Thermodynamics and mechanism of vanadium(IV) extraction from sulphate medium with D2EHPA, EHEHPA and CYANEX 272 in kerosene
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2012���2��
�������ߣ����˱� κ�� ��� ����� ��F͢ ��־�� ���ʤ
����ҳ�룺461 - 466
�ؼ��ʣ��ܼ���ȡ��D2EHPA��EHEHPA��CYANEX 272
Key words��vanadium; solvent extraction; D2EHPA; EHEHPA; CYANEX 272
ժ Ҫ��
�����л�������ȡ����D2EHPA��EHEHPA��CYANEX 272��ú����ϵ�д������������ȡ��(IV)��������ҺpHֵ����ȡ��Ũ�ȡ�������Ũ�ȡ��¶ȶԷ���ȡ���ܵ�Ӱ�죬��ȷ���ͺ������ɡ��������������ˮ��pHֵ����ȡ��Ũ�Ⱥ��¶ȵ����ߣ���(IV)�ķ��������D2EHPA�����ڸ��͵�pHֵ����ȡ��(IV)��������Է�����ȡ��������EHEHPA��CYANEX272����ȡ�����о����������3���л�������ȡ���Է�����ȡ���������ӽ����������ڵ�pHֵ�������ͺ������ΪVOR2(HR)2���ڸ�pHֵ���ͺ������ΪVOR2 (R��ʾ��ȡ��)��
Abstract:
Extraction of vanadium(IV) from sulphate acid solution was studied using organophosphorous-based extractants D2EHPA, EHEHPA and CYANEX 272 in kerosene. The different parameters affecting the extraction of vanadium(IV) under equilibrium conditions were separately investigated to elucidate the stoichiometry of the extracted species. The distribution ratio of vanadium increased with increasing equilibrium pH of the aqueous phase, concentration of the extractants and temperature. D2EHPA was found to be a stronger extractant, having greater pH functionality than EHEHPA and CYANEX 272. Extraction of vanadium(IV) by these organophosphorous-based extractants involved cation exchange mechanism, and the extracted species appear to be VOR2(HR)2 in the low equilibrium pH and VOR2 in the higher equilibrium pH, where HR refers to the three acidic extractants.

![]()

Trans. Nonferrous Met. Soc. China 22(2012) 461-466
LI Xing-bin,WEI Chang, WU Jun, LI Cun-xiong, LI Min-ting, DENG Zhi-gan, XU Hong-sheng
Faculty of Metallurgical and Energy Engineering,Kunming University of Science and Technology, Kunming 650093, China
Received 7 January 2011; accepted 29 September 2011
Abstract: Extraction of vanadium(IV) from sulphate acid solution was studied using organophosphorous-based extractants D2EHPA, EHEHPA and CYANEX 272 in kerosene. The different parameters affecting the extraction of vanadium(IV) under equilibrium conditions were separately investigated to elucidate the stoichiometry of the extracted species. The distribution ratio of vanadium increased with increasing equilibrium pH of the aqueous phase, concentration of the extractants and temperature. D2EHPA was found to be a stronger extractant, having greater pH functionality than EHEHPA and CYANEX 272. Extraction of vanadium(IV) by these organophosphorous-based extractants involved cation exchange mechanism, and the extracted species appear to be VOR2(HR)2 in the low equilibrium pH and VOR2 in the higher equilibrium pH, where HR refers to the three acidic extractants.
Key words: vanadium; solvent extraction; D2EHPA; EHEHPA; CYANEX 272
1 Introduction
Vanadium is a strategic metal and is used in the production of high-quality metal alloys, like strengthened steel, for its property as an active grain refiner and as a strong deoxidant [1]. Recently, substantial attention has been attracted to vanadium oxides because of their perspectives for design of novel materials with predetermined properties for electro-, radio-, and microwave mechanisms. Solvent extraction is one of the techniques being increasingly used for recovery of vanadium from leaching solution of black shale [2,3], oil fly ash [4,5], and spent catalyst [6]. Many reagents have been tested for the extraction of vanadium from various aqueous solutions. The 5,8-diethyl-7-hydroxydodecane- 6-oxime (LIX 63) was used to extract and separate vanadium(IV) and molybdenum(VI) from sulfuric acid solution containing aluminum(III), cobalt(II), nickel(II) and iron (III). Vanadium(IV) was extracted preferentially at low pH (around 1.5), and selectively stripped by 2 mol/L sulfuric acid solution [7].
It is recognized that tetravalent vanadium can be extracted by acidic organophosphorous reagents via cation exchange from weak acidic solutions. Among those, di-(2-ethylhexyl)-phosphoric acid (abbreviated as D2EHPA, commercially as P204) has been used widely for extraction of vanadium from chloride solution [8, 9], nitric acid solution [10], and sulphuric acid solution [11, 12]; 2-ethylexyl phosphonic acid mono 2-ethylexhyl ester (abbreviated as EHEHPA, commercially as PC88A or P507) was used for extraction of vanadium(V) from both acidic and sulfuric solutions [13]; and bis(2,4,4-trimethylpentyl) phosphinic acid (commercially as CYANEX 272) was used for extraction of vanadium(IV) from sulfuric acid solution [14].
D2EHPA, EHEHPA and CYANEX 272 are widely used organophosphorous acidic extractants in hydrometallurgy. They have similar structures. D2EHPA and EHEHPA differ only in the number of alkoxy(��RO) group directly attached to the phosphorus atom. This structure difference significantly affects the extractability and separation efficiency of metal ions.
The present work aims at determining the vanadium(IV) extraction thermodynamics and mechanism by D2EHPA, EHEHPA and CYANEX 272 from sulfuric acid solution. The extracted species are calculated from slop analysis and by graphic method, and thermodynamic functions ��H, ��G and ��S of those investigated systems have been determined.
2 Experimental
2.1 Reagents and apparatus
The stock solution of VOSO4 (0.01 mol/L) was prepared from vanadyl sulfate by dissolving in dilute sulfuric solution with distilled water. The vanadium(IV) concentration in the aqueous solution was standardized by 0.01 mol/L ferrous ammonium sulfate solution using 2-(phenylamino)-benzoic acid as an indicator. A pHS-3C digital pH meter (Shanghai Rex Instruments Factory, China) was employed for pH measurements. The ICP (inductively coupled plasma, Perkin-Elmer Optima 5300, USA) was used for determination of the concentration of vanadium in the aqueous phase. The extractants of D2EHPA and EHEHPA with the purity of 95.3% and 96.5% were supplied by Luoyang Zhongda Chemical Co., Ltd., China. The commercial CYANEX 272 (88%) was supplied by Cyanamid Canada, Inc. D2EHPA, EHEHPA and CYANEX 272 were purified by the three-phase method [15]. The purities of refined products were 98.5%, 98.7% and 97.2% for D2EHPA, EHEHPA and CYANEX 272, respectively. Kerosene was bought from local market and distilled to collect the fraction distilling at 200-260 ��C, which was colorless and mostly aromatic free. All of these refined extractants were used without purification and dissolved in distilled kerosene to the required concentration. All other reagents were of analytical grade and used without purification.
2.2 Extraction and analytical procedures
The solvent extraction experiments were carried out by mechanically shaking equal volume (20 mL) of organic and aqueous phases in a separating funnel at (25��0.5) ��C. But, the temperature variation experiments were carried out in stoppered glass tubes immersed in a thermostatic water bath (��0.5 ��C) by vigorously shaking. The preliminary experiments revealed that 15 min was sufficient to achieve equilibrium for VO2+ distribution between the organic and aqueous phases. The pH of the aqueous solution was initially adjusted to the desired value by adding 2.0 mol/L H2SO4 or 4 mol/L NaOH solution and measured after reaching equilibrium. After phase separation, the metal concentration in the aqueous phase was analyzed by ICP�COES method [16], and the metal concentration in organic phase was calculated by the difference in the aqueous phase before and after extraction. The distribution ratio (D) was calculated as the ratio of the vanadium(IV) concentration in organic phase to that in aqueous phase.
3 Theory and calculation
The thermodynamic diagram for vanadium(IV)�C water system at 298.15 K has been drawn by POST and ROBINS [17] and ZHOU et al [18] (Fig. 1). To simplify the calculation, molar concentrations of metal ion can be used instead of activities. As can be seen from the activity��pH diagram, the predominant existence of vanadium(IV) species in sulfuric acid medium is VO2+ as pH is lower than 3.0 at vanadium(IV) activity of 0.01. In our solvent extraction experiments, the equilibrium pH was lower than 3.0 for the tetravalent vanadium (0.01 mol/L) which presents as VO2+ ion in the aqueous solution.
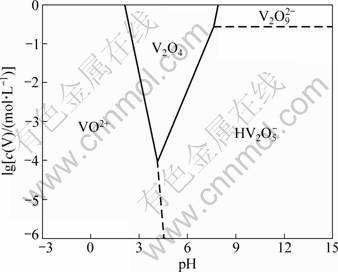
Fig. 1 Activity��pH diagram for vanadium(IV)�Cwater system at 298.15 K [18]
It is well known that organophosphorous-based acidic extractants exit as dimmers in nonpolar organic diluents. Generally, the extraction of vanadium in its tetravalent state from aqueous sulfuric media by organophosphorous-based acidic extractants can be expressed by the following cation exchange reaction [10]:
![]()
![]()
![]() (1)
(1)
The extraction equilibrium constant Kex is
![]() (2)
(2)
where HR represents the extractant species D2EHPA, EHEHPA or CYANEX 272 that acts as a liquid cationic ion exchanger, m is the number of H2R2 consumed in the reaction; and n is the number of H+ liberated due to the reaction.
There are two kinds of vanadium complex ions in vanadium(IV)-H2O system, VO2+ and V2O4, in the acidic solution. Correspondingly, their equilibrium equation is expressed as:
![]()
![]()
![]() (3)
(3)
The equilibrium constant KV is
![]() (4)
(4)
The total vanadium concentration [V]T in the aqueous solution is expressed as:
![]()
![]() (5)
(5)
Therefore,
![]() (6)
(6)
Inserting Eq. (6) into Eq. (2), and substituting the distribution ratio D, Eq. (2) becomes
![]() (7)
(7)
By taking logarithm and rearranging, Eq. (7) can be written as:
![]()
![]() (8)
(8)
4 Results and discussion
4.1 Extraction of vanadium(IV) using D2EHPA, EHEHPA and CYANEX 272
The extraction of vanadium(IV) (0.01 mol/L) from aqueous sulfuric acid solution was studied using the extractants D2EHPA, EHEHPA, CYANEX 272 in their acidic forms in distilled kerosene at phase ratio O/A of 1:1. The extraction of vanadium(IV) increased with increasing the equilibrium pH of the aqueous phase in the range of 0.9-2.0. The relationships (Fig. 2) show that the distribution ratio, D, of vanadium(IV) increased with the increase of equilibrium pH value with the slop of 1.98, 2.01 and 1.93 for 0.1 mol/L D2EHPA, EHEHPA, and CYANEX 272 extractants, respectively, indicating the release of 2 mole of H+ ions in the aqueous medium with 1 mole of vanadium(IV). SATO and TAKEDA [12] obtained a slope of about -2 in the plot of lg D vs lg[H+] for extraction of vanadium(IV) from sulphuric acid solution by D2EHPA. This work is therefore practically consistent with the previous work. The corresponding pH50 values were 1.17, 1.42 and 1.72 for 0.1 mol/L D2EHPA, EHEHPA and CYANEX 272 at this investigated condition, respectively. It means the extraction ability of alkylphosphorus extractants decreases in the following order: D2EHPA>EHEHPA> CYANEX 272. It is obvious that the extraction ability decreases with decreasing the acidity of the organophosphorus acids. The increasing of pH5,0 value for D2EHPA, EHEHPA and CYANEX 272 can be explained that the former molecule contains more alkoxy than the later molecule. The presence of alkoxy may increase the electric charge of central atom P, resulting in the decreasing acidity of the organophosphorus acids.
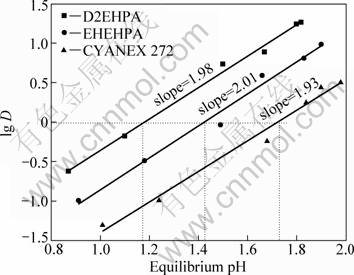
Fig. 2 Plot of lg D vs equilibrium pH for extraction of vanadium(IV) with D2EHPA , EHEHPA and CYANEX 272
The plots of lg D vs lg{[V]/(mol��L-1)} for the three extractants give a linear relation with the slopes of -1.06, -1.03 and -1.07 for D2EHPA, EHEHPA and CYANEX 272, respectively. The equilibrium [H+] was maintained at a concentration of 0.01 mol/L in the equilibrium aqueous phase. According to Eq. (8), the plot of lg D vs lg[V] gave straight lines with negative slopes of about -1.0, indicating that a monomeric metal species is extracted into the organic phase (Fig. 3).
The influence of extractant concentration on the extraction of vanadium(IV) was studied in the range of 0.02-0.8 mol/L. The corresponding equilibrium pH was in the range of 0.9-1.1 and 2.0-2.1, respectively. The extraction rate increased steadily with the increase of extractant concentration. The plots of lg D vs lg[H2R2] (Fig. 4) provide the metal/extractant ratio of the extracted
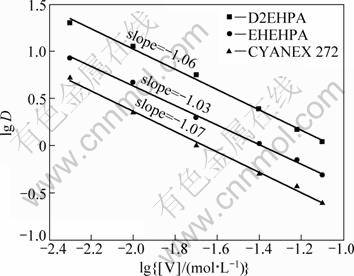
Fig. 3 Plots of lg D vs lg{[V]/(mol��L-1)} for extraction of vanadium(IV) with D2EHPA , EHEHPA and CYANEX 272
species. It can be seen from Fig. 4 that, the plots of lg D vs lg[H2R2] are linear with the slope of about 1.0 (equilibrium pH about 2.02) and about 2.0 (equilibrium pH about 1.05) in the extraction systems, indicating the association of one mole of the dimmer organophosphorous-based acidic extractants with the extracted species at higher equilibrium pH value and two mole of the dimmer organophosphorous-based acidic extractants with the extracted species at lower equilibrium pH value. ISLAM and BISWAS [11] found the similar phenomenon when extracting vanadium (IV) by D2EHPA both in kerosene and benzene systems from the sulphuric acid solution, and the pH dependence of extraction mechanism may be explained by the reason that the suppression of ionization of the dimeric acid is less than that of monomeric acid with increasing acidity. Therefore, the overall cation exchange extraction reaction for the three extractants may be represented by reaction (9) at high equilibrium pH value or by reaction (10) at low equilibrium pH value.
![]() (9)
(9)
![]() (10)
(10)
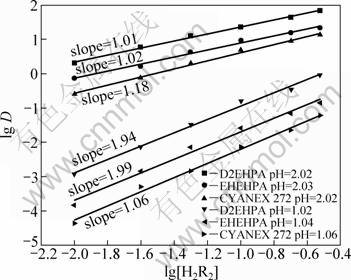
Fig. 4 Plots of lg D vs lg[H2R2] for extraction of vanadium(IV) with D2EHPA , EHEHPA and CYANEX 272
4.2 Effect of sulfate ions
The effect of sulfate ions on the extraction equilibrium was tested in sulfate ion concentration range of 0.1-2.5 mol/L. The results (Fig. 5) show that the distribution ratio of vanadium(IV) in sodium sulfate solutions does not change with varying the concentration of sulfate ion in the investigated range, which indicates that sulfate ion does not participate in the extracted complexes. This may be due to the fact that vanadium(IV) ion is unlikely complex with sulfate ion because the complex formation constant of this metal ions with sulfate ions is very small.
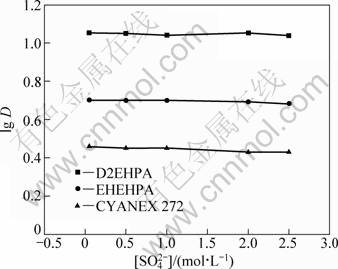
Fig. 5 Effect of sulfate ion on extraction of vanadium(IV) with D2EHPA , EHEHPA and CYANEX 272
4.3 Effect of temperature
The effect of temperature on the extraction of 0.01 mol/L vanadium(IV) from aqueous sulfuric acid solution containing 0.5 mol/L Na2SO4 using 0.1 mol/L D2EHPA, EHEHPA and CYANEX 272 was investigated at phase ratio O/A of 1:1, respectively. The initial aqueous pH was maintained at 2.2 for all the experiments, and the equilibrium pH was 2.04-1.8 for D2EHPA, 2.06-1.82 for EHEHPA and 2.10-1.86 for CYANEX 272 as the temperature increased from 293 K to 333 K, respectively. It was observed that the extraction rate increased with the increase of temperature in the range of 293-333 K. The results are shown in Fig. 6. When acidic CYANEX 272 was used as extractant, the extraction of vanadium(IV) increased form 57.1% to 76.9% as the temperature increased from 293 K to 333 K. While, there were no significant change on vanadium(IV) extraction when D2EHPA or EHEHPA was used.
From the slopes obtained from Fig. 6 and applying the Van��t Hoff equations [19], the thermodynamic parameters are calculated as:

Fig. 6 Plots of lg D vs 1/T for extraction of vanadium(IV) with D2EHPA , EHEHPA and Cyanex272
![]() (11)
(11)
where R is the gas constant, T is the absolute temperature and C is a conditional constant. Activity coefficient for other components are assumed to be constant under the experimental conditions [20]. The slopes equal �C��H/2.303R. On the basis of Eq. (11) and Fig. 6, the value of ��H are calculated to be 3.51, 4.68 and 19.90 kJ/mol for vanadium(IV) extraction using D2EHPA, EHEHPA and CYANEX 272, respectively, indicating that the extraction reactions are endothermic.
The value of ��H is used to calculate the corresponding free energy change (��G) and entropy change (��S) for extraction reaction at 298 K, respectively.
![]() (12)
(12)
![]() (13)
(13)
The calculated thermodynamic parameters are presented in Table 1. From these values, the negative ��G indicates that the extraction reactions by acidic organophosphorus extractants proceed spontaneously, while the positive value of ��S shows that the extraction process is more random in nature.
Table 1 Thermodynamic parameters for extraction process at 298 K

4.4 Effects of diluents
The extraction of 0.01 mol/L vanadium(IV) in the three organiphosphorous-based extractants was studied using various aliphatic and aromatic diluents like kerosene, n-hexane, cyclohexane, carbon tetrachloride, benzene, and toluene at the initial pH value of 2.45 and O/A ratio of 1:1. It was found that the extraction of vanadium(IV) was quantitative when both D2EHPA and EHEHPA were dissolved in kerosene and n-hexane, while benzene, toluene, cyclohexane and carbon tetrachloride were comparatively less efficient as solvents (Table 2). However, the CYANEX 272 extracted vanadium(IV) quantitatively when dissolved in benzene, toluene and carbon tetrachloride, while was less efficient when dissolved in cyclohexane, kerosene and n-hexane. The incomplete extraction of vanadium(IV) by some diluents may be due to the formation of extractant diluent species in the organic phase, which produces a low concentration of a free extractant for the metal extraction with a consequent decrease in the extraction rate [21, 22].
Kerosene was preferred as the diluent for both D2EHPA and EHEHPA, while toluene should be a good candidate for CYANEX 272, as those provided better vanadium extraction.
Table 2 Effect of diluents on extraction of vanadium(IV) with D2EHPA, EHEHPA and CYANEX 272

5 Conclusions
1) Solvent extraction of vanadium(IV) was carried out in sulfuric acid medium using the acidic organophosphorus extractant of D2EHPA, EHEHPA and CYANEX 272. The extraction of vanadium(IV) was dependent on the pH values of aqueous phase and extractant concentration. The extraction rate increased with increasing equilibrium pH and extractant concentration. The corresponding pH50 values for 0.1 mol/L D2EHPA, EHEHPA and CYANEX 272 were 1.17, 1.42 and 1.72, respectively.
2) The extraction of vanadium(IV) by the three acidic organophosphorus extractant was proceeded via a cation exchange mechanism. The species were found to be VOR2(HR)2 in the high acidity and VOR2 in low acidity. Sodium sulfate in the solution had a negligible effect on the vanadium extraction under the experimental condition.
3) The extraction process was an endothermic reaction, which was confirmed from the positive value of ��H for all the extraction systems. The negative value of ��G indicated that the extraction reactions proceed spontaneously, and the positive value of ��S showed the extraction process is more random in nature.
References
[1] TANER F, ROBERT A, GAULT T, ERCIT S. Vanadium mineralization and its industry in Canada [J]. The Gangue, 2000, 65: 1-4.
[2] LI Min-ting, WEI Chang, FAN Gang, LI Cun-xiong, DENF Zhi-gan, LI Xing-bin. Extraction of vanadium from black shale using pressure acid leaching [J]. Hydrometallurgy, 2009, 98: 308-313.
[3] DENG Zhi-gan, WEI Chang, FAN Gang, LI Min-ting, LI Cun-xiong, LI Xing-bin. Extraction vanadium from stone-coal by oxygen pressure acid leaching and solvent extraction [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(s1): s118-s122.
[4] NAVARRO R, GUZMAN J, SAUCEDO I, REVILLA J, GUIBAL E. Vanadium recovery from oil fly ash by leaching, precipitation and solvent extraction processes [J]. Waste Management, 2007, 27: 425-438.
[5] VITOLO S, SEGGIANI M, FALASCHI F. Recovery of vanadium from a previously burned heavy oil fly ash [J]. Hydrometallurgy, 2001, 62: 145-150.
[6] CHEN Yun, FENG Qi-ming, SHAO Yan-hai, ZHANG Guo-fan, OU Le-ming, LU Yi-ping. Investigations on the extraction of molybdenum and vanadium from ammonia leaching residue of spent catalyst [J]. International Journal of Mineral Processing, 2006, 79: 42-48.
[7] ZHANG Ping-wei, INOUE K, YOSHIZUKA K, TSUYAMA H. Extraction and selectively stripping of molybdenum (VI) and vanadium(IV) from sulfuric acid solution containing aluminum(III), cobalt(II), nickel(��) and iron(III) by LIX 63 in Exxsol D80 [J]. Hydrometallurgy, 1996, 41: 45-53.
[8] RIGG T, GAMER J O. Solvent extraction of vanadium from chloride solutions using di-(2-ethylhexyl)-phosphoric acid [J]. Journal of Inorganic and Nuclear Chemistry, 1967, 29(8): 2019-2025.
[9] SATO T, NAKAMURA T, KAWAMURA M. The extraction of vanadium(IV) from hydrochloric acid solutions by di-(2-ethylhexyl)-phosphoric acid [J]. Journal of Inorganic and Nuclear Chemistry, 1978, 40(5): 853-856.
[10] BRUNETTE J P, RASTEGAR F, LEROY M J F. Solvent extraction of vanadium(V) by di-(2-ethylhexyl)-phosphoric acid from nitric acid solutions [J]. Journal of Inorganic and Nuclear Chemistry, 1979, 41(5): 735-737.
[11] ISLAM F, BISWAS R K. The solvent-extraction of vanadium(IV) from sulphuric acid solutions with bis-(2-ethyl hexyl)- phosphoric acid in benzene and kerosene [J]. Journal of Inorganic and Nuclear Chemistry, 1980, 42(3): 415-420.
[12] SATO T, TAKEDA T. The extraction of vanadium(IV) from sulphuric acid solutions by di-(2-ethylhexyl) -phosphoric acid [J]. Journal of Inorganic and Nuclear Chemistry, 1970, 32(10): 3387-3396.
[13] SAJI J, REDDY M L P. Solvent extraction separation of vanadium(V) from multivalent metal chloride solutions by di-2-ethylhexyl phosphoric acid mono-2-ethylheyl ester [J]. Journal of Chemical Technology and Biotechnology, 2002, 77: 1149-1256.
[14] ZHANG Ping-wei, INOUE K, YOSHIZUKA K, TSUYAMA H. Solvent extraction of vanadium (IV) from sulfuric acid solution by bis(2,4,4-trimethylpentyl) phosphinic acid in EXXSOL D80 [J]. Journal of Chemical Engineering of Japan, 1996, 29: 82-87.
[15] HU Zheng-shui, PAN Ying, MA Wan-wu, FU Xun. Purification of organophosphorus acid extractants [J]. Solvent Extraction and Ion Exchange, 1995, 13(5): 965-976.
[16] WUILOUD R G, WUILOUD J C, OLSNA R A, MARTINEZ L D. Sepciation and preconcentration of vanadium(V) and vanadium(IV) in water samples by flow injection-inductively coupled plasma optical emission spectrometry and ultrasonic nebulization [J]. Analyst, 2001, 126: 715-719.
[17] POST K, ROBINS R G. Thermodynamic diagrams for the vanadium�Cwater system at 298.15 K [J]. Electrochimica Acta, 1967, 21(6): 401-405.
[18] ZHOU Xue-jiao, WEI Chang, LI Min-ting, QIU Shuang, LI Xing-bin. Thermodynamics of vanadium-sulfur-water systems at 298 K [J]. Hydrometallurgy, 2010, 106(1-2): 104-112.
[19] ISLAM F, RAHMAN H, ALI M. Solvent extraction separation study of Ti(IV), Fe(III) and Fe(II) from aqueous solution with di-2-ethylexyl phosphoric acid in benzene [J]. Journal of Inorganic and Nuclear Chemistry, 1979, 41: 217-221.
[20] MOHAPATRA D, KIM H I, NAM C W, PARK K H. Liquid-liquid extraction of aluminium(III) from mixed sulphate solution using sodium salts of CYANEX 272 and D2EHPA [J]. Separation and Purification Technology, 2007, 56: 311-318.
[21] SINGH R K, DHADKE P M. Extraction and separation of titanium(IV) with D2EHPA and PC-88A from aqueous perchloric acid solutions [J]. Journal of the Serbian Chemical Society, 2002, 67(7): 507-521.
[22] SINGH R K, DHADKE P M. Extraction and separation study of scandium(III) from perchlorate media by D2EHPA and PC 88A [J]. Bulletin of the Chemists and Technologists of Macedonia, 2002, 22(1): 1-11.
���˱�κ �ƣ��� ��������֣���F͢����־�ң����ʤ
����������ѧ ұ������Դ����ѧԺ������ 650093
ժ Ҫ�������л�������ȡ����D2EHPA��EHEHPA��CYANEX 272��ú����ϵ�д������������ȡ��(IV)��������ҺpHֵ����ȡ��Ũ�ȡ�������Ũ�ȡ��¶ȶԷ���ȡ���ܵ�Ӱ�죬��ȷ���ͺ������ɡ��������������ˮ��pHֵ����ȡ��Ũ�Ⱥ��¶ȵ����ߣ���(IV)�ķ��������D2EHPA�����ڸ��͵�pHֵ����ȡ��(IV)��������Է�����ȡ��������EHEHPA��CYANEX272����ȡ�����о����������3���л�������ȡ���Է�����ȡ���������ӽ����������ڵ�pHֵ�������ͺ������ΪVOR2(HR)2���ڸ�pHֵ���ͺ������ΪVOR2 (R��ʾ��ȡ��)��
�ؼ��ʣ������ܼ���ȡ��D2EHPA��EHEHPA��CYANEX 272
(Edited by YUAN Sai-qian)
Foundation item: Project (2009AA06Z106) supported by the High-tech Research and Development Program of China; Project (51174104) supported by the National Natural Science Foundation of China; Project (20115314110001) supported by the Doctoral Fund of Ministry of Education of China
Corresponding author: WEI Chang; Tel/Fax: +86-871-5188819; E-mail: weichang2008@sina.com
DOI: 10.1016/S1003-6326(11)61199-0

