
Synthesis of LiVPO4F with high electrochemical performance by sol-gel route
ZHONG Sheng-kui(钟胜奎)1, 2, CHEN Wei(陈 伟)1, LI Yan-hong(李艳红)3, 4,
ZOU Zheng-guang(邹正光)1, 2, LIU Chang-jiu(刘长久)1, 2
1. College of Chemistry and Bioengineering, Guilin University of Technology, Guilin 541004, China;
2. Key Laboratory of New Processing Technology for Nonferrous Metals and Materials, Ministry of Education,Guilin University of Technology, Guilin 541004, China;
3. Guangxi Key Laboratory of Environmental Engineering, Protection and Assessment,Guilin University of Technology, Gulin 541004, China
4. College of Environmental Science and Engineering, Guilin University of Technology, Guilin 541004, China
Received 6 July 2009; accepted 30 December 2009
_____________________________________________________________________________________________________
Abstract: LiVPO4F was synthesized by a novel sol-gel method under Ar atmosphere using V2O5・nH2O, LiF, NH4H2PO4, and citric acid as starting materials, and its physicochemical properties were characterized using X-ray diffractometry (XRD), SEM, and electrochemical methods. The XRD patterns show that LiVPO4F displays a triclinic structure with a space group of  . The SEM results indicate that the particle size of LiVPO4F is about 0.8 ?m together with homogenous distribution. The optimal sintering temperature and sintering time are 600 ℃ and 20 h, respectively, in order to obtain pure triclinic LiVPO4F with good electrochemical performance. LiVPO4F has the discharge capacity of 134 mA?h/g in the range of 3.0-4.4 V at the first cycle, and the discharge capacity remains 125 mA?h/g after 30 cycles. The sol-gel method is suitable for the preparation of LiVPO4F cathode materials with good electrochemical performance for lithium ion batteries.
. The SEM results indicate that the particle size of LiVPO4F is about 0.8 ?m together with homogenous distribution. The optimal sintering temperature and sintering time are 600 ℃ and 20 h, respectively, in order to obtain pure triclinic LiVPO4F with good electrochemical performance. LiVPO4F has the discharge capacity of 134 mA?h/g in the range of 3.0-4.4 V at the first cycle, and the discharge capacity remains 125 mA?h/g after 30 cycles. The sol-gel method is suitable for the preparation of LiVPO4F cathode materials with good electrochemical performance for lithium ion batteries.
Key words: lithium-ion batteries; LiVPO4F; cathode material; sol-gel method
_____________________________________________________________________________________________________
1 Introduction
Recently, phosphate-based compounds, such as Li3V2(PO4)3[1-3], LiVPO4F[4-10], LiFePO4[11-14] and LiMnPO4[15], have been proposed as a new class of cathode materials for lithium-ion batteries. Lithium vanadium fluorophosphate, LiVPO4F, is a highly promising cathode material and has attracted particular interests. LiVPO4F is isostructural with the naturally-occurring mineral tavorite, LiFePO4・OH or ambylgonite, LiAlPO4F, crystallizing with a triclinic structure (space group ). The reversible Li extraction/insertion reaction for Li1-xVPO4F, based on the V3+/4+ redox couple, operates at about 4.2 V versus Li+/Li. However, the conductivity of lithiated transition metal phosphates is not good. To improve the conductivity of lithiated transition metal phosphates, an effective way is to decrease the particle size by changing synthesis routes. The reduction of particle size may shorten the diffusion path of Li ion, and accordingly enhance the electrochemical performance. In this work, LiVPO4F was synthesized by a novel sol-gel method using citric acid as both reduction agent and carbon sources. The results showed that the particle size of LiVPO4F was decreased dramatically, and the electrochemical performance especially the reversibility is improved.
). The reversible Li extraction/insertion reaction for Li1-xVPO4F, based on the V3+/4+ redox couple, operates at about 4.2 V versus Li+/Li. However, the conductivity of lithiated transition metal phosphates is not good. To improve the conductivity of lithiated transition metal phosphates, an effective way is to decrease the particle size by changing synthesis routes. The reduction of particle size may shorten the diffusion path of Li ion, and accordingly enhance the electrochemical performance. In this work, LiVPO4F was synthesized by a novel sol-gel method using citric acid as both reduction agent and carbon sources. The results showed that the particle size of LiVPO4F was decreased dramatically, and the electrochemical performance especially the reversibility is improved.
2 Experimental
2.1 Synthesis of LiVPO4F
Firstly, V2O5・nH2O hydro-gel was prepared. V2O5 was melted in a crucible by heating at 700 ℃ for 2 h in the furnace, and then the molten V2O5 was poured into water and a red brown solution was formed in a stainless steel container. Product was obtained by keeping the solution at room temperature for 10 h. Secondly, citric acid of equivalent mole to V2O5 and stoichiometric amounts of NH4H2PO4 and LiF were added to the above V2O5・nH2O hydro-gels. The mixture was strongly stirred at room temperature for 2 h, and finally dried at 80 ℃ in an oven. The mixture was pressed into pellets and heated at 300 ℃ in a tubular furnace with flowing argon gas for 4 h to decompose and obtain precursor. The precursor was then ground again, pressed into pellets, heated at 600 ℃ and held for 20 h in flowing argon gas to obtain LiVPO4F sample.
2.2 Physical characterization
The powder X-ray diffractometry (XRD, Rint-2000, Rigaku) measurement using Cu Kα radiation was employed to identify the crystalline phase of the synthesized materials, and recorded at room temperature. The particle size and morphology of the LiVPO4F powders were observed by scanning electron microscope (JEOL, JSM-5600 LV) with an accelerating voltage of 20 kV. The carbon content of samples was determined by a carbon-sulfur analyser (Mlti EA2000).
2.3 Electrochemical test
The LiVPO4F electrodes were prepared by mixing with acetylene black, PVDF, and 1-methl-2-pyrrolidone in acetone to ensure homogeneity. The mass ratio of active material, acetylene black and PVDF in the electrodes was 80:10:10 except for special statement. The resulting slurry was pasted on an aluminium foil current collector with a diameter of 12 mm. After being dried in a vacuum oven at 120 ℃ for 24 h, the resulting electrodes with an active material loading of about 8 mg/cm2 were transferred to an Ar-filled glove box to assemble testing cells. The testing cells comprise Li3V2(PO4)3 cathode, lithium metal anode and Celguard-2300 separator placed between the cathode and anode. The electrolyte was 1 mol/L LiPF6 in a mixture of ethylene carbonate (EC) and dimethyl carbonate (DMC) with a volume ratio of 1?1. The cells were charged and discharged over a voltage range of 3.0-4.4 V versus Li+/Li electrode. The electrochemical impendence spectrum (EIS) measurements were carried out with a CHI 600A electrochemical analyzer. Electrochemical impendence spectrum (EIS) measurements were performed in the frequency range of 10 kHz-10 mHz with an AC voltage of 5 mV. The EIS experiments were performed in the three-electrode system using metallic foils as both counter and reference electrode.
3 Results and discussion
Fig.1 shows the XRD patterns of LiVPO4F synthesized at different temperatures. LiVPO4F with triclinic structure appears at sintering temperature of 600 ℃, which suggests that there is sufficient sintering for the samples prepared at 600 ℃. When the sintering temperature is higher than 700 ℃, the diffraction peaks of the samples become sharp. Moreover, the impurity phase Li3V2(PO4)3 can be observed and the diffraction intensities become strong with the increase of the sintering temperature.
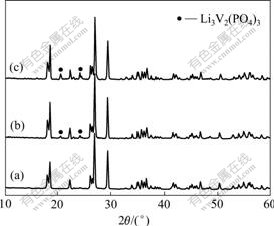
Fig.1 XRD patterns for LiVPO4F synthesized at different temperatures: (a) 600 ℃; (b) 700 ℃; (c) 800 ℃
The first charge-discharge curves of LiVPO4F samples prepared at various temperatures with 0.2C rate are shown in Fig.2. For these samples, the shape of charge and discharge curves is similar, while the initial charge and discharge capacities of LiVPO4F depend on sintering temperature. The sample synthesized at 600 ℃ exhibits the highest initial charge and discharge capacities of 140 and 134 mA?h/g, respectively. The columbic efficiency is about 95.71%.
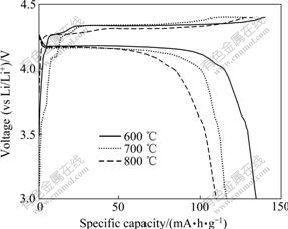
Fig.2 First charge-discharge curves of LiVPO4F synthesized at different temperatures
In order to explain why the first charge-discharge capacities of the sample depend strongly on sintering temperature, the SEM images are used to identify the morphologies of samples, as shown in Fig.3. When the samples were sintered at 700 ℃ and 800 ℃, the non- uniform particles with a smooth surface appear to be somewhat larger, which are agglomerated due to a higher sintering temperature and an abrupt decrease in capacity is observed.

Fig.3 SEM images of LiVPO4F synthesized at 600 ℃(a), 700 ℃(b), and 800 ℃(c)
Based on the above-mentioned results, it is necessary to study further the electrochemical properties of the composite prepared at 600 ℃. The cyclic performance of LiVPO4F synthesized at 600 ℃ is presented in Fig.4. It can be found that the LiVPO4F sample exhibits good rate capability with initial discharge capacities of 134 mA?h/g (0.2C), 132 mA?h/g (0.5C) and 127 mA?h/g (1C). At a rate of 0.2C, the discharge capacity reaches 125 mA?h/g after 30 cycles, which reaches the previously published result for the LiVPO4F samples prepared by two-step solid-state reactions[5]. When discharge rate increases to 1C, the discharge capacity still approaches to 111 mA?h/g after 30 cycles.
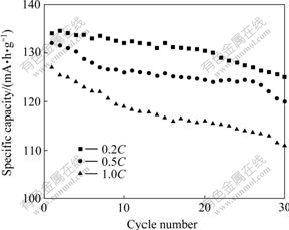
Fig.4 Discharge capacities vs cycle number at various rates between 3.0 and 4.4 V for LiVPO4F sample synthesized at 600 ℃
The electrochemical impedance spectra (EIS) of LiVPO4F electrodes materials were measured at different charging states. The typical Nyquist plots of EIS are presented in Fig.5 for three samples. Similar EIS patterns are observed for LiVPO4F samples. A semicircle is observed to center on the real axis in the higher frequency range. In the lower frequency range, a straight line with an angle of about 45? to the real axis corresponds to the Warburg impedance. The higher frequency semicircle is related to the charge-transfer resistance (Rct) and the double-layer capacitance. The lower frequency tails are resulted from the diffusion of lithium ions in the bulk active mass. In the case of LiVPO4F synthesized at 600 ℃, the diameter of the semicircle significantly depends on the potential during the charging, which indicates that the film formation process is dependent on the lithium ion content. On the other hand, the charge transfer resistance, Rct, shows a greater dependence on the lithium insertion and extraction levels. In the highly charged states, the sample is found to give lower Rct values. By comparing the diameter of the semicircle of the above two systems, it can be found that LiVPO4F synthesized at 600 ℃ shows lower Rct value than LiVPO4F synthesized at 700 ℃ or 800 ℃, which indicates that the proper synthesized temperature may increase the electronic conductivity and improve the Li+ kinetic behavior.
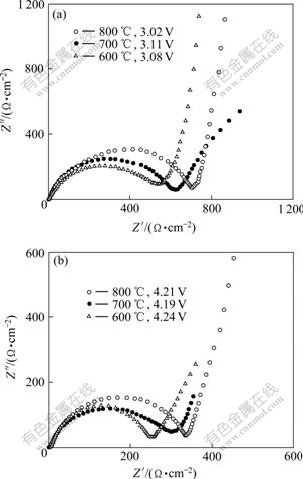
Fig.5 Nyquist plots for EIS of LiVPO4F synthesized at different temperatures at different charge states
4 Conclusions
1) LiVPO4F sample can be synthesized by a novel sol-gel method under Ar atmosphere when the sintering temperature is higher than 600 ℃. The electrochemical properties of LiVPO4F samples are seriously influenced by the sintering temperatures. Among the samples synthesized at various temperatures, the sample synthesized at 600 ℃ presents the highest initial capacity of 134 mA?h/g and a capacity of 125 mA?h/g after 30 cycles at 0.2C. At the discharge rate of 0.5C and 1C, the initial discharge capacities are 120 and 111 mA?h/g, respectively.
2) These results indicate that the sol-gel method is suitable for the preparation of LiVPO4F cathode materials with good electrochemical performance for lithium ion batteries.
References
[1] CHEN Q Q, WANG J M, TANG Z, HE W C, SHAO H B, ZHANG J Q. Electrochemical performance of the carbon coated Li3V2(PO4)3 cathode material synthesized by a sol-gel method [J]. Electrochim Acta, 2007, 52(16): 5251-5257.
[2] ZHONG S K, YIN Z L, WANG Z X, CHEN Q Y. Cathode material Li3V2(PO4)3: Low temperature solid-state reaction synthesis and performance [J]. Chinese Inorg Chem, 2006, 22(10): 1843-1846.
[3] LI Y Z, LIU X, YAN J. Study on synthesis routes and their influences on chemical and electrochemic performances of Li3V2(PO4)3/carbon [J]. Electrochim Acta, 2007, 53(2): 473-479.
[4] BARKER J, SAIDI M Y, SWOYER J L. Lithium metal fluorophosphates materials and preparation thereof: US patent 6 387[P]. 2002-05-14.
[5] ZHONG S K, YIN Z L, WANG Z X, CHEN Q Y. Synthesis and characterization of the triclinic structural LiVPO4F as possible 4.2 V cathode materials for lithium batteries [J]. Cent South Univ Techno, 2007, 14(3): 340-343.
[6] LI Y Z, ZHOU Z, GAO X P, YAN J. A novel sol-gel method to synthesize nanocrystalline LiVPO4F and its electrochemical Li intercalation performances [J]. J Power Sources, 2006, 160(1): 633-637.
[7] BARKER J, SAIDI M Y, SWOYER J L. Electrochemical insertion properties of the novel lithium vanadium fluorophosphates, LiVPO4F [J]. J Electrochem Soc, 2003, 150(10): A1394-A1398.
[8] BARKER J, SAIDI M Y, SWOYER J. A comparative investigate of the li insertion properties of the novel fluorophosphates phases, NaVPO4F and LiVPO4F [J]. J Electrochem Soc, 2004, 151(10): A1670-A1677.
[9] BARKER J, SAIDI M Y, GOVER R K B, BURNS P, BRYAN A. The effect of Al substitution on the lithium insertion properties of lithium vanadium fluorophosphate, LiVPO4F [J]. J Power Sources, 2007, 174(2): 927-931.
[10] BARKER J, GOVER R K B, BURNS P, BRYAN A, SAIDI M Y, SWOYER J L. Structural and electrochemical properties of lithium vanadium fluorophosphates, LiVPO4F [J]. J. Electrochem Soc, 2005, 152(9): A1776-A1779.
[11] CHEN J M, HSU C H, LI Y R, HSIAO M H. High-power LiFePO4 cathode materials with a continuous nano carbon network for lithium-ion batteries [J]. J Power Sources, 2008, 184(2): 498-502.
[12] JIN B, JIN E M, HEE K, GU H B. Electrochemical properties of LiFePO4-multiwalled carbon nanotubes composite cathode materials for lithium polymer battery [J]. Electrochem Commun, 2008, 10(10): 1537-1540.
[13] HIROSE K, HONMA T, DOI Y, HINATSU Y, KOMATSU T. M?ssbauer analysis of Fe ion state in lithium iron phosphate glasses and their glass-ceramics with olivine-type LiFePO4 crystals [J]. Solid State Commun, 2008, 146(5/6): 273-277.
[14] MI C H, ZHANG X G, ZHAO X B, LI H L. Effect of sintering time on the physical and electrochemical properties of LiFePO4/C composite cathodes [J]. J Alloys Compd, 2006, 424(1/2): 327-333.
[15] FANG H S, LI L P, LI G S. Hydrothermal synthesis of electrochemically active LiMnPO4 [J]. Chem Lett, 2007, 36(3): 436-437.
___________________________
Foundation item: Project(0991025) supported by the Natural Science Foundation of Guangxi Province, China; Project(GuiJiaoRen 2007-71) supported by the Research Fund of Key Laboratory of Environmental Engineering, Protection, and Assessment of Guangxi to Sponsor Teams for Innovation in the Construction of Talent Highlands in Guangxi Institutions of Higher Learning, China
Corresponding author: ZHONG Sheng-kui; Tel: +86-773-5896839; Fax: +86-773-5896839; E-mail: zhongshk@glite.edu.cn
(Edited by CHEN Ai-hua)