
Initial atmospheric corrosion of zinc in presence of Na2SO4 and (NH4)2SO4
QU Qing(�� ��)1, LI Lei(�� ��)1, BAI Wei(�� ��)2, YAN Chuan-wei(�ϴ�ΰ)3
1. Department of Chemistry, Yunnan University, Kunming 650091, China;
2. Department of Chemistry, Yunnan Nationalities University, Kunming 650092, China;
3. Stake Key Laboratory for Corrosion and Protection, Institute of Metal Research,
Chinese Academy of Sciences, Shenyang 110016, China
Received 14 December 2005; accepted 15 March 2006
Abstract: Initial atmospheric corrosion of zinc in the presence of Na2SO4 and (NH4)2SO4 was investigated via quartz crystal microbalance(QCM) in laboratory at relative humidity(RH) of 80% and 25 ��. The results show that both Na2SO4 and (NH4)2SO4 can accelerate the initial atmospheric corrosion of zinc. The combined effect of Na2SO4 and (NH4)2SO4 on the corrosion of zinc is greater than that caused by (NH4)2SO4 and less than that caused by Na2SO4. Fourier transform infrared spectroscopy(FTIR), X-ray diffractometry(XRD) and scanning electron microscopy(SEM) were used to characterize the corrosion products of zinc. (NH4)2Zn(SO4)2, Zn4SO4(OH)6��5H2O and ZnO present on zinc surface in the presence of (NH4)2SO4 while Zn4SO4(OH)6��5H2O and ZnO are the dominant corrosion products on Na2SO4-treated zinc surface. Probable mechanisms are presented to explain the experimental results.
Key words: zinc; Na2SO4; (NH4)2SO4; atmospheric corrosion
1 Introduction
In view of the widespread use of zinc, as metallic sheet or zinc coatings, it was desirable to study its corrosion behaviour in the wide variety of atmospheres. The atmospheric corrosion of zinc has been studied in field exposures as well as in laboratory with controlled environments[1-12]. The tarnishes of zinc often start at points where dust particles have settled on the surface. ASKEY et al[13] have given a review of studies on the effect of particulate matter on the corrosion of zinc and pointed out that the corrosivity was related to the quantity of water-soluble ionic species in the particles. Major components in aerosols include sea salt and other ionic substances such as ammonium salts, sulfates and nitrates created by acid rain processes in the atmosphere. However, except the studies on the effect of NaCl, investigations of the influence of other atmospheric particulates on the corrosion rates of zinc are scare. Additionally, the initial atmospheric corrosion of zinc in the presence of atmospheric particulates is rarely reported due to measurement difficulties. The information of the initial atmospheric corrosion is very important and helpful to understand the corrosion mechanism.
With the development of highly sensitive instrument quartz crystal microbalance(QCM) in recent years, corrosion effects can be observed after several hours�� exposure. Several studies on atmospheric corrosion of different metals using QCM have been carried out in laboratory exposure and field exposure[9,14,15], their studies show that QCM can be well used to survey the atmospheric corrosion of metals. Thus it is possible to study the initial atmospheric corrosion in the presence of some particles via QCM.
In the present work, the initial atmospheric corrosion of zinc in the presence of Na2SO4 and (NH4)2SO4 is investigated by means of QCM. The corrosion products are characterized by Fourier transform infrared spectroscopy, X-ray diffractometry and scanning electron microscopy. Meanwhile, probable corrosion mechanisms are presented to explain the experimental observations.
2 Experimental
The equipment used in the experiments was described in detail in Ref.[9]. It was made entirely of glass and Teflon. Relative humidity was kept at (80��2)% which was achieved by mixing flows of dry air and the air saturated with water vapor at 60 ��. And the experimental temperature was kept at 25 �� in the whole test process by means of a thermostatically controlled water tank. The environmental temperature was kept at (25��1)�� to avoid condensation in the parts of the system outside the water tank.
To study the initial atmospheric corrosion of zinc in the presence of Na2SO4 and (NH4)2SO4, the QCM (EG&G) with a sensitivity better than 10-8 g/cm2 was applied to measure in situ the small mass changes of zinc. The QCM method for mass change monitoring is based on the inverse piezoelectric effect in which a voltage applied to an ionic crystalline solid, such as quartz, will produce physical distortions of the crystal. Piezoelectric materials have been used as stable oscillators, and it was soon recognized that the addition of mass to an oscillator would change its frequency. For rigid deposits, the change in resonant frequency of the oscillating crystal (D?) is proportional to the change in mass per unit area (Dm/A) of the working electrode. As long as D? is less than a few percent of the resonant frequency of crystal, the linear relation between D? and Dm/A is constant. The equation is expressed as[14, 15]
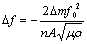 (1)
(1)
where f0 is the fundamental frequency of the crystal, n is order of the harmonic (for rigid deposits, n=1), m is the shear modulus of quartz (2.94?1011 g��cm-1��s-2), and r is the density of quartz (2.648 g/cm3). Thus, for a 9 MHz crystal operating in the fundamental mode, Eqn.(1) can be rewritten as
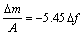 (2)
(2)
Platinum was evaporated on each side of the AT-cut quartz crystal disk to form circular electrodes with an area of 0.2 cm2 and a thickness of around 300 ?m. Before plating zinc, the electrodes were rinsed in ethanol and dried with warm air. Then zinc was plated in an acid bath to the platinum surface of one side of the AT-cut quartz crystal with a thickness of 1.3 ?m. The purity of platinum and zinc used for the QCM electrodes is 99.99%. After washed with ethanol and dried with a hair drier, they were stored in a desiccator over silica gel. Na2SO4, (NH4)2SO4 and Na2SO4+(NH4)2SO4 were pre-deposited on the sample surface by spraying solution of different percent of ethanol saturated with Na2SO4, (NH4)2SO4 and Na2SO4+(NH4)2SO4, respectively. The amount of salt applied was equal on a molar basis with respect to 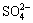 ion (about 0.34 ?mol/cm2), and the amount of Na2SO4 was equal to (NH4)2SO4 in the mixed electrolyte. The samples were stored dry for about 12 h before the start of exposure to test atmosphere. The change in resonant frequency was recalculated into mass variation per unit area (ng/cm2) using Eqn.(2). Corrosion products were then analyzed by SEM(XL30PHILIPS), XRD(PW1700) and FTIR(Magna-IR 560), respectively.
ion (about 0.34 ?mol/cm2), and the amount of Na2SO4 was equal to (NH4)2SO4 in the mixed electrolyte. The samples were stored dry for about 12 h before the start of exposure to test atmosphere. The change in resonant frequency was recalculated into mass variation per unit area (ng/cm2) using Eqn.(2). Corrosion products were then analyzed by SEM(XL30PHILIPS), XRD(PW1700) and FTIR(Magna-IR 560), respectively.
3 Results and discussion
Fig.1 shows the mass gain of zinc as a function of the exposure time at 25 �� and 80% RH. All the mass gain in Fig.1 has subtracted the amount of salt deposition and the amount of irreversible water absorbed by corresponding salt[9]. Fig.1 indicates that the mass gain of zinc increases with the exposure time with and without salt deposition. But the mass gain of zinc in the absence of salt deposition is very small. The mass gain of zinc follows the order as: Na2SO4��Na2SO4+(NH4)2SO4��(NH4)2SO4��no Na2SO4 and (NH4)2SO4. So the combined effect of Na2SO4 and (NH4)2SO4 on the corrosion of zinc is greater than that caused by (NH4)2SO4 and less than that caused by Na2SO4.
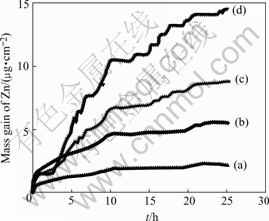
Fig.1 Curves of mass gain ��s exposure time for zinc samples treated with different salts at RH 80% and 25 ��: (a) No Na2SO4 and (NH4)2SO4; (b) 0.34 ��mol/cm2 (NH4)2SO4; (c) 0.17 ��mol/cm2 (NH4)2SO4 and 0.17 ��mol/cm2 Na2SO4; (d) 0.34 ��mol/cm2 Na2SO4
The initial atmospheric corrosion products on zinc surface in the present of Na2SO4 and (NH4)2SO4 were respectively analyzed by XRD and FTIR. Analysis of the samples by XRD[16] under that condition revealed that zinc hydroxysulfate, Zn4SO4(OH)6��5H2O and zincite, ZnO were the main corrosion products in the presence of Na2SO4 while ammonium zinc sulfate, (NH4)2Zn(SO4)2, Zn4SO4(OH)6��5H2O and ZnO were detected in the products in the presence of (NH4)2SO4. The position and relative intensities for the three compounds agree well with the standard values obtained from the database. Figs.2(a), (b) and (c) give the FTIR spectra of the products on zinc deposited with Na2SO4, (NH4)2SO4 and Na2SO4+(NH4)2SO4 exposed to air after 25 h, respectively. The different reference compounds have characteristic absorption bands at different positions[17]. Zincite only has the bands in the 350 to 600 cm-1 region corresponding to the zinc-oxygen bond. Hydrozincite has strong bands at 1 380 and 1 510 cm-1, corresponding to the asymmetric stretching vibrations of carbonate ion, and hydrozincite also has bands at 830, 735 and 675 cm-1. In Fig.2(a), strong bands at 1 510 and 1 380 cm-1 and weak bands at 830 and 710 cm-1 suggest that hydrozincite may exist in the products of zinc in the presence of Na2SO4. The broader band at 3 420 cm-1 is for OH stretching, and the absorption bands at 1 120 cm-1 and 1 040 cm-1 are attributed to 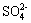 ion, which indicates that hydroxysulfate also exists in the products. In Fig.2(b), the absorption bands around 3 500, 1 630, 1 120, 1 070 and 980 cm-1 can be assigned to the characteristic absorptions of Zn4SO4(OH)6��5H2O, the strong bands around 3 330 cm-1 and weak band at 1 426 cm-1 are attributed to NH4+ bending, and the absorption band around 1 750 cm-1 corresponds to N��H, which probably originates from an ammonia-zinc complex, (NH4)2Zn(SO4)2. The characteristic curve in Fig.2(c) is similar to that in Fig.2(b), and the transmittance of each band in Fig.2(c) is lower than that in Fig.2(b). These indicate that the corrosion products on zinc deposited with Na2SO4+(NH4)2SO4 are the same as the corrosion products on zinc deposited with (NH4)2SO4, but the amount of corrosion products on zinc deposited with Na2SO4+(NH4)2SO4 are more than that deposited with (NH4)2SO4. With the FTIR it was possible to observe the formation of phase that was not detectable with XRD (hydrozincite) after exposures to air. This may be attributed to the higher surface sensitivity of FTIR; That is, due to the air used in the experiment has been cleaned, only a little amount of CO2 is present in the air, hydrozincite formed on the corrosion products is so minor that it cannot be detected by XRD in the present of Na2SO4. Furthermore, due to the spectra in the experiment were obtained in the region from 650 cm-1 to 4 000 cm-1, and zincite only has the bands in the 350 cm-1 to 600 cm-1 region corresponding to the zinc-oxygen bond, so zincite can not be detected by FTIR in this study. The combined results obtained from XRD and FTIR indicate that (NH4)2Zn(SO4)2, Zn4SO4(OH)6��5H2O and ZnO present on zinc surface in the presence of (NH4)2SO4 while Zn4SO4(OH)6��5H2O and ZnO are the dominant corrosion products on Na2SO4-treated zinc surface.
ion, which indicates that hydroxysulfate also exists in the products. In Fig.2(b), the absorption bands around 3 500, 1 630, 1 120, 1 070 and 980 cm-1 can be assigned to the characteristic absorptions of Zn4SO4(OH)6��5H2O, the strong bands around 3 330 cm-1 and weak band at 1 426 cm-1 are attributed to NH4+ bending, and the absorption band around 1 750 cm-1 corresponds to N��H, which probably originates from an ammonia-zinc complex, (NH4)2Zn(SO4)2. The characteristic curve in Fig.2(c) is similar to that in Fig.2(b), and the transmittance of each band in Fig.2(c) is lower than that in Fig.2(b). These indicate that the corrosion products on zinc deposited with Na2SO4+(NH4)2SO4 are the same as the corrosion products on zinc deposited with (NH4)2SO4, but the amount of corrosion products on zinc deposited with Na2SO4+(NH4)2SO4 are more than that deposited with (NH4)2SO4. With the FTIR it was possible to observe the formation of phase that was not detectable with XRD (hydrozincite) after exposures to air. This may be attributed to the higher surface sensitivity of FTIR; That is, due to the air used in the experiment has been cleaned, only a little amount of CO2 is present in the air, hydrozincite formed on the corrosion products is so minor that it cannot be detected by XRD in the present of Na2SO4. Furthermore, due to the spectra in the experiment were obtained in the region from 650 cm-1 to 4 000 cm-1, and zincite only has the bands in the 350 cm-1 to 600 cm-1 region corresponding to the zinc-oxygen bond, so zincite can not be detected by FTIR in this study. The combined results obtained from XRD and FTIR indicate that (NH4)2Zn(SO4)2, Zn4SO4(OH)6��5H2O and ZnO present on zinc surface in the presence of (NH4)2SO4 while Zn4SO4(OH)6��5H2O and ZnO are the dominant corrosion products on Na2SO4-treated zinc surface.
Fig.3 shows the SEM morphologies of the Na2SO4
Fig.2 IR spectra of products on zinc deposited with Na2SO4(a), (NH4)2SO4(b) and Na2SO4+(NH4)2SO4(c)
and (NH4)2SO4-treated sample after exposure to air for about 30 h at 25 �� and RH 80%, respectively. As can be seen from Fig.3(a) that the zinc surface in the presence of Na2SO4 is covered by an inhomogeneous corrosion products layer. The uneven distribution of the corrosion products is due to the migration of aqueous ions in the electrical field established between anodic and cathodic areas on zinc surface. In contrast, the distribution of the corrosion products on zinc in the presence of (NH4)2SO4 is rather even (Fig.3(b)).
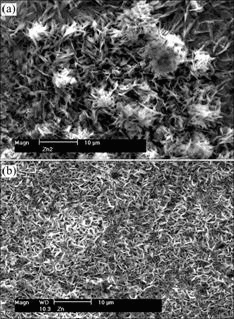
Fig.3 SEM morphologies of zinc samples treated with Na2SO4(a) and (NH4)2SO4(b) after exposure to air at 25 �� and RH 80%
The present study shows that the presence of Na2SO4 and (NH4)2SO4 can accelerate the initial atmospheric corrosion of zinc, which can be explained by using the electrochemical process that takes place in the corrosion of zinc. When the surface of zinc is wet, the anodic dissolution of zinc is balanced by oxygen reduction in the cathodic areas. During the corrosion process of the sample treated with Na2SO4, cations, i.e. Na+, Zn2+ will migrate towards the cathodic areas while anions, i.e. 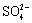 and OH- will move towards the zinc dissolution sites. Then the pH and the concentration of
and OH- will move towards the zinc dissolution sites. Then the pH and the concentration of 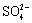 gradually rise in the anodic sites, so the insoluble zinc hydroxysulfate, Zn4SO4(OH)6��5H2O, forms gra- dually:
gradually rise in the anodic sites, so the insoluble zinc hydroxysulfate, Zn4SO4(OH)6��5H2O, forms gra- dually:
3ZnO+Zn2++8H2O+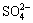 ��Zn4SO4(OH)6?5H2O (3)
��Zn4SO4(OH)6?5H2O (3)
In the cathodic areas where sulfate activities are low and pH is high, ZnO tends to form. So the corrosion products in the cathodic areas are different from those in the anodic sites, which leads to the uneven distribution of the products (Fig.3(a)).
According to studies by LOBNIG et al[18], the corrosion of metals in the presence of  may be separated to anodic and cathodic sites. The following anodic reaction leads to a decrease in pH:
may be separated to anodic and cathodic sites. The following anodic reaction leads to a decrease in pH:
Zn+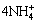 ��
��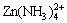 +4H++2e (4)
+4H++2e (4)
The cathodic reaction of oxygen, which in this initial stage, is most likely the reduction of oxygen, occurs at the outer edges, and leads to an increase in pH
O2+2H2O+4e��4OH- (5)
Resembling the initial process in the presence of Na2SO4, Zn4SO4(OH)6��5H2O tends to form gradually in the anodic sites. However in the cathodic sites where pH is higher,  will react with OH-:
will react with OH-:
 +OH-��NH3+H2O (6)
+OH-��NH3+H2O (6)
The above reaction decreases the pH of the electrolyte, and the following reactions will take place in these areas
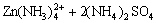 �� (NH4)2Zn(SO4)2+6NH3+2H+ (7)
�� (NH4)2Zn(SO4)2+6NH3+2H+ (7)
4(NH4)2Zn(SO4)2+11H2O�� Zn4SO4(OH)6?5H2O+8NH3+7H2SO4 (8)
So ammonium zinc sulfate, (NH4)2Zn(SO4)2 and zinc hydroxysulfate, Zn4SO4(OH)6��5H2O present in zinc corrosion layer.
As can be seen from the above analyses, Zn4SO4(OH)6��5H2O can accumulate both in anodic and cathodic sites on zinc surface, so the distribution of the corrosion products in the presence of (NH4)2SO4 is more even than that in the presence of Na2SO4. The insoluble and insulating zinc hydroxysulfate is unfavorable to oxygen diffusion and blocks the active areas, so the corrosion of zinc becomes more difficult.
Therefore, it is suggested that the lower corrosion rate of zinc in the presence of (NH4)2SO4 be attributed to the acidity of the ions and the formation of the insoluble zinc hydroxysulfate, Zn4SO4(OH)6��5H2O. Equilibrium 2 and 4 imply that the formation of 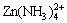 and NH3 decreases the concentration of
and NH3 decreases the concentration of  at the beginning of the corrosion, resulting in a decrease of conductivity in the electrolyte. Furthermore, Zn4SO4(OH)6��5H2O, which may inhibit the electrochemical corrosion, can form in both cathodic and anodic sites. This can be confirmed by the more even corrosion attack in the presence of (NH4)2SO4.
at the beginning of the corrosion, resulting in a decrease of conductivity in the electrolyte. Furthermore, Zn4SO4(OH)6��5H2O, which may inhibit the electrochemical corrosion, can form in both cathodic and anodic sites. This can be confirmed by the more even corrosion attack in the presence of (NH4)2SO4.
4 Conclusions
1) The present study by QCM shows that both Na2SO4 and (NH4)2SO4 dissolved in the absorbed water present on the zinc surface give rise to corrosion attack. Mass gain of zinc increases with the exposure time, and mass gain decreases in the order as: Na2SO4�� Na2SO4+(NH4)2SO4��(NH4)2SO4��no Na2SO4 and (NH4)2SO4.
2) The corrosion products are evener distributing on the zinc surface in the presence of (NH4)2SO4 than that in the presence of Na2SO4. The lower corrosion rate of zinc in the presence of (NH4)2SO4 may be attributed to the decrease of conductivity in the electrolyte and precipitation of insoluble zinc hydroxysulfate both in anodic and cathodic sites. (NH4)2Zn(SO4)2, Zn4SO4(OH)6��5H2O and ZnO present on zinc surface in the presence of (NH4)2SO4 while Zn4SO4(OH)6��5H2O and ZnO are the dominant corrosion products on Na2SO4-treated zinc surface.
References
[1] ALMEIDA E, MORCILLO M, ROSALES B. Atmospheric corrosion of zinc- Part 1: Rual and urban atmospheres [J]. British Corrosion Journal, 2000, 35(4): 284-288.
[2] ALMEIDA E, MORCILLO M, ROSALES B. Atmospheric corrosion of zinc-Part 2: Marine atmospheres [J]. British Corrosion Journal, 2000, 35(4): 289-296.
[3] GRAEDEL T E. Corrosion mechanisms for zinc exposed to the atmosphere [J]. J Electrochem Soc, 1989, 136(4):193C-203C.
[4] SVENSSON J E, JOHANSSON L G. Laboratory studies on the atmospheric corrosion of zinc in the presence of O3, NO2 and SO2 [J]. J Electrochem Soc, 1993, 140(8): 2210-2216.
[5] SVENSSON J E, JOHANSSON L G. A laboratory study of the initial stages of the atmospheric corrosion of zinc in the presence of NaCl; Influence of SO2 ad NO2 [J]. Corrosion Science, 1993, 34(5): 721-740.
[6] HAYNIE F H, UPHAM J B, Effects of atmospheric sulfur dioxide on the corrosion of zinc [J]. Material Performance, 1970, 9(8): 35-40.
[7] LINDSTR?M R, SVENSSON J E, JOHANSSON L G. The atmospheric corrosion of zinc in the presence of NaCl [J]. J Electrochem Soc, 2000, 147(5): 1751-1757.
[8] QU Q, YAN C W, BAI W, CAO C N. Influence of NaCl deposition on atmospheric corrosion of zinc in the presence of SO2 [J]. Acta Metallurgica Sinica, 2001, 37(11): 72-763. (in Chinese)
[9] QU Q, YAN C W, WAN Y, CAO C N. Effects of NaCl and SO2 on the initial atmospheric corrosion of zinc [J]. Corrosion Science, 2002, 44(11): 2789-2803.
[10] QU Q, YAN C W, ZHANG L, LIU G H, CAO C N. Initial atmospheric corrosion of zinc sprayed with NaCl [J]. Trans Nonferrous Met Soc China, 2003, 13(5): 1243-1246.
[11] QU Q, LI L, BAI W, YAN C W, CAO C N. Effects of NaCl and NH4Cl on the initial atmospheric corrosion of zinc [J]. Corrosion Science, 2005, 47(11): 2832-2840.
[12] NEUFELD A K, COLE I S, BOND A M, FURMAN S A. The initial atmospheric mechanism of corrosion of zinc by sodium chloride particle desposition [J]. Corrosion Science, 2002, 44(3): 555-572.
[13] ASKEY A, LYON S B, THOMPSON G E, JOHNSON J B, WOOD G C, SAGE P W, COOKE M J. The effect of fly-ash particulates on the atmospheric corrosion of zinc and mild steel [J]. Corrosion Science, 1993, 34(7): 1055-1081.
[14] ZAKIPOUR S, LEYGRAF C. Quartz crystal microbalance applied to studies of atmospheric corrosion of metals [J]. British Corrosion Journal, 1992, 27(4): 295-298.
[15] DEAKIN M R, MELROY O R. Monitoring the growth of an oxide film on aluminum in situ with the quartz crystal microbalance [J]. J Electrochem Soc, 1989, 136(2): 349-352.
[16] QU Qing. The Role of Several Soluble Salts and SO2 in the Initial Atmospheric Corrosion of Metals [D]. Shenyang: Institute of Metal Research, Chinese Academy of Sciences, 2002. (in Chinese)
[17] ZHU F, PERSSON D, THIERRY D, et al. Formation of corrosion products on open and confined zinc surfaces exposed to periodic wet/dry conditions [J]. Corrosion, 2000, 56(12): 1256-1265.
[18] LOBING R E, SICONOLFI D J, GRUNDREIER G, STRECKEL H, FRANKENTHAL R P, STRATMANN M, SINCLAIR J D. Atmospheric corrosion of aluminum in the presence of ammonium sulfate particles [J]. J Electrochem Soc, 1996, 143(4): 1175-1182.
(Edited by LONG Huai-zhong)
Foundation item: Project(50261004) supported by the National Natural Science Foundation of China
Corresponding author: QU Qing; Tel: +86-871-5035789; E-mail: quqing@ynu.edu.cn