Trans. Nonferrous Met. Soc. China 22(2012) s153-s156
Effect of Mg and Co co-doping on electrochemical properties of LiFePO4
DING Yan-huai1, 2, ZHANG Ping2
1. College of Chemical Engineering, Xiangtan University, Xiangtan 411105, China;
2. Institute of Rheology Mechanics, Xiangtan University, Xiangtan 411105, China
Received 9 July 2012; accepted 18 September 2012
Abstract: LiFePO4 co-doped with Mg2+ and Co4+ ions was synthesized by a solid state reaction method. The structure and electrochemical properties of the prepared LiFe0.99Mg0.005Co0.005PO4 were investigated by X-ray diffraction (XRD), galvanostatic charge-discharge experiment and cyclic voltammograms (CV). Specific discharge capacity of LiFePO4 co-doped with Mg and Co ions reach 147.2 mA・h/g at 0.1C and 133.3 mA・h/g at 1C. The results of CV show that the reversibility of lithium extraction/insertion in LiFePO4 can be promoted by (Mg2+, Co4+) multiple-ion doping.
Key words: LiFePO4; multiple-ion doping; Fe-site doping; solid state reaction; electrochemical performance
1 Introduction
Since they are first reported by the SONY corporation in the early 1990s, lithium-ion batteries have rapidly taken over the high-performance rechargeable battery market due to their high energy and power densities [1-3]. The improvement of the performance and cost of cathode materials is an important task in the lithium-ion batteries study. Recently, olivine-structured LiFePO4 was investigated intensively as a potential cathode material for rechargeable Li-ion batteries because of its low cost and improved safety [4-6]. The main disadvantage of LiFePO4 is its low electronic conductivity and diffusion coefficient of lithium ions which lead to its poor rate capability and hinder its commercialization as cathode material for lithium ion batteries.
The electronic and ionic transport limitations were overcome by doping with metal atoms [7,8], reducing the particle sizes [9] or coating electrically conductive agents [10,11]. Among them, heterogeneous doping at Li-site was widely adopted for optimization of electrochemical performance. HU et al [12] reported that LiFePO4 turned into a good P-type semiconductor material and its electric conductivity was promoted by metal ion doping at Li-site. However, there are fewer reports on the study of LiFePO4 doped by low-level aliovalent cation in Fe-sites [13,14]. Fe-site doped samples LiFe0.99Mg0.005- Co0.005PO4 were prepared by a solid-state reaction method using oxalate as precursor. And the effect of Mg and Co co-doping on electrochemical properties of LiFePO4 was studied. This is the first report of LiFePO4 doping with multiple-ion at Fe-sites. This method may open up new avenues for application of LiFePO4 in high-performance lithium-ion batteries.
2 Experimental
LiFePO4 (LFP) was prepared by a solid-state reaction method. A mixture of Li2CO3, FeC2O4・2H2O and NH4H2PO4 with mole ratio of 0.5:1:1 was ground and mixed by ball-milling for 5 h. The mixture was calcinated in a tube furnace at 350 °C for 10 h with flowing high-pure nitrogen gas. After being cooled down to room temperature, the sample was pressed to flake product and sintered at 700 °C for 24 h in the tube furnace under a protective atmosphere. The sample of LiFe0.99Mg0.005Co0.005PO4 (LMCFP) was prepared following the same procedures. MgC2O4・2H2O and Co(C2O4)2・4H2O were used as precursors for doping samples.
X-ray diffraction (XRD) measurements were carried out with a Bruker D8 using filtered Cu Kα radiation.
The electrochemical experiments were performed using two-electrode coin-type cells. The working electrode composed of active materials, carbon black and poly (vinyl difluoride) (PVdF) with mass ratio of 80:10:10 was pasted on Nickel foam. The metallic lithium foil was used as the counter electrode, 1 mol/L LiPF6 was dissolved in ethylene carbonate/dimethyl carbonate with volume ratio of 1:1 as electrolyte, and Celgard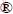 2 400 as separator. Test cells were assembled in an argon-filled glove box with less than 1×10-6 of oxygen, water and nitrogen contents. The discharge and charge measurements were carried out under an identical electrochemical condition on a Neware battery tester. Cyclic voltammograms (CV) were performed on a PARSTAT 2273 at a slow sweep rate of 0.1 mV/s and voltage between 2.7 and 4.3 V.
2 400 as separator. Test cells were assembled in an argon-filled glove box with less than 1×10-6 of oxygen, water and nitrogen contents. The discharge and charge measurements were carried out under an identical electrochemical condition on a Neware battery tester. Cyclic voltammograms (CV) were performed on a PARSTAT 2273 at a slow sweep rate of 0.1 mV/s and voltage between 2.7 and 4.3 V.
3 Results and discussion
XRD patterns of the prepared LiFePO4 and LiFe0.99Mg0.005Co0.005PO4 are shown in Fig.1. XRD patterns for the samples indicate that pure phases with an ordered olivine structure indexed in orthorhombic Pnmb are exclusively formed [15]. LiFePO4is chemically stable in the presence of Mg2+ and Co4+ doping. No extra diffraction peaks from related secondary phases or impurities are found. It is obvious that multiple-ion doping with a low amount does not affect the structure of the samples.
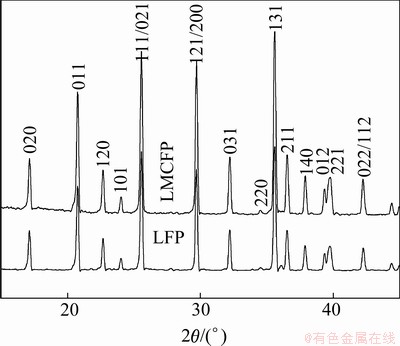
Fig. 1 XRD patterns of LiFePO4 and LiFe0.99Mg0.005Co0.005 PO4 samples
Figure 2 displays the charge-discharge curves of LiFePO4 and LiFe0.99Mg0.005Co0.005PO4 samples with cutoff voltages of 2.5-4.0 V (vs Li+/Li) at a current density of 0.1C. As can be seen from the figure, the cells show very smooth and monotonous charge-discharge curves. The initial discharge capacity of raw LiFePO4 is 114.8 mA・h/g with a discharge efficiency of 93.1%. For doped samples, the initial discharge capacity and discharge efficiency increase to 147.2 mA・h/g and 97.8%, respectively. Mg2+ has approximately the same size as Fe3+, but is larger than Fe2+. So, Mg-doping causes lattice defect in LiFePO4. The lattice defects promote the spread of lithium ions, and then improve the electrical conductivity of the LiFePO4 [13,14]. Furthermore, Co doping makes the Li-O interaction weakened, which leads to high ionic mobility and diffusion coefficient [13]. This is probably the main reason for the dramatic increase of the initial discharge capacity from raw LiFePO4 to doping samples.
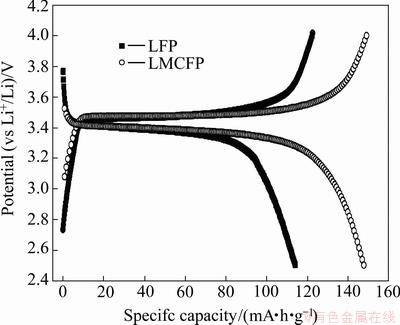
Fig. 2 Charge-discharge curves of LiFePO4 and LiFe0.99 Mg0.005Co0.005PO4 samples
The cycling performance of raw and co-doping LiFePO4 is shown in Fig. 3. The raw LiFePO4 shows a poor cycling performance, with capacity fading of more than 22% after 50 cycles at the current density of 0.1C. One possible reason for this rapid capacity fade is the constantly generated inactive material causes by the structure collapse during Li+ insertion and extraction [16,17]. In the case of doping materials LMCFP, the discharge capacities at the 50th cycle hold at 136.5 mA・h/g with capacity retention rate of 92.3%. This indicates that the structural stability of LiFePO4 is improved by Mg and Co co-doping.
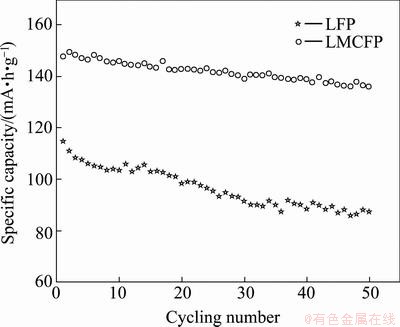
Fig. 3 Cycling performance of raw and co-doping LiFePO4
Figure 4 shows the cyclic voltammograms of raw and doped LiFePO4 measured at a sweep rate of 0.1 mV/s at room temperature and voltage between 2.7 and 4.3 V (vs Li/Li+). Anodic and cathodic peaks appear at about 3.6 and 3.3 V, which correspond to the two phase charge-discharge reaction of the Fe2+/Fe3+ redox couple [18]. The potential separation of doped LiFePO4 between anodic and cathodic peaks is 0.29 V, whereas that of the virginal LiFePO4 is 0.45 V. Furthermore, the shape of anodic/cathodic peaks of doped LiFePO4 is more symmetrical and sharper than that of pure LiFePO4, which indicates that the reversibility of lithium extraction/insertion in LiFePO4 can be promoted by (Mg2+and Co4+) multiple-ion doping.
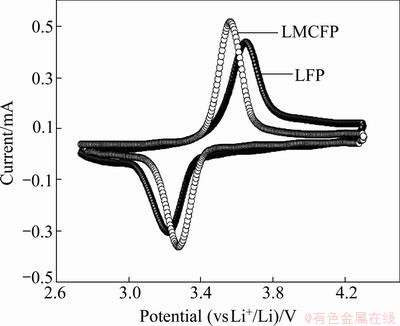
Fig. 4 Cyclic voltammetry profiles of undoped LFP and LMCFP
The rate capabilities of undoped LiFePO4 and LiFe0.99Mg0.005Co0.005PO4 were measured at different discharge current densities. By increasing the discharge rate, utilization of the active material is decreased, which causes the decrease of discharge capacity [19]. As shown in Fig.5, pure LiFePO4 has the discharge specific capacities of 78.4, 68.4, 60.1 and 51.2 mA・h/g at 1, 2, 3 and 4C, respectively. Doping samples show good rate capability compared with raw LiFePO4, and the discharge specific capacities increase to 133.3, 121.4, 114.3 and 108.7 mA・h/g at the same discharge current density.
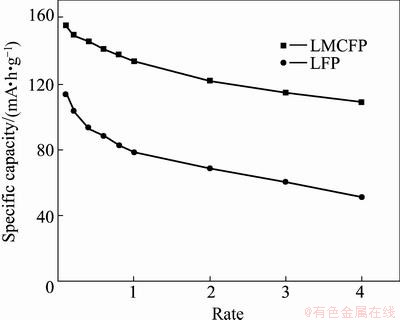
Fig. 5 Rate capability of undoped LiFePO4 and LiFe0.99-Mg0.005Co0.005PO4
4 Conclusions
1) Mg-doping causes lattice defect in LiFePO4, and Co-doping makes the Li-O interaction weakened, which leads to high ionic mobility and diffusion coefficient.
2) The initial discharge capacity and discharge efficiency of doping samples increase to 147.2 mA・h/g and 97.8% compared to pure LiFePO4. The discharge capacities of LMCFP at the 50th cycle also hold at 136.5 mA・h/g with capacity retention rate of 92.3%.
3) Discharge specific capacity of doped LiFePO4 increases to 133.3, 121.4, 114.3 and 108.7 mA・h/g at 1, 2, 3 and 4C, respectively. And cycling performance and rate capability of LiFePO4 are improved by Mg and Co co-doping.
References
[1] MIZUSHIMA K, JONES P C, WISEMAN P J, GOODENOUGH J B. LixCoO2 (0<x<1): A new cathode material for batteries of high energy density [J]. Materials Research Bulletin, 1980, 15(6): 783-789.
[2] YANG Y, PENG W, GUO H, WANG Z, LI X, ZHOU Y, LIU Y. Effects of modification on performance of natural graphite coated by SiO2for anode oflithium ion batteries [J]. Transactions of Nonferrous Metals Society of China, 2007, 17: 1339-1342.
[3] MEGAHED S, EBNER W. Lithium-ion battery for electronic applications [J]. Journal of Power Sources, 1995, 54(1): 155-162.
[4] PADHI A K, NANJUNDASWAMY K S, MASQUELIER C, OKADA S, GOODENOUGH J B. Effect of structure on the Fe3+/Fe2+ redox couple in iron phosphates [J]. Journal of theElectrochemicalSociety, 1997, 144: 1609-1613.
[5] YANG Y, LIAO X Z, MA Z F, WANG B F, HE L, HE Y S. Superior high-rate cycling performance of LiFePO4/C-PPy composite at 55 °C [J]. Electrochemistry Communications, 2009, 11: 1277- 1280.
[6] PARK K S, SON J T, CHUNG H T, KIM S J, LEE C H, KIM H G. Synthesis of LiFePO4 by co-precipitation and microwave heating [J]. Electrochemistry Communications, 2003, 5: 839-842.
[7] CHUNG S Y, BLOKING J T, CHIANG Y M. Electronically conductive phospho-olivines as lithium storage electrodes [J]. Nature Mater, 2002, 1: 123-128.
[8] TIAN Y, KANG X, LIU L, XU C, QU T. Research on cathode material of Li-ion battery by yttrium doping [J]. Journal of Rare Earths, 2008, 26(2): 279-283.
[9] PARK K S, SCHOUGAARD S B, GOODENOUGH J B. Improved electrode performance of porous LiFePO4 using RuO2 as an oxidic nanoscale interconnect [J]. Adv Mater, 2007, 19: 848-851.
[10] PROSINI P P, CAREWSKA M, SCACCIA S, WISNIEWSKI P, PASQUALI M. Long-term cyclability of nanostructured LiFePO4 [J]. Electrochimica Acta, 2003, 48: 4205-4211.
[11] DING Y, JIANG Y, XU F, YIN J, REN H, ZHUO Q, LONG Z, ZHANG P. Preparation of nano-structured LiFePO4/graphene composites by co-precipitation method [J]. Electrochemistry Communications, 2010, 12: 10-13.
[12] HU G, GAO X, PENG Z, DU K, TAN X, LIU Y. Influence of Ti4+ doping on electrochemical properties of LiFePO4/C cathode material for lithium-ion batteries [J]. Transactions of Nonferrous Metals Society of China, 2007, 17: 296-300.
[13] WANG D, LI H, SHI S, HUANG X, CHEN L. Improving the rate performance of LiFePO4 by Fe-site doping [J]. Electrochimica Acta, 2005, 50: 2955-2958.
[14] SUN C S, ZHOU Z, XU Z G, WANG D G, WEI J P, BIAN X K, YAN J. Improved high-rate charge/discharge performances ofLiFePO4/C via V-doping [J]. Journal of Power Sources, 2009, 193(2): 841-845.
[15] LIU Y, LI X, GUO H, WANG Z, PENG W. YANG Y, LIANG R. Effect of carbon nanotube on the electrochemical performance of C-LiFePO4/graphite battery [J]. Journal of Power Sources, 2008, 184: 522-526.
[16] GAO X, HU G, PENG Z, DU K. LiFePO4 cathode power with high energy density synthesized by water quenching treatment [J]. Electrochimica Acta, 2009, 54: 4777-4782.
[17] NI J F, ZHOU H H, CHEN J T, ZHANG X X. LiFePO4 doped with ions prepared by co-precipitation method [J]. Materials Letters, 2005, 59: 2361-2365.
[18] MI C H, ZHANG X G, LI H L. Electrochemical behaviors of solid LiFePO4 and Li0.99Nb0.01FePO4 in Li2SO4 aqueous electrolyte [J]. Journal of Electroanalytical Chemistry, 2007, 602: 245-254.
[19] YAN X, YANG G, LIU J, GE Y, XIE H, PAN X, WANG R. An effective and simple way to synthesize LiFePO4/C composite [J]. Electrochimica Acta, 2009, 54: 5770-5774.
Mg-Co复合掺杂对LiFePO4电化学性能的影响
丁燕怀1, 2,张 平2
1. 湘潭大学 化工学院,湘潭 411105;
2. 湘潭大学 流变力学研究所,湘潭 411105
摘 要:通过固相反应制备了Mg2+和Co4+复合掺杂的LiFePO4电极材料。采用X射线衍射、恒电流充放电和循环伏安研究复合掺杂对LiFePO4结构和电化学性能的影响。结果表明:复合掺杂能够提高LiFePO4的首次放电比容量,0.1C和1C的放电容量分别达到147.2 mA・h/g和133.3 mA・h/g。循环伏安测试结果表明:复合掺杂改善了LiFePO4的导电性能,增强了Li+的脱嵌可逆性。
关键词:LiFePO4;复合离子掺杂;Fe位掺杂;固相法;电化学性能
(Edited by LI Yan-hong)
Foundation item: Project (51002128) supported by the National Natural Science Foundation of China; Project (2012M511737) supported by the National Science Foundation for Post-Doctor of China
Corresponding author: Ding Yan-huai; Tel: +86-731-58293861; E-mail: yhding@xtu.edu.cn