�Ӣɰ�����������������еķֽ��ѧ
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2019���9��
�������ߣ����� �ξ� ��˧ ������ ��ΰ ����
����ҳ�룺1948 - 1955
�ؼ��ʣ��Ӣɰ���ֽ⣻����ѧ�������Σ��������
Key words��zircon sand; decomposition; kinetics; sub-molten salt; sodium zirconium silicate
ժ Ҫ�����Ӣɰ�����������������еķֽ��ѧ�����о������췴Ӧ�¶ȡ���Ӧʱ�䡢NaOH�����������ٶȡ����ȵ����ض��Ӣɰ�ֽ��ʵ�Ӱ�졣�о�����������Ӣɰ�ķֽ����淴Ӧ�¶ȡ���Ӧʱ���NaOH���������Ӷ����ӡ��ֽ���̷�������ģ�ͣ����滯ѧ��ӦΪ���Ʋ��裬���ۻ��Ϊ77.98 kJ/mol�����ʵ�������µ�ת���ʸ���99%���ֽ����Ϊ�������(Na2ZrSiO5)��
Abstract: Sub-molten salt was applied to the decomposition of zircon sand (ZrSiO4). The kinetics of the decomposition of zircon sand and the effects of reaction temperature, reaction time, NaOH content, agitation speed, and the NaOH/ore mass ratio on the decomposition rate of zircon sand in NaOH sub-molten salt were investigated. The results indicate that the decomposition rate of zircon sand increases with the increase in the reaction temperature, reaction time, and NaOH content. The shrinking-core model with surface chemical reaction-controlled process is the most applicable for the decomposition of zircon sand, with the apparent activation energy of 77.98 kJ/mol. The decomposition product is sodium zirconium silicate (Na2ZrSiO5), and the decomposition rate is higher than 99% under the optimal conditions.
Trans. Nonferrous Met. Soc. China 29(2019) 1948-1955
Hong-qian SUN1,2, Jing SONG2, Shuai SUN2, Jing-kui QU2, Wei L��1, Tao QI2
1. School of Chemistry and Chemical Engineering, Shandong University, Jinan 250100, China;
2. National Engineering Laboratory for Hydrometallurgical Cleaner Production Technology, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100190, China
Received 19 December 2018; accepted 28 May 2019
Abstract: Sub-molten salt was applied to the decomposition of zircon sand (ZrSiO4). The kinetics of the decomposition of zircon sand and the effects of reaction temperature, reaction time, NaOH content, agitation speed, and the NaOH/ore mass ratio on the decomposition rate of zircon sand in NaOH sub-molten salt were investigated. The results indicate that the decomposition rate of zircon sand increases with the increase in the reaction temperature, reaction time, and NaOH content. The shrinking-core model with surface chemical reaction-controlled process is the most applicable for the decomposition of zircon sand, with the apparent activation energy of 77.98 kJ/mol. The decomposition product is sodium zirconium silicate (Na2ZrSiO5), and the decomposition rate is higher than 99% under the optimal conditions.
Key words: zircon sand; decomposition; kinetics; sub-molten salt; sodium zirconium silicate
1 Introduction
Zirconium is a metal with high hardness, high ductility, and wide applicability. Zirconium compounds are widely used in ceramics, chemicals, electronics, military industry, and other industries [1]. Zirconium oxychloride octahydrate (ZrOCl2��8H2O) is an important intermediate product of zirconium salt decomposited from zircon sand (ZrSiO4). ZrOCl2��8H2O not only produces zirconium compounds such as industrial-grade and high-purity zirconium, zirconium carbonate, zirconium sulfate, zirconium nitrate, and zirconium hydroxide, but also yields zirconium metal usable in the nuclear power industry for subsequent processing. Zircon sand is therefore an important raw material for obtaining zirconium and compounds [2-5].
The decomposition methods for zircon sand include thermal decomposition, chlorination, and sintering. The thermal decomposition method decomposes zircon sand in a high-temperature electric furnace into zirconium dioxide (ZrO2) and silicon dioxide (SiO2). This method has the disadvantages of a high decomposition temperature and poor economic benefits [6]. The chlorination of zircon sand yields ZrCl4 and then zirconium oxychloride, but it requires specific equipment [7,8]. In sintering, zircon sand reacts with a sintering agent at a high temperature to obtain zirconium oxychloride. Sintering agents include calcium carbonate (CaCO3), sodium carbonate (Na2CO3), sodium hydroxide (NaOH), and so on. Compared with CaCO3 and Na2CO3, NaOH has the advantages of simplicity, easy treatment, and a low sintering temperature, so it is extensively used [1,9-16].
Zirconium compounds are most commonly prepared by sintering with NaOH. Sintering is the most important step in the preparation of zirconium compounds, as it decomposes ZrO2 and SiO2 in zircon sand into sodium zirconate (Na2ZrO3) and sodium silicate (Na2SiO3), which are easily treated after reacting zircon sand and NaOH at high temperatures. After washing, trans- formation, and crystallization, the final product of ZrOCl2��8H2O is obtained [9,13]. However, this operation occurs in the sintering pot and is performed discontinuously in open spaces, which entails operator safety risks and wasted labor. Many liquids used in washing are difficult-to-handle and potential pollutants. In order to solve these problems, it is imperative to develop a new technology for the decomposition of zircon sand.
Sub-molten salt is a high-concentration medium with high chemical activity, high ionic activity for oxygen anions, high boiling point, low vapor pressure, and good fluidity, with a concentration between that of electrolytes and molten salts [17-19]. Recently, new sub-molten salt extraction processes have been proposed and widely studied to improve the efficiency of ore transformation. Many basic research results have shown that the sub-molten salt medium has some advantages relative to conventional media. In the sub-molten salt region, the reactant activity is greatly improved and the sub-molten salt medium has stronger reaction thermodynamics compared with traditional processes because of the increased boiling points of high- concentration media. In the sub-molten salt medium, the reaction, flow, and transfer characteristics are better than those in traditional reaction media, greatly improving the decomposition rates of reactants [20-24].
In the present work, sub-molten salt was applied to the decomposition of zircon sand. This method has the advantages of low reaction temperature and low pollution. The process of substituting sub-molten salt for a solid base permits the recycling of NaOH and more effective environmental protection. The flow chart of the decomposition process of zircon sand in the NaOH sub-molten salt solution is illustrated in Fig. 1. In this study, the effects of reaction temperature, reaction time, NaOH content, agitation speed, and the NaOH/ore mass ratio on zirconium extraction efficiency were investigated.
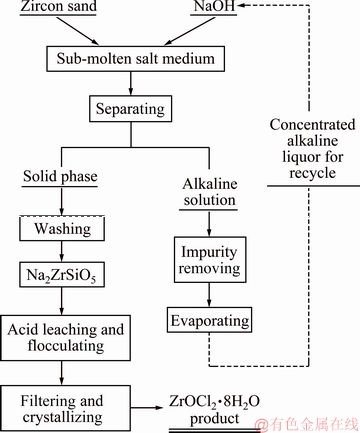
Fig. 1 Schematic flow chart of decomposition process of zircon sand in NaOH sub-molten salt solution
Finally, an empirical rate equation for the decomposition process of zircon sand in the NaOH sub-molten solution was obtained.
2 Experimental
2.1 Materials
The NaOH used in this work was of analytical grade and obtained from the Beijing Chemical Plant, China. The deionized water was super-purified using a water super-purification system (Milli-Q, Millipore, USA). The zircon sand used in the present work was provided by Kingan Hi-tech. Co., Ltd., Jiangxi Province, China. The detailed chemical composition of the zircon sand, as examined by X-ray fluorescence (XRF) (AXIOS), is listed in Table 1. The particle size (expressed as average diameter d50: 22.565 ��m) of the zircon sand was measured by a laser particle sizer, as shown in Fig. 2. The sample analysis by X-ray diffraction (XRD, Philips 1140, Cu K�� radiation, 40 mA, and 30 kV) is shown in Fig. 3. The results indicate that the main crystalline phase of the zircon sand is ZrSiO4.
Table 1 Chemical composition of zircon sand (wt.%)
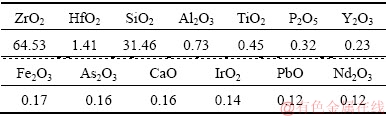

Fig. 2 Size distribution of zircon sand
2.2 Experimental apparatus and procedure
A diagram of the experimental set-up is shown in Fig. 4. All experiments were performed in a 1.0 L-cylindrical stainless steel-lined nickel autoclave. The autoclave was equipped with temperature control units, an electric heating furnace, a magnet-driven agitator, and an internally mounted cooling pipe. The internal temperature of the reactor was measured by a thermocouple that fed back to the furnace regulator with a digital read-out to allow adjustment of the heating power. The reaction temperature can be held constant with a precision of ��1 ��C.
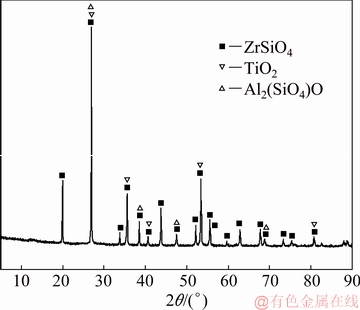
Fig. 3 XRD pattern of zircon sand

Fig. 4 Schematic diagram of experimental set-up
A certain quantity of NaOH was placed in the reactor with a certain amount of deionized water according to the required concentration. The zircon sand was added to reach a certain NaOH/ore ratio. Heating began after sealing, and time t was set to be 0 when the specified temperature was reached. Cooling began after a certain reaction time. The product was filtered at high temperature; some of the solid phase was washed with deionized water and dried at 105 ��C for 12 h. The filtrate was recycled by adding fresh NaOH to decompose fresh zircon sand. The reaction of zircon sand with NaOH is as follows:
ZrSiO4(s)+2NaOH(l)=Na2ZrSiO5(s)+H2O(l) (1)
ZrSiO4(s)+4NaOH(l)=Na2ZrO3(s)+Na2SiO3(l)+2H2O(l) (2)
To obtain the decomposition rate of the zircon sand, a certain amount of solid phase was added into hot water to remove the soluble substance, and excess 3:1 hydrochloric acid (diluted four times with deionized water) (at the solid-liquid ratio of 5:1) was added and reacted at 50 ��C for 4 h. After 1 h, the supernatant was dumped and the residue was washed with deionized water repeatedly to remove the silica gel. After filtration, the solid (unreacted ZrSiO4) was dried at 105 ��C for 12 h and weighed. All experimental data reported are the average values of three parallel examinations. The decomposition rate of zircon sand was calculated using the following equation:
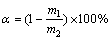 (3)
(3)
where m1 is the mass of the residue (unreacted ZrSiO4), and m2 is the total mass of the reacted zircon sand.
The reaction of hydrochloric acid leaching is as follows:
Na2ZrSiO5(s)+4HCl(1)=H2SiO3(s)+ZrOCl2(l)+2NaCl(l)+H2O(l) (4)
3 Results and discussion
3.1 Effect of reaction temperature
The temperature dependence of the sub-molten salt decomposition process can be used to estimate the apparent free energy and elucidate the macrokinetics of the process. The effect of the reaction temperature on the decomposition rate of zircon sand was investigated at 200-275 ��C, agitation speed of 400 r/min, NaOH/ore mass ratio of 3:1, initial NaOH content of 70%, and reaction time of 8 h, as shown in Fig. 5.
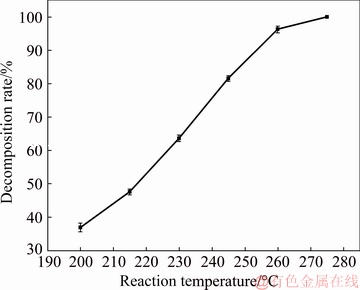
Fig. 5 Effect of reaction temperature on decomposition rate of zircon sand
Figure 5 shows that the decomposition rate of zircon sand is increased significantly with increasing reaction temperature. The temperature critically affects the decomposition rate of zircon sand. And the decomposition rate of zircon sand reached the maximum value of 99.0% at 275 ��C after 8 h reaction. This demonstrated that the zircon sand was almost completely decomposed. This behavior is attributed to the effect of the reaction temperature on both the rate constant and the diffusion coefficient. As the reaction temperature increases, both the diffusion and chemical reaction rates of the reactants and reaction products are increased, thereby increasing the decomposition rate of zircon sand.
3.2 Effect of initial NaOH content
The initial NaOH content of the sub-molten salt solution is important in determining the decomposition rate of zircon sand. The effect of the initial NaOH content on the decomposition rate was investigated using the initial NaOH content of 50%, 60%, 70% and 80% by mass fraction. In the experiments, the reaction temperature and agitation speed were fixed at 260 ��C and 400 r/min respectively and the reaction time was 8 h.
Figure 6 summarizes the effect of the initial NaOH content on the decomposition rate of zircon sand. According to the results shown in Fig. 6, the decomposition rate apparently increases as the initial NaOH content increases in the studied range. According to the law of mass action, with the increase in the initial NaOH content, the number of molecules participating in the reaction increases, thus promoting the reaction. Therefore, the increase of the initial NaOH content increases the decomposition rate of zircon sand. However, it can also be demonstrated that the reaction is not controlled by external diffusion. If the reaction is controlled by external diffusion, the increase of the initial NaOH content will increase the viscosity of the system and weaken the mass transfer, thereby suppressing the reaction.
3.3 Effect of reaction time
The effect of reaction time on the decomposition rate of zircon sand was investigated at 260 ��C, agitation speed of 400 r/min, NaOH/ore mass ratio of 3:1, and initial NaOH content of 70%. The experimental results after 2, 4, 6, 8, and 10 h reaction were obtained, as shown in Fig. 7. The decomposition rate at t=0 is not equal to 0 because the reaction of zircon sand begins during heating. For equal temperatures, the time necessary to reach a specific temperature is constant.
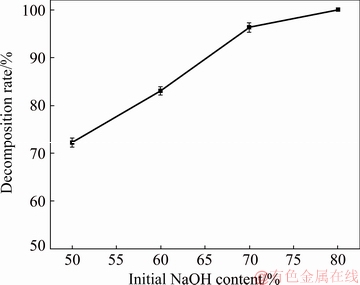
Fig. 6 Effect of initial NaOH content on decomposition rate of zircon sand
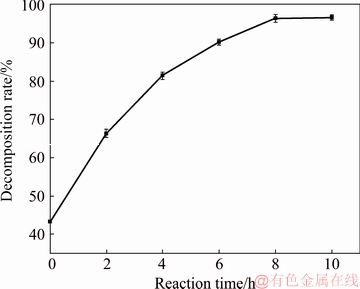
Fig. 7 Effect of reaction time on decomposition rate of zircon sand
Figure 7 shows that the decomposition rate of zircon sand increases significantly with increasing reaction time, but the rate of growth decreases with increasing reaction time. This is because the reaction initially occurs on the surface of the zircon sand particles. As the surface product layer accumulates on the zircon sand, the diffusion rate decreases and the mass transfer slows, thus decreasing the growth of the decomposition rate.
3.4 Effect of agitation speed
The effect of agitation speed on the decomposition rate of zircon sand was investigated under the following conditions: NaOH/ore mass ratio of 3:1, reaction temperature of 260 ��C, initial NaOH content of 70%, and reaction time of 4 h. The effect of agitation speed on the decomposition rate of zircon sand was studied. The results are shown in Fig. 8. The results indicate that the decomposition rate of zircon sand is not obviously affected by changes in agitation speed. This also demonstrates that the decomposition reaction of zircon sand is not controlled by external diffusion. If the reaction is controlled by external diffusion, changes in the agitation speed will obviously change the thickness of the diffusion layer and affect the mass transfer rate, thus influencing the decomposition rate. Therefore, to exclude the agitation intensity as a variable in the kinetics study, the agitation speed was maintained at 400 r/min in subsequent experiments.

Fig. 8 Effect of agitation speed on decomposition rate of zircon sand
3.5 Effect of NaOH/ore mass ratio
The amount of NaOH during the reaction is an important factor affecting mass transport and the degree of recycling throughout the process. The effect of the NaOH/ore mass ratio on the decomposition rate of zircon sand was investigated under the conditions of the initial NaOH content of 70%, reaction temperature of 260 ��C, agitation speed of 400 r/min, and reaction time of 4 h. The results are shown in Fig. 9, indicating that the decomposition rate of zircon sand does not change significantly with the increase in the NaOH/ore mass ratio, but only slightly increases. The increase of the NaOH/ore mass ratio is equivalent to the increase of NaOH content in the reactants, which promotes the decomposition reaction.

Fig. 9 Effect of NaOH/ore mass ratio on decomposition rate of zircon sand
3.6 Morphology and phase of products
To investigate the extraction behavior of zircon sand in NaOH sub-molten salt solution, the decomposed products under the optimal conditions (reaction temperature of 260 ��C, initial NaOH content of 70%, agitation speed of 400 r/min, and NaOH/ore mass ratio of 3:1) at different reaction time were analyzed by SEM and XRD. The results are shown in Figs. 10 and 11, respectively. As shown in Fig. 10, zircon sand is irregular and blocky in shape, while the product morphology is hexagonal columnar. The initial reaction occurs on the surface of the particle; the product is attached to the particle surface and gradually attains a regular shape. With increasing reaction time, the product surrounds the reactant particle, and the NaOH must be transported through the product layer to reach the reaction interface. As time progresses, a product with clear outlines and a regular shape is obtained. The corresponding XRD patterns in Fig. 11 indicate that the reaction of zircon and NaOH occurs according to Reaction (1), yielding the product Na2ZrSiO5. As the reaction proceeds, the phase ratio of ZrSiO4/Na2ZrSiO5 is gradually reduced. The solid particles gradually change from ZrSiO4 to Na2ZrSiO5. At the reaction time of 8 h, the product is almost all Na2ZrSiO5. The average particle size of the product is about 13 ��m, which is slightly reduced compared to that of zircon sand, as shown in Fig. 12.

Fig. 10 SEM images of decomposed products for different reaction time
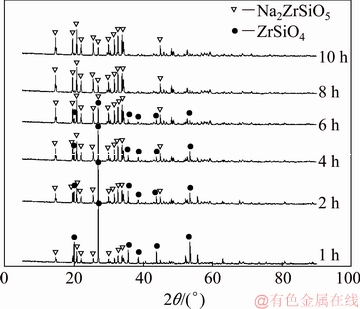
Fig. 11 XRD patterns of decomposed products at different reaction time

Fig. 12 Size distribution of zircon sand and its decomposed product at different reaction time
3.7 Kinetics analysis
The decomposition of zircon sand in NaOH sub-molten salt is a typical liquid�Csolid reaction that can be analyzed by the shrinking-core model in the temperature range of 200-275 ��C. Assuming spherical zircon sand particles, the reaction rate is mainly controlled by the following steps: the diffusion of reactants through the ash layer and the chemical reaction on the surface of the unreacted core materials. When the process rate is controlled by the chemical reaction, the following equation can be used to calculate the kinetic rate of the reaction [25]:
1-(1-X)1/3=k1t (5)
If internal diffusion is the rate-controlling step, the following equation can be used to calculate the kinetic rate of the reaction [25]:
1+2(1-X)-3(1-X)2/3=k2t (6)
where X is the ZrSiO4 decomposition rate at time t, and k1 and k2 are the apparent rate constants.
In order to determine the rate-controlling steps of the reaction, the experimental data were fitted with the above equations. The dependency of these models on the kinetic data was evaluated using the correlation coefficient (R2) values. The process corresponding to the equation with R2 values closer to 1 is the rate-controlling step. The relationship between the decomposition rate and reaction time at 200, 215, 230, 245 and 260 ��C was obtained at the initial NaOH content of 70%, the NaOH/ore mass ratio of 3:1, and the agitation speed of 400 r/min, as shown in Fig. 13. The experimental data were fitted according to the above equations, with results as shown in Figs. 14 and 15.
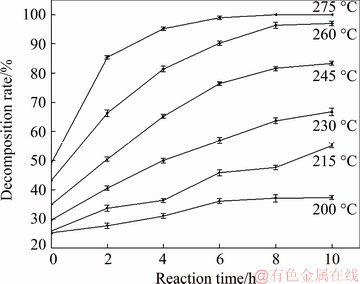
Fig. 13 Relationship between decomposition rate and reaction time for decomposition process of zircon sand at different reaction temperatures
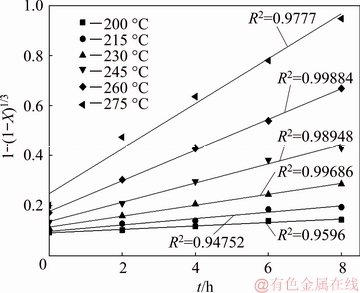
Fig. 14 Plots of 1-(1-X)1/3 versus reaction time (t) at various reaction temperatures according to Fig. 13

Fig. 15 Plots of 1+2(1-X)�C3(1-X)2/3 versus reaction time (t) at various reaction temperatures according to Fig. 13
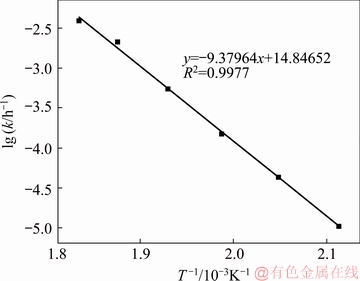
Fig. 16 Arrhenius plot for decomposition process of zircon sand
Comparing Figs. 14 and 15, the reaction-controlled model Eq. (5) yields a better fit than the diffusion- controlled model Eq. (6). Each temperature is well fitted, especially those of 230, 245 and 260 ��C. Therefore, the surface chemical reaction is considered the rate- controlling step for the decomposition of zircon sand. The apparent rate constant is determined from the straight lines in Fig. 14 and plotted according to the Arrhenius equation as shown in Fig. 16. The activation energy of the pressure decomposition process is 77.98 kJ/mol. In summary, the following kinetic expression can be applied to describing the decomposition process of zircon sand in the 70% NaOH sub-molten salt solution:
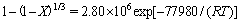 (7)
(7)
4 Conclusions
(1) With the closed cycle of NaOH, the decomposition process of zircon sand can realize a reduced reaction temperature, enhanced worker safety, and reduced pollution. Zircon sand was completely decomposed under the NaOH sub-molten salt system to form Na2ZrSiO5.
(2) The effects of the reaction temperature, initial NaOH content, agitation speed, and NaOH/ore mass ratio on the decomposition rates of zircon sand were obtained. Under the optimal conditions of 80% NaOH, 260 ��C, and 3:1 NaOH/ore mass ratio, the decomposition rate of zircon sand approached 99.0% after reacting for 8 h.
(3) The kinetics investigation indicated that the shrinking-core model suitably described the reaction of zircon sand in NaOH sub-molten salt and identified the surface chemical reaction as the rate-controlling step. The results showed the apparent activation energy of 77.98 kJ/mol.
References
[1] LIU Ran, QU Jing-kui, SONG Jing, QI Tao, DU Ai-ling. Analysis of water leaching and transition processes in zirconium oxychloride octahydrate production [J]. Ceramics International, 2014, l40(1): 1431-1438.
[2] ABDELKADER A M, DAHER A, EL-KASHEF E. Novel decomposition method for zircon [J]. Journal of Alloys and Compounds, 2008, 460(1): 577-580.
[3] LIU Jing-chong, SONG Jing, QI Tao, ZHANG Chang-qiao, QU Jing-kui. Controlling the formation of Na2ZrSiO5 in alkali fusion process for zirconium oxychloride production [J]. Advanced Powder Technology, 2016, 27(1): 1-8.
[4] CHANG Qi-bing, ZHOU Jian-er, WANG Yong-qing, MENG Guang-yao. Preparation and characterization of unique zirconia crystals within pores via a sol-gel-hydrothermal method [J]. Advanced Powder Technology, 2009, 20(4): 371-374.
[5] EJTEMAEI M, TAVAKOLI A, CHARCHI N, BAYATI B, BABALUO A A, BAYAT Y. Synthesis of sulfated zirconia nanopowders via polyacrylamide gel method [J]. Advanced Powder Technology, 2014, 25(3): 840-846.
[6] SINGH B P, BHATTACHARJEE S, BESRA L. Optimisation of performance of dispersants in aqueous plasma dissociated zircon suspension [J]. Ceramics International, 2008, 28(4): 413-417.
[7] BIDAYE A C, VENKATACHALAM S, GUPTA C K. Kinetic studies on the chlorination of zircon [J]. High Temperature Materials and Processes, 2002, 21(1-2): 1-10.
[8] BIDAYE A C, VENKATACHALAM S, GUPTA C K. Studies on the chlorination of zircon: Part I [J]. Metallurgical and Materials Transactions B, 1999, 30(2): 205-213.
[9] BISWAS R K, HABIB M A, KARMAKAR A K, ISLAM M R. A novel method for processing of Bangladeshi zircon: Part I: Baking, and fusion with NaOH [J]. Hydrometallurgy, 2010, 103(1): 124-129.
[10] ZHANG Jian-dong, WANG Li-jun, JIANG Dong-min. Decomposition process of zircon sand concentrate with CaO-NaOH [J]. Rare Metals, 2012, 31(4): 410-414.
[11] LI Zhong-jun, HUANG Yin-xia, YAO Hong-chang, YANG Min-le, CHEN Jian-li. Preparation of zirconium oxychloride via alkali fusing of zirconium carbide [J]. Chinese Journal of Rare Metals, 2006, 30(3): 415-418. (in Chinese)
[12] ABDEL-REHIM A M. A new technique for extracting zirconium from Egyptian zircon concentrate [J]. International Journal of Mineral Processing, 2005, 76(4): 234-243.
[13] MANHIQUE A, KWELA Z, FOCKE W W. De wet process for the benification of zircon: Optimization of the alkali fusion step [J]. Industrial and Engineering Chemistry, 2003, 42(2): 777-783.
[14] SILVA R J F D, DUTRA A J B, AFONSO J C. Alkali fusion followed by a two-step leaching of Brazilianzircon concentrate [J]. Hydrometallurgy, 2012, 117(2): 93-100.
[15] CHEN Wei-dong, YAN Guo-ping, ZHAO Jian-xiang, ZHANG Ya-zeng, YAN Shu-fang. Research on the decomposed process of zircon sand by NaOH and CaCl2 [J]. Titanium Industry Progress, 2013, 30(1): 37-40.
[16] SONG Jing, FAN Jing-fei, LIU Jing-chong, LIU Ran, QU Jing-kui, QI Tao. A two-step zircon decomposition method to produce zirconium oxychloride: Alkali fusion and water leaching [J]. Rare Metals, 2015, 34(1): 1-7.
[17] XU Hong-bin, ZHANG Yi, LI Zuo-hu, ZHENG Shi-li, WANG Zhi-kuan, QI Tao, LI Hui-quan. Development of a new cleaner production process for producing chromic oxide from chromite ore [J]. Journal of Cleaner Production, 2006, 14(2): 211-219.
[18] ZHENG Shi-li, ZHANG Yi, LI Zuo-hu, QI Tao, LI Hui-quan, XU Hong-bin. Green metallurgical processing of chromite [J]. Hydrometallurgy, 2006, 82(3): 157-163.
[19] ZHANG Yi, LI Zuo-hu, QI Tao, ZHENG Shi-li, LI Hui-quan, XU Hong-bin. Green manufacturing process of chromium compounds [J]. Environmental Progress and Sustainable Energy, 2005, 24(1): 44-50.
[20] WANG Shao-na, WANG Ya-ru, DU Hao, ZHENG Shi-li, ZHANG Yi. Decomposition mechanism of vanadium slag by sub-molten salt medium with active carbon catalytic oxidation [J]. The Chinese Journal of Nonferrous Metals, 2017, 27(8): 1729-1737. (in Chinese)
[21] CHEN Gang, WANG Jia-jun, WANG Xiao-hu, ZHENG Shi-li, DU Hao, ZHANG Yi. An investigation on the kinetics of chromium dissolution from Philippine chromite ore at high oxygen pressure in KOH sub-molten salt solution [J]. Hydrometallurgy, 2013, 139(3): 46-53.
[22] MENG Yan-rui, ZHAO Bin, ZHANG Hong-qi, LIU Xiu-wu, CAO Ji-lin. Synthesis of zeolite W from potassic rocks activated by KOH sub-molten salt method [J]. Crystal Research and Technology, 2018, 53: 1700216.
[23] LIU Long-jie, DU Hao, ZHANG Yang, ZHENG Shi-li, ZHANG Yi. Leaching of chromite ore in concentrated KOH by catalytic oxidation using CuO as catalys [J]. Transactions of Nonferrous Metals Society of China, 2017, 27: 891-900.
[24] CHEN Gang, WANG Xiao-hui, DU Hao, ZHANG Yang, WANG Jia-jun, ZHENG Shi-li, ZHANG Yi. A clean and efficient leaching process for chromite ore [J]. Mineral Engineering, 2014, 60(1): 60-68.
[25] LEVENSPIEL O. Chemical reaction engineering [M]. New York: Wiley, 1972.
����1,2���� ��2���� ˧2��������2���� ΰ1���� ��2
1. ɽ����ѧ ��ѧ�뻯��ѧԺ������ 250100��
2. �й���ѧԺ ���̹����о��� ʪ��ұ����������������ҹ���ʵ���ң����� 100190
ժ Ҫ�����Ӣɰ�����������������еķֽ��ѧ�����о������췴Ӧ�¶ȡ���Ӧʱ�䡢NaOH�����������ٶȡ����ȵ����ض��Ӣɰ�ֽ��ʵ�Ӱ�졣�о�����������Ӣɰ�ķֽ����淴Ӧ�¶ȡ���Ӧʱ���NaOH���������Ӷ����ӡ��ֽ���̷�������ģ�ͣ����滯ѧ��ӦΪ���Ʋ��裬���ۻ��Ϊ77.98 kJ/mol�����ʵ�������µ�ת���ʸ���99%���ֽ����Ϊ�������(Na2ZrSiO5)��
�ؼ��ʣ��Ӣɰ���ֽ⣻����ѧ�������Σ��������
(Edited by Wei-ping CHEN)
Foundation item: Project (51704270) supported by the National Natural Science Foundation of China; Project (ZDRW-ZS-2018-1) supported by the Key Research Program of Chinese Academy of Sciences; Project (KFJ-STS-ZDTP-040) supported by the Science and Technology Service Network Plan of Chinese Academy of Sciences
Corresponding author: Wei L��, Tel: +86-18678300166, E-mail: lwei@sdu.edu.cn; Jing-kui QU, Tel: +86-10-62631710, E-mail: jkqu@home.ipe.ac.cn
DOI: 10.1016/S1003-6326(19)65102-2

