混合酸溶液浸出刚玉烟尘灰中的镓
来源期刊:中国有色金属学报(英文版)2018年第9期
论文作者:文康 姜锋 周向阳 孙召明
文章页码:1862 - 1868
关键词:镓;浸出;刚玉烟尘灰;硫酸;氢氟酸;二氧化硅
Key words:gallium; leaching; corundum flue dust; sulphuric acid; hydrofluoric acid; silica
摘 要:为了从高硅含量的刚玉冶炼烟尘中提取镓,提出一种硫酸与氢氟酸混合酸溶液浸出的方法,并对浸出过程的各项实验参数进行优化。结果显示,当单独使用硫酸作为浸出剂时,刚玉烟尘中镓的浸出率仅为38%。从刚玉烟尘灰的微区成分分析结果中可知,烟尘中的富硅相镓含量较高;这部分镓无法溶入硫酸溶液。氢氟酸的加入对二氧化硅产生侵蚀,从而显著提高了刚玉烟尘灰中镓的浸出率。研究反应时间、温度、氢氟酸浓度和硫酸浓度对镓浸出率的影响。结果表明,在最佳浸出条件下,烟尘中镓的浸出率可达到91%。最佳的浸出参数如下:反应时间4 h、反应温度80 °C、混合酸溶液中硫酸浓度1.5 mol/L、氢氟酸浓度6.4 mol/L和液固比5:1 (mL/g)。
Abstract: In order to extract gallium from a high-silica-content flue dust generated in corundum production, a mixed acid solution of H2SO4 and HF was used for leaching, and test parameters of the leaching process were optimized. Experimental results show that the leaching rate of gallium was only 38% when H2SO4 was used as leaching agent. Composition analysis results of micro areas in this corundum flue dust indicate that the content of gallium in silica-enriched phases was high; this portion of gallium was insoluble in H2SO4 solution. The leaching rate of gallium increased significantly with addition of HF due to corrosion of silica. Effects of reaction time, temperature, and concentrations of HF and H2SO4 on leaching rates of gallium were investigated. The leaching rate of gallium reached 91% when this corundum flue dust was leached in a mixed acid solution of H2SO4 and HF for 4 h, at a temperature of 80 °C, with a liquid-to-solid ratio of 5:1 (mL/g). The optimal concentrations of H2SO4 and HF in the mixed acid solution were 1.5 and 6.4 mol/L, respectively.
Trans. Nonferrous Met. Soc. China 28(2018) 1862-1868
Kang WEN1, Feng JIANG1, Xiang-yang ZHOU2, Zhao-ming SUN2
1. School of Materials Science and Engineering, Central South University, Changsha 410083, China;
2. School of Metallurgy and Environment, Central South University, Changsha 410083, China
Received 30 May 2017; accepted 30 September 2017
Abstract: In order to extract gallium from a high-silica-content flue dust generated in corundum production, a mixed acid solution of H2SO4 and HF was used for leaching, and test parameters of the leaching process were optimized. Experimental results show that the leaching rate of gallium was only 38% when H2SO4 was used as leaching agent. Composition analysis results of micro areas in this corundum flue dust indicate that the content of gallium in silica-enriched phases was high; this portion of gallium was insoluble in H2SO4 solution. The leaching rate of gallium increased significantly with addition of HF due to corrosion of silica. Effects of reaction time, temperature, and concentrations of HF and H2SO4 on leaching rates of gallium were investigated. The leaching rate of gallium reached 91% when this corundum flue dust was leached in a mixed acid solution of H2SO4 and HF for 4 h, at a temperature of 80 °C, with a liquid-to-solid ratio of 5:1 (mL/g). The optimal concentrations of H2SO4 and HF in the mixed acid solution were 1.5 and 6.4 mol/L, respectively.
Key words: gallium; leaching; corundum flue dust; sulphuric acid; hydrofluoric acid; silica
1 Introduction
Gallium is an important scattered metal that enjoys vast applications in various high-tech fields. The use of GaAs and GaN in integrated circuits accounts for about 74% of total U.S. gallium consumption [1-3]. GaAs (or GaN) based integrated circuits offer low energy consumption and high computation speed. Therefore, they are mainly employed in telecommunications and military applications. Besides, gallium can also be used in optoelectronic devices such as solar cells, LEDs and photodetector [4-7].
Gallium is chemically similar to aluminum and zinc. Primary ores of gallium are extremely rare (except for gallite in Namibia and germanite in Tsumeb) and recovery of gallium from these sources is difficult [8]; it is generally recovered as a byproduct from alumina and zinc industries. OKUDAN et al [9] and ZHAO et al [10] recovered gallium from industrial waste generated in Bayer process, and similar researches have been carried out during the past decades. WU et al [11] and LIU et al [12-14] used acid leaching method to recover gallium from zinc residues. To a less extent, gallium is also recovered from coal fly ash and phosphorus flue dust [15,16]. In addition, flue dust obtained from dust collecting system in corundum manufacturer is another important source of gallium. This kind of dust was generated during the process of roasting bauxite to produce brown corundum, and gallium in the bauxite could be enriched through this process; it usually contains 0.05%-0.2% Ga (mass fraction) with SiO2, Al2O3, K2O and Fe2O3 as main components. So far, only a few researches have been carried out to extract gallium from corundum flue dust, and they mainly focused on acid leaching method since it was easy to operate. Some researchers used hydrochloric acid to extract gallium from a corundum flue dust, but the leaching rate of gallium was only about 77% [17]. In another research [18], a corundum flue dust containing 35% SiO2, 40% Al2O3 and 17% K2O (mass fraction) was treated with sulfuric acid, and the leaching rate of gallium exceeded 80% under normal pressure; this process was simple and low-cost, and it was performed under mild condition. However, the portion of gallium entrapped in silica in flue dust could not be extracted, and the filter velocity was very low due to the formation of silica gel during the process of acid leaching. Another problem of this method is the liquid-to-solid ratio was higher than 10:1 (mL/g), leading to high consumption of water. TIAN et al [19] proposed a process of alkali roasting- grinding-alkali leaching-separation-alkali leaching to recover gallium from a corundum flue dust collected in Guizhou province, China. This was an environmental benign method, and the silicon-calcium slag generated from recovering process could be recycled and used as raw materials in the cement industry. However, this process is complicated, flue dust should be mixed with CaCO3 and K2CO3, and then roasted at 1200 °C, so the energy consumption is high. Moreover, pH of the solution in separation stage should be strictly controlled, and the dosage of KOH used for leaching is very high.
In this study, an improved method of using mixed acid of H2SO4 and HF to leach gallium from a high-silica-content corundum flue dust was developed, and the related mechanisms were discussed. The whole process was easy to operate under mild conditions. Over 90% of the gallium in corundum flue dust could be effectively extracted. On the other hand, considering the large-scale production of corundum in China, this research also contributes to effective recovery of valuable metals from industrial waste.
2 Experimental
2.1 Materials and reagents
The corundum flue dust used in this study was collected from Shanxi Province, China. It was generated during the process of smelting bauxite in electric arc furnace to produce corundum. The flue dust sample and all chemicals were used as received without any treatment. Chemicals used for leaching, including sulfuric acid, hydrochloric acid and hydrofluoric acid were of analytical grade.
2.2 Equipment and leaching procedure
The leaching procedure was performed in a container equipped with a rotary stirrer. At first the leaching agent was added into the vessel and stirred for 30 min. Water bath equipment was used to heat the solution and maintained a constant temperature during the process of leaching. Corundum flue dust was added into the acid solution under continuously stirring when the solution was heated to required temperature. When the leaching process was finished, the suspension was filtered and the filter cake was washed with water until the filtrate was colorless. Finally, the filter cake was dried overnight in an oven at 60 °C. All leaching tests using HF should be performed in a Teflon container with a fitted lid. The liquid-to-solid ratio (L/S) of all leaching tests in this study was fixed at 5:1 (mL/g), which ensured a good fluidity of the suspension.
2.3 Analysis method
The particle size distribution of corundum flue dust was analyzed by using Malvern Mastersizer 2000 particle size analyzer. Phase characterization of flue dust was performed on an X-ray diffractometer (XRD, Rigaku-TTRIII, Cu Kα) with a scan step of 10 (°)/min, in the 2θ range from 5° to 70°. The XRD pattern was analyzed with MDI JADE software. The concentration of gallium in the leachate was analyzed by inductivity coupled plasma atomic emission spectroscopy (ICP-AES, IRIS Intrepid Ⅱ XSP), and the composition of leaching residues was determined by chemical analysis.
JXA-8230 electron probe microanalyzer (EPMA) equipped with a wavelength dispersive spectrometer (WDS) was used to determine the composition of micro areas in corundum flue dust sample. Qualitative analysis was carried out to identify phases, and then over 10 spots were chosen randomly in different silica-enriched micro areas and the composition of each spot was analyzed.
3 Results and discussion
3.1 Characterization of corundum flue dust
The main composition of the corundum flue dust is shown in Table 1, and it should be noted that the silica content in this sample is much higher than that of other corundum flue dust reported before. Moreover, the content of gallium in this flue dust is also higher than that in traditional gallium sources.
Table 1 Main chemical composition of corundum flue dust (mass fraction, %)

Particle size distribution of the corundum flue dust is presented in Table 2. It shows that only about 15% of the flue dust particles are larger than 100 μm, and about 7% of the particles are larger than 200 μm. Therefore, this corundum flue dust could be used in the leaching experiments without grinding.
X-ray diffraction (XRD) analysis was used to identify the existence of alumina and quartz in the flue dust sample. A broad peak centered at 2θ value around 21.6° was also recorded in the pattern (Fig. 1), indicating that the major silica phase in this material was in an amorphous state [20,21]. Although a peak of quartz could be observed at 2θ value of 26.6° in the XRD pattern of this flue dust sample, the quartz content was estimated to be very low. No special gallium-bearing minerals were identified in the pattern. The ion radius of Ga3+ is close to that of Al3+, and chemical properties of gallium and aluminum are similar. Gallium exists in bauxite in the form of isomorphism, so it probably exists as oxide in the flue dust, just as aluminum does.
Table 2 Particle size distribution of corundum flue dust


Fig. 1 XRD pattern of corundum flue dust
3.2 Leaching gallium from corundum flue dust using H2SO4
To investigate the feasibility of using sulfuric acid to extract gallium from corundum flue dust, tests were carried out under different H2SO4 concentrations and reaction time. The temperature for this set of leaching tests was fixed at 90 °C. The main chemical reactions in the process of H2SO4 leaching are shown in Eqs. (1) and (2):
Ga2O3+3H2SO4=Ga2(SO4)3+3H2O (1)
Al2O3+3H2SO4=Al2(SO4)3+3H2O (2)
According to result illustrated in Fig. 2(a), the leaching rate of gallium rose slightly with the increase of H2SO4 concentration, and the increase of leaching rate could be negligible when the H2SO4 concentration exceeded 2.9 mol/L. In these tests, only about 38% of the gallium in flue dust could be dissolved into the H2SO4 solution at the most.
The effect of reaction time on the extraction of gallium was also studied. Tests were performed at fixed temperature of 90 °C and H2SO4 concentration of 2.9 mol/L, with L/S of 5:1 mL/g. Figure 2(b) shows that the leaching rate of gallium increased slightly when the reaction time was prolonged, and there was almost no further dissolution of gallium after 6 h. The leaching rate of gallium was still below 40%.
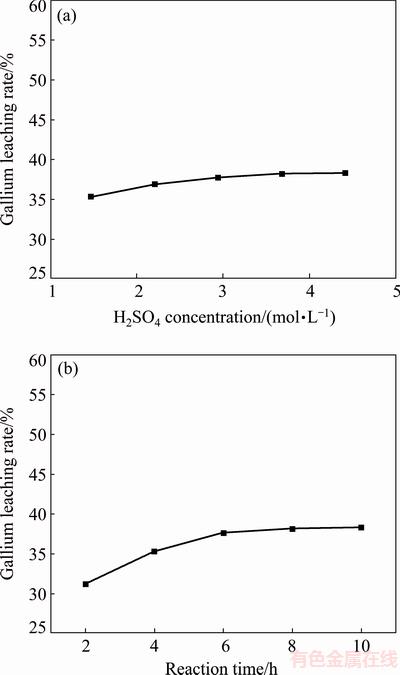
Fig. 2 Influence of H2SO4 concentration on gallium extraction in H2SO4 solution at reaction time of 6 h (a) and influence of reaction time on gallium extraction in 2.9 mol/L H2SO4 solution (b)
It is obvious that the leaching rate of gallium increased slightly with notable increase of H2SO4 concentration or reaction time. As stated in literatures, in the presence of potassium sulfate, the gallium sulfate gave alums, KGa(SO4)2・12H2O [8]. For this reason, the gallium content in H2SO4 leachate may be reduced. Hence, a leaching test using HCl as leaching agent was performed (temperature: 50 °C; reaction time: 6 h; L/S: 5:1 mL/g; HCl concentration: 5.8 mol/L), but the leaching rate of gallium was only raised slightly to 40%, indicating that the formation of alum was not the major cause of low leaching rate. Besides, another problem in these tests was the low filtration velocity (about 0.12 m/h).
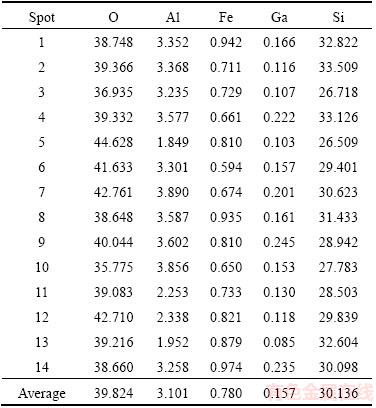
Obviously, the results of H2SO4 leaching tests were quite different from those reported in former studies [19], which may be ascribed to differences in composition of raw materials and occurrence condition of gallium. According to research results in Ref. [22], vitreous eutectic mixtures of silica and alumina (may also contain other elements, such as K and Fe) were formed during the process of manufacturing corundum in electric arc furnace at a temperature of over 2000 °C. Considering chemical similarities between Ga and Al, it is probable that gallium exists as oxide in this flue dust and occurs in vitreous eutectic mixtures, just as Al does. As shown in Table 1, the silica content of this corundum flue dust exceeds 50% (mass fraction), so it is highly probable that a large portion of gallium in this flue dust was wrapped in silica-enriched vitreous eutectic mixtures, which is insoluble in H2SO4 or HCl solution. As a result, gallium in these phases could not be extracted into the leachate. As illustrated in Table 3, the content of gallium in different silica-enriched areas ranges from 0.085% to 0.235%, and the average gallium content of these spots is 0.157%, which is close to that in raw materials. This confirms the assumption that there exists a large portion of gallium in the corundum flue dust sample in the silica-enriched phases.
Table 3 EPMA analysis of silica-enriched areas (mass fraction, %)
Besides, in aqueous solution, water molecules were adsorbed on silica surface and colloidal silica particles were formed. Then, condensation of these particles leads to the formation of silica gel [23], resulting in low filtration velocity.
3.3 Leaching gallium from corundum flue dust using H2SO4-HF mixed acid
The leaching rate of gallium was low when H2SO4 or HCl was used individually as leaching agent. As discussed above, it was because the portion of gallium wrapped in silica-enriched phases could not be extracted. It is important to corrode the silica phases to release this portion of gallium. Therefore, a mixed solution of HF and H2SO4 was used. The main reactions occurring during the process of leaching corundum flue dust in H2SO4-HF mixed acid solution can be expressed as follows [24,25]:
SiO2+6HF=H2SiF6+2H2O (3)
Al2O3+H2SiF6=2AlF3+SiO2+H2O (4)
Ga2O3+6HF=2GaF3+3H2O (5)
The reactions illustrated in Eqs. (1) and (2) also happened during this process. Moreover, GaF3 could be dissolved in hydrofluoric acid to form a hydrate GaF3・3H2O [26].
3.3.1 Effect of HF concentration
The effect of HF concentration on leaching rate of gallium was studied by varying the HF concentration in the range of 4-7.2 mol/L in mixed acid solution at 80 °C. The H2SO4 concentration was set to be 2.9 mol/L, and the reaction time was 6 h. It should be noted that the leaching rate of gallium in the mixed solution is much higher than that in H2SO4 solution, because HF molecules in solution could break the Si―O bonds and corrode vitreous silica phases. Therefore, gallium wrapped in these phases could be released into leachate. Figure 3 shows that the leaching rate of gallium increased from 59% to 93% with the rise of HF concentration from 4 to 6.4 mol/L, demonstrating that the dosage of HF is an important factor that influences the recovery of gallium from corundum flue dust. The leaching rate of gallium remained almost constant when the HF concentration was increased from 6.4 to 7.2 mol/L. Therefore, the optimal concentration of HF in this study was 6.4 mol/L.
3.3.2 Effect of reaction time

Fig. 3 Influence of HF concentration on gallium extraction in H2SO4-HF solution
In order to determine the optimal reaction time, a set of tests were performed in a mixed acid solution of 2.9 mol/L H2SO4 and 6.4 mol/L HF at 80 °C, by varying the duration of leaching. Figure 4 shows that the leaching rate of gallium increased steadily with prolongation of reaction time within 4 h; the leaching rate of gallium reached 92.5%, and it could hardly be increased by further prolonging the reaction time to 6 h. Therefore, the reaction time for subsequent tests was fixed to be 4 h.

Fig. 4 Influence of reaction time on gallium extraction in H2SO4-HF solution
3.3.3 Effect of H2SO4 concentration
The effect of H2SO4 concentration on gallium extraction was determined by leaching the corundum flue dust at 80 °C for 4 h in a mixed acid solution with HF concentration of 6.4 mol/L. The concentration of H2SO4 was varied from 0 to 3.6 mol/L. It can be seen from Fig. 5 that gallium could be dissolved in 6.4 mol/L HF solution without adding H2SO4 owing to dissolution of GaF3 in HF acid, and the leaching rate was 65%. The leaching rate of gallium could be increased greatly by raising H2SO4 concentration from 0 to 1.5 mol/L and it reached 91% when the H2SO4 concentration was 1.5 mol/L. It is obvious that the addition of H2SO4 enhanced the extraction of gallium effectively because H2SO4 in the mixed solution promoted the dissolution of silica, facilitating the release of gallium from silica-enriched phases. This could be attributed to some reasons. First of all, the addition of H2SO4 decreased pH value of the solution and prevented the precipitation of silicate in the form of calcium, sodium and potassium salts. Secondly, H3O+ ions in the mixed solution shifted the equilibrium of silica distribution towards silicon and aluminum complexes coordinated by fewer fluorine atoms, thus reducing the consumption of HF. Thirdly, according to researches, the dissolution of silica in the mixed solution of HF and strong acid consists of two parallel surface reactions: attack by adsorbed HF molecules and a reaction catalyzed by adsorbed H3O+ ions, which means that H3O+ ions in the mixed solution could act as an effective catalyst to increase the dissolution rate of silica [27-30]. Further increasing the concentration of H2SO4 could only slightly improve the gallium leaching rate, indicating that 1.5 mol/L H2SO4 in the mixed solution was sufficient for leaching.
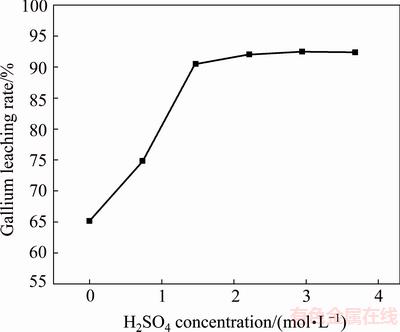
Fig. 5 Influence of H2SO4 concentration on gallium extraction in H2SO4-HF solution
3.3.4 Effect of leaching temperature
Corundum flue dust was leached at 50, 60, 70, 80 and 90 °C in a mixed solution of 1.5 mol/L H2SO4 and 6.4 mol/L HF for 4 h to determine the optimal temperature for leaching. It is shown in Fig. 6 that the curve of gallium leaching rate shows a slight upward trend with the increase of temperature. Similar leaching rates (about 91%) were obtained at both 80 °C and 90 °C. Therefore, a moderate temperature of 80 °C was chosen as the optimum.

Fig. 6 Influence of temperature on gallium extraction in H2SO4-HF solution
3.3.5 Characterization of leaching residues
Chemical compositions of the residues obtained from H2SO4 leaching (test conditions: 100 g flue dust, 2.9 mol/L H2SO4, 90 °C, 6 h, L/S=5:1) and mixed acid leaching test (test conditions: 100 g flue dust, 1.5 mol/L H2SO4, 6.4 mol/L HF, 80 °C, 4 h, L/S=5:1) are shown in Table 4. It can be seen that the mass of flue dust sample only decreased by 15% after leaching in H2SO4 solution; the mass of silica in the sample remained almost unchanged, and about 40% of alumina in the flue dust was dissolved into H2SO4 solution (the portion of alumina remained undissolved is probably α-Al2O3). Relative content of silica in the residue increased due to the mass loss of the flue dust sample, and the content of gallium in the leaching residue was still high. The mass of acid soluble phases such as K2O, Na2O, and Fe2O3 decreased obviously.
Table 4 Chemical composition of leaching residues

When a mixed acid solution of H2SO4 (1.5 mol/L) and HF (6.4 mol/L) was used as leaching agent, the mass of the corundum flue dust sample decreased markedly (from 100 to 43.9 g), and the total mass of silica in the sample reduced significantly (from 55.11 to 17.35 g). Moreover, the content of gallium in this residue was very low, indicating that most of gallium in the flue dust sample was dissolved into the leachate. Besides, the masses of Al2O3 remaining in both H2SO4 and H2SO4-HF leaching residues were almost the same.
According to results obtained from leaching tests and composition analysis of leaching residues, as the mass of silica in flue dust sample decreased, the leaching rate of gallium increased correspondingly. This further illustrates that H2SO4 could only dissolve gallium occurring in acid soluble phases (may also include small amount of gallium in silica-enriched phases but was not completely covered by silica), and the addition of HF enabled the dissolution of silica-enriched phases, so that the portion of gallium wrapped in these phases could be dissolved into the leachate.
Moreover, the addition of HF also increased the filtration velocity of the leaching suspension because the fluorine-containing ions in the mixed solution attacked the silica surface by breaking the Si―O bond of surface hydroxyl groups. The removal of hydroxyl groups brought hydrophobic properties to the surface of silica; therefore, the formation of silica gel was effectively inhibited [29,31].
4 Conclusions
1) Corundum flue dust used in this study contains high levels of silica, and the analysis result shows that there is a relatively high proportion of gallium in this flue dust in silica-enriched phases.
2) Less than 40% of the gallium in this corundum flue dust could be extracted by using H2SO4 or HCl individually as leaching agent, because the portion of gallium entrapped in silica-enriched phases could not be dissolved.
3) The leaching rate of gallium could be increased significantly to over 90% with addition of HF due to the corrosion of silica. In certain ranges, the leaching rate of gallium increased obviously with the rise of reaction time, temperature and concentration of H2SO4 and HF. The leaching rate of gallium was more than 91% under optimal leaching conditions: H2SO4 concentration 1.5 mol/L, HF concentration 6.4 mol/L, temperature 80 °C, reaction time 4 h and liquid-to-solid ratio 5:1 (mL/g).
References
[1] TANG Wu-jun, FENG Jun-cong. China rare metals [M]. Beijing: Metallurgical Industry Press, 2014: 2-5. (in Chinese)
[2] MOSKALYK R R. Gallium: The backbone of the electronics industry [J]. Minerals Engineering, 2003, 16(10): 921-929.
[3] FRENZEL M, KETRIS M P, SEIFERT T, GUTZMER J. On the current and future availability of gallium [J]. Resources Policy, 2016, 47: 38-50.
[4] SINCLAIR W D. Electronic metals (In, Ge and Ga): Present and future resources [J]. Acta Geologica Sinica (English Edition), 2014, 88(S2): s463-s465.
[5] QIN Shen-jun, SUN Yu-zhuang, LI Yan-heng, WANG Jin-xi, ZHAO Cun-liang, GAO Kang. Coal deposits as promising alternative sources for gallium [J]. Earth-Science Reviews, 2015, 150: 95-101.
[6] LOVIK A N, RESTREPO E, MULLER D B. The global anthropogenic gallium system: Determinants of demand, supply and efficiency improvements [J]. Environmental Science & Technology, 2015, 49(9): 5704-5712.
[7] NAUMOV A V. Modern state of the world market of gallium [J]. Russian Journal of Nonferrous Metals, 2014, 55(3): 270-276.
[8] GRAY F, KRAMER D A, BLISS J D. Gallium and gallium compounds [C]//Encyclopedia of Chemical Technology. New York: John Wiley & Sons, Inc., 2013: 1-26.
[9] OKUDAN M D, AKCIL A, TUNCUK A, DEVECI H. Recovery of gallium and aluminum from electrofilter dust of alumina calcination plant in Bayer process [J]. Separation Science and Technology, 2015, 50(16): 2596-2605.
[10] ZHAO Zhuo, YANG Yong-xiang, XIAO Yan-ping, FAN You-qi. Recovery of gallium from Bayer liquor: A review [J]. Hydrometallurgy, 2012, 125-126(8): 115-124.
[11] WU Xue-lan, WU Shun-ke, QIN Wen-qing, MA Xi-hong, NIU Yin-jian, LAI Shao-shi, YANG Cong-ren, JIAO Fen, REN Liu-yi. Reductive leaching of gallium from zinc residue [J]. Hydrometallurgy, 2012, 113-114(3): 195-199.
[12] LIU Fu-peng, LIU Zhi-hong, LI Yu-hu, LIU Zhi-yong, LI Qi-hou, ZENG Li. Extraction of gallium and germanium from zinc refinery residues by pressure acid leaching [J]. Hydrometallurgy, 2016, 164: 313-320.
[13] LIU Fu-peng, LIU Zhi-hong, LI Yu-hu, LIU Zhi-yong, LI Qi-hou. Leaching mechanism of zinc powder replacement residue containing gallium and germanium by high pressure acid leaching [J]. The Chinese Journal of Nonferrous Metals, 2014, 24(4): 1091-1098. (in Chinese)
[14] LIU Fu-peng, LIU Zhi-hong, LI Yu-hu, LIU Zhi-yong, LI Qi-hou, WEN Da-min. Sulfuric leaching process of zinc powder replacement residue containing gallium and germanium [J]. The Chinese Journal of Nonferrous Metals, 2016, 26(4): 908-918. (in Chinese)
[15] FANG Zheng, GESSER H D. Recovery of gallium from coal fly ash [J]. Hydrometallurgy, 1996, 41(2): 187-200.
[16] XU Ke, DENG Tong, LIU Jun-tan, PENG Wei-gong. Study on the recovery of gallium from phosphorus flue dust by leaching with spent sulfuric acid solution and precipitation [J]. Hydrometallurgy, 2007, 86(3-4): 172-177.
[17] ZHOU Ling-zhi. Rare metals metallurgy [M]. Beijing: Metallurgical Industry Press, 1988: 178-199. (in Chinese)
[18] LI Wang-xing, LU Pei-qian, YIN Zhong-lin, LI Xiao-ping, HAN Dong-zhan, YANG Qiao-fang. Method for recovering gallium from corundum flue dust: China Patent, 102676829A [P]. 2012-09-19. (in Chinese)
[19] TIAN Yuan-jiang, LI Hui-wen, ZENG Tian-yu, DENG Hua-xing. Method for recovering gallium from corundum smelting dust in arc furnace: China Patent, 1375564 [P]. 2002-10-23. (in Chinese)
[20] SHINOHARA Y, KOHYAMA N. Quantitative analysis of tridymite and cristobalite crystallized in rice husk ash by heating [J]. Industrial Health, 2004, 42(2): 277-285.
[21] WADDELL W H, EVANS L R. Silica, amorphous silica [C]// Encyclopedia of Chemical Technology. New York: John Wiley and Sons, Inc., 2000: 1-25.
[22] LI Hui-wen, WANG Ning, CHEN Ye-cai, TIAN Yuan-jiang. The environmental significance of the removal of corundum dusts and the mineralogical study of such dusts [J]. Acta Petrrologica et Mineralogica, 1999, 18(4): 348-356. (in Chinese)
[23] CARMEN P C. Constitution of colloidal silica [J]. Transactions of the Faraday Society, 1940, 36: 964-973.
[24] AIGUEPERSE J, MOLLARD P, DEVILLIERS D, CHEMLA M, FARON R, ROMANO R, CUER J P. Fluorine compounds, inorganic [M]. Weinheim: Wiley-VCH Verlag GmbH & Co. KGaA, 2005: 411-412.
[25] SHRIVER D, WELLER M, OVERTON T, ROURKE J, ARMSTRONG F. Inorganic chemistry [M]. 6th ed. New York: W. H. Freeman and Company, 2014: 374-375.
[26] DOWNS A J. Chemistry of aluminium, gallium, indium and thallium [M]. Glasgow: Blackie Academic & Professional, 1993: 128-129.
[27] KNOTTER D M. Etching mechanism of vitreous silicon dioxide in HF-based solutions [J]. Journal of the American Chemical Society, 2000, 122(18): 4345-4351.
[28] FRADEN J. Handbook of modern sensors: Physics, designs, and applications [M]. 5th ed. Cham: Springer International Publishing, 2016: 711-713.
[29] SPIERINGS G. Wet chemical etching of silicate glasses in hydrofluoric acid based solutions [J]. Journal of Materials Science, 1993, 28(23): 6261-6273.
[30] LE TIEC Y. Chemistry in microelectronics [M]. Hoboken: John Wiley & Sons, Inc., 2013: 202-209.
[31] MONK D J, SOANE D S, HOWE R T. A review of the chemical reaction mechanism and kinetics for hydrofluoric acid etching of silicon dioxide for surface micromachining applications [J]. Thin Solid Films, 1993, 232(1): 1-12.
文 康1,姜 锋1,周向阳2,孙召明2
1. 中南大学 材料科学与工程学院,长沙 410083;
2. 中南大学 冶金与环境学院,长沙 410083
摘 要:为了从高硅含量的刚玉冶炼烟尘中提取镓,提出一种硫酸与氢氟酸混合酸溶液浸出的方法,并对浸出过程的各项实验参数进行优化。结果显示,当单独使用硫酸作为浸出剂时,刚玉烟尘中镓的浸出率仅为38%。从刚玉烟尘灰的微区成分分析结果中可知,烟尘中的富硅相镓含量较高;这部分镓无法溶入硫酸溶液。氢氟酸的加入对二氧化硅产生侵蚀,从而显著提高了刚玉烟尘灰中镓的浸出率。研究反应时间、温度、氢氟酸浓度和硫酸浓度对镓浸出率的影响。结果表明,在最佳浸出条件下,烟尘中镓的浸出率可达到91%。最佳的浸出参数如下:反应时间4 h、反应温度80 °C、混合酸溶液中硫酸浓度1.5 mol/L、氢氟酸浓度6.4 mol/L和液固比5:1 (mL/g)。
关键词:镓;浸出;刚玉烟尘灰;硫酸;氢氟酸;二氧化硅
(Edited by Wei-ping CHEN)
Foundation item: Projects (51274240, 51204209) supported by the National Natural Science Foundation of China
Corresponding author: Xiang-yang ZHOU; Tel: +86-731-88836329; E-mail: hncsyjy308@163.com
DOI: 10.1016/S1003-6326(18)64831-9

