以白云石和菱镁石的混合物为原料真空铝 热还原法炼镁过程的动力学
来源期刊:中国有色金属学报(英文版)2014年第3期
论文作者:傅大学 冯乃祥 王耀武 彭建平 狄跃忠
文章页码:839 - 847
关键词:镁;动力学;铝热还原;菱镁石;白云石
Key words:magnesium; kinetics; aluminothermic reduction; magnesite; dolomite
摘 要:研究真空铝热还原煅烧白云石和煅烧菱镁石的混合物炼镁过程的动力学,提出一种满足真空铝热过程的等温还原方法。实验在4 Pa的真空下进行。实验结果表明:还原率随着温度、制团压力和铝粉添加量的提高而增加。对不同还原时间的还原渣进行XRD分析,结果表明,还原过程能够粗略地分为3个阶段:MgAl2O4和Ca12Al14O33的形成阶段;CaAl2O4的生成阶段;CaAl4O7的形成阶段。根据动力学模型将获得的实验数据分为3部分,这3部分的活化能分别为98.2、133.0和223.3 kJ/mol。
Abstract: The vacuum aluminothermic reduction of the mixture of calcined magnesite and calcined dolomite was studied. An isothermal reduction method satisfying the vacuum aluminothermic reduction was proposed. The experiments were carried out at 4 Pa. The results indicate that the reduction rate is increased with increasing temperature, content of aluminum and pellet forming pressure. The XRD patterns of pellets at different reduction stages confirm that the reduction process can be roughly classified into three stages: the formation of MgAl2O4, and Ca12Al14O33 phases; the phase transformation from MgAl2O4 and C12A7 to CaAl2O4; the formation of CaAl4O7 phase. The experimental data were divided into three parts according to the kinetic models. The apparent activation energies of the three parts were determined to be 98.2, 133.0 and 223.3 kJ/mol, respectively.
Trans. Nonferrous Met. Soc. China 24(2014) 839-847
Da-xue FU, Nai-xiang FENG, Yao-wu WANG, Jian-ping PENG, Yue-zhong DI
School of Materials and Metallurgy, Northeastern University, Shenyang 110819, China
Received 5 March 2013; accepted 4 July 2013
Abstract: The vacuum aluminothermic reduction of the mixture of calcined magnesite and calcined dolomite was studied. An isothermal reduction method satisfying the vacuum aluminothermic reduction was proposed. The experiments were carried out at 4 Pa. The results indicate that the reduction rate is increased with increasing temperature, content of aluminum and pellet forming pressure. The XRD patterns of pellets at different reduction stages confirm that the reduction process can be roughly classified into three stages: the formation of MgAl2O4, and Ca12Al14O33 phases; the phase transformation from MgAl2O4 and C12A7 to CaAl2O4; the formation of CaAl4O7 phase. The experimental data were divided into three parts according to the kinetic models. The apparent activation energies of the three parts were determined to be 98.2, 133.0 and 223.3 kJ/mol, respectively.
Key words: magnesium; kinetics; aluminothermic reduction; magnesite; dolomite
1 Introduction
Magnesium is the lightest metal among the commonly used structured metals, and its wide application in many fields, such as metallurgy, chemistry, automobile industry [1,2], implies the huge demands for magnesium in the world. Magnesium is produced by two principal processes: electrolysis of molten magnesium chloride and thermal reduction of magnesia. The proportion of electrolysis is getting smaller because this process is characterized by high-energy consumption [3] and high cost [4]. The Pidgeon process, which has been in the mature stage [5-7], is widely used in thermal reduction of magnesia. Although this process is better than electrolysis in terms of energy consumption, cost, etc, it is still disadvantaged in these parts [8]. Several works [9,10] proposed the process of carbothermic reduction of magnesia. However, the well-known primary issue with the carbothermal process is the easy reverse reaction. A new vacuum thermal reduction method, by which magnesium is produced through aluminothermic reduction of a mixture of calcined dolomite and calcined magnesite, was proposed by FENG and WANG [11]. Compared with the Pidgeon process, this method can reuse the reduction residue and greatly cut down energy consumption and CO2 emissions. The optimal parameters of this process were studied by HU [12] et al and WANG et al [13]. However, the kinetics of this process is not well studied.
On the other hand, the kinetics of aluminothermic reduction of magnesia was reported. By use of the isothermal reduction, YANG et al [14] investigated the effects of temperature, carrier gas flow rate, pellet forming pressure on the reduction ratio of MgO. The apparent activation energy of the pellets containing magnesium oxide and aluminum at a molar ratio of 4:2 was calculated and the value was 109 kJ/mol. LAN et al [15] used the non-isothermal reduction method to calculate the apparent activation energy of the pellets containing magnesium oxide and aluminum at a molar ratio of 3:2, which was 151.2 kJ/mol. In addition, they also proved that the lime addition accelerated the reaction. However, the apparent activation energy of the pellets with the lime addition was not calculated in their work.
As for the conventional isothermal reduction methods, such as aluminothermic reduction of titanium oxide [16], the reacting substances were preliminarily placed in an alumina crucible and were preheated at a lower temperature. Before starting an experiment, the temperature was increased to the prescribed one as quickly as possible by applying full power of induction furnace. However, this method cannot be applied to a rapid reduction process such as the aluminothermic reduction of magnesium oxide. YANG et al [14] proposed an isothermal aluminothermic reduction method. They promptly inserted a graphite tube charged with the samples into a high-temperature zone to start the experiment and the tube was rapidly lifted up to a low temperature zone to end the experiment. The inert atmosphere was maintained in the furnace by blowing argon gas. However, the method cannot satisfy the vacuum reduction condition.
In this work, a method is proposed in order to obtain the isothermal reduction process under the vacuum condition. Effects of temperature, MgO-to-Al mole ratio and pellet forming pressure on the reduction ratio are investigated by the isothermal reduction method. The apparent activation energy is obtained. The reduction mechanism of extracting magnesium from the mixture of dolomite and magnesite is preliminarily analyzed by the X-ray diffraction.
2 Experimental
2.1 Raw materials
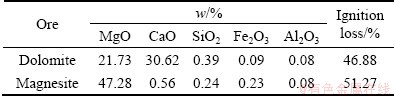
The raw materials used in this study were obtained from Dashiqiao in Liaoning Province, China. Table 1 shows the major compositions of magnesite and dolomite. The purity of aluminum power used as reduction agent is 99% in mass fraction.
Table 1 Major chemical composition of raw materials
2.2 Apparatus
The experimental apparatus is shown in Fig. 1. A resistance furnace with SiC heating elements was used to heat the tube which was made of high-temperature alloyed steel. The temperature was measured with NiCr-NiSi thermocouple.
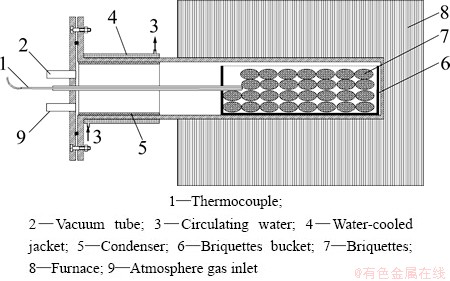
Fig. 1 Schematic of experimental apparatus
2.3 Procedures
The dolomite and magnesite were calcined at 1323 K and 1023 K for 90 min, respectively. They were pulverized (10 μm in average diameter) and mixed at a molar ratio of calcined magnesite to calcined dolomite to aluminum to be 5:1:4 since the reaction was expected as follows:
CaO・MgO+5MgO+4Al→CaO・2Al2O3+6Mg↑ (1)
The mixed powders were compacted into 25 mm- diameter and 23 mm-height cylindrical pellets under a compacting pressure of 90 MPa using a cold isostatic press. The pellet mass was (20±0.2) g before reduction and the change in pellet mass after reduction was measured with an electronic balance with a detection precision of 1 mg. The vacuum thermal reduction experiments were carried out at 4 Pa. The reduction residues at different stages of reduction were qualitatively investigated by XRD (PW3040/60 PANALYICAL B.V, using the Cu Kα target).
The reduction ratio of magnesium oxide (η) is defined as the ratio of magnesium mass loss in the reduction (Δm) to the initial magnesium mass in the pellets (m0). Since release of the produced magnesium vapor is the only reason of the change in the pellet mass, the change in the pellet mass is taken as the magnesium mass loss.
η= (Δm/m0)×100% (2)
2.4 Isothermal reduction method
For the isothermal vacuum reduction, if the samples are placed in the vacuum tube at the experimental temperature, the reductant will be oxidized in the operating time for sealing the tube. The other method is that the samples are placed in the tube at the ambient temperature and the atmosphere in the tube is adjusted to the experimental vacuum. The tube is preheated at a lower temperature. Before starting an experiment, the temperature is increased to the prescribed one as quickly as possible by applying full power of the furnace. However, the elapsed time for temperature increasing from the preheated one to the prescribed one is too long to satisfy the isothermal reduction condition for rapid reduction processes such as the aluminothermic reduction.
In the present experiments, two same furnaces were used. The temperature of the first one was kept at 1273 K. The temperature of the second one was at the experimental one. The samples were placed in the tube at room temperature. The tube was outgassed down to 0.5 Pa and then filled with argon to the pressure of 120 kPa. After the temperature of the tube reached 1273 K, the tube was promptly transferred into the second furnace. The release valve was regulated to maintain the pressure at 120 kPa during the heating process. The vacuum valve was opened when the temperature reached the prescribed one. The pressure in the tube was reduced to 4 Pa within 20 s by using the mechanical pump and the roots pump. After the experiment, the pressure in the tube was promptly recovered to 120 kPa by filling argon and the tube was taken out of the furnace to end the reduction. Figure 2 shows the relationship between the elapsed time for elevating temperature from 1273 K to the experimental one and the reduction ratio during this process. The reduction reaction is so slow at 120 kPa during the heating process that it is appropriate to consider the method as an isothermal reduction one.
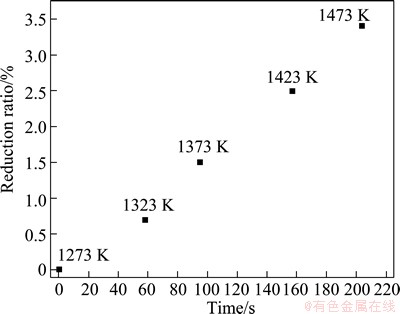
Fig. 2 Relationship between elapsed time from 1273 K to experimental one and reduction ratio during this process
3 Results and discussion
3.1 Effect of temperature on reduction ratio
Figure 3 shows the changes in reduction ratio with time at different temperatures. The compacting pressure of pellets was 90 MPa. The lines are the calculated results, which will be explained later.
At the temperature of 1473 K, the reduction ratio increased very fast at first, reached 77.11% at only 10 min and increased slowly at subsequent soaking time. The final reduction ratio was 85.39%. With decreasing temperature to 1423 K and 1373 K, the reduction ratio became slow. The reduction ratios were 71.13% and 60.36% at 15 min, and 78.1% and 70.29% at the end of the experiments. When the temperature was further decreased to 1323 K, the reduction behavior was quite different from that at the higher temperatures. The reduction ratio rose fast in the initial 2 min, and then turned into a gradual increase until 5 min, but the reduction accelerated after 5min. The final reduction ratio was 63.46%. The difference in the reduction pattern may be explained by the results of YANG et al [17]. They observed the pellets at the different reduction stages by SEM at 1373 K and considered that alumina film formed on the surface of aluminum particle reduced the aluminothermic reduction ratio in the initial several minutes. The reduction was improved in the subsequent time due to the elimination of the alumina film. However, the phenomenon was observed at 1323 K in the present study. This is because calcium oxide contained in the raw materials reacted with the alumina film, which resulted in the phenomenon observed at a lower temperature.
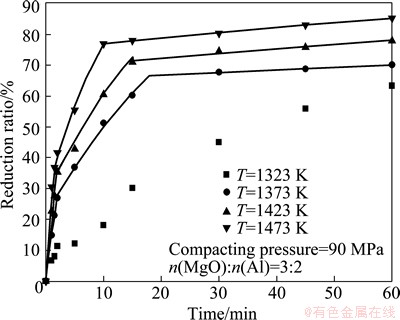
Fig. 3 Effect of temperature on reduction ratio
3.2 Effect of magnesia-to-aluminum mole ratio on reduction ratio
Figure 4 illustrates the effect of the magnesia to aluminum mole ratio on the reduction ratio. There was no apparent increase of the reduction ratio in the first 2 min with the increase of aluminum content. The reduction ratio increased evidently after 2 min. The finial reduction ratio was 68.2% at the magnesia-to-aluminum mole ratio of 3:1.9. With magnesia-to-aluminum mole ratio reaching 3:2.0 and 3:2.1, the finial reduction ratio increased to 78.1% and 82.2%. The reduction ratio increased slowly when magnesia to aluminum mole ratio was further increased to 3:2.2. The contacting area between magnesia and aluminum increases with the increase of aluminum content in the mixture, which accelerates the reaction.
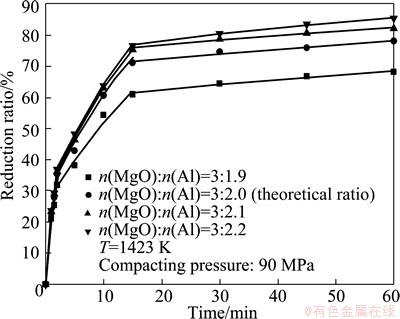
Fig. 4 Effect of MgO-to-Al mole ratio on reduction ratio
3.3 Effect of pellet forming pressure on reduction ratio
Figure 5 shows the effect of the pellet compacting pressure on the isothermal reduction. The reduction ratio increased evidently when the pellet forming pressure increased from 60 MPa to 90 MPa. The reduction ratios were 70.92% and 78.1% at 60 min, respectively. This is because increasing the pellet forming pressure increases the contact area between magnesia and aluminum, and thus improves the penetration of molten aluminum into the magnesium oxide phase [14]. The final reduction ratio only reached 80.5% when the pellet forming pressure was increased to 120 MPa. The escape of the produced magnesium vapor from the reaction site was hindered since the increase of the compacting pressure raised the pellet density. This resulted in the slow increase of the reduction ratio.
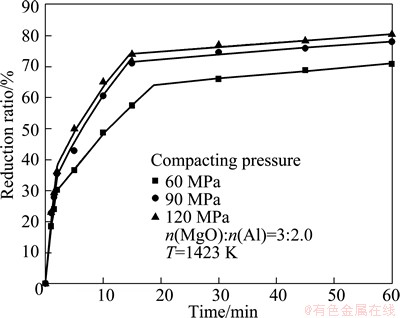
Fig. 5 Effect of compacting pressure on reduction ratio
4 Kinetics and reaction mechanism of isothermal reduction
4.1 Reaction mechanism
The pellets at different reduction stages were analyzed by XRD to clarify the mechanism of the aluminothermic reduction. The results are shown in Fig. 6. The reduction experiments were carried out under the conditions of 1423 K, n(MgO):n(Al) of 3:2 and pellet forming pressure of 90 MPa.
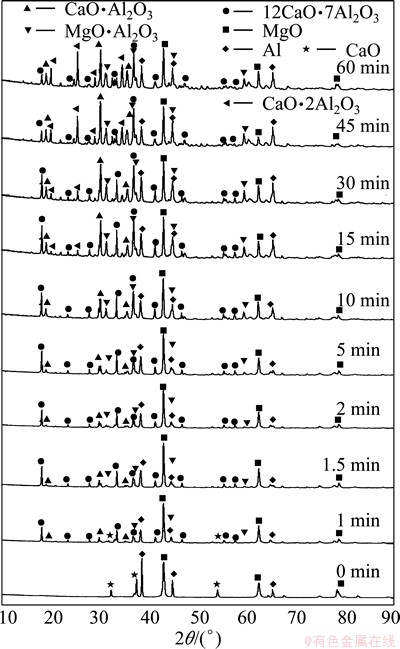
Fig. 6 XRD patterns of reduction residues prepared at 1423 K for different reduction time
Before the reduction, the XRD pattern shows that the pellets contained MgO, CaO and Al. After the reduction, the main phases in the residue were Al, MgO, MgAl2O4, CaAl2O4 (CA), CaAl4O7 (CA2) and Ca12Al14O33 (C12A7). The changes with time in the intensities of peaks of Al (111), MgO (200), MgAl2O4 (311), CA (123), CA2 ( ) and C12A7 (211), which are the strongest peaks of these substances, are presented in Fig. 7. It is clear that the intensities of magnesium oxide and aluminum peaks decreased as the reduction proceeded. This indicated that magnesium oxide and aluminum were continuously consumed by the reduction reaction. The intensity of the C12A7 peak increased only in the first 1.5 min, and then decreased until the end of the experiment. The intensity of the CA peak increased rapidly in the first 15 min, changed slowly in the subsequent 30 min and decreased in the last 15 min. The intensity of the spinel peak increased rapidly in the first 10 min and decreased slowly in the subsequent time. The CA2 phase appeared after 15 min and the peak intensity increased until the end of the experiment.
) and C12A7 (211), which are the strongest peaks of these substances, are presented in Fig. 7. It is clear that the intensities of magnesium oxide and aluminum peaks decreased as the reduction proceeded. This indicated that magnesium oxide and aluminum were continuously consumed by the reduction reaction. The intensity of the C12A7 peak increased only in the first 1.5 min, and then decreased until the end of the experiment. The intensity of the CA peak increased rapidly in the first 15 min, changed slowly in the subsequent 30 min and decreased in the last 15 min. The intensity of the spinel peak increased rapidly in the first 10 min and decreased slowly in the subsequent time. The CA2 phase appeared after 15 min and the peak intensity increased until the end of the experiment.
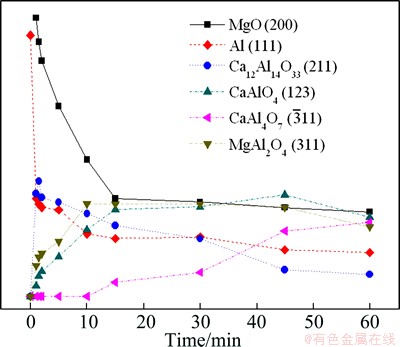
Fig. 7 Changes in intensities of Al(111), MgO(200), MgAl2O4(311), CA(123), 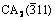 , C12A7(211) in XRD patterns with time
, C12A7(211) in XRD patterns with time
From these results, it is deduced that the C12A7 phase was transferred into the CA phase from 1.5 min to 15 min. The CA phase was consumed and transferred into the CA2 phase after 15 min, but the CA peak increased slowly in the subsequent 30 min due to the phase transformation from C12A7 to CA. It can be believed that the calcium aluminate phases in the reduction residue were formed in the sequence of C12A7→CA→CA2. The results are consistent with that reported by SCIAN et al [18]. The formation rate of spinel was slower than its consumption rate after 10 min. Therefore, the reduction process can be roughly classified into three stages. In the first stage, MgAl2O4, and C12A7 phases were formed rapidly in the initial of 1.5 min. In the second stage, the phase transformation occurred from MgAl2O4 and C12A7 to CA with the fast increase of the reduction ratio. In the third stage, the magnesium oxide and part of the spinel were further reduced. The CA2 phase was formed with the slow increase of the reduction ratio.
As well known, spinel is the main product of the aluminothermic reduction of magnesium oxide. And C12A7 was the main phase in the residue of aluminothermic reduction of calcined dolomite according to LIU et al [19]. The reaction mechanism is summarized as follows. 1) Molten aluminum penetrates into calcined magnesite and calcined dolomite [17] in which the magnesium oxide is reduced to form MgO・Al2O3 and C12A7, respectively. 2) MgO・Al2O3 and C12A7 transfer into CA and further into CA2. The description of the reaction mechanism is superficial and simple. The more specific mechanism needs to be studied further in the future.
4.2 Kinetics
The kinetic formula is expressed as follows:
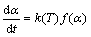 (3)
(3)
where α is the reaction degree, which is defined by
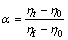 (4)
(4)
where ηt is the reduction ratio at time t, ηf is the final reduction ratio at the end of the experiment, and η0 is the initial reduction ratio. In the present case, α=ηt can be obtained since ηf =1 and η0=0. Integration of Eq. (3) gives the relationship between α and t. The functions F(α) and f(α) are shown in Table 2.
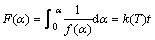 (5)
(5)
The function F(α) in Table 2 is substituted into Eq. (5). Table 3 shows the Adj. R-square values of the fitting lines by using the reduction ratios at 1373 K. The linear relationship is poor in the scale of 0-60 min. If the scale of time is reduced, the linear relationship will become stronger. In the scale of 0-2 min, the linear relationship obtained from the functions A1, R2 and R3 is better than that from other functions. The same method was used to determine the scale of time at temperatures of 1423 K and 1473 K. Good linear relationship is obtained in the scale of 0-2 min and the values of Adj. R-square are shown in Table 4. According to the Adj. R-square values, the function R3 is the best to explain the experimental results in the scale of 0-2 min.
Table 2 Mechanism functions of reduction process
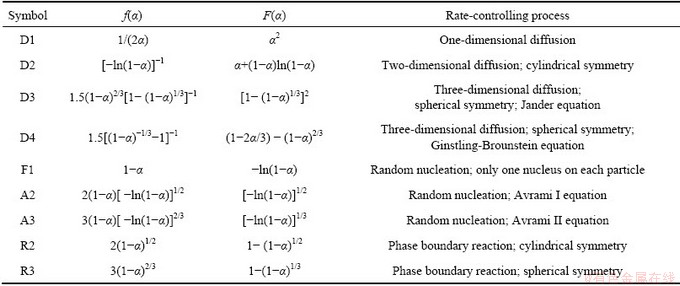
Table 3 Values of Adj. R-square obtained from reduction ratios at 1373 K
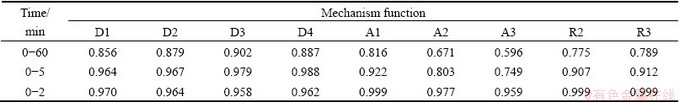
Table 4 Values of Adj. R-square obtained from reduction ratios at 0-2 min
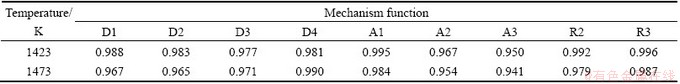
According to the same principle, the experimental results are divided into three parts. The second and third parts are well explained by the functions R3 and D3, respectively. The relationship between F(α) and t at different temperatures is shown in Fig. 8.
A parameter β is defined as follows:
 (6)
(6)
where αt is equal to ηt which is the reduction ratio at time t; αR,f is the final reduction ratio in the experiments. In Figs. 8(b) and (c), the linear relationship between F(α) and t is expressed by Eq. (7) since the intercepts are not equal to zero.
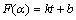 (7)
(7)
The increasing k values with temperature indicate that the reduction is greatly enhanced at a higher temperature. From the obtained apparent rate constants, the change in reduction ratio with time can be calculated according to Eq. (5) and Eq. (7), which are plotted in Fig. 3. The inflection points of the curves can be attributed to the errors of the different parts. The calculated results are in good agreement with the experimental ones. Therefore, the kinetic models are valid for explaining the present experimental results.
According to the Arrhenius equation, the relationship between the apparent rate constant (k) and the absolute temperature (T) is given by
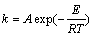 (8)
(8)
where A is the pre-exponential constant (min-1), R is the gas constant (J/(mol・K)), and E is the apparent activation energy (J/mol). Figure 9 shows the relationship between ln k and 1/T at the temperatures of 1373, 1423 and 1473 K. The apparent activation energy values are determined from the slopes of the straight lines to be 98.2, 133.0 and 223.3 kJ/mol for the three stages, respectively. According to the kinetic models, the reduction process of the first and second stages are controlled by phase boundary reaction, while the diffusion process determines the reaction rate of the third stage. The differences of the apparent activation energy for the three stages are due to the different reaction mechanisms.
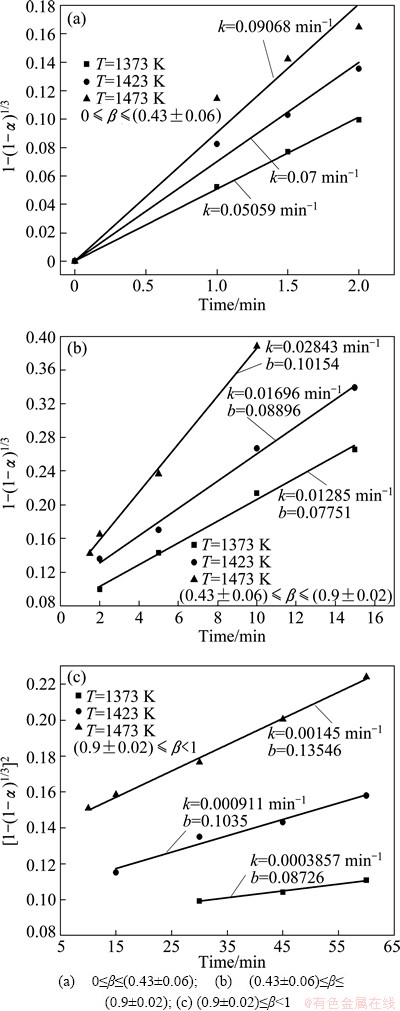
Fig. 8 Relationship between F(α) and time at different temperatures
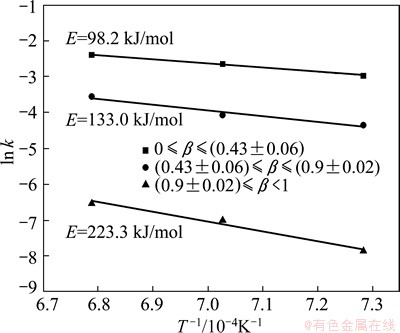
Fig. 9 Relationship between ln k and 1/T
The apparent activation energy of the aluminothermic reduction of magnesium oxide was 151.2 kJ/mol obtained by LAN et al [15] without the lime addition. According to their results, the lime addition can reduce the activation energy. However, the average value of activation energy is 151.5 kJ/mol in this work which is almost equal to that obtained by LAN et al [15]. This may be attributed to the different size of the pellets. The diameter of the pellets used in this study was 25 mm, which was 5 times larger than that used in the study of LAN et al. In addition, LAN et al [15] made experiments under the non-isothermal condition and used the different kinetic equation.
Figure 10 shows the relationship between F(α) and time at the different MgO-to-Al mole ratios. The gaps between the values of k increase with the increasing time. For example, the values of k are 0.06295, 0.07, 0.07252 and 0.07367 min-1 at the stage of 0≤β≤(0.44±0.03). With the decreasing MgO-to-Al mole ratios, the value of k increases by 11%, 15.2% and 17%, respectively. However, the increase of the k values are 45.3%, 81.8% and 272.7% at the stage of (0.9±0.02)≤β<1. With the increasing aluminum content, the increase of the reduction rate in the latter stage is more marked than in the early stage. By use of the obtained apparent rate constants, the calculated changes in reduction ratio with time accord well with the experimental ones at different MgO-to-Al mole ratios as shown in Fig. 4.
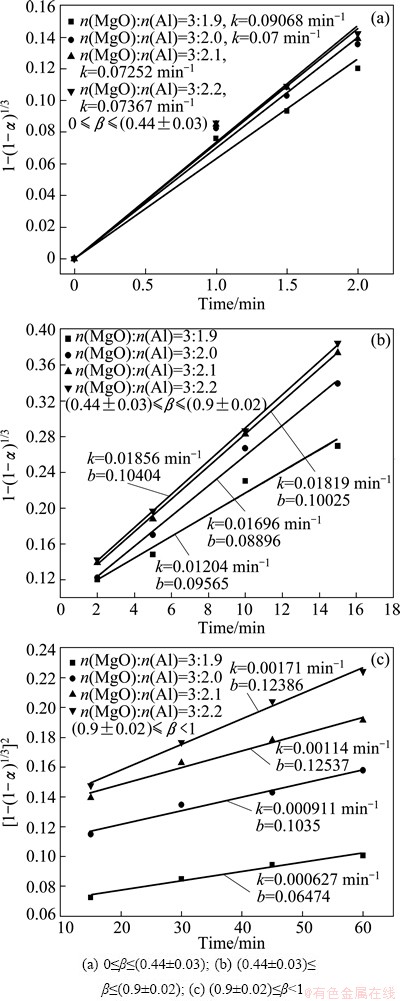
Fig. 10 Relationship between F(α) and time at different MgO-to-Al mole ratios
The relationship between F(α) and time for different pellet forming pressures is shown in Fig. 11. Good linear relationship is obtained. The increasing pellet forming pressure is conducive to enhance the mass transport which has an advantaged effect on the reduction rate. Therefore, the values of k increase with the increasing pellet forming pressure. By use of the obtained apparent rate constants, the calculated reduction ratios are in agreement with the experimental results for the different pellet forming pressures shown in Fig. 5.
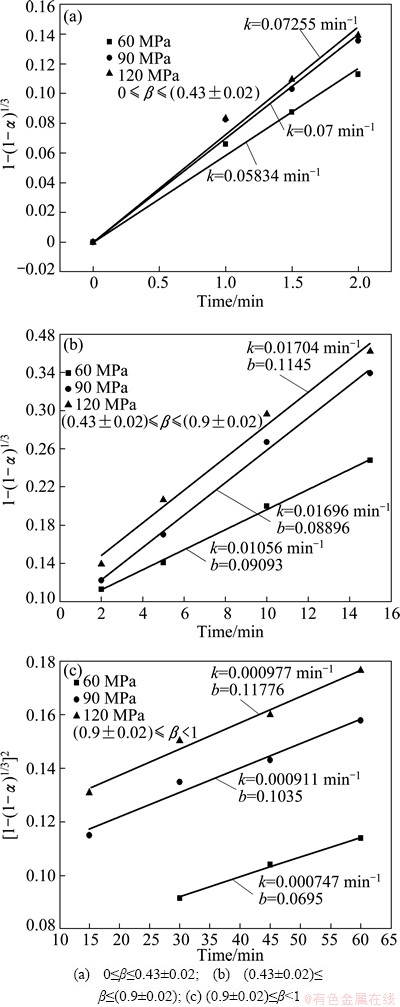
Fig. 11 Relationship between F(α) and time at different pellet forming pressures
5 Conclusions
1) In the sealed tube, air is replaced by argon gas and the pressure is maintained at 120 kPa. The aluminothermic reduction rate is so slow in this condition that there is enough time to elevate the temperature. This method can satisfy with the isothermal aluminothermic reduction.
2) The reduction ratio is increased with the increase of temperature, content of aluminum and pellet forming pressure.
3) The reaction mechanism is summarized as follows. Molten aluminum penetrates into calcined magnesite and calcined dolomite in which the magnesium oxide is reduced to form MgO・Al2O3 and C12A7, respectively; MgO・Al2O3 and C12A7 transfer into CA and further into CA2.
4) The experimental data can be divided into three parts according to the kinetic calculation. The reduction process of the first and second stages is controlled by phase boundary reaction, while the diffusion process determines the reaction rate of the third stage. The apparent rate constants of the three parts are calculated by using the kinetic models. The results indicate that the apparent rate constants increase with the increase of temperature, aluminum content and pellet forming pressure. The apparent activation energy values of the three parts are determined to be 98.2, 133.0 and 223.3 kJ/mol, respectively.
References
[1] ELIEZER D, AGHION E, FROES F H. Magnesium science, technology and applications [J]. Advanced Performance Materials, 1998, 5(3): 201-212.
[2] Hanko G, Antrekowitsch H, Ebner P. Recycling automotive magnesium scrap [J]. JOM, 2002 54(2): 51-54.
[3] Sun Ze, Zhang He-nan, Li Ping, Li Bing, Lu Gui-min. Modeling and simulation of the flow field in the electrolysis of magnesium [J]. JOM, 2009, 61(5): 29-33.
[4] Lebedev O A, Brusakov Y, Shkuryakov N P. Express monitoring of MgCl2 concentration in the electrolyte of magnesium electrolyzers [J]. Russian Journal of Applied Chemistry, 2005, 78(8): 1276-1279.
[5] Morsi I M, Barawy K A E, Morsi M B, Abdel-Gawad S R. Silicothermic reduction of dolomite ore under inert atmosphere [J]. Canadian Metallurgical Quarterly, 2002, 41: 15-28.
[6]  Silicothermic reduction process in magnesium production [J]. Journal of Thermal Analysisi and Calorimetry, 2008, 93: 411-415.
Silicothermic reduction process in magnesium production [J]. Journal of Thermal Analysisi and Calorimetry, 2008, 93: 411-415.
[7] Holywell G C. Magnesium: The first quarter millennium [J]. JOM, 2005, 57: 26-33.
[8] GAO Feng, NIE Zuo-ren, Wang Zhi-hong, Gong Xian-zheng, Zuo Tie-yong. Assessing environmental impact of magnesium production using Pidgeon process in China [J]. Transactions of Nonferrous Metals Society of China, 2008, 18: 749-754.
[9] Winand R, Gysel M V, Fontana A, Segers L, Carlier J C. Production of magnesium by vacuum carbothermic reduction of calcined dolomite [J]. Trans Inst Min Metall, 1990, 99: C105-C112.
[10] LI R T, PAN W, Sano M. Kinetics and mechanism of carbothermic reduction of magnesia [J]. Metall Mater Trans B, 2003, 34: 433-437.
[11] Feng Nai-xiang, Wang Yao-wu. A method of producing magnesium by vacuum thermal reduction using magnesite and dolomite as materials [J]. The Chinese Journal of Nonferrous Metals, 2011, 21(10): 2678-2686. (in Chinese)
[12] Hu Wen-xin, Feng Nai-xiang, Wang Yao-wu, Wang Zhi-hui. Magnesium production by vacuum aluminothemic reduction of a mixture of calcined dolomite and calcined magnesite [C]// Magnesium Technology of TMS, 2011: 43-47.
[13] Wang Yao-wu, Feng Nai-xiang, You Jing, Hu Wen-xin, Peng Jian-ping, DI Yue-zhong, Wang Zhi-hui. Study on extracting aluminum hydroxide from reduction slag of magnesium smelting by vacuum aluminothermic reduction [J]. Light Metal, 2011: 205-209.
[14] Yang J, Kuwabara M, Sawada T, Sano M. Kinetics of isothermal reduction of MgO with Al [J]. ISIJ Int, 2006, 46: 1130-1136.
[15] Lan H, OKUMURA K, Sano M. Nonisothermal gravimetric investigation on kinetics of reduction of magnesia by aluminum [J]. Metall Mater Trans B, 1999, 30: 1003-1008.
[16] Maeda M, Yahata T, Mitugi K, Ikeda T. Aluminothermic reduction of titanium oxide [J]. Mater Trans, 1993, 34: 599-603.
[17] Yang J, Kuwabara M, Liu Z Z, Asano T, Sano M. In situ observation of aluminothermic reduction of MgO with high temperature optical microscope [J]. ISIJ Int, 2006, 46: 202-209.
[18] Scian A N, López J M P, Pereira E. High alumina cements, study of CaO・Al2O3 formation, I. Stoichiometric mechanism [J]. Cement and Concrete Research, 1987, 17: 198-204.
[19] Liu Zhan-qi, Liu Jia-xiang, Jiang Bing, Qiu Qian. Process for preparing magnesium from dolomite by vacuum aluminothermic reduction [J]. Nonferrous Metals, 2010(2): 123. (in Chinese).
傅大学,冯乃祥,王耀武,彭建平,狄跃忠
东北大学 材料与冶金学院,沈阳 110819
摘 要:研究真空铝热还原煅烧白云石和煅烧菱镁石的混合物炼镁过程的动力学,提出一种满足真空铝热过程的等温还原方法。实验在4 Pa的真空下进行。实验结果表明:还原率随着温度、制团压力和铝粉添加量的提高而增加。对不同还原时间的还原渣进行XRD分析,结果表明,还原过程能够粗略地分为3个阶段:MgAl2O4和Ca12Al14O33的形成阶段;CaAl2O4的生成阶段;CaAl4O7的形成阶段。根据动力学模型将获得的实验数据分为3部分,这3部分的活化能分别为98.2、133.0和223.3 kJ/mol。
关键词:镁;动力学;铝热还原;菱镁石;白云石
(Edited by Hua YANG)
Foundation item: Project (MYF2011-34) supported by High-tech R&D Plan of Liaoning Province, China; Project (2011221002) supported by Industrial Research Projects of Liaoning Province, China; Project (N100302009) supported by the Fundamental Research Funds for the Central Universities, China
Corresponding author: Nai-xiang FENG; Tel: +86-24-83686463; E-mail: Fengnaixiang@163.com
DOI: 10.1016/S1003-6326(14)63133-2

