Trans. Nonferrous Met. Soc. China 23(2013) 1501-1505
Mechanism of debismuthizing with calcium and magnesium
Dian-kun LU, Zhe-nan JIN, Yong-feng CHANG, Shu-chen SUN
School of Materials and Metallurgy, Northeastern University, Shenyang 110819, China
Received 9 April 2012; accepted 2 July 2012
Abstract: In order to further understand the debismuthizing mechanism with calcium and magnesium, the influence of adding amount of calcium on the bismuth removal from lead was examined. A part of the debismuthizing dross samples were studied by electron probe microanalysis (EPMA). The results show that the calculated bismuthide governing debismuthizing process is Ca3Mg7Bi8, which may be a mixture of Ca3Bi2, CaBi, CaBi3 and Mg3Bi2. And the bismuthide formed during the debismuthizing process exists in two states: one is free bismuthide in the matrix of dross, and the other is symbiotic with Pb-Ca phase.
Key words: debismuthizing mechanism; Kroll-Betterton process; Ca3Mg7Bi8; Pb-Ca phase; Bi-Ca-Mg phase
1 Introduction
Most of the alkali and alkaline earth metals can form intermetallic compound with bismuth, and this sort of reactions were often used for the research work on the debismuthizing of lead bullion [1-3]. The Kroll- Betterton (K-B) process with calcium and magnesium reagents is the only practical process in operation for pyrometallurgical bismuth removal [4,5]. There still have been arguments on the mechanism of debismuthizing, and the arguments mostly focused on the compounds which act on bismuth removal.
On the mechanism of bismuth removal from lead by the K-B process, BETTERTON and LEBEDEFF [6] suggested that it resulted from the formation of both Ca3Bi2 and Mg3Bi2 compounds according to the microscopic study of the debismuthizing dross. While EVERS [7] believed that it resulted from the formation of CaMg2Bi2. The latter opinion has been accepted later by a majority of researchers [8-11]. According to this view, DAVEY [12] summarized the thermodynamic relationships that govern the removal of bismuth from lead by the K-B process. The removal of bismuth from lead is governed by the reaction equations (1) and (2):
2Mg+Ca+2Bi=Mg2CaBi2 (1)
Ksp=[Mg]2[Ca][Bi]2 (2)
where Ksp is the solubility product of the magnesium, calcium and bismuth in lead bullion, and it should be a constant at the same temperature. But the values of this solubility product obtained by many researchers changed in a very wide range [9-12]. The difference among solubility product values of CaMg2Bi2 obtained by different works indicates that there may be some unknown reasons affecting the debismuthizing results. Apart from these problems, the insufficiency of direct examination on the dross causes the debismuthizing mechanism lack of direct evidence to support.
This study focuses on ascertaining the mechanism governing debismuthizing with calcium and magnesium, especially on ascertaining the bismuthide and its occurence in debismuthizing dross. In addition to revealing debismuthizing mechanism, this research can also play a significant role in understanding the influence of bismuth on the performance of Pb-Ca alloy, which is a very important material in the storage battery industry.
2 Experimental
By addition of bismuth (99.99% purity) into a steel pot containing about 15 kg molten lead (99.99% purity) at 450 ��C, the lead-bismuth alloy (0.023% Bi) was obtained. Calcium was added as lead-calcium alloy (1.36% Ca) and magnesium (99.99% purity) as magnesium piece.
Five experiments were conducted in a sealed vertical tubular crucible electric furnace under the protection of a nitrogen atmosphere. For each test, 500 g of prepared Pb-Bi alloy was used, and the initial Mg concentration was held at 0.155%, the initial Ca concentration varied between 0.040% and 0.135%. The temperature for bismuth removal was set at 330 ��C, and the experimental procedure was the same as that in our previous work [13]. After the separation of lead bullion and dross, the alloys were weighed and sampled for ICP-AES analysis, and four of the dross samples were sent to electron microscopic and electron probe microanalysis. The results are shown in Table 1.
3 Results and discussion
The dross samples were taken from tests No. 2-5. The results show that all the samples are composed of matrix, Pb-Ca phase and Bi-Ca-Mg phase. The matrix and Pb-Ca phase account for about 60% and 30% of the dross, respectively. Bi-Ca-Mg phase is very little in the dross.
3.1 Analysis of matrix of dross
The composition of the matrix obtained by electron probe microanalysis is shown in Table 2. The backscattered electron image of the matrix and Pb-Ca phase is shown in Fig. 1, and the X-ray surface distribution images of Fe, Ca and Pb can be seen in Figs. 2(a-c), respectively.
Table 2 shows that the matrix is basically constituted by lead and iron. A large amount of iron in the matrix may come from the corrosion of stainless net by liquid lead alloy [14] during the separation of dross samples. Bi is enriched in matrix compared with the final lead (see Table 1). Through the average relative mass of Bi, Ca and Mg in matrix, the mass ratios of Ca and Mg to Bi were calculated as 0.243 and 0.234, respectively. They are all higher than 0.096 and 0.116, which are the mass ratios of Ca and Mg to Bi in CaMg2Bi2 compound, respectively. The surplus Ca and Mg may exist as Ca3Mg4Pbx [12].
In Fig. 1 it can be found that the distribution of Pb-Ca phase in the dross is uneven. This uneven distribution is strict in accordance with the surface distribution image of Ca (see Fig. 2(a)), which forms Pb-Ca phase. Pb-Ca phase has a relatively larger grain size, generally larger than 20 ��m, and takes the shape of round, strips or branch-like in the matrix. The boundary between the Pb-Ca phase and the matrix is smooth. Fe in the dross is also unevenly distributed (see Fig. 2(b), and is complementary with the distribution areas of Ca in the X-ray surface distribution images, which means that the Fe contaminated in the dross is corroded by lead bullion matrix, but not by Pb-Ca phase.
3.2 Analysis of Pb-Ca phase in dross
In contrast to the composition of matrix (see Table 2), the content of Ca in Pb-Ca phase (see Table 3) is well above that in the matrix. The mass ratio of Ca to Pb in Pb-Ca phase is about 5.71%, which is a little lower than that of compound CaPb3 (6.45%). So, there must be a little lead left in Pb-Ca phase under the present dross separation conditions, and Pb-Ca phase can be taken as a solid solution of CaPb3 and final lead alloy. Bi content in Pb-Ca phase is also higher than that in the matrix. But there is still some Bi in the matrix. This means that a part of bismuthide floats out of the lead before the formation of Pb-Ca phase during the falling of temperature. So, the compounds governing debismuthizing process consist of both free bismuthide and symbiotic bismuthide with Pb-Ca phase. Fe content in the Pb-Ca phase is well below that in the matrix. This also indicates that the stainless steel net is corroded by the final lead alloy, but not by Pb-Ca phase.

Fig. 1 Backscattered electron image of matrix and Pb-Ca phase (bright part)
Table 1 Initial conditions and analysis of final lead alloy in calcium dependent tests

Table 2 Composition of matrix in debismuthizing dross samples


Fig. 2 X-ray surface distribution images of Ca (a), Fe (b) and Pb (c) in dross (bright part)
Table 3 Composition of Pb-Ca phase in dross samples

3.3 Analysis of Bi-Ca-Mg phase in dross
The Bi-Ca-Mg phase generally has a little grain size, the majority of which is about 10 ��m, and only a little reaches to 20 ��m (see Fig. 3). The X-ray surface distribution images of Ca, Mg and Bi are very similar (see Figs. 4(a-c), which means that elements Bi, Ca and Mg are really enriched in the dross to form Bi-Ca-Mg compounds.

Fig. 3 Backscattered electron image of matrix and Bi-Ca-Mg phase
Apart from these mentioned above, it can be seen that this Bi-Ca-Mg phase is circulated by Pb-Ca phase (see Fig. 3). So, the Bi-Ca-Mg phase should be formed together with Pb-Ca phase. Then the reactivity of the Bi-Ca-Mg phase may decrease and the Bi content in final lead alloy also decreases.
We know that there is a peritectic point in Pb-Ca phase diagram. When the peritectic reaction occurs, a part of Pb-Ca phases (see Fig. 5) will be consumed and some Bi-Ca-Mg phases will be exposed to liquid lead. This will result in a bad debismuthizing effect due to the redissolution of some Bi-Ca-Mg phases. And it leads to the disturbance of Bi content in final lead according to the difference of Pb-Ca phase consumption (see Table 1). This disturbance will cause the change of the solubility product of the Bi-Ca-Mg compound.

Fig. 4 X-ray surface distribution images of Bi (a), Mg (b) and Ca (c) (bright part)
The composition of Bi-Ca-Mg phase is determined as 63.312% Bi, 1.900% Pb, 6.580% Mg, 4.580% Ca, 0.128% Fe, 23.280% O. A large amount of oxygen in this phase shows that the dross is significantly oxidized during the dross separation. If the contents of O, Pb and Fe are ignored, the compositions of the simplified Bi-Ca-Mg phase and CaMg2Bi2 compound can be compared (see Table 4).
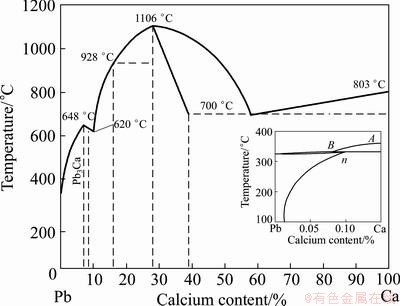
Fig. 5 Pb-Ca phase diagram and Pb-rich region [13]
Table 4 Compositions comparison between of Bi-Ca-Mg phase and CaMg2Bi2 compound

Table 4 shows that the composition of Bi-Ca-Mg phase deviates from the stoichiometric coefficient of CaMg2Bi2 compound. Molecular formula of Ca0.76Mg1.79Bi2 can be derived from the composition of simplified Bi-Ca-Mg phase. When the mole number in Ca0.76Mg1.79Bi2 is changed into round figure, Ca3Mg7Bi8 can be obtained. The total amount of Ca and Mg in Ca3Mg7Bi8 is not enough to maintain the ratio of Bi to Ca and Mg for both Ca3Bi2 and Mg3Bi2. This demonstrates that the compound governing debismuthizing process is not produced by the union of Ca3Bi2 and Mg3Bi2. So, we cannot conclude that CaMg2Bi2 forms during removing bismuth from lead with calcium and magnesium. If we intend to take Ca3Mg7Bi8 taken apart into three CaMg2Bi2, one MgBi2 will be left aside. But there is no MgBi2 compound in Bi-Mg system [15], so the Bi-Ca-Mg phase contains excessive Bi but lacks of Ca. Ca can form a series of compounds with Bi, such as Ca3Bi2, CaBi and CaBi3 [16]. This means that when the Ca content is not enough to form Ca3Bi2, the other two compounds, CaBi and CaBi3 may yield. So, we may view the calculated compound Ca3Mg7Bi8 as the combination of Ca3Bi2, CaBi and CaBi3 with Mg3Bi2. On the other hand, although there is only one compound in the Bi-Mg system, the phase diagram of Bi-Mg system shows that the composition of the compound has more Mg than Mg3Bi2 (see Fig. 6 [15]). It appears that the compound presents more like a solid solution of Mg3Bi2 and Mg. So, the calculated compound Ca3Mg7Bi8 should not be taken as a compound with fixed compositions, and can be taken as a solid solution of Ca3Bi2, CaBi, CaBi3, Mg3Bi2 and Mg, similar to Ca3Mg4Pbx in Pb-Ca-Mg system [12].

Fig. 6 Optimized phase diagram of Mg-Bi system [16]
4 Conclusions
1) The calculated bismuthide governing debismuthizing process is Ca3Mg7Bi8, which can be taken as a solid solution of Ca3Bi2, CaBi , CaBi3, Mg3Bi2 and Mg, similar to Ca3Mg4Pbx in Pb-Ca-Mg system.
2) The bismuthide formed during debismuthizing process exists in two states: one is free compound in the matrix of the dross, and the other is symbiotic bismuthide with the Pb-Ca phase.
3) When the peritectic reaction occurs, a part of the Pb-Ca phase will be consumed and a part of Bi-Ca-Mg phase dissolves into lead. This results in a bad debismuthizing effect and the disturbance of Bi content in final lead, according to the difference of Pb-Ca phase consumption.
References
[1] JOLLIVET L. Equilibria in systems rich in lead and containing alkali and alkaline earth metals [J]. Comptes Rendus, 1949, 228: 129.
[2] CHEN Guo-fa. Lead metallurgy [M]. Beijing: Metallurgy Industry Press, 2000: 126-127. (in Chinese)
[3] XIN H X, QIN X Y, ZHU X G, ZHANG J, KONG M G. Fabrication of nanocrystalline Mg3X2 (X=Bi, Sb) with supersaturated solid solubility by mechanical alloying [J]. Materials Science and Engineering B, 2006, 128: 192-200.
[4] CASTLE J F, RICHARDS J H. Lead refining: Current technology and a new continuous process [C]// DENHOLM W T. Advance in Extractive Metallurgy. London: The Institution of Mining and Metallurgy, 1977: 217-234.
[5] HIBBINS S G, CLOSSET B, BRAY M. Advances in the refining and alloying of low-bismuth lead [J]. Journal of Power Sources, 1995, 53: 75-83.
[6] BETTERTON J O, LEBEDEFF Y. Debismuthing lead with alkaline earth metals [J]. Trans AIME, 1936, 121: 205-225.
[7] EVERS D. Debismuthing by the Kroll-Betterton process [J]. Metallhuttenw, 1949, 2: 129-133.
[8] DAVEY T R A. Debismuthing of lead [J]. Journal of Metals, 1956(3): 341-350.
[9] ILEY J D, WARD D H. Development of a continuous process for the fine debismuthizing of lead [C]//DENHOLM W. Advance in Extractive Metallurgy. London: The Institution of Mining and Metallurgy, 1977: 133-139.
[10] HANCOCK P, HARRIS R. Solubility of calcium-magnesium- bismuth intermetallic in molten lead [J]. Candian Metallurgy Quarterly, 1991, 30: 275-291.
[11] LU Dian-kun, LIU Xue-shan, YE Guo-rui, HE Jia-qi. Thermodynamical analysis of debismuthizing mechanism with calcium and magnesium [J]. Journal of Shenyang Institute of Gold Technology, 1997, 16(2): 110-115. (in Chinese)
[12] DAVEY T R A. The physical chemistry of lead refining [C]// OBERBECKMANN F K, PONEN M. Lead-Zinc-Tin'80. TMS-AIME: Metallurgical Society of AIME, 1980: 477-506.
[13] LU Dian-kun, JIN Ze-nan, JIANG Kai-xi. Fine debismuthizing with calcium, magnesium and antimony [J]. Transactions of Nonferrous Metals Society of China, 2011, 21(10): 2311-2316.
[14] ZHANG J S. A review of steel corrosion by liquid lead and lead�Cbismuth [J]. Corrosion Science, 2009, 51: 1207-1227.
[15] MANAS P, JUNG I H. Thermodynamic modeling of the Mg-Bi and Mg-Sb binary systems and short-range-ordering behavior of the liquid solutions [J]. Computer Coupling of Phase Diagrams and Thermochemistry, 2009, 33: 744-754.
[16] NOTIN M, MEJBAR J, BOUHAJIB A, CHARLES J, HERTZ J. The thermodynamic properties of calcium intermetallic compounds [J]. Journal of Alloys and Compounds, 1995, 220: 62-75.
��þ����Ļ���
·�����������У������棬������
������ѧ ������ұ��ѧԺ������ 110819
ժ Ҫ��Ϊ�������˽��þ����Ļ������о��˸���������Ǧ�����ѳ���ȵ�Ӱ�졣�������������е���̽�����(EPMA)��������������Ƴ�����̵Ļ�������Ca3Mg7Bi8����Pb-Ca-Mg��ϵ�д��ڵ�Ca3Mg4Pbx���������ƣ� Ca3Mg7Bi8��������Կ�������Ca3Bi2��CaBi��CaBi3��Mg3Bi2�γɵĹ����塣ͬʱ���ڳ�����������γɵ�Ca3Mg7Bi8�����������ָ���״̬��һ���������黯��״̬����������Ļ����У���һ������Pb-Ca�����
�ؼ��ʣ��������������-���ض��ٷ���Ca3Mg7Bi8��Pb-Ca�ࣻBi-Ca-Mg��
(Edited by Xiang-qun LI)
Corresponding author: Dian-kun LU; Tel: +86-24-83681319; E-mail: ludk@smm.neu.edu.cn
DOI: 10.1016/S1003-6326(13)62622-9