Trans. Nonferrous Met. Soc. China 22(2012) s697-s701
Modification of mechanical properties by TiO2 nano-particle for biodegradable materials made from palm oil sludge and activated sludge cake
Panithi YUTHAVISUTHI1, Lerpong JARUPAN1,2, Chiravoot PECHYEN1,2
1. Department of Packaging and Materials Technology, Faculty of Agro-Industry, Kasetsart University, Bangkok 10900, Thailand;
2. Center for Advanced Studies in Agriculture and Food (CASAF), KU Institute for Advanced Studies Kasetsart University, Bangkok 10900, Thailand
Received 21 May 2012; accepted 2 November 2012
Abstract: Titanium dioxide (TiO2) nano-particle is widely used in composite materials to its improved mechanical properties. TiO2 nano-particle was used in the composite material that consists of palm oil sludge from the palm oil production and activated sludge cake from a papermaking process. TiO2 was synthesized by a hydrolysis of titanium isopropoxide. The parameter was investigated by the mole ratio of reactant to different solvents (isopropanol and isobutanol). The solution was prepared by adjusting pH to acid solution, resulting in different sizes and distributions of precipitate which was heated at different temperatures. The obtained samples were then morphologically and structurally characterized using X-ray diffraction (XRD) and particle size distribution (PSD). The experimental results show that the TiO2 sample from isopropanol solvent with heated temperature of 600 ��C exhibits the best results. Consequently, different mass fractions of TiO2 (0, 0.5%, 1.0%, 2.0%, 4.0% and 8.0%) were used for specimens that were made from palm oil sludge mixed with activated sludge cake. All samples were later characterized by 3-point bending test and compression test. The results indicate that the adding 0.5% and 1.0% TiO2 particles to the composite material outperforms the other fractions in terms of enhancing mechanical properties. Nonetheless, mechanical properties tends to decrease when adding 2.0% TiO2.
Key words: titanium dioxide; palm oil sludge; active sludge cake; composite; mechanical properties
1 Introduction
The growing production and mass-volume use of petroleum plastics and composites in everyday life has led the accumulation of tremendous amount of non-degradable wastes, which have become a serious and urgent threat to the environment [1]. Therefore, environmental friendly materials has been chosen. Composite materials are focused because they usually have higher mechanical properties and longer lifetime than other materials. Composite materials are engineered or naturally occurring materials that are made from two or more constituent materials with significant difference in physical or chemical properties. Those properties remain separated and distinctive at the macroscopic or microscopic scale within the finished structure [2]. Recycling process utilizes old or waste materials to produce new products. Examples of recycled products can be composite materials from waste product [1,3-5] and biodegradable adhesive [6-8]. One main purpose of utilizing waste materials in recycling process is to prevent potential wastes to be worthlessly discarded and to reduce the consumption of fresh raw materials as well as to alleviate the environmental problems arisen from polluted resources and landfills.
Titanium dioxide (TiO2) nano-particle is widely used as filler agent to improve mechanical properties of composite materials. TiO2 is the naturally occurring oxide of titanium and has a form of crystal structure of anatase, rutile, or brookite. Presently, TiO2 has been extensively studied for various electronic applications. Its photocatalytic nature and transparent conductivity are strongly dependent upon the crystalline structure, morphology and crystallite size. Due to the photo- semiconductor properties of TiO2, one may find its applications as antibacterial agent for the decomposition of organisms. TiO2 of the anatase crystalline form is a strong bactericidal agent when exposed to near-UV light [9].
The preparation condition of the nanometer TiO2 powder was attempted to optimize by different solvents and calcination temperatures. With an evaluation on compressive strength, an optimal fraction of TiO2 nanoparticle for the composite material that made from palm oil sludge mixed with activated sludge cake was investigated, and tapioca starch was used as biodegradable adhesive.
2 Experimental
2.1 Synthesis of TiO2 nanopaticles
The initial solution was a mixture of 5 mL titanium isopropoxide (Ti[OCH(CH3)2]4) and two kinds of solvents (isopropanal and isobutanal) with the mole ratio between reactant to solvent was 1:30. The volume was adjusted to 250 mL with distilled water. The pH value of solution was adjusted to pH=2 by adding HNO3 or NH4OH. Hydrolysis of the turbid solution was heated up to 70 ��C in water bath for 18-20 h. After process, the solution was dried into a crystalline white solid. The prepared precipitates were washed with ethanol and dried for several hours at 100 ��C. They were dried again in a vacuum system for 3 h to obtain a yellow-white powder. The prepared powder was calcined at varied temperature of 600 ��C and 800 ��C for 2 h [10]. The obtained samples were then structurally characterized by means of X-ray diffraction (Bruker D8 ADVANCE), which was used to identify the crystal phase and estimate the average crystallite size as well.
2.2 Preparation of specimens
The composite material specimens made from palm oil sludge were mixed with activated sludge cake. Both materials were sifted with a sieve sized 500 ��m and a mixing ratio between palm oil sludge to activated sludge cake was 3:1. The biodegradable adhesive was prepared by 10% tapioca starch. Six different types of specimens were prepared based on different mass fractions of TiO2 (anatase phase) at 0, 0.5%, 1.0%, 2.0%, 4.0% and 8.0% (mass fraction). The compression machine was used to fabricate the specimens. Length, width and dept of each specimen are 90 mm, 25.4 mm and 3 mm, respectively. Later, the specimens were characterized by 3-point bending test and compression test.
2.3 Flexural properties
Flexural properties of the specimens were obtained by three-point bending test according to ASTM D-790. The testing was carried out at room temperature (23��1) ��C) by Instron machine No.5530 at crosshead rate of 3.0 mm/min. Flexural stress and strain were measured in three-point bending using specimen with cross-sectioned dimensions 3 mm��25.4 mm and 90 mm in length. The length between support span was 40 mm as recommended by the standard.
2.4 Compression resistance
Compression resistance of the specimens was carried out using Testometric Universal Machine No. 2696 at crosshead rate of 30 mm/min and room temperature of (23��1) ��C. Each specimen was tested according to ASTM D 642. The values of load at peak and load at break were measured.
3 Results and discussion
3.1 Analysis of TiO2 nanopaticles
The preparation of the TiO2 colloids in the nanometer range can be effectively conducted through the hydrolysis and condensation of titanium alkoxides in aqueous media. Alkoxides are hydrolyzed and subsequently polymerized to form, in the presence of water, a three-dimensional oxide network. The reactions can be systematically represented as
Ti(OR)4+4H2O��Ti(OH)4+4ROH (1)
Ti(OH)4��TiO2��xH2O+(2-x)H2O (2)
where R is ethyl, i-propyl, n-butyl, etc [11]. It is common to know that the tetravalent cations are so acidic that the nucleation of stable hydroxide Ti(OH)4 cannot occur.
Water molecules formed according to reaction (2) always bear a positive partial charge [12]. Therefore, oxolation and olation proceed simultaneously during nucleation and growth, leading to the formation of amorphous hydrous oxide (TiO2��nH2O). The precipi- tation of TiO2 leads to rutile or anatase phases [13]. Such structures from aqueous molecular precursors are formed. Non-occurrence of deoxolation (O=Ti��OH2��HO��Ti��Ti��OH) during nucleation olation leads to a linear growth along one of the two equivalent directions in the equatorial plane of [Ti2O2(OH)4(OH2)4]0 dimers. Oxolation between the resulting TiO(OH)2(OH)2 linear chains after an internal proton transfer leads to corner-sharing octahedral chains (Ti3O bridges) characteristics of the rutile structure. The formation of rutile may be associated to the metastability of apical Ti=O bonds within monomers or dimers. If deoxolation occurs prior to olation, condensation can proceed along apical direction, leading to skewed chains typical of the anatase structure. Controlling the stage of deoxolation prior to olation is obtained just by adjusting the pH and initial water concentration. This control leads to precipitation of anatase nanopowder TiO2 in the experimental procedure [10].
Figure 1 shows the XRD pattern of the prepared powder in different solvents at the different calcinations temperature. The plane diffraction peak is used for anatase and rutile. It shows that the volume of anatase phase in isopropanol solvent is more than isobutanol solvent. The volume of anatase phase decreases when the calcination temperature was up to 800 ��C. This temperature has been mostly reported from 600 to 900 ��C for the initiation and finishing temperature respectively [14].

Fig. 1 XRD patterns of TiO2 particles obtained from different solvents and temperatures
It is then possible to assume that the growth of rutile particle starts right after its nucleation. In fact, this reveals that nucleation and growth of rutile phase would have been initiated at temperature somewhere from 400 to 600 ��C [10,15].
3.2 Mechanical properties of composite material
The fabricated composite material specimens made from palm oil sludge mixed with activated sludge cake by compression method is shown in Fig. 2. According to the results of mechanical properties, a comparison of the flexure stress at maximum flexure strain and maximum flexure stress of 3-point bending of the five samples were compared as shown in Fig. 3. According to the results, it is found that the flexure stresses at maximum flexure strain has similar values, but the maximum flexure stress increases by the addition of TiO2 nanopaticles. In the case of 0.5% and 1.0% TiO2, the maximum flexure stresses remarkably increase. However, the maximum flexure stress is not improved as the addition amount of the TiO2 nanoparticles increase. It is obviously seen that the flexural stress tends to decrease when TiO2 is over 2.0%. Figures. 4 and 5 show the maximum flexure load and elastic modulus, respectively. The results are consistent with maximum flexure stress, remarkable increase by the addition of 0.5% and 1.0% TiO2, with the highest at the 1.0% TiO2. However, the elastic modulus decreases when the addition of TiO2 is over 2.0%.

Fig. 2 Specimen made from palm oil sludge mixed with activated sludge cake by compression method
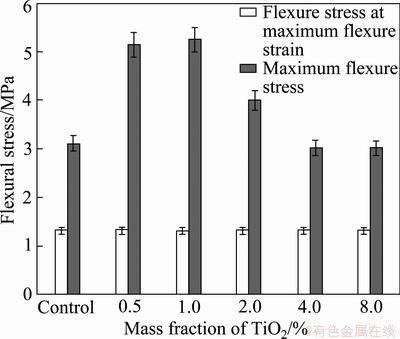
Fig. 3 Comparison of flexure stress at maximum flexure strain and maximum flexure stress of 3-point bending

Fig. 4 Maximum flexure load of composites at different mass fractions of TiO2
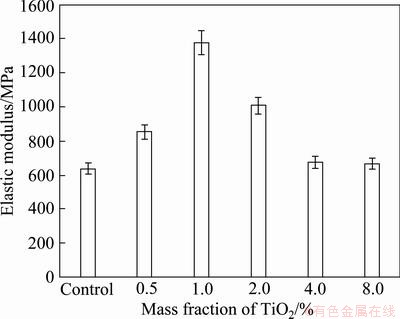
Fig. 5 Elastic modulus of composites at different mass fractions of TiO2
In general, there is a strong argument in the aforementioned literature over the effect of filler content researcher results show that the flexural strength of on the flexural strength of modified composite. Some microparticle-filled composites is reduced with rising filler content [16], while others argue that flexural strength increases with nano particles [17].
Figure 6 shows a comparison of the load at peak and load at break of compression test. Based on the results, the values of load at peak are more than the values of load at break. Both values tend to remarkably increase by adding 0.5% TiO2 nanoparticles. However, the values decrease with the mass fraction of TiO2 increasing.
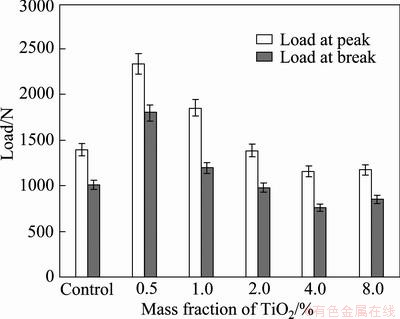
Fig. 6 Mass fraction vs load at peak and break of compression test at different mass fractions of TiO2
In order to explain this phenomenon, the following should be considered. The dispersion state of the nanoparticles and the interface in composites occur. Regarding the first factor, the agglomeration is likely to be formed more at higher filler content, and that may cause embrittling effects. Large agglomerates in the matrix cause a propagation crack and induce the initiation of the final failure [18].
As discussed earlier, the nano-particles interface in composites plays a major role in the improvement of mechanical properties. Stresses transfer and elastic deformation from the matrix to the fillers are governed by the interface quality [19]. The nano-particles with 1% TiO2 are able to induce further mechanisms of failure without blocking matrix deformation. According to the crack pining theory, particles may act as obstacles to crack growth by pinning [20]. When fillers exceed 1% (mass fraction), a large number of fillers dominate and reduce the matrix deformation. This indicates that there is not increasing significantly when the filler content is above 1%.
4 Conclusions
Nanocrystalline TiO2 powder were prepared by the hydrolysis of titanium isopropoxide alcoholic solution. Rutile phase is formed at calcinations temperatures 600 ��C and grew slightly when heated up to 800 ��C. Mechanical properties of the composite material specimens made from palm oil sludge mixed with activated sludge cake, tapioca starch as biodegradable adhesive and TiO2 nanoparticle remarkably increase as the addition proportion of TiO2 nanoparticle increases. However, the additional amount of TiO2 nanoparticles could cause the depletion in mechanical properties. Therefore, it can be concluded that adding 0.5% TiO2 particles can be an optimal proportion for the composite material since it outperforms the other fractions in terms of enhancing mechanical properties.
Acknowledgments
The authors wish to thank the Graduate School Kasetsart University and the Office of the National Research Council of Thailand for their financial support under a research plan ��Increase the value through the production of agriculture��.
References
[1] SONG Y, ZHENG Q. Structure and properties of methylcellulose microfiber reinforced wheat gluten based green composites [J]. Industrial Crops and Products, 2009, 29(2-3): 446-454.
[2] ROSATO D V. Handbook of composites [M]. LUBIN G. New York: Van Nostrand Reinhold, 1982.
[3] DAVIS G, SONG J H. Biodegradable packaging based on raw materials from crops and their impact on waste management [J]. Industrial Crops and Products, 2006, 23(2): 147-161.
[4] MIKI T, TAKEUCHI K, SUGIMOTO H, KANAYAMA K. Material development from wood powder without adhesive by vapor steaming compaction process [J]. Journal of Materials Processing Technology, 2008, 199(1-3): 396-401.
[5] KINOSHITA H, KAIZU K, FUKUDA M, TOKUNAKA H, KOGA K, IKEDA K. Development of green composite consists of woodchips, bamboo fibers and biodegradable adhesive [J]. Composites Part B: Engineering, 2009, 40(7): 607-612.
[6] STEFANI P M, PEN C, RUSECKAITE R A, PITER J C, MONDRAGON I. Processing conditions analysis of Eucalyptus globulus plywood bonded with resol-tannin adhesives [J]. Bioresource Technology, 2008, 99(13): 5977-5980.
[7] KACZMAREK H, SWIATEK M, KAMINSKA A. Modification of polystyrene and poly (vinyl chloride) for the purpose of obtaining packaging materials degradable in the natural environment [J]. Polymer Degradation and Stability, 2004, 83(1): 35-45.
[8] LI X, LI Y, ZHONG Z, WANG D, RATTO J A, SHENG K, SUN X S. Mechanical and water soaking properties of medium density fiberboard with wood fiber and soybean protein adhesive [J]. Bioresource Technology, 2009, 100(14): 3556-3562.
[9] JITKATKUAN S. The catalyst reaction cleaning with titanium dioxide [J]. Journal of Department of Science Service, 2009, 179: 21-24.
[10] MAHSHID S, ASAKI M, GHAMSARI M S. Synthesis of TiO2 nanoparticles by hydrolysis and peptization of titanium isopropoxide solution [J]. Journal of Materials Processing Technology, 2007, 189(1-3): 296-300.
[11] LIVAGE J, HENRY M, SANCHEZ C. Sol-gel chemistry of transition metal oxides [J]. Progress in Solid State Chemistry, 1988, 18(4): 259-341.
[12] SANCHEZ C, LIVAGE J, HENRY M, BABONNEAU F. Chemical modification of alkoxide precursors [J]. Journal of Non-Crystalline Solids, 1988, 100(1-3): 65-76.
[13] SHIN H, JUNG H S, HONG K S, LEE J K. Crystallization process of TiO2 nanoparticles in an acidic solution [J]. Chemistry Letters, 2004, 33: 1382-1383.
[14] OSKAM G, NELLORE A, PENN R L, SEARSON P C. The growth kinetics of TiO2 nanoparticles from titanium (IV) alkoxide at high water/titanium ratio [J]. The Journal of Physical Chemistry B, 2003, 107: 1734-1738.
[15] LI B, WANG X, YAN M, LI L. Preparation and characterization of nano-TiO2 powder [J]. Materials Chemistry and Physics, 2002, 78: 184-188.
[16] NIELSEN L, LANDEL R. Mechanical properties of polymer and composites [M]. New York: Marcel Decker, 1994.
[17] CHATTERJEE A, ISLAM M S. Fabrication and characterization of TiO2�Cepoxy nanocomposite [J]. Materials Science and Engineering A, 2008, 487(1-2): 574-585.
[18] HAMAD A A. Effect of nano TiO2 particle size on mechanical properties of cured epoxy resin [J]. Progress in Organic Coatings, 2010, 69(3): 241-246.
[19] ZHANG M, ZENG H, ZHANG L, LIN L, LIN G, LI R K Y. Fracture characteristics of discontinuous carbon fibre-reinforced PPS and PES-C composites [J]. Polymers and Polymer Composites, 1993, 1: 357-365.
[20] GREEN D J, NICHOLSON P S, EMBURY J D. Fracture of a brittle particulate composite-Part 2 theoretical aspects [J]. Journal of Materials Science, 1979, 14(7): 16571661.
(Edited by LI Yan-hong)
Corresponding author: Lerpong JARUPAN; Tel: +662-562-5045; E-mail: lerpong.j@ku.ac.th
DOI: 10.1016/S1003-6326(12)61789-0