J. Cent. South Univ. Technol. (2007)04-0509-05
DOI: 10.1007/s11771-007-0099-8
Chemical component analysis of volatile oil in drug pair Herba Ephedrae-Ramulus Cinnamomi by GC-MS and CRM
CHEN Yong(�� ��)1��LI Xiao-ru(������)2, ZHAO Jun(�� ��)2, ZHOU Tao(�� ��)2, ZOU Qiao(�� ��)2
(1. The Second Xiangya Hospital, Central South University, Changsha 410011, China;
2. School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China)
_________________________________________________________________________
Abstract: Active volatile components in drug pair (DP) Herba Ephedrae-Ramulus Cinnamomi (HE-RC), single drug HE and RC were analyzed by gas chromatography/mass spectrometry (GC/MS), chemometric resolution method (CRM) and overall volume integration. By means of CRM, the two-dimensional data obtained from GC-MS instruments were resolved into a pure chromatogram and a mass spectrum of each chemical compound. In total, 97, 62, and 78 volatile chemical components in volatile oil of HE, RC, and DP HE-RC, were respectively determined qualitatively and quantitatively, accounting for 90.08%, 91.62%, and 89.76% total contents of volatile oil of HE, RC, and DP HE-RC respectively. It is further demonstrated that the numbers of volatile components of DP HE-RC are almost the sum of those of two single drugs, but some relative contents of them are changed. Some new components, such as 1,6-dimethylhepta-1,3,5-triene, tetracyclo[4.2.1.1(2,5).0(9,10)]deca-3,7-diene, globulol and (E,E)-6,10,14-trimethyl-5,9,13-pentadecatrien-2-one are found in DP HE-RC because of chemical reactions and physical changes during decoction.
Key words: drug pair (DP); Herba Ephedrae-Ramulus Cinnamomi; volatile oil; gas chromatography/mass spectrometry (GC-MS); chemometric resolution method (CRM)
_________________________________________________________________________
1 Introduction
The compatibility theory is the core problem of traditional Chinese medicine (TCM). Drug pair (DP) is the basic unit in TCM prescription, which is commonly used in TCM clinic and its composition relative fixed. The focus of DP is to elucidate the effective relationship and compatibilities between two single selected drugs in DP that is the special form of TCM prescription[1]. Only after chemical reactions and physical changes during the decoction are clarified, can the compatibility mechanism of TCM be elucidated.
DP Herba Ephedrae-Ramulus Cinnamomi (HE-RC) is one of the commonly used DPs for treating exterior syndromes[1]. HE has the function of dispelling pathogenic factors from the exterior of the body by diaphoresis, promoting the flow of the lung-��qi�� to allay asthma and inducing diuress to reduce ederma, and RC dispelling pathogenic factors from the exterior of the body by diaphoresis, warming the channels to relieve pain, and reinforcing ��yang�� to promote the flow of ��qi��[2]. HE and RC used together are capable of inducing sweating to dispel exogenous pathogenic factors and dispersing the lung to relieve asthma and pathogenic cold by warming the meridians, and the DP is a drastic prescription of inducing sweating with pungent and warm drugs. For DPs of treating exterior syndromes, volatile oil is one of pharmacologically active constituents[3], but the volatile components in DP HE-RC have not been reported yet. In this study, the combination of GC-MS and chemometric resolution method (CRM) was employed to analyze, quantitatively and qualitatively, the volatile chemical components in HE, RC and DP HE-RC, their constituents were compared with each other, and chemical changes during the processing of decoction were also discussed.
2 Experimental
2.1 Instruments and reagents
The QP2010 GC-MS spectrometer from Shimadzu, Japan, was employed in this research. The single drugs HE and RC were purchased from Nanyang Medicine Company, Henan Province, and were identified by Institute of Materia Medica, Hunan Academy of Traditional Chinese Medicine and Materia Medica, Changsha, China.
2.2 Extraction of volatile oil
The extraction of volatile oil of DP HE-RC: 100 g of each dried single drug powder were weighted exactly and mixed and then processed according to Chinese Pharmacopoeia (2000 version)[4].
The extraction of volatile oil of single drug was carried out using the same method as mentioned above.
2.3 Analytical conditions of volatile oil
Chromatogram conditions: A 30 m��0.25 mm (length��inner diameter) OV-1 capillary column was used. Temperature-increasing procedure: started at 40 ��, increased to 120 �� at 2 ��/min, then to 230 �� at 10 ��/min, maintained for 20 min. Helium carrier gas was employed at a constant flow-rate of 1.0 mL/min. The inlet temperature was 250 ��, and the interfacial temperature 280 ��. The mass spectrometer conditions: the spectrometers were operated in electron-impact (EI) mode, the scan range was 20-600 amu, the ionization energy was 70 eV and the scan rate was 3.8 scan/s. The ionization source temperature was 230 ��, and the solvent delay was 2 min.
2.4 Data analyses
Data analysis was performed on a Pentium III 850 (Intel) personal computer; all programs were coded in Matlab 6.1 for windows. Resolved spectra were identified by matching against the standard mass spectral database of National Institute of Standards and Technology (NIST).
3 Results and discussion
3.1 CRM
CRM is an efficient method for two-dimensional data matrix of chromatogram and spectra. The chromatogram and spectra information contained in the two-dimensional data matrix was analyzed and the local factor analysis method was adopted to identify the pure chromatogram and spectra of each constituent, the principles and concrete method can be found in Refs.[5-9]. CRM was successfully employed to analyze the volatile constituents in TCM[10-17].
3.2 Qualitative analysis of volatile constituents
The GC/MS TICs of HE, RC and DP HE-RC are shown in Fig.1. There are many overlapped peaks in Fig.1, even some peaks that look like the pure peaks are also overlapped by several components. Peak cluster A in Fig.1(b), whose detention time is from 52.88 to 53.04 min, is taken as an example to show the analytical proceeding.
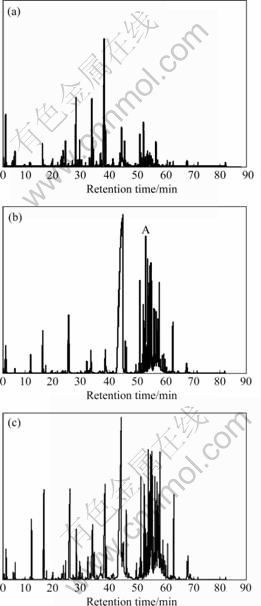
Fig.1 TICs of volatile oils from HE(a), RC(b) and DP HE-RC(c)
From peak cluster A in Fig.1 (b), it can be seen that peak cluster A looks like a pure chromatographic peak, but the curve is not smooth actually. The MS spectra are different at the different points of the left of peak cluster A and their similarities are very low. For example, a compound 1,2,4-benzenetricarboxylic acid, 1,2-dimethyl ester(C11H10O6) was searched directly in NIST, and its similarity is as low as 69%; and 1-nitro-2-propanone��C3H5NO3��was detected in the middle of peak cluster A, its similarity is 72%; and the right side of peak cluster A is also similar to left side, different compounds will be searched out at different points and the similarities are also changed at different points. So, it is obvious that searching directly in NIST will bring qualitative results with low reliability and low accuracy. And it is also difficult to perform the quantitative analysis because of existence of overlapping peaks.
By means of CRM, the results show that peak cluster A consists of 3 constituents (Fig.2), that is, hexadecanal(C16H32O), n-hexadecanoic acid(C16H32O2) and 1,8-dihydroxy-3-methyl-9,10-anthracenedione (C15H10O4) with similarity index of 91.2%��92.35%��95.5% and the relative content of 0.023%��0.089% and 0.045% respectively. The resolved mass spectra and the standard spectra of each compound from the NIST MS library were shown in Figs.3-5. The reliability and accuracy of qualitative analysis are improved greatly because the results are obtained from the pure mass spectrum of each compound.
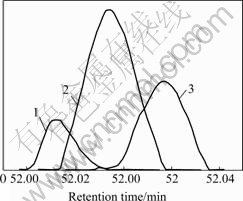
Fig.2 Resolved chromatograms of peak cluster A
1��Hexadecanal; 2��n-hexadecanoic acid;3��1,8-dihydroxy-3-methyl-9,10-anthracenedione
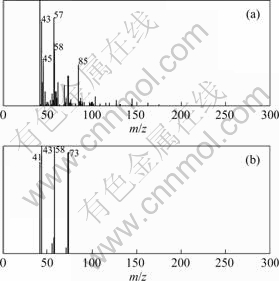
Fig.3 Resolved (a) and standard (b) mass spectra of hexadecanal(C16H32O)
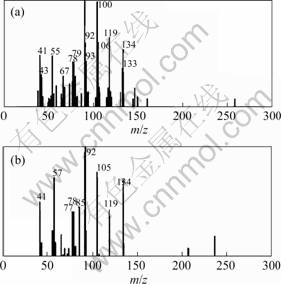
Fig.4 Resolved (a) and standard (b) mass spectra of n-hexadecanoic acid (C16H32O2)
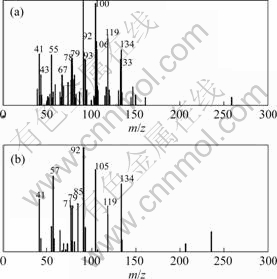
Fig.5 Resolved (a) and standard (b) mass spectra of 1,8-dihydroxy-3-methyl-9,10-anthracenedione (C15H10O4)
The other peak clusters in TIC profiles of RC and all peaks in two chromatographic profiles of HE and DP HE-RC are also resolved in the same way as peaks cluster A. In total, 97��72 and 68 volatile constituents in DP HE-RC, HE and RC are identified respectively.
3.3 Quantitative analysis
The overall volume integration method was adopted to all the analyzed chromatogram peaks in order to obtain the quantitative results of each component. The qualitative components of single drugs HE, RC and DP HE-RC accounting for 93.01%, 92.29% and 91.36% of the total contents of volatile oil, are identified, respectively. The main volatile components in the three systems are shown in Table 1.
Table 1 Main chemical components of volatile oils from HE, RC and DP HE-RC
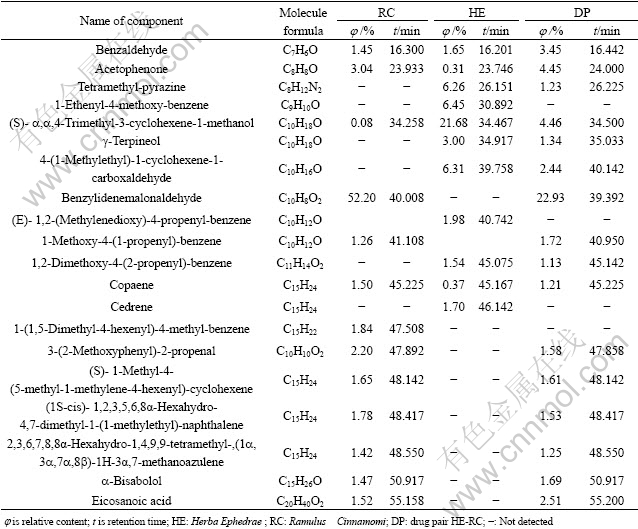
3.4 Comparison of volatile components in DP HE-RC with that in single drugs
Table 1 shows that most of the volatile chemical components of DP are almost from addition of two single drugs. The main volatile constituents in the DP are either from single drug HE, such as a,a,4-trimethyl- (S)-3-cyclohexene-1-methanol, 4-(1-methylethyl)-1- cyclohexene-1-carboxaldehyde, acetophenone, tetra- methyl-pyrazine, or from single drug RC, such as benzylidenemalonaldehyde, acetophenone, copaene, 3-(2-methoxyphenyl)-2-propenal, or from the common constituents in two single drugs such as copaene,��,��-4-trimethyl-3-cyclohexene-1-methanol, ace tophenone, benzaldehyde, but the relative contents of these constituents in the DP are different from those in the single drugs. The experimental results also indicate that 58 new constituents that are not found in both two single drugs appeared in the DP such as (E)- 7,11-dimethyl-3-methylene-1,6,10-dodecatriene,(1S- endo)-a.-caryophyllene,2-methyl-3-methylene-2-(4-methyl-3-pentenyl)-bicyclo[2.2.1]heptane,2-methyl-3-methylene-2-(4-methyl-3-pentenyl)-6,10,14-trimethyl-2-pentadecanone, 2-cyclohexyl-dodecane etc��although the relative contents of these constituents are very low. The physical changes and chemical reactions occurring during the decocting process are probably responsible for the appearance of these new constituents, such as solubilizing effect and co-dissolving effect.
4 Conclusions
1) Totally, 97, 62, and 78 volatile chemical components in HE, RC, and DP HE-RC are respectively determined qualitatively and quantitatively, accounting for 90.08%, 91.62% and 89.76% of total contents of volatile oil of HE, RC, and DP HE-RC, respectively.
2) The numbers of volatile components of DP HE-RC are almost addition of that of two single drugs, but some relative contents of them are changed.
3) Because of chemical reactions and physical changes during the course of decoction, some new components, such as 1,6-dimethylhepta-1,3,5-triene, tetracyclo[4.2.1.1(2,5).0(9,10)]deca-3,7-diene, globulol, are found in DP HE-RC.
References
[1] XU Qing-hua, LIU Li-yun, ZHAO Rui-hua. Collection of Drug Pairs in Traditional Chinese Medicine[M]. Beijing: Traditional Chinese Medicine Press, 1996: 360. (in Chinese).
[2] TIAN Dai-hua. Practical Dictionary of Traditional Chinese Drugs[M]. Beijing: People��s Healthy Press, 2000: 165-167; 984-985. (in Chinese)
[3] SHEN Ying-jun. Study on Exterior-Releasing Drugs and Prescriptions in Traditional Chinese Medicine[M]. Beijing: Traditional Chinese Medicine Press, 2005: 198-199. (in Chinese)
[4] Chinese Pharmacopoeia Committee. Chinese Pharmacopoeia[M]. Beijing: Chemical Industry Press, 2000, Appendis 64. (in Chinese)
[5] KVALHEIM O M, LIANG Y Z. Heuristic evolving latent projections-resolving 2-way multicomponent data.1: Selectivity, latent-projective graph, datascope, local rank and unique resolution[J]. Anal Chem, 1992, 64(8): 936-946.
[6] LIANG Y Z, KVALHEIM O M, KELLER H R et al. Heuristic evolving latent projections-resolving 2-way multicomponent data. 2: Detection and resolution of minor constituents[J]. Anal Chem, 1992, 64(8): 946-953.
[7] LIANG Y Z, KVALHEIM O M, RAHMANI A, et al. Resolution of strongly overlapping two-way multicomponent data by means of heuristic evolving latent projections [J]. J Chemom, 1993, 7(1): 15-43.
[8] LIANG Yi-zeng. White, Gray and Black Multicomponent Systems and Their Chemometric Algorithms[M]. Changsha: Hunan Science and Technology Press, 1996. (in Chinese).
[9] KELLER H R, MASSART D L. Peak purity control in liquid chromatography with photodiode-array detection by a fixed size moving window evolving factor analysis[J]. Anal Chim Acta, 1991, 246(4): 379-390.
[10] GONG Fan, LIANG Yi-zeng, CUI Hui, et al. Determination of volatile components in peptic power by gas chromatography-mass spectrometry and chemometric resolution[J]. J Chromatogr A, 2001, 909: 237-247.
[11] GONG F, LIANG Y Z, XU Q S, et al. Gas chromatography-mass spectrometry and chemometric resolution applied to the determination of essential oils in Cortex Cinnamomi[J]. J Chromatogr A, 2001,905: 193-205.
[12] GONG F, LIANG Y Z, FUNG Y S. Analysis of volatile components from Cortex cinnamomi with hyphenated chromatography and chemometric resolution[J]. J Pharma Biomed Anal, 2004, 34(5): 1029-1047.
[13] LI Xiao-ru, LAN Zheng-gang, LIANG Yi-zeng. Analysis of the volatile chemical constituents of Radix Paeoniae Rubra by GC-MS and chemometric resolution[J]. J Cent South Univ Technol, 2007��14(1): 57-61.
[14] LI Xiao-ru, LIANG Yi-zeng, GUO Fan-qiu. Analysis of volatile oil in rhizoma ligustici chuanxiong�Cradix paeoniae rubra by gas chromatography�Cmass spectrometry and chemometric resolution[J]. Acta Pharmacologica Sinica, 2006��27(4)��491-498.
[15] WU Ming-jian, SUN Xian-jun, DAI Yuan-hui, et al. Determination of constituents of essential oil from Angelica sinensis by gas chromatography -mass spectrometry[J]. J Cent South Univ Technol, 2005, 12(4): 430-436.
[16] LI Xiao-ru, LIANG Yi-zeng, GUO Fan-qiu, et al. Analysis of volatile oil in Semen Persicae��Flos Carthami by gas chromatography-mass spectrometry and chemometric resolution method[J]. Anal Chem, 2007, 25(4): 532-536.(in Chinese)
[17] LI Xiao-ru, ZHOU Tao, LIANG Yi-zeng, et al. Common volatile constituent analysis between herbal pair Chuangxiong Rhizome-Notopterygium Root and its single herbs[J]. Acta Pharmacentica Sinica, 2007, 42(9): 1023-1027. (in Chinse)
__________________________
Foundation item: Project (01962502) supported by the Natural Science Foundation of Hunan Province, China
Received date: 2006-12-24; Accepted date: 2007-03-28
Corresponding author: LI Xiao-ru, Professor; Tel: +86-731-8836376; E-mail: xrli@mail.csu.edu.cn
(Edited by CHEN Wei-ping)