Trans. Nonferrous Met. Soc. China 20(2010) s768-s773

Microstructural evolution of Mg9AlZnY alloy with vibration in lost foam casting during semi-solid isothermal heat treatment
ZHAO Zhong(赵 忠), FAN Zi-tian(樊自田), JIANG Wen-ming(蒋文明), DONG Xuan-pu(董选普)
State Key Laboratory of Material Processing and Die & Mould Technology,
Huazhong University of Science and Technology, Wuhan 430074, China
Received 13 May 2010; accepted 25 June 2010
Abstract: The nearly equiaxed grains of Mg9AlZnY alloy were obtained by vibrating solidification in lost foam casting(LFC) and the microstructure of Mg9AlZnY alloy was analyzed. On this basis, the morphology and size of a-Mg grains fabricated by semi-solid isothermal heat treatment(SSIT) at 530 ℃ and 570 ℃ holding different time were studied. The results show that the main constituent phases of Mg9AlZnY alloy are a-Mg, β-Mg17Al12 and Al2Y, and the Y can greatly refine a-Mg grains. The distribution of a-Mg grains equivalent diameters between 20 and 100 μm is up to 87%, and the average roundness of a-Mg grains reaches 1.37 in the specimen obtained at 570 ℃ and holding time 60 min. According to the analysis of solidification kinetics and thermodynamic, binary eutectic with low melting point melts firstly on SSIT process. As the liquid fraction increases with the solute diffusibility, both of the shape and size of a-Mg grains change ceaselessly. When the liquid fraction reaches equilibrium, the a-Mg grains are gradually spheroidized under the interfacial tension, and then the a-Mg grains begin to combine and grow. Evolution of a-Mg dendritic grains on SSIT process is obviously different from that of equiaxed grains.
Key words: lost foam casting (LFC); semi-solid isothermal heat treatment (SSIT); microstructural evolution; magnesium alloy
1 Introduction
Research of semi-solid metal forming (SSF) technology began in the 1970s, which mainly included rheoforming and thixoforming[1-2]. The core technology of SSF is that non-dendritic structure is obtained by solid-liquid mixed slurry or billet, and solid-phase generally contains spherical grain, nearly spherical grain, and equiaxed grain[3]. Semi-solid isothermal heat treatment (SSIT) [4-6] researched in the mid-1990s omits the non-dendritic ingot fabricated in thixotropic semi-solid forming. Semi-solid non-dendritic crystallization is achieved by holding for some times at semi-solid temperature, and more satisfactory thixotropic semi-solid microstructure is obtained. SSIT process is simple and low cost, so it has a certain future development[7-8].
However, it is difficult to complete complex forgings forming because of the simple shape of semi-solid billets prepared using SSIT. If the semi-solid billets which close to the shape of the complex forging are prepared by lost foam casting which has high precision, low cost and flexible design[9], and then forging formability will be greatly enhanced.
Thus, the microstructure of nearly equiaxed grains has been obtained by the technology of lost foam casting (LFC) with vibration and by methods that rare earth Y refined AZ91D alloy. On this basis, the nearly forging shape semi-solid billets of magnesium alloy were prepared using SSIT, and the evolution of α-Mg grains was analyzed by the solidification kinetics and thermodynamics.
2 Experimental
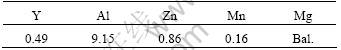
AZ91D magnesium alloy was melted in 7.5 kW resistance furnace, which was protected using SF6+CO2 mixed gas. Mg-30Y master alloy was added at 720 ?C and evenly mixed. The chemical composition of Mg9AlZnY alloy in the experiment is shown in Table1. It was poured to 45 mm×45 mm×100 mm samples at 680 ?C by the technology of lost foam casting with vibration, which the vibration frequency was 50 Hz, the amplitude was 0.34 mm and vibration time was 100 s. Then the samples were water quenched after casting 5 min. The relative vacuum level was -0.03 MPa during the pouring and solidifying process[10-11]. The microstructure of Mg9AlZnY alloy as-cast was analyzed by SEM and EDS.
Table 1 Chemical composition of Mg9AlZnY alloy(mass fraction, %)
The six specimens (20 mm×20 mm×20 mm) were cut in center of the samples, and they were wrapped with aluminum foil to place in a small tray. The specimens were placed into KSX2-15 heat treatment furnace at 400 ?C, and were heated up with 8 ?C/min and protected by argon gas, and the controlled temperature precision was ±1 ?C.
According to DSC curve analysis of the Mg9AlZnY alloy (see Fig.1), liquidus temperature of the alloy is about 598.3 ?C and solidus temperature is about 435.3 ?C. If the temperature of SSIT was selected at 530?C and 570 ?C in the experiments, the solid fraction was about 75% and 51%, respectively, and the solid fraction are calculated by Scheil equation[12].
fs=1-[(Tm-T)/(Tm-Tl)]-1/(1-k) (1)
where fs is the solid fraction, k is the balance distribution coefficient (constant), Tm is the melting point of pure metal, Tl is liquidus temperature of the alloy, and T is the temperature of SSIT.
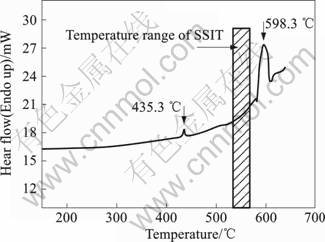
Fig.1 DSC analysis of Mg9AlZnY alloy
Holding time of SSIT was set to 0, 30, 60, 90 and 120 min, respectively. After the time being got, the specimens were water quenched quickly.
The specimens were sawed from the centre, for grinding and polishing, and then corroded with 4% nitric acid alcohol, and the microstructure of the specimens was observed with XJL 02-type optical microscope. Metallography of 100 times obtained from five regions of the center and corners around of square specimens were analyzed, and the liquid within the grains was ignored. The mean equivalent diameter and roundness of a-Mg grains which were hold for different time at 530 ?C and 570 ?C were analyzed by JX-2000 analysis software. The mean equivalent diameter is[2]
d=2(A/π)1/2 (2)
where d is the mean equivalent diameter, and A is the grain cross-section area.
The distribution rate of equivalent diameters ξ is
ξ=(nd1d2/N)×100% (3)
where nd1d2 is the number of grains between d1 and d2, and N is the total number of grains.
The shape of the grains in the semi-solid micro- structure is evaluated by the roundness, which is[2]:
λ=4πA/L2 (4)
where λ is the roundness, and L is the perimeter of grain.
3 Results and analysis
The SEM image of Mg9AlZnY alloy and EDS spectra of A and B phases are shown in Fig.2. There are mainly α-Mg phase, β-Mg17Al12 phase and some small granulated Al2Y compounds in Mg9AlZnY alloy. Al2Y compounds can play an important role in heterogeneous nucleation to refine α-Mg phase because the Al2Y is generated earlier than the α-Mg.
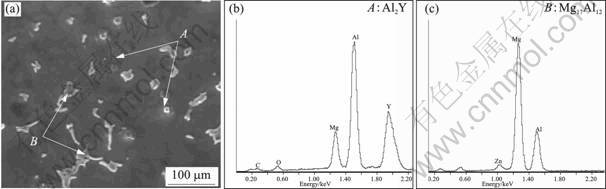
Fig.2 SEM image of Mg9AlZnY alloy (a), and EDS spectra of point A(b) and point B(c)
The microstructures of as-cast AZ91D alloy in LFC without vibration and Mg9AlZnY alloy with vibration in LFC are shown in Fig.3. The α-Mg grains are refined and the coarse dendrites are reduced because of the addition Y, strong vibration and rapid cooling. Compared to the sample of AZ91D alloy in Fig.3(a), there are little dendrite grains but more equiaxed grains in the as-cast sample of Mg9AlZnY alloy and the α-Mg grains size mainly distributes in 20- 250 μm, as shown in Fig.3(b).

Fig.3 Microstructures in LFC : (a) AZ91D without vibration; (b) Mg9AlZnY with vibration
Microstructure evolutions of Mg9AlZnY alloy with vibration in LFC on SSIT at 530 ?C holding different time are shown in Fig.4. Microstructure of water quenching at 530 ?C holding time 0-60 min shows that the β-Mg17Al12 phases of the grains boundary are melted and the α-Mg grains begin rapidly consolidation and spheroidization, the a-Mg grains are 2-3 times larger than that of as-cast, as shown in Figs.4(a)-(c). Microstructure of water quenching holding time 90 min at 530 ?C shows that liquid continuously distributes as network in grain boundary, and the a-Mg grains are surrounded by liquid. Meanwhile, the grains are continuously spheroidized as shown in Fig.4(d). The a-Mg grains holding time 120 min at 530 ?C combine and grow as shown in Fig.4(e).
Microstructure evolutions of Mg9AlZnY alloy with vibration in LFC on SSIT at 570 ?C holding different time are essentially the same as at 530 ?C, but it is slightly ahead of schedule as shown in Fig.5. The liquid phases within the grains on SSIT at 570 ?C are significantly more than that at 530 ?C. The roundness of the grains at 570 ?C holding time 60 min is better as shown in Fig.5(c). The part of bigger grains is melted to break because of the increase of liquid phase within the grains, which leads to the cluster of grains, as shown in Fig.5(d). The increasing liquid phase results in its composition change from the eutectic to the hypeoeutectic. Many small α-Mg grains of the newborn are generated in the liquid phase by water quenching as shown in Fig.5(e).
The distribution rates of equivalent diameters in water quenching specimens at 530 ?C and 570 ?C holding different time are shown in Fig.6. The distribution rate of equivalent diameters is about 58% between 20 and 100 μm in the specimens held 90 min at 530 ?C as shown in Fig.6(a). Fig.6(b) shows that the distribution rate of equivalent diameters between 20 and 100 μm is about 87% in the specimens held time 60 min at 570 ?C. Thus, the grains size is more uniform and the distribution of grains size is more centralized using SSIT at 570 ?C.
The average roundness change of α-Mg grains for different holding times is shown in Fig.7. The average roundness at 530 ?C with holding time between 60 and 120 min is smaller, that is 1.59 at 530 ?C with holding 90 min. While the average roundness at 570 ?C with holding time between 30 and 90 min is smaller, that is 1.37 at 570 ?C holding 60 min.
Therefore, the best solidified billet of Mg9AlZnY alloy with vibration in LFC can be obtained at 570?C holding time 60 min according to the analysis of the distribution rate of equivalent diameters and the average roundness. It reaches basic requirements of semi-solid structure with spherical grains, nearly spherical grains and equiaxed grains between 10 and 100 μm[2]. However, the grains size at SSIT is larger due to a small amount of the long strip or dendritic grains in Mg9AlZnY alloy with vibration in LFC.
4 Discussion
4.1 Microstructure evolution of Mg9AlZnY alloy by SSIT
It is the first stage that the samples are heated at 530 ?C for 0-60 min and for 0-30 min at 570 ?C. The eutectic phases (a-Mg + β-Mg17Al12) of low melting point in the grains or in its boundary begin to remelt in the first stage, and the divorced eutectic β-Mg17Al12 in the grains boundary is melted and connected each other into a network.
As the solutes of Al, Zn and Y diffuse in the liquid phase, the liquid fraction of the alloy would increase. The solute diffusion caused by concentration fluctuation should change shape and size of the a-Mg grains in very large extent. Many a-Mg grains with the same crystal orientation would combine and grow. The size of α-Mg grains in the first stage increases significantly as shown in Figs.4 and 5.

Fig.4 Microstructures of Mg9AlZnY by SSIT at 530 ?C for different holding time: (a) 0 min; (b) 30 min; (c) 60 min; (d) 90 min; (e) 120 min
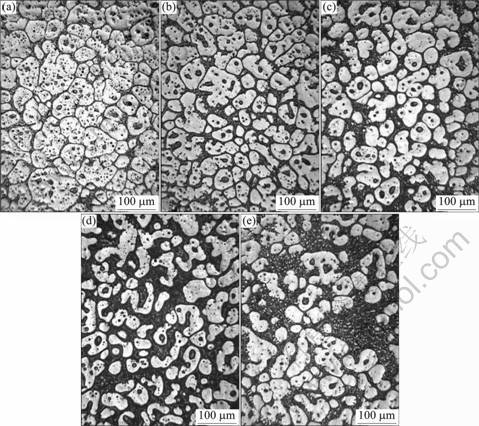
Fig.5 Microstructures of Mg9AlZnY by SSIT at 570 ?C for different holding time: (a) 0 min; (b) 30 min; (c) 60 min; (d) 90 min; (e) 120 min
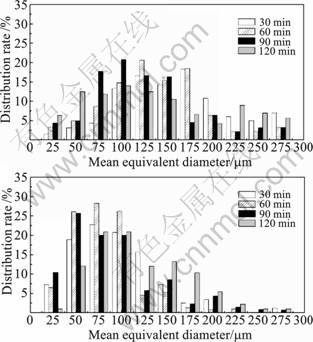
Fig.6 Distribution of equivalent diameter held different time at different temperatures: (a) 530 ?C; (b) 570 ?C
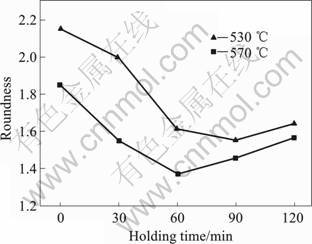
Fig.7 Average roundness of grains at 530 ?C and 570 ?C with different holding time
In the second stage, the samples are heated at 530 ?C for 60-90 min and at 570 ?C for 30-90 min. The grains grow slowly in this stage and the liquid fraction of the alloy would reach equilibrium. According to the free energy changing rule of metal solidification, the formula is as[12]
DF=-DFvV +σS (5)
where DFv=DH-TDS is the free energy difference of liquid-solid phase, V is the grain volume, σ is the solid-liquid interfacial tension, and S is the grain surface area.
If the temperature T is certain, the melting heat DH and the melting entropy DS are also fixed, then DFv is a certain value. DFv, V and σ are constant, then DF of the alloy calculated from Eq.(5) would reduce as S decreases. Therefore, the grain surface area tends to reduce under the action of solid-liquid interfacial tension, while the α-Mg grains shape is gradually spheroidized because of the temperature fluctuation and concentration fluctuation in the liquid phase.
The third stage is that the samples are heated for 120 min at 530 ?C and 90-120 min at 570 ?C. With the increase of holding time, the liquid volume in grains boundary and within α-Mg grains increases and integrates, which grandly changes the shape of grains and makes the grains continue to combine and grow. The roundness of the grains is decreased.
In a word, the proliferation of solute redistribution plays a leading role in the initial stage, while the interfacial tension of the grains plays a dominant role in the later stage in SSIT process[13].
4.2 Evolution of equiaxed and dentritic grain at SSIT
According to solidification thermodynamics[14], the formula is
r=2Tsσ/(DHTs+DHTl) (6)
where Ts is the melting point of the alloy, Tl is the alloy temperature, and DH is the melting enthalpy. When the average curvature radius of the grains r is small, the energy which atoms overcome the solid-phase interfacial tension r calculated from Eq.(6) is small. Therefore, the small equiaxed grains are easily melted to disappear with the solute diffusion and temperature fluctuations in the liquid phase as shown in Fig.8. The large equiaxed grains could be moved and rotated easily compared to the dentritic grains, which could be combined and grown by crashing when the crystal orientation is the same. Thus, the average size of the equiaxed grains gradually increases with extending the holding time.
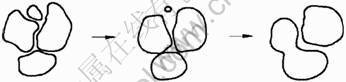
Fig.8 Evolution of equiaxed grains on SSIT
Because the dendrite has a large surface area and it occupies a large space, it is difficult that the crystal is rotated and moved[15]. The dendrite is not easy to combine and grow with other grains. However, the secondary dendrite arms have the same crystal orientation, and they could be easily combined and grown by crashing. The dendrite is gradually spheroidized by the interfacial tension as the holding time is extended[15]. When the solute proliferation within the grains reaches a limit concentration, it begins to be melted to form the liquid within grains. The liquid will grow and propagate and eventually break one grain into two as shown in Fig.9.
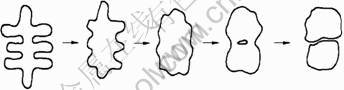
Fig.9 Evolution of dendrites on SSIT
5 Conclusions
1) Nearly equiaxed grains of Mg9AlZnY alloy can be obtained by vibration solidification and rapid cooling in lost foam casting (LFC). The semi-solid microstructure with a good roundness can be obtained by SSIT at 530 ?C and 570 ?C.
2) The distribution rate of equivalent diameters and the average roundness of grains are analyzed. The results show that the semi-solid microstructure at 570 ?C is better than that at 530 ?C on SSIT. The best solidified billet of Mg9AlZnY alloy with vibration in LFC is obtained at 570 ?C with holding time 60 min, and the distribution rate of equivalent diameters is about 87% between 20 and 100 μm and the average roundness is 1.37.
3) Microstructure evolution of dentritic grains at SSIT is different from that of equiaxed grains. Evolution of dentritic grains depends on its secondary dendrite arm spacing, the concentration of solute in liquid phase and the diffusivity, the temperature fluctuations and the boundary surface tension, and so on. The evolution of equiaxed grains shows that small grains tend to be melted and large equiaxed grains tend to grow and spheroidize.
References
[1] FLEMINGS M C. Behavior of metal alloys in the semisolid state[J]. Metallurgical Transaction A , 1991, 22 (5): 957-981.
[2] KANG Y L, MAO W M, HU Z Q. Semi-solid metallurgy of metal materials[M]. Beijing: Science Press, 2004: 5.(in Chinese)
[3] KANG Y L, MAO W M, HU Z Q. Semi-solid metallurgy of metal materials[M]. Beijing: Science Press, 2004: 5.(in Chinese)
[4] HARDY S C, VOORHEES P W. Ostwald ripening in a system with high volume fraction of coarsening phase[J]. Met Trans A, 1988, 19 (11): 2713-2721.
[5] LI Y D, HAO Y, YAN Y F. Structural evolution of AZ91D magnesium alloy during semisolid isothermal heat treatment[J]. The Chinese Journal of Nonferrous Metals, 2001, 11(4): 571-570. (in Chinese)
[6] MAO W M, ZHONG X Y, LI L Q. Microstructural evolution of AlSi7Mg non-dendritic alloys during semi-solid remelting[J]. Foundry, 1998(8): 10-12. (in Chinese)
[7] YANG H L, ZHANG Z L, OHNAK I. Structure evolution and compressive behavior of semi-solid Al-Si hypoeutectic alloy with re-melting heat treatment[J]. Journal of Materials Processing Technology, 2004, 151(1/3) :155-164.
[8] ZHAI Q Y, YUAN S, JIANG B L. Microstructural characteristics of semisolid AZ91 magnesium alloy based on SIMA process[J]. The Chinese Journal of Nonferrous Metals, 2005, 15(1): 123-128 (in Chinese)
[9] YAN F Y, QIANG X D, ZHANG Y H, HUANG X F, FAN S Y. Die-casting process of semi-solid thixo-forming of AZ91D Mg alloy[J]. The Chinese Journal of Nonferrous Metals, 2008, 18(4): 595-600.(in Chinese)
[10] BROWN J R. The lost foam casting process[J]. Metals Mater, 1992, 8 (10): 550-530.
[11] WANG Y Q, FAN Z T, LI J Q. Evolution of microstructure of AZ91D magnesium alloy in lost foam casting under vibration and semi-solid heattreatment[J]. Special Casting & Nonferroius Alloys, 2006, 26(8): 506-510.(in Chinese)
[12] LI J Q, FAN Z T, WANG Y Q, DONG X P. Effects of isothermal heat treatment on m icrostructure and mechanical properties of lost foam casting AZ91D magnesium alloy[J]. Metal Heat Treatment, 2008, 3(1): 97-100. (in Chinese)
[13] HU H Q. The solidification theory of metals[M]. Beijing: Metallurgical Industry Press, 2001: 58.(in Chinese)
[14] YANG G Y, HAO Q T, JIE W Q. Microstructure and properties of Mg-5Al-1.5Ca-0.4Zn based alloy by semi-solid isothermal heat treatment die pressure casting process[J]. The Chinese Journal of Nonferrous Metals, 2001, 15 (4): 615-621.(in Chinese)
[15] LI Q C. Theoretical basis for the formation of casting[M]. Beijing: Chine Machine Press,1982: 77.(in Chinese)
[16] ZOQUI E J, ROBERT M H. Contribution to the study of mechanisms involved in the formation of rheocast structure[J]. Journal of Materials Processing Technology, 2001, 109 (1): 215-219.
(Edited by YANG You-ping)
Foundation item: Project(2007AA03Z113) supported by the National High-Tech Research and Development Program of China; Project (50775085) supported by the National Natural Science Foundation of China
Corresponding author: FAN Zi-tian; Tel: +86-27-87540094; E-mail: fanzt@mail.hust.edu.cn