微波热解法制备的炭涂层对LiNi1/3Mn1/3Co1/3O2性能的影响
来源期刊:中国有色金属学报(英文版)2013年第10期
论文作者:韩亚梅 张正富 张利波 彭金辉 傅梦笔 C. SRINIVASAKANNAN 杜 江
文章页码:2971 - 2976
关键词:锂离子电池;正极材料;炭涂层;微波热解;电化学性能
Key words:lithium-ion battery; cathode material; carbon coating; microwave pyrolysis method; electrochemical performance
摘 要:报道了炭包覆锂离子电池正极材料LiNi1/3Mn1/3Co1/3O2的新工艺。炭涂层由前驱体葡萄糖通过微波热解而形成。采用X射线粉末衍射(XRD)、扫描电镜、X射线荧光测试和恒流充放电测试来表征所制备的材料。XRD结果表明,炭包覆没有改变LiNi1/3Mn1/3Co1/3O2材料的相结构。SEM结果表明,炭包覆的LiNi1/3Mn1/3Co1/3O2颗粒表面变得粗糙。充放电测试结果显示,炭包覆的LiNi1/3Mn1/3Co1/3O2的循环性能与未包覆的相比得到提高。炭包覆的LiNi1/3Mn1/3Co1/3O2在0.2C倍率下循环50次的容量保持率由84.8%提升到95.5%,且高倍率下材料的容量保持率得到提高。
Abstract: A novel synthesis method of carbon-coated LiNi1/3Mn1/3Co1/3O2 cathode material for lithium-ion battery was reported. The carbon coating was produced from a precursor, glucose, by microwave-pyrolysis method. The prepared powders were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), X-ray fluorescence (XRF) and charge/discharge tests. XRD results indicate that the carbon coating does not change the phase structure of LiNi1/3Mn1/3Co1/3O2 material. SEM results show that the surface of spherical carbon-coated material becomes rough. Electrochemical performance results show that the carbon coating can improve the cycling performance of LiNi1/3Mn1/3Co1/3O2. The specific discharge capacity retention of the carbon-coated LiNi1/3Mn1/3Co1/3O2 reached 85.0%-96.0% at the 50th cycle at 0.2C rate, and the specific discharge capacity retention is improved at a high rate.
Trans. Nonferrous Met. Soc. China 23(2013) 2971-2976
Ya-mei HAN1,2,3, Zheng-fu ZHANG1,2,3, Li-bo ZHANG2,3, Jin-hui PENG2,3, Meng-bi FU2,3, C. SRINIVASAKANNAN4, Jiang DU1,2,3
1. Faculty of Materials Science and Engineering, Kunming University of Science and Technology, Kunming 650093, China;
2. National Local Joint Engineering Laboratory of Engineering Applications of Microwave Energy and Equipment Technology, Kunming University of Science and Technology, Kunming 650093, China;
3. Key Laboratory of Unconventional Metallurgy, Ministry of Education, Kunming University of Science and Technology, Kunming 650093, China;
4. Chemical Engineering Department, The Petroleum Institute, P.O. Box 2533, Abu Dhabi, United Arab Emirates
Received 22 August 2012; accepted 19 April 2013
Abstract: A novel synthesis method of carbon-coated LiNi1/3Mn1/3Co1/3O2 cathode material for lithium-ion battery was reported. The carbon coating was produced from a precursor, glucose, by microwave-pyrolysis method. The prepared powders were characterized by scanning electron microscopy (SEM), X-ray diffraction (XRD), X-ray fluorescence (XRF) and charge/discharge tests. XRD results indicate that the carbon coating does not change the phase structure of LiNi1/3Mn1/3Co1/3O2 material. SEM results show that the surface of spherical carbon-coated material becomes rough. Electrochemical performance results show that the carbon coating can improve the cycling performance of LiNi1/3Mn1/3Co1/3O2. The specific discharge capacity retention of the carbon-coated LiNi1/3Mn1/3Co1/3O2 reached 85.0%-96.0% at the 50th cycle at 0.2C rate, and the specific discharge capacity retention is improved at a high rate.
Key words: lithium-ion battery; cathode material; carbon coating; microwave pyrolysis method; electrochemical performance
1 Introduction
Lithium ion battery has become the state-of-power sources for portable appliances such as cellular phones, notebook computers, digital cameras and also a prime candidate for hybrid electric vehicles (HEVs), plug-in hybrid electric vehicles (PHEVs) and electric vehicles (EVs) for reasons of high power density, high energy density, safety and cycling performance [1-3]. Literatures related to lithium transition metal oxides and their derivatives, such as LiMn2O4, LiCoO2 and Li(Ni,Co)O2, have been extensively reported, which are commercially available as 4 V class cathode materials for lithium ion batteries. Among them, LiCoO2 is widely used as positive electrode in commercial lithium secondary batteries. However, utilization of LiCoO2 has the drawback due to the scarcity of cobalt, being expensive and toxic. The cathode material synthesized by OHZUKU and MAKIMURA [4], LiNi1/3Mn1/3Co1/3O2, attracts lots of attention for its lower cost, less toxicity and higher capacity, which is superior to the commercial material of LiCoO2. It was reported that even 200 mA・h/g can be attainable by charging up to 2.8 and 4.6 V, with superior cycle performance [5]. Although LiNi1/3Mn1/3Co1/3O2 exhibits excellent performance comparatively, it has drawbacks, such as low electronic conductivity, low tap density and relatively low cycling performance at high-rate, which impede its industrial acceptance. Some measures have been reported to improve the cycle performance of LiNi1/3Mn1/3Co1/3O2, such as doping small amounts of additional ions [6-8], and modification of the surfaces [9-11]. In particular, a thin carbon coating on particles is known to be effective not only in enhancing the conductivity of the cathode material but also in protecting the particles from chemical attack by the electrolyte. However, only a few studies on the carbon-coated LiNi1/3Co1/3Mn1/3O2 material were reported. Diverse methods of carbon coatings, such as carbon black compounds or pyrolysis of adsorbed organic compounds [12,13], were employed. Nevertheless, all of these methods need some hours for providing carbon coating on the particles at an elevated temperature. It is well known that microwave process is widely used to synthesize many inorganic materials and advanced sinter materials [14,15], such as LiMn2O4 [16-18], LiNiO2 [19], LiFePO4 [20] and LiCoO2 [21], due to its short reaction time, low energy consumption, high efficiency of synthesis and product crystal structure [22].
In this study, in order to decrease the process time, reduce the synthesis cost and improve the cycle performance of cathode material, carbon was coated on the surface of LiNi1/3Mn1/3Co1/3O2 powder by microwave pyrolysis method in a novel microwave reactor (as shown in Fig. 1). Coin cells were prepared with the carbon-coated LiNi1/3Mn1/3Co1/3O2 powder and lithium metal, and their electrochemical performance was measured.
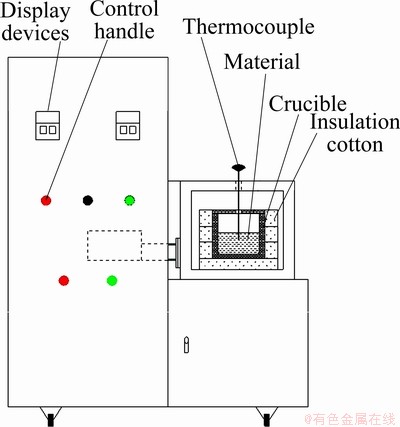
Fig. 1 Schematic of microwave reactor
2 Experimental
2.1 Sample preparation
The spherical (Ni1/3Mn1/3Co1/3)(OH)2 (Lanzhou Jinchuan) and Li2CO3 (China Lithium, 99.9%) powders with molar ratio of 1:1.06 were ball-milled thoroughly. The excess amount of Li was used to compensate for the loss of Li during calcination. The powder after drying at 323 K was sintered in the microwave reactor at 1243 K for 30 min. LiNi1/3Mn1/3Co1/3O2 powder was mixed with glucose of 2% or 3% in mass fraction by mechanically mixing for 16 h, and then pyrolyzed in flowing nitrogen in a microwave reactor at 673 K for 15 min.
2.2 Experimental equipment
Experiments were carried out using a novel microwave reactor. The schematic is shown in Fig. 1. The microwave reactor with a frequency of 2450 MHz and a maximum power output of 1500 W was made by Kunming University of Science and Technology, China.
2.3 Characterization
X-ray diffraction (XRD) data for the finely ground samples were collected at 298 K using a Bruker D8 X-ray diffractometer with Cu Kα radiation (λ=1.5406 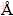 ). It was operated at 40 kV and 300 mA in the 2θ range of 10°-80° in the continuous scan mode with the step size of 0.01° and the scan rate 1.0 (°)/min. The particle shapes and morphologies of the bare and carbon-coated LiNi1/3Mn1/3Co1/3O2 materials were obtained using scanning electron microscopy (SEM, Philips XL-30E). Rigaku ZSX100e X-ray fluorescence spectrometer was applied to multi-element determination in the bare and carbon-coated LiNi1/3Mn1/3Co1/3O2 materials. The electrodes were fabricated from a 85:10:5 (mass fraction) mixture of active materials, acetylene black as the current conductor and polyvinylidene difluoride (PVDF) as the binder. The PVDF was dissolved in n-methyl pyrrolidinone before the active material and the conductor mixture were added. After homogenization, the slurry was evacuated for 20 min to remove the residual air. The slurry was then coated on a thin aluminum foil (20-μm thick) and dried overnight at 60-70 °C. The electrode was pressed under a pressure of 10 MPa and punched into 12-mm diameter disks. The electrochemical cells were prepared as 2025-coin cell hardware with lithium metal foil as both the counter and reference electrodes. The cells were assembled in a Ar-filled glove box. The electrolyte used for analysis was 1 mol/L LiPF6 in ethylene carbonate/diethyl carbonate (1:1).
). It was operated at 40 kV and 300 mA in the 2θ range of 10°-80° in the continuous scan mode with the step size of 0.01° and the scan rate 1.0 (°)/min. The particle shapes and morphologies of the bare and carbon-coated LiNi1/3Mn1/3Co1/3O2 materials were obtained using scanning electron microscopy (SEM, Philips XL-30E). Rigaku ZSX100e X-ray fluorescence spectrometer was applied to multi-element determination in the bare and carbon-coated LiNi1/3Mn1/3Co1/3O2 materials. The electrodes were fabricated from a 85:10:5 (mass fraction) mixture of active materials, acetylene black as the current conductor and polyvinylidene difluoride (PVDF) as the binder. The PVDF was dissolved in n-methyl pyrrolidinone before the active material and the conductor mixture were added. After homogenization, the slurry was evacuated for 20 min to remove the residual air. The slurry was then coated on a thin aluminum foil (20-μm thick) and dried overnight at 60-70 °C. The electrode was pressed under a pressure of 10 MPa and punched into 12-mm diameter disks. The electrochemical cells were prepared as 2025-coin cell hardware with lithium metal foil as both the counter and reference electrodes. The cells were assembled in a Ar-filled glove box. The electrolyte used for analysis was 1 mol/L LiPF6 in ethylene carbonate/diethyl carbonate (1:1).
3 Results and discussion
3.1 Structure
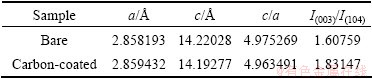
Figure 2 shows the XRD patterns and Miller indices of the bare and carbon-coated LiCo1/3Ni1/3Mn1/3O2. In Fig. 2, the XRD patterns of the two materials indicate a single phase of α-NaFeO2 structure with space group of 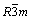 . The splits in the (006/012) and (108/110) are around at 38° and 65° doublets, indicating the formation of a highly ordered layered structure. The ratios of intensities of the I(003)/I(104) of the bare and carbon-coated material are 1.60759 and 1.83147, respectively, above the values reported for compounds like LiNi1-xCoxO2 to deliver good electrochemical performance [23]. No other impurity phases were observed in the XRD patterns for the carbon-coated material, which suggests that the structure of LiNi1/3Co1/3Mn1/3O2 is not affected by the carbon coating. The structural parameters of the two samples are summarized in Table 1 according to the XRD data and the hexagonal crystal spacing calculation equation:
. The splits in the (006/012) and (108/110) are around at 38° and 65° doublets, indicating the formation of a highly ordered layered structure. The ratios of intensities of the I(003)/I(104) of the bare and carbon-coated material are 1.60759 and 1.83147, respectively, above the values reported for compounds like LiNi1-xCoxO2 to deliver good electrochemical performance [23]. No other impurity phases were observed in the XRD patterns for the carbon-coated material, which suggests that the structure of LiNi1/3Co1/3Mn1/3O2 is not affected by the carbon coating. The structural parameters of the two samples are summarized in Table 1 according to the XRD data and the hexagonal crystal spacing calculation equation:
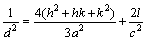 (1)
(1)

Fig. 2 XRD patterns of bare LiNi1/3Mn1/3Co1/3O2 (a) and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 (b)
Table 1 Lattice structural parameters of bare and 2% carbon- coated LiNi1/3Mn1/3Co1/3O2
3.2 Morphology
The SEM images of the bare and carbon-coated LiNi1/3Mn1/3Co1/3O2 particles are shown in Fig. 3. For the bare LiNi1/3Mn1/3Co1/3O2, the rod-shaped particles with a submicron size are agglomerated to form a sphere- shaped particle with diameter of 10-14 μm. A smooth and clean surface is observed in the bare particles. After coating carbon, the surface of LiNi1/3Mn1/3Co1/3O2 particles is rough. Similar SEM images were also observed in ZrFx-coated Li[Ni1/3Co1/3Mn1/3]O2 [24] and the carbon-coated LiNi1/3Mn1/3Co1/3O2 [25]. Compared with the average size of the primary grain of bare particles, that of the carbon-coated material increased from 540 nm to 750 nm. The primary grain of the carbon-coated LiNi1/3Mn1/3Co1/3O2 particles treated by microwave is more distinct than that of the bare one.
3.3 Elemental analysis
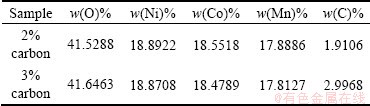
Table 2 shows the X-ray fluorescence results of 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 and 3% carbon coated LiNi1/3Mn1/3Co1/3O2. The XRF analysis indicates that O, Ni, Co, Mn and C are in the carbon-coated LiNi1/3Mn1/3Co1/3O2 particle. The practical contents of carbon are 1.9106% and 2.9968% after microwave pyrolysis of 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 and 3% carbon-coated LiNi1/3Mn1/3Co1/3O2, respectively.

Fig. 3 SEM images of bare LiNi1/3Mn1/3Co1/3O2 (a) and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 (b)
Table 2 XRF results of 2% carbon and 3% carbon coated LiNi1/3Mn1/3Co1/3O2
3.4 Electrochemical performance
Figure 4 shows the discharge curves of bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes cycled between 2.75 and 4.3 V at 0.2C rate. The specific discharge capacities for the LiNi1/3Mn1/3Co1/3O2 electrode without carbon coating are 161.7 mA・h/g at the first time and 137.4 mA・h/g at the fiftieth time at 0.2C rate, while they are 159.3 mA・h/g at the first time and 152.9 mA・h/g at the fiftieth time at 0.2C rate for the 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode. As can be seen, the first specific discharge capacity of carbon- coated sample at 0.2C reduces slightly because a layer of barrier, carbon, is covered on the positive electrode material surface, which blocks cathode materials to contact with electrolyte.

Fig. 4 Discharge curves of bare (a) and 2% carbon-coated (b) LiNi1/3Mn1/3Co1/3O2 electrodes tested at 0.2C rate in 2.75-4.3 V
Figure 5 shows the discharge curves of bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes cycled between 2.75 and 4.3 V at 2C rate. The specific discharge capacities for the LiNi1/3Mn1/3Co1/3O2 electrode without carbon coating are 105.5 mA・h/g at the first time and 86.4 mA・h/g at the 50th time at 2C rate, while they are 130.8 mA・h/g at the first time and 112.6 mA・h/g at the 50th time at 2C rate for the 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode.
Comparison of the cycling performance between the bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes at 0.2C rate are shown in Fig. 6. The specific discharge capacity of the bare LiNi1/3Mn1/3Co1/3O2 electrode at 0.2C rate at the first circle is about 161.7 mA・h/g, while the 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode exhibits comparable specific discharge capacity of 152.9 mA・h/g. However, after the 50 cycles the specific capacity retentions of the bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes are 84.8% and 95.5%, respectively.

Fig. 5 Discharge curves of bare (a) and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 (b) LiNi1/3Mn1/3Co1/3O2 electrodes tested at 2C rate in 2.75-4.3 V
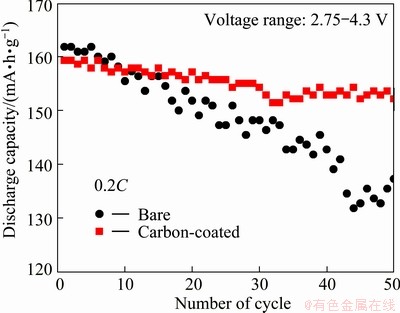
Fig. 6 Cyclic performance of bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes in 2.75-4.3 V at 0.2C rate
Figure 7 shows the specific discharge capacities and cyclic properties of the bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes at 0.2C, 2C, 4C and 8C rates in the voltage range of 2.75-4.3V. At 2C rate, after the 50 cycles the specific capacity retention of 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode is 86.1%, while that of the bare LiNi1/3Mn1/3Co1/3O2 electrode is 81.9%.

Fig. 7 Specific discharge capacities and cyclic performance of bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes in 2.75-4.3 V at 0.2C, 2C, 4C and 8C rates
Figure 8 displays the capacity retention (ratio of the discharge capacity at an assigned discharge rate to the discharge capacity at a 0.2C rate) as a function of the rate. After 50 cycles the specific capacity retention of the bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes are 69.7% and 69.9% at 4C rate, respectively. At 8C rate, after 50 cycles the capacity retentions of the bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes are 50.0% and 59.8%, respectively. The specific capacity retention of the carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode is better than that of bare LiNi1/3Mn1/3Co1/3O2 electrode at any discharge rates because the carbon coating blocks the contact between cathode materials and electrolyte, which reduces the corrosion of positive electrode material.
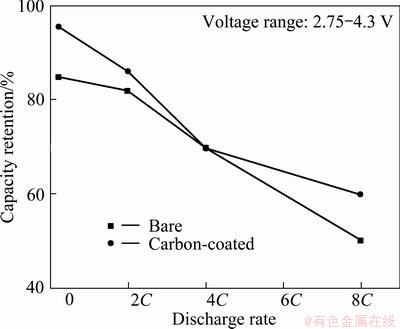
Fig. 8 Specific capacity retentions of bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes as function of discharge rate
Although the specific discharge capacities of the bare and 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrodes decrease with the increase of charge-discharge cycles, the specific capacity retention of the 2% carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode remains better than the bare LiNi1/3Mn1/3Co1/3O2 electrode. The result affirms that the carbon-coated LiNi1/3Mn1/3Co1/3O2 electrode exhibits good discharge rate capability and cycle performance. Perhaps, it could be attributed to the increase in electronic conductivity of the carbon-coated material, reducing the cell polarization and oxygen evolution from the cathodes at the end of charge [25]. The carbon coating could act as a protection layer of positive material, which restrains the dissolution of metal ions and reduces the impedance of the charge transfer [26]. The improved electronic conductivity of the carbon-coated material surface may contribute to the improvement in electrical contact of the material among particles, thereby improving the electrochemical performance of the material [27].
4 Conclusions
1) The carbon-coated LiNi1/3Mn1/3Co1/3O2 material was successfully synthesized by microwave-pyrolysis method using glucose as carbon source. The carbon coating does not change the phase structure of LiNi1/3Mn1/3Co1/3O2 material.
2) The experimental results show that the carbon coating enhances the rate capability and cycling performance of the LiNi1/3Mn1/3Co1/3O2 electrode.
References
[1] LIU H K, WANG G X, GUO Z P,WANG J Z,KOSTA K. Nanomaterials for lithium-ion rechargeable batteries [J]. Journal of Nanoscience and Nanotechnology, 2006, 6: 1-15.
[2] WHITTINGHAM M S. Lithium batteries and cathode materials [J]. Chemical Reviews, 2004, 104: 4271-4301.
[3] TARASCON J M, ARMAND M. Issues and challenges facing rechargeable lithium batteries [J]. Nature, 2001, 414: 359-367.
[4] OHZUKU T, MAKIMURA Y. Layered lithium insertion material of LiCo1/3Ni1/3Mn1/3O2 for lithium ion batteries [J]. Chemistry Letters, 2001, 68: 642-643.
[5] CHOI J, MANTHIRAM A.Comparison of the electrochemical behaviors of stoichiometric LiNi1/3 Co1/3Mn1/3O2 and lithium excess Li1.03(Ni1/3Co1/3Mn1/3)0.79O2 [J]. Electrochemical and Solid State Letters A, 2004, 7(10): 365-368.
[6] GUO J, JIAO L F, YUAN H T, WANG L Q, LI H X, ZHANG M, WANG Y M. Effect of structural and electrochemical properties of different Cr-doped contents of Li[Ni1/3Mn1/3Co1/3]O2 [J]. Electrochimica Acta, 2006, 51: 6275-6280.
[7] ZHAI Jing, ZHAO Min-shou, WANG Dan-dan. Effect of Mn-doping on performance of Li3V2(PO4)3/C cathode material for lithium ion batteries [J]. Transactions of Nonferrous Metals Society of China, 2011, 21: 523-528.
[8] YE S Y, XIA Y Y, ZHANG P W, QIAO Z Y. Al, B, and F doped LiNi1/3Co1/3Mn1/3O2 as cathode material of lithium-ion batteries [J]. Journal of Solid State Electrochemistry, 2007, 11: 805-810.
[9] YANG Z, LI X H, WANG Z X, ZHU Y J. Surface modification of spherical LiNi1/3Co1/3Mn1/3O2 with Al2O3 using heterogeneous nucleation process [J]. Transactions of Nonferrous Metals Society of China, 2007, 17: 1319-1323.
[10] LI J G, WANG L, ZHANG Q, HE X M. Electrochemical performance of SrF2-coated LiNi1/3Co1/3Mn1/3O2 cathode materials for Li-ion batteries [J]. Journal of Power Sources, 2009, 190: 149-153.
[11] WANG H Y, TANG A D, HUANG K L, LIU S Q. Uniform AlF3 thin layer to improve rate capability of LiNi1/3Co1/3Mn1/3O2 material for Li-ion batteries [J]. Transactions of Nonferrous Metals Society of China, 2010, 20: 803-808.
[12] KIM H S, KIM K, MOON S I, KIM I J, GU H B. A study on carbon-coated LiNi1/3Mn1/3Co1/3O2 cathode material for lithium secondary batteries [J]. Journal of Solid State Electrochemistry, 2008, 12: 867-872.
[13] GUO R, SHI P F, CHENG X Q, DU C Y. Synthesis and characterization of carbon-coated LiNi1/3Co1/3Mn1/3O2 cathode material prepared by polyvinyl alcohol pyrolysis route [J]. Journal of Alloys and Compounds, 2009, 473: 53-59.
[14] YANG Y J, SHEU C I, CHENG S Y, CHANG H Y. Si-Ca species modification and microwave sintering for NiZn ferrites [J]. Journal of Magnetism and Magnetic Materials, 2004, 284: 220-226.
[15] BHAT M H, CHAKRAVARTHY B P, RAMAKRISHNAN P A, LEVASSEUR, RAO J. Microwave synthesis of electrode materials for lithium batteries [J]. Bulletin of Materials Science, 2000, 23(6): 461-466.
[16] NASANOBU M, WATANABE K, IKUTA H, UCHIMOTO Y, WAKIHARA M. Grain size control of LiMn2O4 cathode material using microwave synthesis method [J]. Solid State Ionics, 2003, 164: 35-42.
[17] YANG S T, JIA J H, DING L, ZHANG M C. Studies of structure and cycleability of LiMn2O4 and LiNd0.01Mn1.99O4 as cathode for Li-ion batteries [J]. Electrochimica Acta, 2003, 48: 569-573.
[18] YAN H W, HUANG X J, CHEN L Q. Microwave synthesis of LiMn2O4 cathode material [J]. Journal of Power Sources, 1999, 81-82: 647-650.
[19] KALYANI P, KALAISELVI N, RENGANNATHAN N G. Microwave-assisted synthesis of LiNiO2―A preliminary investigation [J]. Journal of Power Sources, 2003, 123: 53-60.
[20] ZHANG Y, FENG H, WU X B, WANG L Z, ZHANG A Q, XIA T C, DONG H C, LIU M H. One-step microwave synthesis and characterization of carbon-modified nanocrystalline LiFePO4 [J]. Electrochimica Acta, 2009, 54(11): 3206-3210.
[21] SUBRAMANIAN V, CHEN C L, CHOU H S, TEY G T K. Microwave-assisted solid-state synthesis of LiCoO2 and its electrochemical properties as a cathode material for lithium batteries [J]. Journal of Materials Chemistry, 2001, 11: 3348-3353.
[22] SHEN B J, MA J S, WU H C, LU C H. Microwave-mediated hydrothermal synthesis and electrochemical properties of LiNi1/3Co1/3Mn1/3O2 powders [J]. Materials Letters, 2008, 62: 4075-4077.
[23] SHAJU K M, RAO S G V, CHOWDARI B V R. X-ray photoelectron spectroscopy and electrochemical behaviour of 4 V cathode, Li(Ni1/2Mn1/2)O2 [J]. Electrochimica Acta, 2003, 48: 1505-1514.
[24] YUN S H, PARK K S, PARK Y J. The electrochemical property of ZrFx-coated Li[Ni1/3Co1/3Mn1/3]O2 cathode material [J]. Journal of Power Sources, 2010, 195: 6108-6115.
[25] LIN B, WEN Z Y, HAN J , WU X W. Electrochemical properties of carbon-coated Li[Ni1/3Co1/3Mn1/3]O2 cathode material for lithium-ion batteries [J]. Solid State Ionics, 2008, 179: 1750-1753.
[26] SHIN H C,CHO W I,JANG H.Electrochemical properties of the carbon-coated LiFePO4 as a cathode material for lithium ion secondary batteries [J]. Journal of Power Sources, 2006, 159: 1383-1388.
[27] SHIN H C, CHO W I, JANG H. Electrochemical properties of carbon-coated LiFePO4 cathode using graphite,carbon black,and acetylene black [J]. Electrochimica Acta, 2006, 52: 1472-1476.
韩亚梅1,2,3,张正富1,2,3,张利波2,3,彭金辉2,3,傅梦笔2,3,C. SRINIVASAKANNAN4, 杜 江1,2,3
1. 昆明理工大学 材料科学与工程学院,昆明 650093;
2. 昆明理工大学 微波工程应用及装备技术国家地方联合工程实验室,昆明 650093;
3. 昆明理工大学 非常规冶金教育部重点实验室,昆明 650093;
4. Chemical Engineering Department, The Petroleum Institute, P.O. Box 2533, Abu Dhabi, United Arab Emirates
摘 要:报道了炭包覆锂离子电池正极材料LiNi1/3Mn1/3Co1/3O2的新工艺。炭涂层由前驱体葡萄糖通过微波热解而形成。采用X射线粉末衍射(XRD)、扫描电镜、X射线荧光测试和恒流充放电测试来表征所制备的材料。XRD结果表明,炭包覆没有改变LiNi1/3Mn1/3Co1/3O2材料的相结构。SEM结果表明,炭包覆的LiNi1/3Mn1/3Co1/3O2颗粒表面变得粗糙。充放电测试结果显示,炭包覆的LiNi1/3Mn1/3Co1/3O2的循环性能与未包覆的相比得到提高。炭包覆的LiNi1/3Mn1/3Co1/3O2在0.2C倍率下循环50次的容量保持率由84.8%提升到95.5%,且高倍率下材料的容量保持率得到提高。
关键词:锂离子电池;正极材料;炭涂层;微波热解;电化学性能
(Edited by Hua YANG)
Foundation item: Project (U1202272) supported by the National Natural Science Foundation of China
Corresponding author: Zheng-fu ZHANG; Tel: +86-871-5109952; E-mail: zhang-zhengfu@163.com
DOI: 10.1016/S1003-6326(13)62822-8

