Separation of nickel, cobalt and copper by solvent extraction with P204
栖坿豚侵⦿嶄忽嗤弼署奉僥烏(哂猟井)2001定及5豚
胎猟恬宀⦿熱叫 藍蝕吼 果敢爽 屠敢埔 牝嗤功 蒐署乢
猟嫗匈鷹⦿803 - 805
Key words⦿non-equilibrium solvent extraction; equilibrium solvent extraction; nickel; cobalt; copper; di(2-ethylhexyl) phosphate
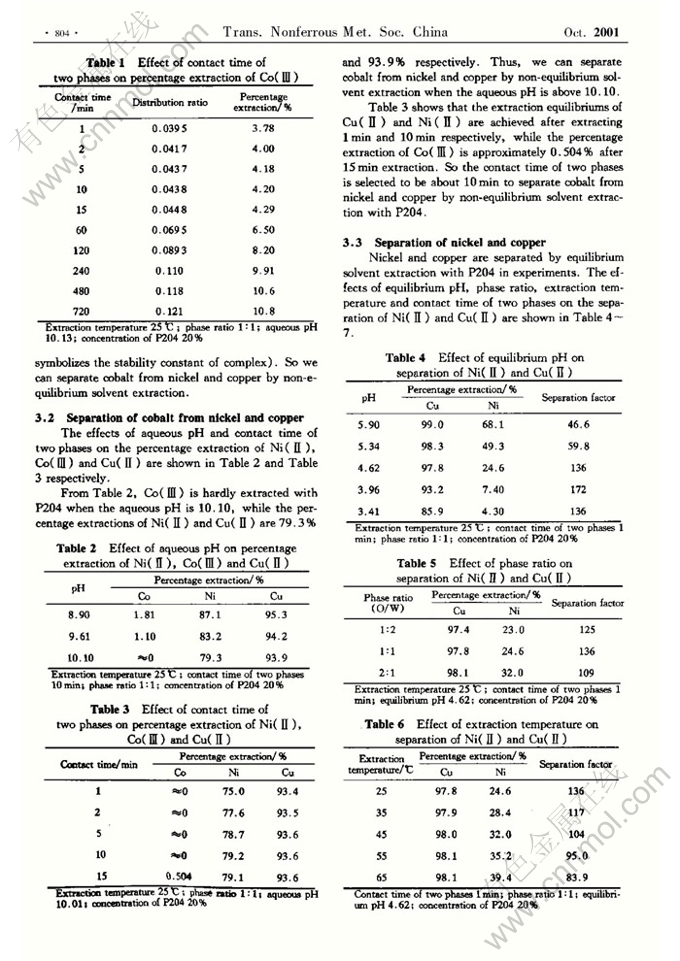
Abstract: Nickel, cobalt and copper were separated by solvent extraction with P204. The experimental results show that ∓Co(NH3)6ℏ3+ is an inert complex in extraction kinetics, therefore cobalt can be separated from nickel and copper by non-equilibrium solvent extraction. Under the conditions of temperature 25≧,Žcontact time of two phases 10min, phase ratio 1|1, aqueous pH 10.10 and concentration of P204 20%,∓Co(NH3)6ℏ3+ is hardly extracted by P204, while the percentage extractions of nickel and copper are 79.3%Žand 93.9% respectively. Nickel and copper are separated by equilibrium solvent extraction with P204. Under the conditions of temperature 25≧,Žcontact time of two phases 1 min,Žphase ratio 1|1, equilibrium pH 4.01 and concentration of P204 20%,the separation factor of copper and nickel is 216.