�ۼ䱽��������η���Ч�ϳɼ���Cr(VI)����������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2013���11��
�������ߣ������� ����Ԫ ����Դ ����ӥ
����ҳ�룺3490 - 3498
Key words��poly (m-phenylenediamine); chromium (VI); sodium carbonate; adsorption
ժ Ҫ��������о���Na2CO3��η���ѧ�����ϳɾۼ䱽�������·��������л�̼(TOC)�IJⶨ�����ʾ������NaOH��η��ϳɵľۼ䱽������������Һ�е�TOC�ﵽ120.9 mg/L��������Na2CO3��η��ϳɵľۼ䱽������������Һ�е�TOCС��8 mg/L�����Ե���ǰ�ߺ���ˮ�ۺ��ŷű�(20 mg/L)�����ʺ���ˮ���������Ӧ�á�����Na2CO3��Ũ�ȿ�ʹ�ۼ䱽����������̬���ͣ�������ߵ�84%�����Ҷ�Cr(VI)���ѳ����������Ը��ơ��ۼ䱽������Cr(VI)������������ɴ�666.8 mg/g��Զ����Ŀǰ�����ⱨ���������������ϡ�
Abstract: Chemically oxidative polymerization of m-phenylenediamine was improved through adding the weak alkaline, Na2CO3. Results show that the poly (m-phenylenediamine) (PmPD) possesses a weak solubility in acidic solution according to total organic carbon (TOC) that the TOC is less than 8 mg/L, which is much lower than the discharge standard (20 mg/L). The TOC of the PmPD synthesized with NaOH can be as high as 120.9 mg/L. This very weak solubility of PmPD synthesized with Na2CO3 facilitates its application in water purification. The oxidation state of PmPD is decreased and the yield is increased with a maximum of 84%, promoting the concentration of Na2CO3 in the synthesis. Moreover, the Cr(VI) performance of PmPD was marvelously enhanced with Na2CO3 to improve the synthesis. The largest Cr(VI) adsorbance can reach as high as 666.8 mg/g, which is far more than the performance of other common adsorbents.
Trans. Nonferrous Met. Soc. China 23(2013) 3490-3498
Wan-ting YU1, Li-yuan CHAI1,2, Li-yuan ZHANG1, Hai-ying WANG1,2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. National Engineering Research Center for Heavy Metals Pollution Control and Treatment, Central South University, Changsha 410083, China
Received 27 December 2012; accepted 21 May 2013
Abstract: Chemically oxidative polymerization of m-phenylenediamine was improved through adding the weak alkaline, Na2CO3. Results show that the poly (m-phenylenediamine) (PmPD) possesses a weak solubility in acidic solution according to total organic carbon (TOC) that the TOC is less than 8 mg/L, which is much lower than the discharge standard (20 mg/L). The TOC of the PmPD synthesized with NaOH can be as high as 120.9 mg/L. This very weak solubility of PmPD synthesized with Na2CO3 facilitates its application in water purification. The oxidation state of PmPD is decreased and the yield is increased with a maximum of 84%, promoting the concentration of Na2CO3 in the synthesis. Moreover, the Cr(VI) performance of PmPD was marvelously enhanced with Na2CO3 to improve the synthesis. The largest Cr(VI) adsorbance can reach as high as 666.8 mg/g, which is far more than the performance of other common adsorbents.
Key words: poly (m-phenylenediamine); chromium (VI); sodium carbonate; adsorption
1 Introduction
Recently, aromatic amine/diamine polymers have attracted great attention mainly because of their multifunctionality, stability and low-cost production [1]. As effective adsorption materials, poly (phenylene- diamine) has attracted much attention due to its powerful redox reversibility and chelation ability. For instance, poly (p-phenylenediamine) was demonstrated to bear a high Ag+ adsorbance of 2890 mg/g [2], while the adsorption capacity of Pb2+ onto PmPD can reach 242.7 mg/g [3]. HAN et al [4] also reported that the poly (o-phenylenediamine) hollow structures bear a profound adsorbance of 4700 mg/g toward Pb2+. ZHANG et al [5] have recently discovered the PmPD nanoparticles possess powerful adsorption ability toward Orange G.
PmPD, as well as polyaniline and its other derivatives, are believed to be primarily composed of quinoid imine (Q) and benzenoid amine (B) units [6]. The ratio of Q/B is commonly described by oxidation state and the increase of Q/B means the increase of oxidation state [7]. Actually, the performance of these polymers is intensively affected by oxidation state. For instance, decreasing the oxidation state of polyaniline promotes the reduction kinetics of Cr(VI) and declining oxidation state of PmPD and poly (o-phenylenediamine) facilitates their Ag+ redox adsorption performance [8-10]. However, the high yield which is the native advantage of the chemically oxidative polymerization, cannot maintain when lowering the oxidation state of these polymers [11,12]. In this context, ZHANG et al [13] have recently in-situ tuned the oxidation state of PmPD through controlling the reduction potential of oxidant by concurrently adding NaOH solution during the addition of oxidant. Meanwhile, the synthesized PmPD still has high yield. And superior ability for Ag+ removal with adsorbance of 1693 mg/g was displayed [13]. Unfortunately, with increasing the NaOH concentration, the solubility of the final product increases obviously in acidic wastewater, e.g., Cr(VI) acid solution, which in-turn limited its application in wastewater treatment.
In the present research, we adopt the weak alkaline, Na2CO3, to improve the polymerization to produce the PmPD with rather weak solubility in acidic solution and decreased oxidation state. The products are characterized by total organic carbon (TOC) detection, X-ray diffraction (XRD) and Fourier transformed infrared spectroscopy (FTIR). The Cr(VI) adsorption ability is also investigated.
2 Experimental
2.1 Materials
All solutions were prepared with distilled water. Sodium carbonate, m-phenylenediamine, ammonium peroxydisulfate (APS), ammonia, ethanol were in analytical grade. Potassium dichromate was guarantee reagent. HCl and NaOH were used to adjust the pH.
2.2 Preparation of PmPD
The PmPD was prepared by chemical oxidation method. Distilled water was the reaction solution. 5 g of m-phenylenediamine was added in to 87.5 mL distilled water and was vigorously stirred at 25 ��C for 30 min. 10.5 g of APS was dissolved in 27.5 mL distilled water. Certain concentration of Na2CO3 solution was added dropwise with the same amount and speed as the oxidant solution to initiate and adjust the polymerization. After concurrently adding the oxidant and alkaline solutions, the reaction mixtures were kept stirring for another 5 h at 25 ��C. The products were collected by filtration, and washed with distilled water, ammonia water (1:1, v/v), absolute ethanol, respectively, to remove the oligomers, inorganic salts and other impurities. Then the products were dried at 60 ��C under vacuum for 12 h. The product was denoted as PmPD(Na2CO3)x, where the x represents the concentration of Na2CO3. The polymer was named PmPD(NaOH)x when using NaOH instead of Na2CO3. The product synthesized without adding NaOH and Na2CO3 was named PmPD(NM).
2.3 Sample characterization
The solubility of the PmPD in acid solution (pH=2) was measured with the TOC value. 15 mg PmPD was added into 50 mL acid aqueous solution (pH=2), and then filtrated after shaking for 5 h at 30 oC to get the filtrate. The TOC value was tested in a TOC-VCPH analyzer using high pure air as working environment. XRD and FTIR were applied to analyzing the structure of the products. The XRD pattern was obtained with a D/Max 2500 VB+X X-ray diffractometer using Cu (40 kV, 300 mA) radiation. The FTIR spectra were recorded on a Nicolet 1S10 spectrometer at 4 cm-1 resolution.
2.4 Batch experiments
The effects of initial solution pH, Cr(VI) concentration and time on the Cr(VI) adsorption were investigated in batch experiments. The Cr(VI) adsorption experiments were carried out in Teflon bottles containing 50 mL Cr(VI) solution. 0.015 g of adsorbent was added. The adsorption reaction was proceeded by shaking at 120 r/min and 25 ��C for 5 h.
For studying the effect of pH on adsorption, the initial pH value of the solution was controlled from 1.8 to 10 and the initial concentration of Cr(VI) solution was set to be 150 mg/L. In the adsorption isotherm experiments, the initial concentration of Cr(VI) solution was varied from 50 to 300 mg/L, and the initial solution pH was adjusted to 2. The adsorption kinetics experiments were carried out with the initial Cr(VI) concentration of 150 mg/L and the initial pH value of 2. Adsorption time was ranged from 1 min to 540 min.
1,5-diphenylcarbohydrazide spectrophotometric technique was used for determining the Cr(VI) concentration in solution after adsorption [14]. In the sample the Cr(VI) concentration was controlled to be lower than 0.2 mg/L, then the visible spectrum was measured at a wave length of 540 nm using a 722 visible spectrophotometer.
3 Results and discussion
3.1 Characterization of PmPD
3.1.1 Solubility of PmPD
TOC was adopted to quantitatively measure the solubility of PmPD in aqueous solution, as shown in Fig. 1. The initial pH of the aqueous solution was set to be 2.0. From Fig. 1, when using NaOH to synthesize the PmPD, the TOC of the PmPD increased with increasing the concentration of NaOH solution. Typically, as the NaOH concentration was promoted to 3 mol/L and further to 4 mol/L, the TOC went up to 50.4 [PmPD(NaOH)3] and 120.9 mg/L [PmPD(NaOH)4]. It is interesting that when replacing the NaOH with Na2CO3 with half the NaOH concentration (note: 0.5 mol  or 1 mol OH- can consume 1 mol H+), the TOC values of all the PmPD synthesized with different Na2CO3 concentrations were less than 8 mg/L. This is at least 6 times less than that of the PmPD(NaOH)3 and 10 times that of the PmPD(NaOH)4. Furthermore, this low concentration of filtrate satisfies the direct discharge standard (20 mg/L) according to GB 8978��1996, implying the superior application prospect of PmPD synthesized with Na2CO3 for wastewater treatment.
or 1 mol OH- can consume 1 mol H+), the TOC values of all the PmPD synthesized with different Na2CO3 concentrations were less than 8 mg/L. This is at least 6 times less than that of the PmPD(NaOH)3 and 10 times that of the PmPD(NaOH)4. Furthermore, this low concentration of filtrate satisfies the direct discharge standard (20 mg/L) according to GB 8978��1996, implying the superior application prospect of PmPD synthesized with Na2CO3 for wastewater treatment.
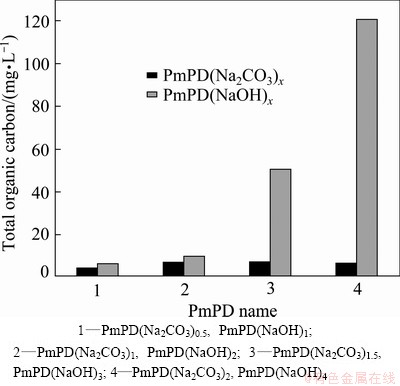
Fig. 1 Total organic carbon of filtrate obtained after Cr(VI) adsorbed by PmPD
3.1.2 Yield
To further understand the advantage of using Na2CO3 over NaOH to improve the synthesis of PmPD, yield was calculated and compared here. The relevant result is provided in Fig. 2. The yield of the PmPD synthesized with NaOH began to decrease when the concentration of NaOH was equal to or more than 3 mol/L and the maximum yield was 81%. In terms of using Na2CO3, the yield enhanced with promoting the concentration of Na2CO3 and when it increased to 2 mol/L, the yield reached up to 84 %. Up to the best of our knowledge, this is much higher than the data reported elsewhere. Based upon these results, the high yield of PmPD prepared with Na2CO3 endows it with higher prospect in industrialization production.
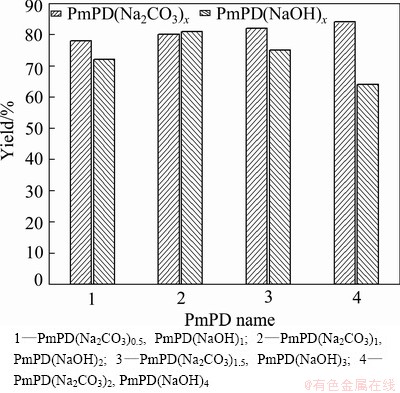
Fig. 2 Yield of PmPD synthesized with NaOH and Na2CO3
3.2 Characterization of PmPD synthesized with Na2CO3
The supermolecular structure of PmPD was characterized by XRD (Fig. 3). As shown in Fig. 3, there is one broad peak in these five patterns, which is a typical characteristic for amorphous structure [15]. The diffraction peak for PmPD(NM), which was prepared without using alkaline, is located at ~23��. However, the diffraction peak for the PmPD synthesized with Na2CO3 gradually dropped from ~22�� [PmPD(Na2CO3)0.5] to ~18�� [PmPD(Na2CO3)2]. According to the Brag equation, the decreased diffraction peak should be related to the increased intermolecular spacing and amorphousness [16]. Generally speaking, an amorphous structure is conducive to the penetration and then adsorption of ions onto the macromolecules due to the loose and disordered piles of the polymeric chains in the amorphous structure [1,15]. In other words, the PmPD(Na2CO3)x may be more suitable for adsorption applications.
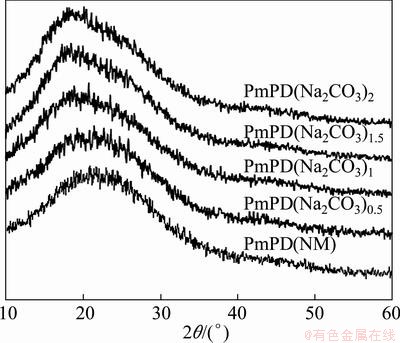
Fig. 3 XRD patterns of PmPD(Na2CO3)x
Figure 4 gives the FTIR spectra of PmPD(Na2CO3)x. The broad bands between 3500 and 3000 cm-1 are due to the characteristic stretching vibration of ��NH�� [17-19]. The two peaks at 1620 and 1500 cm-1 are associated with quinoid imine and benzenoid amine units, respectively [6]. Another typical adsorption band of PmPD is found at 1257 cm-1 which should be attributed to the C��N in the benzenoid amine units [20]. More interestingly, the relative intensity of the benzenoid amine unit of the PmPD synthesized with Na2CO3 with increasing concentration is enhanced compared with the quinoid imine unit. This means that the oxidation state declined when using Na2CO3 and increasing its concentration. Consequently, Na2CO3 is also an appropriate chemical to tune the oxidation state of PmPD.
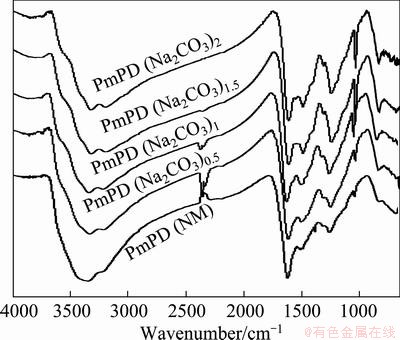
Fig. 4 FTIR spectra of PmPD(Na2CO3)x
3.3 Cr(VI) performance of PmPD
To further understand the application potential of PmPD in wastewater treatment, Cr(VI) adsorbability of them was investigated under the same initial adsorption conditions (Fig. 5). As can be seen, the performance of the PmPD is good that the Cr(VI) adsorbance of PmPD(Na2CO3)0.5 reached ~341.9 mg/g. When using the PmPD synthesized with an increased Na2CO3 concentration, the Cr(VI) adsorbance rapidly increased from ~454.1 mg/g [PmPD(Na2CO3)1], to ~556.8 mg/g [PmPD(Na2CO3)1.5] and further to ~598.5 mg/g [PmPD(Na2CO3)2].
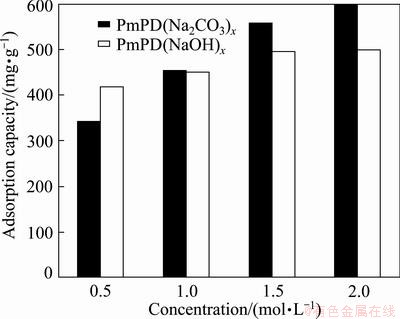
Fig. 5 Cr(VI) adsorption capacity of PmPD synthesized with Na2CO3 and NaOH under adsorption conditions of 50 mL of 300 mg/L Cr(VI), initial pH 2, 0.015 g of PmPDs, 5 h, 30 ��C
In a comparison, the Cr(VI) adsorption performance of PmPD(NaOH)x was also tested to indirectly reveal the prospect of PmPD(Na2CO3)x. It can be found that the Cr(VI) adsorption performance of PmPD(NaOH)x increased with promoting the NaOH concentration for the synthesis. The PmPD(NaOH)2 possesses the highest adsorbance among the PmPD(NaOH)x in the present research, which is ~498.8 mg/g. It can be calculated that the highest performance of PmPD(Na2CO3)2 is nearly 100 mg/g more than that of the PmPD(NaOH)2, demonstrating the much more superior prospect of PmPD(Na2CO3)x, typically the PmPD(Na2CO3)2 for Cr(VI) adsorption. Here, a deep investigation on the Cr(VI) behavior onto the PmPD(Na2CO3)2 was conducted and given below.
3.4 Factors on Cr(VI) adsorption
3.4.1 Effect of pH
The effect of pH on Cr(VI) removal by PmPD(Na2CO3)2 is shown in Fig. 6. The maximum Cr(VI) adsorptivity (96.6%) of PmPD(Na2CO3)2 was observed at pH 2.0. The Cr(VI) adsorptivity did not change much when further declining the solution pH. However, increasing the initial solution pH to higher than 2.0, the Cr(VI) adsorbability decreased obviously. Typically, the Cr(VI) adsorptivity dropped to 39.8% when the initial pH was 3 and furthermore the adsorptivity of Cr(VI) declined to nearly 0 in alkaline media. It clearly revealed that acidic condition benefits to Cr(VI) adsorption, especially when the initial pH is 2.
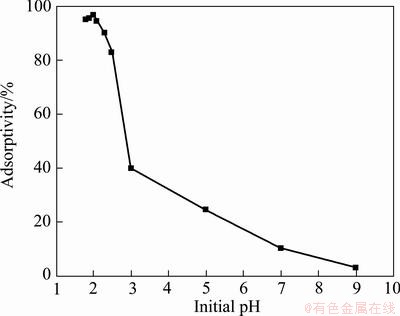
Fig. 6 Effect of initial pH on Cr(VI) adsorption with PmPD(Na2CO3)2
Generally, aromatic amine polymers, such as PmPD, contain abundant =N�� functional group [3,21]. The lone pair of electrons from the nitrogen atom can attract H+ by electrical attraction force, and =N+H�� groups are formed. Due to the charge neutralization, the counter Cr(VI) anions will be attracted and immobilized by the =N+H�� groups on the polymer powders. Therefore, when decreasing the initial pH of Cr(VI) solution, the number of protonated =N+H�� groups would increase and thus the number of adsorption sites on the PmPD surface would enhance, resulting in an raising in Cr(VI) anions adsorption. In addition, few Cr(VI) anions were adsorbed in the pH of 7 and 9, because =N+H�� groups could not form and there were nearly no adsorption sites provided for Cr(VI) anions.
3.4.2 Adsorption kinetics
Figure 7 shows the effect of the contact time on the removal of Cr(VI) with PmPD(Na2CO3)2 at different temperatures. It is obvious that the Cr(VI) adsorptivity increased with the contact time. Especially, a nearly linear growth of Cr(VI) adsorptivity was found within the initial 20 min. Then, the increment gradually became slow and the adsorption tended to be equilibrium when the contact time was 3 h. The initial rapid step may be attributed to the physical adsorption and surface reaction [1]. But the subsequent slow step may be ascribed to the penetration process of adsorbate into the inner structures of adsorbents [22]. According to Fig. 7, promoting the temperature did not shorten the equilibrium time. So, the effect of temperature on the adsorption rate is negligible.
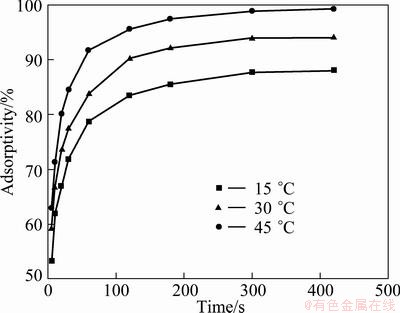
Fig. 7 Effect of adsorption time on Cr(VI) adsorption with PmPD(Na2CO3)2 at different temperatures
Furthermore, the pseudo-first-order and pseudo- second-order kinetic equations were employed to analyze the Cr(VI) adsorption behaviors onto the PmPD powders [23].
The pseudo-first-order is written as follows:
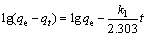 (1)
(1)
The pseudo-second-order is written as follows:
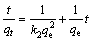 (2)
(2)
where qt and qe are the amounts of Cr(VI) adsorbed at time t and at equilibrium, respectively; k1 and k2 are the rate constants of the pseudo-first-order and pseudo- second-order adsorption reaction.
The fitting results of the pseudo-first-order and pseudo-second-order kinetic models and corresponding parameters are given in Table 1. The correlation coefficient for the pseudo-second-order model is greater than 0.999, but lower for the pseudo-first-order model. Therefore, the pseudo-second-order model is more appropriately to describe kinetic behavior of chromium adsorption. The hypothesis that the chemically covalent reaction is the main interaction manner between adsorbent and adsorbate is the establishment basis of the pseudo-second-order model [24]. Thus, it strongly suggests the Cr(VI) adsorption by PmPD is primarily built on the chemisorptions.
Moreover, to describe the adsorption process, intraparticle diffusion model was also applied. Intraparticle diffusion model was given below and the relevant results can be seen in Fig. 8 [25].
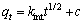 (3)
(3)
where kint is the intraparticle diffusion rate constant. According to Eq. (3), a plot of qt versus t1/2 should be a straight line with a slope kint and intercept c when the adsorption follows the intraparticle diffusion process.
As shown in Fig. 8, the adsorption process tends to be followed by two phases, which indicates that the adsorption process proceeds by surface adsorption and the intraparticle diffusion during the Cr(VI) removal with PmPD(Na2CO3)2. No plot does not pass through the origin, which suggests that the intraparticle diffusion was not the sole rate-controlling step [26,27].
3.4.3 Adsorption isotherms
The effect of initial Cr(VI) concentration on the removal of Cr(VI) by PmPD(Na2CO3)2 at different temperatures was studied (Fig. 9). As shown in Fig. 9, the adsorbance rose significantly when Cr(VI) concentration increased from 75 to 200 mg/L, and the increase of Cr(VI) adsorbance became slow when further enhancing the initial Cr(VI) concentration. Meanwhile, the Cr(VI) adsorbance increased with temperature increasing, which accounts for the endothermic nature of the adsorption process. For instance, when the initial Cr(VI) concentration was 300 mg/L, the adsorption capability of PmPD(Na2CO3)2 for Cr(VI) was 666.8 mg/g at 45 ��C, 598.5 mg/g at 30 ��C, and 510.9 mg/g at 15 ��C.
Table 1 Kinetics model equation for Cr(VI) adsorption at different temperatures
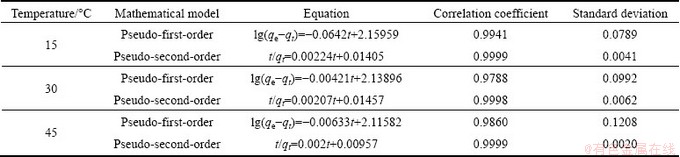
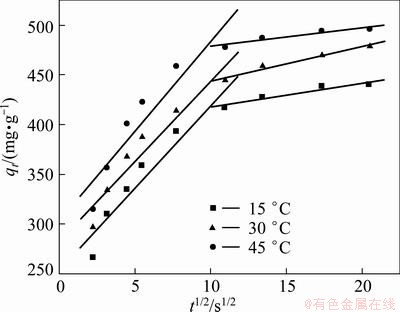
Fig. 8 Intraparticle diffusion plots for different temperatures
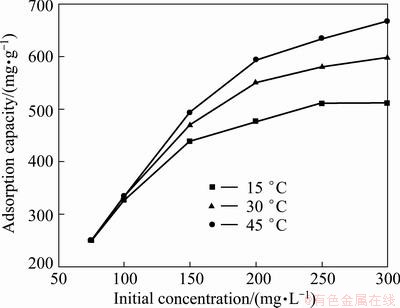
Fig. 9 Effect of initial Cr(VI) concentration on Cr(VI) adsorption at different temperatures
To establish a quantitative relationship between Cr(VI) concentration and the adsorption process, two mathematical models proposed by LANGMUIR [28] and FREUNDLICH [29] (Eqs. (4) and (5)) were used to describe and analyze the adsorption isotherm.
 (4)
(4)
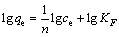 (5)
(5)
where ce is the equilibrium liquid phase concentration; qe is the amount of sorbent adsorbed per unit mass; qm and b are the Langmuir constants related to adsorption capacity and the rate of adsorption, respectively; KF is the equilibrium constant indicating adsorption capacity; n is the adsorption equilibrium constant.
Isotherm parameters for Langmuir and Freundlich models are shown in Table 2. The correlation coefficient value for Langmuir isotherm is higher than that for Freundlich isotherm. This confirms that Langmuir model can better explain the Cr(VI) adsorption by PmPD(Na2CO3)2.
To further display the superior performance of PmPD(Na2CO3)2 toward Cr(VI) removal, a comparison between it and other adsorbents was carried out and given in Table 3. Activated carbon and salvaged material (red mud and wheat bran) bear a relatively low adsorbance less than 36 mg/g toward Cr(VI) removal. Tetragonal FeS, nanoscale zerovalent iron (nZVI), polyaniline and chitosan possess higher performance. But these data are still much lower than the performance of PmPD(Na2CO3)2 with adsorbance of 666.8 mg/g. Taking the high yield and low cost into account, the PmPD(Na2CO3)2 synthesized with Na2CO3 to modify the polymerization shows a much more brilliant prospect in Cr(VI) adsorption application over other materials.
3.4.4 Thermodynamic parameters
According to Fig. 9, it is indicated that the adsorption was an endothermic process, which can be explained on the thermodynamic parameters such as free energy (��G), enthalpy (��H), and entropy (��S) change of adsorption. The equations are as follows [39]:
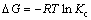 (6)
(6)
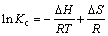 (7)
(7)
where Kc(=b) is the equilibrium constant, T is temperature and R is the mole gas constant. The values of ��H and ��S can be calculated from the slope and intercept of the Van��t Hoff plot of ln K versus 1/T from Eq. (7) and Fig. 10. The values of ��G, ��H and ��S are shown in Table 4.
The values of ��G are negative at all temperatures, which indicates the spontaneous nature of Cr(VI) onto
Table 2 Isotherm model equations for Cr(VI) adsorption at different temperatures
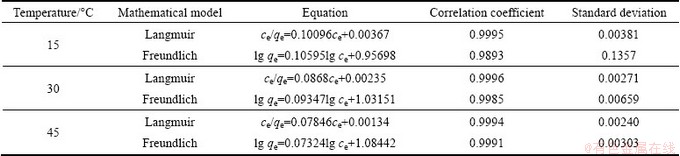
Table 3 Comparison of some adsorbents for Cr(VI) removal
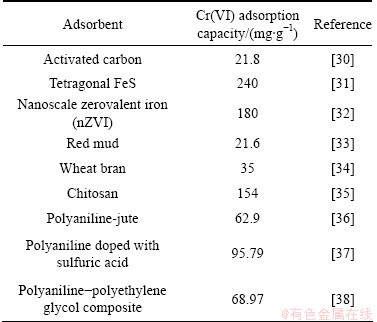
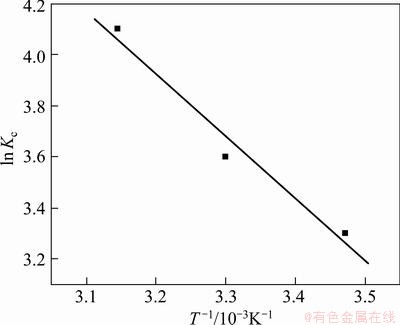
Fig. 10 Van��t Hoff plot for adsorption of PmPD(Na2CO3)2
Table 4 Thermodynamic parameters for adsorption of Cr(VI) in aqueous solutions onto PmPD(Na2CO3)2
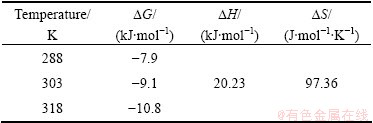
The absolute value of ��G increases with the rise of the temperature, which shows that the adsorbance at equilibrium should enhance with increasing temperature. The positive value of ��H shows an endothermic nature of Cr(VI) adsorption with PmPD(Na2CO3)2. The positive value of ��S suggests the increased randomness at the solid�Csolution interface during the adsorption of metal ions on PmPD(Na2CO3)2 [40,41]. As the free energy values are negative and accompanied by positive entropy changes, the adsorption reactions are spontaneous with a high affinity.
4 Conclusions
1) PmPD was synthesized by chemically oxidative polymerization modified with concurrently by adding Na2CO3 solution during giving APS solutions. As quantitatively revealed by TOC technique, the solubility of the PmPD is very weak, of which the TOC is ~7 mg/L. This value is much less than the discharge standard value of 20 mg/L. The maximum yield of PmPD can be 84 %.
2) XRD and FTIR were used to characterize PmPD. The results show that PmPD synthesized by adding Na2CO3 solution has amorphous structure which is more suitable for adsorption and lower oxidation state.
3) The maximum Cr(VI) adsorptivity was observed at pH 2.0. The Cr(VI) adsorption behavior of PmPD can be described by the pseudo-second-order model and Langmuir model. The adsorption becomes equilibrium at 3 h and the maximum adsorbance reaches as high as 666.8 mg/g. According to the calculation of thermodynamic parameters, the values of ��G are negative, and ��H and ��S are positive, which indicates that the adsorption reactions are spontaneous and endothermic.
References
[1] LI X G, MA X L, SUN J, HUANG M R. Powerful reactive sorption of silver(I) and mercury(II) onto poly(o-phenylenediamine) microparticles [J]. Langmuir, 2009, 25: 1675-1684.
[2] WANG J, JIANG J, HU B, YU S H. Uniformly shaped poly (p-phenylenediamine) microparticles: Shape-controlled synthesis and their potential application for the removal of lead ions from water [J]. Adv Funct Mater, 2008, 18: 1105-1111.
[3] HUANG M R, LU H J, LI X G. Efficient multicyclic sorption and desorption of lead ions on facilely prepared poly (m-phenylenediamine) particles with extremely strong chemoresistance [J]. J Colloid Interface Sci, 2007, 313: 72-79.
[4] HAN J, DAI J, GUO R. Highly efficient adsorbents of poly (o-phenylenediamine) solid and hollow sub-microspheres towards lead ions: A comparative study [J]. J Colloid Interface Sci, 2011, 356: 749-756.
[5] ZHANG L Y, WANG H Y, YU W T, SU Z, CHAI L Y, LI J H, SHI Y. Facile and large-scale synthesis of functional poly (m-phenylenediamine) nanoparticles by Cu2+-assisted method with superior ability for dye adsorption [J]. J Mater Chem, 2012, 22: 18244-18251.
[6] LI X G, HUANG M R, DUAN W, YANG Y L. Novel multifunctional polymers from aromatic diamines by oxidative polymerizations [J]. Chem Rev, 2002, 102: 2925-3030.
[7] STEJSKAL J, KRATOCHVIL P, JENKINS A D. The formation of polyaniline and the nature of its structures [J]. Polymer, 1996, 37: 367-369.
[8] ANSARI R. Application of polyaniline and its composites for adsorption/ recovery of chromium (VI) from aqueous solutions [J]. Acta Chim Slov, 2006, 53: 88-94.
[9] STEJSKAL J,  J, SAPURINA I. The reduction of silver ions with polyaniline: The effect of the type of polyaniline and the mole ratio of the reagents [J]. Mater Lett, 2009, 63: 709-711.
J, SAPURINA I. The reduction of silver ions with polyaniline: The effect of the type of polyaniline and the mole ratio of the reagents [J]. Mater Lett, 2009, 63: 709-711.
[10] LI X G, FENG H, HUANG M R. Redox sorption and recovery of silver ions as silver nanocrystals on poly (aniline-co-5-sulfo- 2-anisidine) nanosorbents [J]. Chem Eur J, 2010, 16: 10113-10123.
[11] LI X G, HUANG M R, LI S X. Facile synthesis of poly (1, 8-diaminonaphthalene) microparticles with a very high silver-ion adsorbability by a chemical oxidative polymerization [J]. Acta Mater, 2004, 52: 5363-5374.
[12] LI X G, HUANG M R. Facile synthesis and highly reactive silver ion adsorption of novel microparticles of sulfodiphenylamine and diaminonaphthalene copolymers [J]. Chem Mater, 2005, 17: 5411-5419.
[13] ZHANG L Y, CHAI L Y, LIU J, WANG H W, YU W T, SANG P L. pH manipulation: A facile method for lowering oxidation state and keeping good yield of poly(m-phenylenediamine) and its powerful Ag+ adsorption ability [J]. Langmuir, 2011, 27: 13729-13738.
[14] DUPONT L, GUILLON E. Removal of hexavalent chromium with a lignocellulosic substrate extracted from wheat bran [J]. Environ Sci Technol, 2003, 37: 4235-4241.
[15] HUANG M R, PENG Q Y, LI X G. Rapid and effective adsorption of lead ions on fine poly (phenylenediamine) microparticles [J]. Chem Eur J, 2006, 12: 4341-4350.
[16] LI X G, DUAN W, HUANG M R, YANG Y L. Preparation and characterization of soluble terpolymers from m-phenylenediamine, o-anisidine, and 2,3-xylidine [J]. J Polym Sci Part A, 2001, 39: 3989-4000.
[17] SANG Pei-lun, WANG Yun-yan, ZHANG Li-yuan, CHAI Li-yuan, WANG Hai-ying. Effective adsorption of sulfate ions with poly (m-phenylenediamine) in aqueous solution and its adsorption mechanism [J]. Transactions of Nonferrous Metals Society of China, 2013, 23: 243-252.
[18] CHAI L Y, ZHANG L Y, WANG H Y, YU W T, SANG P L. An effective and scale-up self-assembly route to prepare the rigid and smooth oligo (o-phenyulenediamine) microfibers in acidic solution by NaClO2 [J]. Mater Lett, 2010, 64: 2302-2305.
[19] ZHANG L Y, CHAI L Y, WANG H Y, YANG Z H. Facile synthesis of one-dimensional self-assembly oligo (o-phenylenediamine) materials by ammonium persulfate in acidic solution [J]. Mater Lett, 2010, 64: 1193-1196.
[20] HUANG M R, LI X G, YANG Y L. Oxidative polymerization of o-phenylenediamine and pyrimidylamine [J]. Polym Degrad Stab, 2001, 71: 31-38.
[21] HUANG M R, LU H J, SONG W D, LI X G. Dynamic reversible adsorption and desorption of lead ions through a packed column of poly (m-phenylenediamine) spheroids [J]. Soft Mater, 2010, 8: 149-163.
[22] LI X G, FENG H, HUANG M R. Strong adsorbability of mercury ions on aniline/sulfoanisidine copolymer nanosorbents [J]. Chem Eur J, 2009, 15: 4573-4581.
[23] CHEN Run-hua, CHAI Li-yuan, WANG Yun-yan, LIU Hui, SHU Yu-de, ZHAO Jing. Degradation of organic wastewater containing Cu-EDTA by Fe-C micro-electrolysis [J]. Transactions of Nonferrous Metals Society of China, 2012, 22: 983-990.
[24] DENG S, YU G, XIE S, YU Q, HUANG J, KUWAKI Y, ISEKI M. Enhanced adsorption of arsenate on the aminated fibers: Sorption behavior and uptake mechanism [J]. Langmuir, 2008, 24: 10961-10967.
[25] HU Z H, CHEN H, JI F, YUAN S J. Removal of congo red from aqueous solution by cattail root [J]. J Hazard Mater, 2010, 173: 292-297.
[26] CHOWDHURY S, MISHRA R, SAHA P, KUSHWAHA P. Adsorption thermodynamics, kinetics and isosteric heat of adsorption of malachite green onto chemically rice husk [J]. Desalination, 2011, 265: 159-168.
[27] DOGAN M, ABAK H, ALKAN M. Adsorption of methylene blue onto hazelnut shell: Kinetics, mechanism and activation parameters [J]. J Hazard Mater, 2009, 164: 172-181.
[28] LANGMUIR I. The constitution and fundamental properties of solids and liquids [J]. J Am Chem Soc, 1916, 38: 2221-2295.
[29] FREUNDICH H M F. Uber die adsorption in losungen [J]. Zeitsch Phys Chem, 1906, 57: 385-470.
[30] MOHAN D, SINGH K P, SINGH V K. Removal of hexavalent chromium from aqueous solution using low-cost activated carbons derived from agricultural waste materials and activated carbon fabric cloth [J]. Ind Eng Chem Res, 2005, 44: 1027-1042.
[31] MULLET M, BOURSIQUOT S, EHRHARDT J J. Removal of hexavalent chromium from solution by mackinawite, tetragonal FeS [J]. Colloids and Surfaces A, 2004, 244: 77-85.
[32] LI X Q, CAO J S, ZHANG W X. Stoichiometry of Cr(VI) immobilization using nanoscale zerovalent iron (nZVI): A study with high-resolution X-ray photoelectron spectroscopy (HR-XPS) [J]. Ind Eng Chem Res, 2008, 47: 2131-2139.
[33] GUPTA V K, GUPTA M, SHARMA S. Process development for the removal of lead and chromium from aqueous solutions using red mud-an aluminum industry waste [J]. Water Res, 2001, 35: 1125-1134.
[34] DUPONT L, GUILLON E. Removal of hexavalent chromium with a lignocellulosic substrate extracted from wheat bran [J]. Environ Sci Technol, 2003, 37: 4235-4241.
[35] BODDU V M, ABBURI K, TALBOTT J L, SMITH E D. Removal of hexavalent chromium from wastewater using a new composite chitosan biosorbent [J]. Environ Sci Technol, 2003, 37: 4449-4456.
[36] KUMAR P A, CHAKRABORTY S, RAY M. Removal and recovery of chromium from wastewater using short chain polyaniline synthesized on jute fiber [J]. Chemical Engineering Journal, 2008, 141: 130-140.
[37] ZHANG R, MA H, WANG B. Removal of chromium(VI) from aqueous solutions using polyaniline doped with sulfuric acid [J]. Ind Eng Chem Res, 2010, 49: 9998-10004.
[38] SAMANI M R, BORGHEI S M, OLAD A, CHAICHI M J. Removal of chromium from aqueous solution using polyaniline-poly ethylene glycol composite [J]. J Hazard Mater, 2010, 184: 248-254.
[39] ANIRUDHAN T S, RADHAKRISHNAN P G. Thermodynamics and kinetics of adsorption of Cu(II) from aqueous solution onto a new cation exchanger derived from ramarind fruit shell [J]. J Chem Thermodynamics, 2008, 40: 702-709.
[40] LIU Y, LIU Y J. Biosorption isotherms, kinetics and thermodynamics [J]. Separation and Purification Technology, 2008, 61: 229-242.
[41] ANIRUDHAN T S, RADHAKRISHNAN P G. Kinetics, thermodynamics and surface heterogeneity assessment of uranium(VI) adsorption onto cation exchange resin derived from a lignocellulosic residue [J]. Applied Surface Science, 2009, 255: 4983-4991.
������1������Ԫ1,2������Դ1������ӥ1,2
1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. ���ϴ�ѧ �����ؽ�����Ⱦ���ι��̼����о����ģ���ɳ 410083
ժ Ҫ��������о���Na2CO3��η���ѧ�����ϳɾۼ䱽�������·��������л�̼(TOC)�IJⶨ�����ʾ������NaOH��η��ϳɵľۼ䱽������������Һ�е�TOC�ﵽ120.9 mg/L��������Na2CO3��η��ϳɵľۼ䱽������������Һ�е�TOCС��8 mg/L�����Ե���ǰ�ߺ���ˮ�ۺ��ŷű�(20 mg/L)�����ʺ���ˮ���������Ӧ�á�����Na2CO3��Ũ�ȿ�ʹ�ۼ䱽����������̬���ͣ�������ߵ�84%�����Ҷ�Cr(VI)���ѳ����������Ը��ơ��ۼ䱽������Cr(VI)������������ɴ�666.8 mg/g��Զ����Ŀǰ�����ⱨ���������������ϡ�
�ؼ��ʣ��ۼ䱽��������(VI)��̼���ƣ�����
(Edited by Hua YANG)
Foundation item: Project (2011467062) supported by National Public Welfare Research Project of Environmental Protection Industry, China; Project (50925417) supported by National Science Fund for Distinguished Young Scholars of China; Project (50830301) supported by the National Natural Science Foundation of China
Corresponding author: Li-yuan CHAI; Tel: +86-731-88836921; E-mail: lychai@csu.edu.cn
DOI: 10.1016/S1003-6326(13)62893-9

