϊ���ں��ӹ����У����ø��Ⱥ͵����������������ֺ���ͭ/��ͭ�壬���Ⱥ͵�������ֱ�Ϊ710 r/min��16 mm/min��450 r/min��25 mm/min��ͨ����·��λ(OCP)�������绯ѧ�迹����(EIS)��Tafel����ʵ��������λ�����Һ�������ĵ绯ѧ��Ϊ������������ɨ���������������������ò���й۲졣����������������������ļ�С���µ�NZ��ʴ���㺸�ۺ����ľ����ߴ�͵������ߵķ��Ƚ��͡�Tafel����ʵ��͵绯ѧ�迹��������������������ͭ�ͻ�ͭ��ͬ���ǣ��ں��ӹ����У�NZ������������ļ��ٸ����˸�ʴ��Ϊ�����⣬���ӹ���������������Ӷ��γɾ��нϸ߱������ܱ�����Ĥ������������
�ؼ��ʣ�����Ħ������ͭ����ͭ�Ͻ𣻸�ʴ��Ϊ��������Һ�����ֺ���ͷ
Received date: 2018-02-09; Accepted date: 2018-10-25
Corresponding author: Kamran AMINI, PhD, Associate Professor; Tel: +98-9131651659; E-mail: Kamran_amini1978@hotmail.com; ORCID: 0000-0002-7689-4579