
Detoxification of chromium-containing slag by Achromobacter sp. CH-1 and selective recovery of chromium
CHAI Li-yuan(柴立元), WANG Yun-yan(王云燕), YANG Zhi-hui(杨志辉),
WANG Qing-wei(王庆伟), WANG Hai-ying(王海鹰)
School of Metallurgical Science and Engineering, Central South University, Changsha 410083, China
Received 6 July 2009; accepted 1 February 2010
Abstract: Based on the principle of bacterial leaching and bacterial metallurgy, a novel technology for the detoxification of chromium-containing slag by Achromobacter sp. CH-1 and chromium recovery was proposed. Strain CH-1 cell morphology before and after Cr(Ⅵ) reduction was observed with a scanning electron microscope (SEM), and the reduction product is found to adhere to terminals of CH-1 cells. Energy-dispersive X-ray (EDX) and electron paramagnetic resonance (EPR) analyses reveal that the main component of the reduction product is Cr(Ⅲ). Furthermore, small and large-scale demonstration projects reveal that Achromobacter sp. can be used to detoxify chromium-containing slag and to selectively recover chromium by using this novel technique. Chromium recovery rate increases with decreasing particle sizes of chromium-containing slag and slagheap height. Chromium recovery rates in 10 t/batch and 20 t/batch of on-site demonstration projects for chromium-containing slag detoxification are more than 90%.
Key words: chromium(Ⅵ) reduction; resource recovery; Achromobacter sp. CH-1
1 Introduction
Chromium-containing slag is generated in the production process of chromium salts and it contains about 1% of the carcinogenic toxic hexavalent chromium. In China, the accumulated chromium-containing slag piling in chromate industries amounts to 6 million tons. In addition, about 600 thousand tons is discharged annually[1]. The inappropriate disposal of chromium-containing slag has caused serious Cr(Ⅵ) contamination of water and soil[2-4]. For instance, the Cr(Ⅵ)-contaminated soil in China reached up to 125 million tons.
In literatures, several chemical methods were introduced for the detoxification of chromium-containing slag such as pyro-based reduction and hydro-based reduction. For example, the high temperature calcination method was commonly used to detoxify chromium- containing slag in Japan, Russian and Roumania[5]. Consequently, the detoxified slag was utilized to make artificial aggregates and fire-resistant materials[6-7]. Those methods were based on the principle of converting Cr(Ⅵ) into Cr(Ⅲ) since Cr(Ⅲ) is less toxic than Cr(Ⅵ)[8]. Chemicals, such as FeSO4?7H2O, Na2SO3, NaHSO3 and Na2S2O5, were commonly used for Cr(Ⅵ) reduction[9-10]. Besides, commercial chemical reductants, various natural minerals or industrial wastes containing iron can be used to reduce toxic Cr(Ⅵ) to less toxic Cr(Ⅲ). PARK et al[11] reported that waste slag generated from iron making industry can effectively convert Cr(Ⅵ) into Cr(Ⅲ). However, these chemical methods were not widely applied due to high cost and incomplete Cr(Ⅵ) removal. In addition, chromium recovery from chromium- containing slag is scantly taken into account. Moreover, calcic calcination is the predominant method to produce chromate in China, which generates a large amount of slag containing high concentration of both water soluble and acid soluble Cr(Ⅵ). In particular, the acid soluble Cr(Ⅵ) is difficult to remove with chemical reduction. Therefore, more and more attention has been paid on microbial method for the detoxification of chromium-containing slag[12]. Although there were plenty bacterial strains which were isolated from sewage sludge or the contaminated site[13-14], those strains were not yet used in large scale of projects for the detoxification of chromium-containing slag. Therefore, a novel technique of chromium- containing slag granulation and a consequently microbial leaching was proposed in our previous research. The laboratory-scale experiments were proved to be feasible to realize both the detoxification of chromium-containing slag and chromium recovery by using the above technique[15-16]. In this study, small and large-scale on-site demonstration projects were established to further demonstrate its feasibility.
2 Experimental
2.1 Chromium-containing slag
Chromium-containing slag was collected from the previous Chromate Factory of Changsha, China. The main chemical compositions of chromium-containing slag are listed in Table 1.
Table 1 Main chemical compositions of chromium-containing slag(mass fraction, %)

2.2 Bacterial strains and growth conditions
The bacterial strain used for Cr(Ⅵ) detoxification in this study was isolated from chromium-containing slag disposal sites in Changsha, China. The strain was identified as Achromobacter sp. by gene sequencing of 16S rRNA, and then nominated as strain CH-1. It was a Gram negative bacillus with a round and movable flagellum. Cells were grown in Luria Broth medium containing 10 g tryptone, 10 g NaCl, 5 g yeast extract, and 0.1 g glucose in 1 L distilled water at pH 10 with shaking (120 r/min). Incubation temperature was 30 °C. Prior to the experiment, a single colony was inoculated into Luria Broth medium and cultured under aerobic conditions for 18 h in order to obtain bacterial cells in log-phase that had the highest reduction activity. All media were autoclaved at 121 °C for 18 min before the experiment.
2.3 Experimental procedure
Based on our previously established technique of chromium-containing slag granulation and a consequently microbial leaching[9], a series of demonstration projects with different scales of 1, 2, 10, 20 t/batch were practically carried out to detoxify chromium-containing slag and selectively recover chromium. The demonstration projects were established at the previous Chromate Factory of Changsha, China.
Chromium-containing slag was passed through a 0.38 mm sieve and then granulated with 3%-5% adhesive and 5%-10% water in a pelletizer to yield granulated slag with particle sizes of 3-15 mm in diameter. The slag heap was built up by the granulated chromium-containing slag according to the different scales and heights. The tap water continuously sprayed the slag heap for 1 d. The leachate was collected in a grit chamber and then pumped into the biochemical pond. 20% of the cultivated bacteria, Achromobacter sp.CH-1, were inoculated into the leachate in the above biochemical pond to reduce Cr (Ⅵ) to Cr (Ⅲ). The supernatant in the biochemical pond was recycled to spray the slag for 2 d until Cr (Ⅵ) was not detected in the supernatant. The precipitation, sludge, was filtered under a certain pressure, collected and then weighed. The contents of chromium in the sludge and the detoxified slag were determined. The flowchart of chromium recovery from chromium-containing slag is shown in Fig.1.

Fig.1 Flowchart of the detoxification chromium recovery of chromium- containing slag
2.4 Scanning electron microscopy of strain CH-1
Bacterial cells after Cr(Ⅵ) reduction were harvested by centrifugation (5 000 r/min for 20 min at 4 °C) after 24 h of incubation. Cell pellets were washed with distilled water and fixed in 2.5% (volume fraction) glutaraldehyde in phosphate buffer (pH 7.0) for 2 h and then fixed with 1% osmium tetroxide for 2 h. The fixed samples were critical point-dried and sputter-coated with gold. Specimens were viewed using a JEOL JSM-6360LV scanning electron microscope.
2.5 Energy-dispersive X-ray analysis
The products of Cr(Ⅵ) reduction were examined under SEM with an energy-dispersive X-ray (EDX) probe to determine the elemental distribution.
2.6 Electron paramagnetic resonance (EPR)analysis
The product of the chromate reduction was analyzed by EPR spectrometry with a Bruker 200D-SRC EPR spectrometer (Bruker Instruments, Billerica, MA) in a Teflon tube (0.5 mm [inside diameter] ×20 cm). All spectra were measured at room temperature with the following EPR settings: 9.53 GHz microwave frequency, 5 mW microwave power, 200 ms time constant, 1 G modulation amplitude, 4 100 G center field, 8 000 G sweep width and 100 s sweep time.
3 Results and discussion
3.1 Identification of Cr(Ⅵ) reduction product
The morphology of CH-1 cells with and without Cr(Ⅵ) reduction was observed by the SEM. As shown in Fig.2(a), the surface of the initial cells without Cr(Ⅵ) reduction is granulated and flagella are loose (most are lost during centrifugation). The morphology of the cells after Cr(Ⅵ) reduction is different (Fig.2(b)). A heap of amorphous compounds accumulate at the terminals of CH-1 cells. This amorphous substance is presumed to be a precipitate of Cr(Ⅲ).
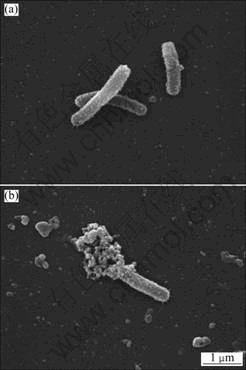
Fig.2 Morphologies of Achromobacter sp.CH-1 cell before (a) and after (b) Cr(Ⅵ) reduction
To further determine the components of the reduction products accumulated at the terminals of CH-1 cells, the precipitate from the detoxifcation process was collected and washed 4 times by distilled water to remove water soluble inorganic salts such as NaCl and KCl. The washed precipitate was dried at 120 °C for 8 h and analyzed by EDAX.
Fig.3 shows that the maximum content in precipitate is O element, second is Cr, and then is C element. The precipitate is composed of reduced production and bacteria cells. Since O and C are the components of bacteria cells, the main substance in reduced production is compound of chromium. In order to further determine the valence of Cr in the precipitate, the reduced production was analyzed by electron paramagnetic resonance (EPR).
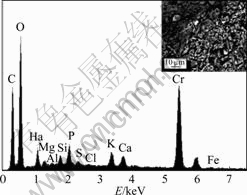
Fig.3 EDAX analysis result of precipitates produced during chromate reduction (Insert: SEM image of precipitation)
The precipitation of Cr(Ⅵ) reduction by CH-1 was collected, dried and ground before EPR analysis. The powders of K2CrO4 and CrCl3 used as standards for Cr(Ⅵ) and Cr(Ⅲ) were also detected under the identical conditions. In Fig.4, there is no signal for the Cr(Ⅵ) solution because no single electron is present. However, a broadened EPR spectrum centered at 3 500 Gs in the Cr(Ⅲ) standard solution was obtained. The reduction product of Cr(Ⅵ) by Achromobacter sp. strain CH-1 exhibits a strong signal that is similar to the Cr(Ⅲ) standard solution. The results suggest that the precipitate generated from Cr(Ⅵ) reduction and deposited on the
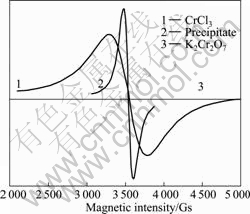
Fig.4 EPR patterns of Cr(Ⅵ) reduction products
surface of CH-1 cells is trivalent chromium and conjecturally presented in the Cr(OH)3 form due to the high pH of the reduction mixture.
3.2 Effect of particle sizes of chromium-containing slag on chromium recovery
Chromium-containing slag with particle sizes of 3-8 mm and 10-15 mm in diameter were used to constitute 1 t (4.48 m×1.00 m×0.28 m) of slag heap, respectively. Two tons of tap water was explored to spray the slag for 1 d with a flow speed of 3.3 L/(min?m2). The leachate was collected in a biochemical pond. Thereafter, 20% of the cultivated bacteria were inoculated into leachate in the above biochemical pond to reduce Cr(Ⅵ) to Cr(Ⅲ) under the proper aeration condition. After Cr(Ⅵ) reduction, the supernatant liquid was recycled to spray slag by a pump until chromium-containing slag was completely detoxified. Cr(Ⅲ) was precipitated as the formation of Cr(OH)3 at the bottom of the biochemical pond and then chromium-containing sludge was collected. As seen in Table 3, after detoxification of slag with the sizes of 3-8 mm and 10-15 mm in diameter, the collected chromium-containing sludge amounts are 32.48 kg and 33.60 kg, respectively. However, the slag with smaller particle size (3-8 mm in diameter) has higher chromium content in sludge and higher chromium recovery rate than the slag with lager particle size (10-15 mm in diameter). Although slag with small particle size has less sludge, its high chromium content in sludge results in high chromium recovery. The results imply that the smaller particle size slag favors Cr(Ⅵ) leaching and chromium recovery.
Table 3 Chromium recovery from chromium-containing slag with different particle sizes

3.3 Effect of heap height on recovery of chromium from chromium-containing slag
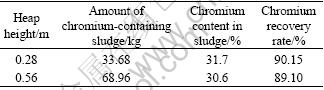
Chromium-containing slag with particle size of 3-8 mm in diameter was chosen to constitute 2 t (4.48 m×1.00 m× 0.56 m) and 1 t (4.48 m×1.00 m× 0.28 m) of slag heap, respectively. The heights of the above slag heap were 0.56 m and 0.26 m. The same treatment procedures of slag and leachate were described as those mentioned above. As shown in Table 4, 33.68 kg and 68.96 kg sludge are obtained from chromium-containing slag with the heap heights of 0.28 m and 0.56 m after the detoxification of slag by bacteria, respectively. The
chromium recovery rates reach up to 90.15% and 89.10%, respectively. It is obviously noted that high slag heap results in low chromium recovery rate. This could contribute to different leaching of Cr(Ⅵ) in high slag heap.
Table 4 Chromium recovery from chromium-containing slag with different heap heights
3.4 Chromium recovery from large-scale chromium- containing slag treatments of 10 t/batch and 20 t/batch
In this study, the optimization parameters for large-scale chromium-containing slag bio-detoxification were obtained as follows. The chromium-containing slag was granulated and yield particles with granular size of 3-8 mm in diameter, sprayed with tap water for 1 d followed by spraying with culture media inoculated with Achromobacter sp. for 2 d. Under the above optimal condition, on-site demonstrating projects to treat 10 and 20 t of slag were run. The results reveal that chromium recovery rates in demonstration projects with the scale of 10 and 20 t/batch reach 90.51% and 92.04%, respectively, which are higher than those in small scale experiments (see Table 5). The results indicate that the bio-detoxification of chromium-containing slag by bacteria and selective recovery of chromium has the amplification effect. Both bio-detoxification and chromium recovery can be simultaneously achieved.
Table 5 Chromium recovery from chromium-containing slag in demonstration projects

3.5 Balance of chromium in process of detoxification of chromium-containing slag
The species of Cr in chromium-containing slag include water-soluble Cr, acid-soluble Cr and enwrapped Cr in crystal lattice. The enwrapped Cr produced in the high temperature process accounts for a small portion of total Cr in chromium-containing slag. The enwrapped Cr cannot be leached by water leaching, acidic leaching, alkali leaching, culture media leaching and bacterial leaching. Therefore, this part of Cr is not considered in the Cr balance calculation in the present study. In the process of Cr balance calculation (Table 6), total Cr consists of chromium in sludge and residue Cr in the detoxified slag. The residue Cr could contain a part of Cr reduced by Achromobacter sp. and consequently intermixed in the slag heap in the form of Cr(OH)3. Besides, a small part of Cr is not reduced or not leached, which also attributes to the residue Cr. As shown in Table 6, more than 90% chromium in chromium- containing slag is recovered in the sludge in large scale demonstration projects of 10 t/batch and 20 t/batch. Furthermore, chromium is almost balanced in this study, although there is less than 3% of Cr loss.
Table 6 Chromium balance in process of detoxification of chromium-containing slag by bacteria

4 Conclusions
1) Chromium is the predominant composition of the Cr(Ⅵ) reduction product, and exists in trivalent chromium form, which verifies that Cr(Ⅵ) is reduced to Cr(Ⅲ) by Achromobacter sp..
2) Achromobacter sp. can be used to detoxify chromium-containing slag and to selectively recover chromium by using this novel technique. Chromium recovery rate is affected by particle sizes of chromium-containing slag and slag heap height.
3) Chromium recovery rates in 10 t/batch and 20 t/batch of on-site demonstration projects for chromium-containing slag detoxification are more than 90%.
References
[1] ZHOU Yue. Chromate pollution [J]. Chromate Industry, 2003, 5(3): 37-38. (in Chinese)
[2] JARDIN P M, FENDOR S E, MAYES M A, LARSEN I L, BROOKS S C, BAILEY W B. Fate and transport of hexavalent chromium in undisturbed heterogenous soil [J]. Environmental Science & Technology, 1999, 33(29): 2939-2944.
[3] GEELHOED J S, MEEUSSEN J C L, ROE M J, HILLIER S, THOMAS R P, FARMER J G. Chromium remediation or release? Effect of iron(Ⅱ) sulfate addition on Cr(Ⅵ) leaching from columns of chromite ore processing residue[J]. Environmental Science & Technology, 2003, 37(3): 3206-3213.
[4] BURKE T, FAGLIANO J, GOLDOFT M, HAZEN R E, IGLEWICZ R, MCKEE T. Chromite ore processing residue in Hudson County, New Jersey [J]. Environmental Health Perspectives, 1991, 92(5): 131-137.
[5] RHA C Y, KANG S K, KIM C E. Investigation of the stability of hardened slag paste for the stabilization/solidification of waste containing heavy metal ions [J]. Journal of Hazardous Materials, 2000, 73(3): 255-267
[6] LAN Si-guo, YIN Hui-min, DI Yi-an, REN Jian-zhang. Detoxication techniques for chromium slag [J]. Research of Environmental Sciences, 1998, 11(3): 51-54 (in Chinese)
[7] CHAI Li-yuan, HE De-wen, YU Xia, LIU Hui, MIN Xiao-bo, CHEN Wei-liang. Technological progression detoxification and comprehensive utilization of chromium containing slag [J]. Transaction of Nonferrous Metals Society of China, 2002, 12(3): 514-519.
[8] MOON D H, WAZNE M, DERMATAS D, CHRISTODOULATOS C, SANCHEZ A M, GRUBB D G, CHRYSOCHOOU M, KIM M G. Long-term treatment issues with chromite ore processing residue (COPR): Cr6+ reduction and heave [J]. Journal of Hazardous Materials, 2007, 143(5): 629-635.
[9] QIN G, MCGUIRE M J, BLUTE N K, SEIDEL C, FONG L. Hexavalent chromium removal by reduction with ferrous sulfate, coagulation, and filtration: A pilot-scale study [J]. Environmental Science and Technology, 2005, 39: 6321-6327.
[10] KURNIAWAN, CHAN G Y S, LO W H, BADEL S. Physico-chemical treatment techniques for wastewater laden with heavy metals [J]. Chemical Engineering Journal, 2006, 118: 83-98.
[11] PARK Dong-hee, LIM Seong-rin, LEE Hae-woo, PARK Jong-moon. Mechanism and kinetics of Cr(Ⅵ) reduction by waste slag generated from iron making industry[J]. Hydrometallurgy, 2008, 93: 72-75.
[12] ACKERLEY D F, GONZALEZ C F, PARK C H, BLAKE R, KEYHAN M, MATIIN A. Chromate reducing properties of soluble flavoproteins from pseudomonas putida and Escherichia coli [J]. Applied & Environmental Microbiology, 2004, 70: 873-882.
[13] CAMARGO F A O, OKEKE B C, BENTO F M, FRANKENBERGER W T. Chromate reduction by chromium-resistant bacteria isolated from soils contaminated with dichromate [J]. Journal of Environmental Quality, 2003, 32: 1228-1233.
[14] HILLOL G, JAYACHANDRAN K, MAURRASSE F. Microbiological reduction of chromium(Ⅵ) in presence of pyrolusite-coated sand by Shewanella alga Simidu ATCC 55627 in laboratory column experiments[J]. Chemosphere, 2003, 52: 175-183.
[15] MA Ze-min, ZHU Wen-jie, LONG Huai-zhong, CHAI Li-yuan, WANG Qing-wei. Chromate reduction by resting cells of Achromobacter sp CH-1 under aerobic conditions [J]. Process Biochemistry, 2007, 42: 1028-1032.
[16] ZHU Wen-jie, CHAI Li-yuan, MA Ze-min, WANG Yun-yan, XIAO Hai-juan, ZHAO Kun. Anaerobic reduction of hexavalent chromium by bacterial cells of Achromobacter sp. Strain CH-1 [J]. Microbiological Research, 2008, 163(6): 616-623.
(Edited by LI Xiang-qun)
Corresponding author: CHAI Li-yuan; Tel: +86-731-88836921; E-mail: lychai@mail.csu.edu.cn
DOI: 10.1016/S1003-6326(09)60328-9