
Polyaniline anode for zinc electrowinning from sulfate electrolytes
HUANG Hui(黄 惠)1, ZHOU Ji-yu(周继禹)2, CHEN Bu-ming(陈步明)1, GUO Zhong-cheng(郭忠诚)1
1. Faculty of Metallurgical and Energy Engineering, Kunming University of Science and Technology,Kunming 650093, China;
2. The No. 705 research Institute, Kunming 650118, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: Polyaniline (Pani) anode is tested to highlight the feasibility of reduction of both energy consumption and capital costs in zinc electrowinning from sulfate solution without any modification to the existing plant. Current density, electrolyte temperature, added gelatin, added Mn2+, oxygen-evolution potential, cell potential and long duration tests were investigated. The zinc deposits were also studied by means of scanning electron microscope (SEM) and X-ray diffraction (XRD). The results show that current density and added gelatin change the preferred crystal orientations of the zinc deposits. Compared with Pb-Ag(1%) anode used in industry, the cell voltage decreases by 0.15-0.30 V, energy consumption of Zn is 2.46-2.70 kW?h/kg which results in 20% energy savings. Long duration tests show that Pani anode can represent a good alterative ability for zinc electrowinning. Zinc deposits obtained have no Pb pollution. The additions of Mn2+ ions and gelatin also change the surface morphology and deposit quality of the electrodeposited zinc, affecting the crystal orientation. These researches demonstrate that Pani anode has distinct advantages over acidic electrowinning process.
Key words: polyaniline anode; zinc electrowinning; morphology; crystal orientation
_____________________________________________________________________________________________________
1 Introduction
Zinc is an important member of the non-ferrous metals and primarily produced from its ores of sulfide, oxide and siliceous types via pyro-electrometallurgical and hydro-electrometallurgical routes. Zinc electro- winning is the main process step of hydro- electrometallurgical route both from primary and secondary sources and about 80% of the world zinc production is carried out by this technique[1-2]. Reduction electrolysis of zinc is extremely sensitive to the impurities present in the electrolyte[3-4]. Thus, the process requires a very pure electrolyte with an approximate composition of 65-70 g/L Zn2+ and 120-180 g/L H2SO4. Energy consumption values of Zn of 3.25-3.40 kW?h/kg are typical for zinc electrowinning operations with 90%-95% current efficiency. Recent investigations in zinc industry have been mainly focused on decreasing the energy costs in reduction electrolysis, which is an energy intensive process.
It is well-known that any insoluble anode material used for zinc electrowinning from sulfate solutions, should possess at least three characteristics: high electrical conductivity[5] good electrocatalytical capabilities (for oxygen evolution)[6], and good stability (corrosion resistance)[7-9]. For various electrode materials and electrolyte process, Pb-based alloy anodes [10], Ti-based electro-catalytic coating anodes[11-12], H2 anode[13] and SO2 anode[14] are researched. Evidently, they do not satisfy these requirements. Despite the large number of studies, the most useful material appears to be Pb-Ag alloy in which the Ag content ranges from 0.5% to 1.0% (mass fraction)[6]. Oxygen over-potential, anode corrosion rate and current efficiency of the Pb-Ag-Sb, Pb-Ag-Ca, Pb-Ca-Sn and Pb-Co are investigated, but have no industrial application [15-17].
In search of a suitable alternative to Pb-Ag anode, polyaniline (Pani) anode has been found to be potential in the anodes. It notably reduces the oxygen overpotential and the corrosion rate of anode. In this work, we focused on investigation of current density, electrolyte temperature, oxygen-evolution potential, cell potential and long duration tests on Pani anode during zinc electrowinning in acidic zinc sulfate electrolytes.
2 Experimental
2.1 Materials
The zinc electrolyte used in this work was prepared from analytical grade ZnSO4・7H2O and H2SO4. The foreign cation Mn2+ ions were added as the sulfate salt to get the desired electrolyte composition. Stock solutions of 65 g/L Zn2+ and 150 g/L H2SO4 were prepared separately using analytical grade reagents in doubly distilled water.
The synthesis of Pani was similar to that described previously in Ref.[18].Pani anode was prepared by mixing active materials with 5% (mass fraction) polyvinyl to form slurry and titanium net as framework. The slurry was pressed under 10 MPa by oil press (area: 4 cm2). The prepared anode was dried at 40 ℃ for 6 h under vacuum.
2.2 Electro-deposition experiments
The optimum conditions of zinc electrowinning were investigated through a series of laboratory scale experiments. Electrolyte composition of 65 g/L Zn2+ and 150 g/L H2SO4 was utilized and electrolyte temperatures of 30, 40 and 50 ℃ were adopted. Moreover, current densities of 500, 1 000 and 1 500 A/m2 were tried in a lidded 500 cm3 double wall beaker where two anodes and a cathode were placed 3.5 cm away from each other. Anode and cathode materials were Pani and aluminium alloy with dimensions of 6.0 cm×4.5 cm×0.2 cm, respectively. Electrolyte circulation (1.5 L/min) was carried out by a pump and current was supplied by a DC. power supply. Electrolyte temperature was kept constant with ±0.2 ℃ precision through two heat exchangers coupled in series. After electrolysis, the cathode was washed thoroughly with water and acetone and dried. The current efficiency (±0.3%) was calculated from the mass of zinc gained by the cathode. Fig.1 displays the schematic diagram of the experimental setup.
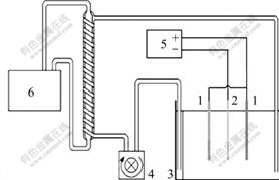
Fig.1 Schematic diagram of experimental setup: 1―Anode; 2―Cathode; 3―Electrolysis cell; 4―Pump; 5―Power supply; 6―Heat exchanger
2.3 Characterization and electrochemical tests
X-ray diffraction (XRD) studies were performed using a powder X-ray diffractometer (D/max-3BX) at a scan rate of 0.5 (?)/min. The surface morphology of the deposits was examined by scanning electron microscopy (SEM) using an XL 30 ESEM TEP+EDAX microscope.
3 Results and discussion
3.1 Effect of current density
As it can be seen from Fig.2, at the end of 1 h electrolysis, the average cell potential values of 3.1, 3.3 and 3.7 V were obtained for the current densities of 500, 1 000 and 1 500 A/m2 at 40 ℃, respectively. The zinc electro-deposits, obtained on the aluminium alloy cathode in the different current densities were examined by XRD to determine the preferred crystal orientations and the relative growth of zinc on the preferred planes and the results are given in Fig.3. The current density had a significant influence on the orientation of zinc crystals in the zinc electrowinning. The crystal orientations of all the zinc deposits showed a hexagonal crystal structure, with the 2θ positions of the peaks remaining constant. Zinc had a strong preferred orientation of the basal plane in the deposits prepared at 500 A/m2.
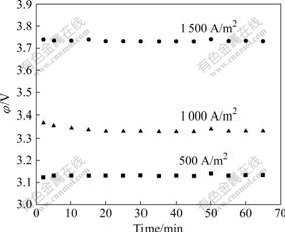
Fig.2 Effect current density on cell potential (65 g/L Zn2+, 150 g/L H2SO4, 40 ℃)
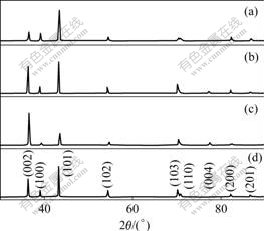
Fig.3 XRD patterns of deposits prepared at different current densities and in the presence of gelatin: (a) 500 A/m2; (b) 1 000 A/m2; (c) 1 500 A/m2; (d) In the presence of gelatin
It was observed that the order of crystal orientations for zinc electro-deposition at 500 A/m2 were (101), (002), (100), (102) and (103) (Fig.3(a)). With the increase of current density, the growth of (002) and (103) crystal planes were promoted and the growth of (100) and (101) crystal planes were suppressed. Thus, the order of preferred orientation was changed to (002), (101), (103), (100) and (102) accompanied with the latter three planes exhibiting a lower relative intensity at 1 500 A/m2 (Fig.3(c)). This result indicates that the zinc deposits result in a more random orientation of the crystals at 1 000 A/m2.
3.2 Effect of temperature
As a result of the experiments carried out at different temperatures, the average cell potential values dropped to 3.15, 3.09 and 2.87 V by increasing the temperature from 35 to 45 and 55 ℃ at 500 A/m2, respectively. To understand the effect of temperature on the electrodeposited zinc morphology, the zinc deposits were examined using SEM. Typical SEM photo- micrographs are shown in Fig.4. As it can be seen from Fig.4, morphological structure of the deposited zinc proves that the characteristics of crystal growth change from layered flake to nodular shape.

Fig.4 SEM micrographs showing effect of temperature and Mn2+ on surfaces of zinc electrodeposits at 500 A/m2: (a) 30 ℃; (b) 40 ℃; (c) 50 ℃; (d) In the presence of Mn2+
3.3 Effect of gelatin
For the deposited zinc from solutions without gelatin and containing 2 mg/L gelatin at temperature of 40 ℃ and current density of 500 A/m2, the effect of gelatin on crystal orientations using Pani anode was investigated. Representative XRD patterns are redrawn and shown in Fig.3(d). It was seen that in the presence of gelatin, the order of preferred (hkl) planes of the zinc deposits still were (101), (002), (100), (102) and (103) with the latter three planes exhibiting a similar relative intensity, but (002) plane exhibiting stronger relative intensity than that of Fig.3(a). So, the addition of gelatin to the electrolytes resulted in random orientation of the zinc crystals. The presence of gelatin had no influence on the cell potential.
The presence of gelatin caused significant changes in the deposit microstructure and improved the hardness of the pure zinc deposits. This result was attributed to the alteration in the crystallographic orientation of the zinc deposit. It is well known that zinc exhibits anisotropic mechanical properties due to its hexagonal crystal structure, i.e., the crystallographic orientation has considerable influence on the mechanical properties. In this work, the addition of gelatin promoted an increasingly random orientation of zinc crystals, resulting in a lower fraction of basal planes parallel to the substrate surface. As a result, the orientation of the zinc deposit limited basal plane slip and increased the microhardness of the pure zinc deposit. The observed relationship between zinc coating hardness and texture is consistent with that observed by other researchers[19].
3.4 Effect of Mn2+
For the deposited zinc from solutions without Mn2+ and containing 5 g/L Mn2+ at temperature of 40 ℃ and current density of 500 A/m2, the effect of Mn2+ on morphology using Pani anode was investigated. To understand the effect of Mn2+ on the electrodeposited zinc morphology, the zinc deposits were also examined using SEM. Typical SEM photomicrographs are shown in Fig.4. It can be seen from Fig.4 that the addition of Mn2+ signi?cantly changes the morphology of the zinc deposits as compared with that obtained from solutions without Mn2+. The zinc deposit obtained from manganese free electrolyte was bright but not smooth and consists of spinulose platelets of moderate size (Fig.4(b)). The addition of Mn2+ produced a more compact deposit with a ?ake structure (Fig.4(d)). Thus, addition of Mn2+ did not change the morphology but affected its compactness.
3.5 Long duration tests
Long duration tests were carried out at 500 A/m2 and room temperature (40 ℃) using an electrolyte solution containing 65 g/L Zn2+, 150 g/L H2SO4, 5 g/L Mn2+ and 2 mg/L gelatin. The most interesting results of the long duration tests are summarized in Table 1, where the average current and voltage efficiencies were given, together with energy yields and specific energy consumption over 30 d of the electrowinning tests. One may immediately note the good results shown by Pani anode.
Table 1 Average results of electrolysis tests with Pani and Pb-Ag(1%) anodes
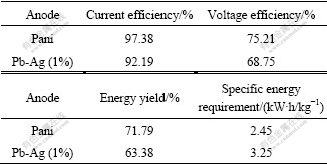
The comparison was carried out with Pani and Pb-Ag (1%) anodes. It can be seen that, there is pronounced difference in the current efficiency. There is a wider one as far as voltage and energetic efficiency is concerned. Specific energy consumption is affected by cell voltage, due, in our case, principally to the fact that the cell voltage of Pani is lower (0.15-0.30 V) than that of Pb-Ag (1%) alloy anode. Long duration tests showed that Pani anode can represent a good alterative ability for zinc electrowinning. Considerably high current efficiencies (97%-99%) were obtained with energy consumptions of Zn of 2.46-2.70 kW?h/kg as compared with 3.25-3.40 kW?h/kg of industrial electrowinning, which resulted in 20% energy savings. The specific energy consumption is smaller than that of Pb-Ag (1%) anode and the lower material costs.
4 Conclusions
Pani anode was studied to solve the problem of reducing the capital costs of zinc electrowinning plant and, if possible, the specific energy consumption without any modifications to the existing plant. The following observations are confirmed by the acidic zinc electrowinning experiments:
1) It is possible to operate Pani anode zinc electrowinning with an energy consumption of Zn of 2.46-2.70 kW?h/kg as compared with 3.25-3.40 kW?h/kg of Pb-Ag(1%) anode used in industry. Hence, around 20% of energy can be saved by electrowinning used Pani anode.
2) Cell potential increases as the current density increases. Cell potential drops as the temperature increases. Compared with Pb-Ag(1%) anode used in industry, the cell voltage decreases by 0.15-0.30 V.
3) The preferred orientations of the zinc deposits change from (101) to (002) with the increase of current density, but the current density of 1 000 A/m2 favors (101) and (002) orientations.
4) The presence of gelatin results in random orientation of the zinc crystals and improves the microhardness of the pure zinc coatings.
5) The manganese-containing electrolytes produce smooth and compacted zinc deposits.
6) Cathode rinsing waters must be recycled back to the electrolysis step in a controlled manner in order to avoid evaporation losses and to comply with the environmental responsibilities. The Pani anode appears to be the most promising technological alternative for industrial zinc production in the future.
References
[1] JHA K M, KUMAR V, SINGH J R. Review of hydrometallurgical recovery of zinc from industrial wastes[J]. Resources Conservation and Recycling, 2001, 33(1): 1-22.
[2] BESTETTI M, DUCATI U, KELSALL G H, LI G, GUERRA E. Use of catalytic anodes for zinc electrowinning at high current densities from purified electrolytes[J]. Canadian Metallurgical Quarterly, 2001, 40(4): 451-458.
[3] SABA A E, ELSBERRY A E. Continuous electrowinning of zinc[J]. Hydrometallurgy, 2000, 54(2/3): 91-106.
[4] STEFANOV Y, VANE I. The in?uence of nickel ions and triethylbenzyl-ammonium chloride on the electrowinning of zinc from sulphate electrolytes containing manganese ions[J]. Hydrometallurgy, 2002, 64(3): 193-203.
[5] CLAASSEN J O, MAYER E H O, RENNIE J, SANDENBERGH R F. Iron precipitation from zinc-rich solutions: Defining the zincor process[J]. Hydrometallurgy, 2002, 67(1/3): 87-108.
[6] RASHKOV S T, DOBREV T S, NONCHERVA Z, STEFANOV Y, RASHKOVA B, PETROVA M. Lead-cobalt anodes for electrowinning of zinc from sulphate electrolytes[J]. Hydrometallurgy, 1999, 52(3): 223-230.
[7] MOSKALYK R R, ALFANTAZI A, TOMBALKIAN A S, VALIC D. Anode effects in electrowinning[J]. Minerals Engineering, 1999, 12(1): 65-73.
[8] XIA Xu-li, ZHITOMIRSKY I, MCDERMID J R. Electrodeposition of zinc and composite zinc-yttria stabilized zirconia coatings[J]. Journal of Materials Processing Technology, 2009, 209(5): 2632-2640.
[9] ALEJANDRO R, IGNACIO G, JOS? L N. Current efficiency studies of the zinc electrowinning process on aluminum rotating cylinder electrode (RCE) in sulfuric acid medium: Influence of different additives[J]. Electrochimica Acta, 2007, 52(24): 6880-6887.
[10] IVANOV I, STEFANOV Y, NONCHEVA Z, PETROVA M, DOBREV T, MIRKOVA L, VERMEERSCH R, DEMAEREL J P. Insoluble anodes used in hydrometallurgy: Part I. Corrosion resistance of lead and lead alloy anodes[J]. Hydrometallurgy, 2000, 57(2): 109-124.
[11] HU Ji-ming, ZHANG Jian-qing, CAO Chu-nan. Oxygen evolution reaction on IrO2-based DSA type electrodes: Kinetics analysis of Tafel lines and EIS[J]. International Journal of Hydrogen Energy, 2004, 29(8): 791-797.
[12] STEFANOV Y, DOBREV T. Developing and studying the properties of Pb-TiO2 alloy coated lead composite anodes for zinc electrowinning[J]. Transactions of the Institute of Metal Finishing, 2005, 83(6): 291-295.
[13] JIN Bi-jian, YANG Xian-wan. Application of gas diffusion anode for zinc hydrometallurgy[J]. Yunnan Metallurgy, 2007, 36(3): 37-39. (in Chinese)
[14] SU Yi, JIN Zuo-mei, DAI Zu-yuan. SO2 anodic reaction kinetics in zinc electrowinning[J]. The Chinese Journal of Nonferrous Metals, 2001, 11(3): 495-498. (in Chinese)
[15] ZHONG Shui-ping, LAI Yan-qing, JIANG Liang-xing. Research development of new anode and techniques for zinc electrowinning[J]. Materials Review, 2008, 22, 22(2): 86-89. (in Chinese)
[16] RASHKOV S T, STEFANOV Y, NONCHEVA Z, PETROVA M, DOBREV T S, KUNCHEV N, PETROV D, VLAEV S T, MIHNEV V, ZAREV S, GEORGIEVA L, BUTTINELLI D. Investigation of the processes of obtaining plastic treatment and electrochemical behaviour of lead alloys in their capacity as anodes during the electroextraction of zinc[J]. Hydrometallurgy, 1996, 40(3): 319-334.
[17] STEFANOV Y, DOBREV T S. Potentiodynamic and electronmicroscopy investigations of lead-cobalt alloy coated lead composite anodes for zinc electrowinning[J]. Transactions of the Institute of Metal Finishing, 2005, 83(6): 296-299.
[18] HUANG Hui, ZHOU Ji-yu, GUO Zhong-cheng. Organic/inorganic acid-doped polyaniline conductive properties of in situ polymerization[J]. Journal of Chemical Engineering of Chinese Universities, 2009, 23 (6): 984-989. (in Chinese)
[19] SEKAR R, JAYAKRISHNAN S. Characteristics of zinc electrodeposits from acetate solutions[J]. J Appl Electrochem, 2006, 36(5): 591-597.
________________________
Foundation item: Project(50974065) supported by the National Natural Science Foundation of China; Project(2009009) supported by the Analysis and Testing Foundation of Kunming University of Science and Technology, China
Corresponding author: GUO Zhong-cheng, Tel: +86-871-8352598; Fax: +86-871-8352599; E-mail: guozhch@vip.163.com
(Edited by YANG You-ping)