
Effects of microorganisms on surface properties of chalcopyrite and bioleaching
CHEN Ming-lian(陈明莲), ZHANG Lin(张 麟), GU Guo-hua(顾帼华),
HU Yue-hua(胡岳华), SU Li-jun(苏丽君)
School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China
Received 20 September 2008; accepted 5 November 2008
Abstract: The alteration of surface properties of chalcopyrite after biological conditioning with Acidithiobacillus ferrooxidans and Acidithiobacillus caldus was evaluated by Zeta-potential, adsorption studies, FT-IR spectra and contact angle measurement. The Zeta-potential studies show that the iso-electric point(IEP) of chalcopyrite after bacterial treatment moves towards the IEP of pure cells, indicating the adsorption of cells on chalcopyrite surface. The FT-IR spectra of chalcopyrite treated with bacterial cells show the presence of the cell functional groups signifying cells adsorption. Due to the formation of elemental sulfur and intermediate copper sulphides on chalcopyrite surface, the contact angle and surface hydrophobicity of chalcopyrite increase at the initial bioleaching stage. Chalcopyrite bioleaching by Acidithiobacillus ferrooxidans has higher copper extraction, which agrees with the fact that Acidithiobacillus ferrooxidans adsorbed on chalcopyrite surface is much more than Acidithiobacillus caldus. The results support the direct mechanism of sulfide oxidations in bioleaching chalcopyrite.
Key words: chalcopyrite; microorganism; bioleaching; adsorption; surface properties
1 Introduction
Bacterial adhesion to mineral surfaces plays an important role not only in bacterial survival in natural ecosystems, but also in mining industry applications. The bacteria can utilize various minerals, and bacterial adsorption to minerals is an initial step in bacterial leaching for metal recovery[1].
Chalcopyrite, CuFeS2, is the most important copper-bearing mineral in the world and unlike many other ores it is known to be recalcitrant to hydrometallurgical processing[2]. Researchers have strived for decades to accelerate the speed of chalcopyrite in the biological leaching. The selection of suitable microorganisms for leaching tests is one of the important factors[3]. Bacterial adhesion is dependent not only on the biochemical properties of the organism but also on the interfacial properties of the various interfaces existing in a bioleaching system[4]. It has been reported that the microorganism-mineral interaction results in changes in the surface properties of the microorganisms and mineral surface. The surface charge on cells grown in media with soluble iron (Fe2+) was different from that grown on a solid substrate (sulfur, pyrite). The altered cell surface charge was attributed to higher protein content in the latter[5]. The results of surface studies always are related to the bioflotation and bioflocculation of sulphide mineral. EDWARDS and RUTENBERG[6] concluded that small local surface alterations due to the bacterial metabolism could strongly affect the local adhesion parameters and result in bacterial adhesion on mineral surfaces. Sulphides can be separated from quartz through selective flocculation and dispersion after bio-treatment[7-9]. Bacterial adhesion changes the surface properties of pyrite in crushed fine coal from hydrophobic to hydrophilic[10]. The surface change makes it possible to remove pyrite from coal in flotation systems[11]. It has also been shown that conditioning minerals with Acidithiobacillus ferrooxidans (A. ferrooxidans), could induce either hydrophilicity or hydrophobicity, making the minerals either floatable or nonfloatable[12-13].
In this work, the alteration of surface properties of chalcopyrite after biological conditioning with Acidithiobacillus ferrooxidans and Acidithiobacillus caldus was studied, and the changes of surface properties caused by bacterial adsorption were discussed with reference to bioleaching behavior of chalcopyrite.
2 Materials and methods
2.1 Mineral
The sample of chalcopyrite used in this study is from Yushui Mine in Guangdong Province, China. Chemical analyses showed that the ore contained 31.36% Cu, 30.50% Fe, and 34.38% S. The fine copper ores with particle sizes of 0.045-0.074 mm were used for bioleaching experiments. The size less than 5 μm was used for zeta-potential, adsorption and FTIR studies. Pure solid chalcopyrite crystals were cut into certain thin sections, and then polished to the exposed faces. The mineral pieces washed with acetone were used to measure contact angle.
2.2 Microorganisms and culture media
A. ferrooxidans type strain (ATCC23270) and A. caldus (DQ256484) used in the experiments were conserved by the Key Lab of Biometallurgy in Central South University.
A. ferrooxidans and A. caldus were grown in 9K medium at an initial pH of 2.0, optimum temperature 30 ℃ and 45 ℃, respectively. The 9K medium compositions are (NH4)4SO4 3g/L, KCl 0.1 g/L, K2HPO4 0.5 g/L, MgSO4・7H2O 0.5 g/L, Ca(NO3)2 0.01 g/L, FeSO4・7H2O 44.7 g/L or S 10 g/L.
2.3 Zeta-potential measurements
Zeta-potential measurements were made with the DELSA440SⅡType electrokinetic instrument. The chalcopyrite particles had a concentration of 1 g/L with the ionic strength(I) of 10-2 mol/L maintained with NaCl. Chalcopyrite was interacted with cells by magnetic blender for 90 min and a cell concentration of 1×108 cells/mL was used. The bacteria soliquid was put into NaCl solution with ionic strength of 10-3 mol/L and mingled slightly. The cell density was about 2×108 cells/mL. Measurements were performed as a function of pH adjusted with HCl and NaOH.
2.4 Adsorption measurements
Tests were performed with 1 g mineral in 100 mL NaCl solution with the ionic strength of 10-2 mol/L and pH 2.0, containing initial cell concentration of 1×108 cells/mL. Chalcopyrite was interacted with cells for 90 min by conditioning the suspension using a magnetic stirrer. After conditioning, the suspension was filtered to separate the solids and the bacterial cells remaining in the liquid phase were estimated by microscope.
2.5 FTIR measurements
FTIR analyses were performed with Nicolet FTIR-740 instrument. The spectrum of A. ferrooxidans and A. caldus cells alone and chalcopyrite interacted with cells were recorded. The cell mass was collected, washed, dried in a vacuum drying incubator and used for measurements. Chalcopyrite was interacted with cells for 90 min using intensive stirring at pH=2. After conditioning, the suspension was filtered and mineral samples were washed with deionised water to remove the loosely holding cells and air-dried.
2.6 Contact angle measurements
The contact angle of the distilled water on the chalcopyrite surface was measured with the JJC-I wetting angle measurement instrument produced by the Changchun Optics Instrument Factory, China. All the measurements were operated at the room temperature (25 ℃).
2.7 Bioleaching experiments
Bioleaching tests were carried out in 250 mL flasks containing 90 mL 9K medium and 10 mL bacterial inoculum with the cell density of 1×108 cells/mL. The mineral concentration was 1%(w/v). The leaching solutions were periodically analyzed for copper concentration and the cell density everyday.
3 Results and discussion
3.1 Zeta-potential
The surface of a bacterial cell is charged due to the presence of functional groups such as carboxyl(―COOH), amino (―NH2) and hydroxyl (―OH), originating from the cell wall components of lipopolysaccharides, lipoprotein and bacterial surface proteins[14]. The Zeta-potentials of the different types of A. ferrooxidans and A. caldus as a function of pH are shown in Fig.1.
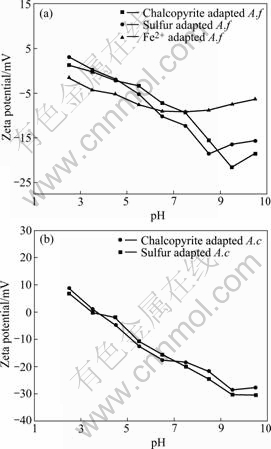
Fig.1 Zeta-potential of A.ferrooxidans (A.f) (a) and A caldus (A.c.) (b) at I=0.001 mol/L
A. ferrooxidans grown in ferrous exhibited an isoelectric point (IEP) at pH 2.0. However, A. ferrooxidans grown on elemental sulfur and chalcopyrite showed IEP around 3.3-3.5. The IEP is generally higher and typical for A. ferrooxidans grown on solid substrate cells with higher amount of EPS and proteins. A. caldus grown on sulfur and chalcopyrite have the similar IEP at pH=3.2-3.5 and beyond this pH the magnitude of negative potential increased with rise in pH.
The Zeta-potentials of chalcopyrite before and after interaction with bacterial cells are presented in Figs.2-3.
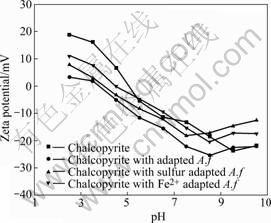
Fig.2 Zeta-potential of chalcopyrite before and after conditioning with A. ferrooxidans at I=0.01 mol/L
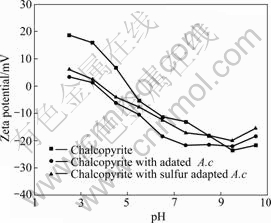
Fig.3 Zeta-potential of chalcopyrite before and after conditioning with A. caldus at I=0.01 mol/L
The IEP of chalcopyrite displayed at pH 5.1, after which interaction with cells moved towards the IEP of cells. In strong acidic pH medium, the Zeta-potential of chalcopyrite decreased and became negative. Beyond pH 5.5, the Zeta-potentials of chalcopyrite interacted with
unadapted cells are similar to pure chalcopyrite potentials. There is hardly any influence of cells on chalcopyrite potentials in the basic pH region. The effects of adapted cells on the Zeta-potential of chalcopyrite were more significant than that of unadapted cells. The observed difference in the electrokinetic behavior of chalcopyrite and cells consequent to their mutual interaction confirms that the
cells grown on different energy resources exhibit different abilities and affinities towards chalcopyrite.
The change of Zeta-potential of chalcopyrite after interaction with microorganisms may be related to the reactions for the oxidation of chalcopyrite as follows:
CuFeS2+2H++(1/2)O2 CuS+Fe2++H2O+S0 (1)
CuS+Fe2++H2O+S0 (1)
Fe2++H++(1/4)O2 Fe3++(1/2)H2O (2)
Fe3++(1/2)H2O (2)
CuS+Fe3++4H2O→Fe2++Cu2++8H++SO42- (3)
2S0+2H2O+3O2 H2SO4 (4)
H2SO4 (4)
Unequal dissolution of mineral surface ion by direct action of A. ferrooxidans and A. caldus on chalcopyrite resulted in the decrease of Zeta-potential. The direct biooxidation mechanism at the initial stage was considered as a main action in chalcopyrite bioleaching.
3.2 Adsorption researches
The adsorption curves of A. ferrooxidans and A. caldus on chalcopyrite are presented in Fig.4.
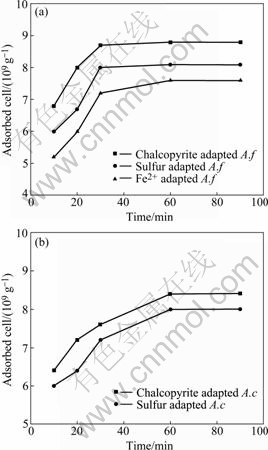
Fig.4 Adsorption curves of A. ferrooxidans (a) and A. caldus (b) on chalcopyrite
The kinetics of adsorption of A. ferrooxidans and A. caldus grown on different energy were similar to chalcopyrite respectively. And the adsorption equilibrium was attained in about 30 min for A. ferrooxidans, 60 min for A. caldus. The adsorption of A. ferrooxidans was faster than A. caldus cells onto chalcopyrite, while the A. ferrooxidans adhered to chalcopyrite was more than A. caldus under the same equilibrium concentration (1× 108 cells/mL). After 60 min, A. ferrooxidans and A. caldus cells adhered to chalcopyrite were about 88%, 84% of the initial cells, respectively. It also could be seen that, compared with unadapted bacterial cells, the adsorption of adapted bacterial cells was faster and higher. This explains the observed difference in the electrokinetic behaviour of chalcopyrite consequent to the interaction with the bacterial cells.
3.3 FTIR studies
The FTIR spectra of A. ferrooxidans and A. caldus (Fig.5) show several peaks for typical organic compounds such as carbohydrates CH3, CH2, CH, nitrogenous substances. The broad and strong band near 3 300 cm-1 represents asymmetric stretching of ―NH2 group. The band near 2 930 cm-1 characterizes ―CH stretching mode of CH2 group. The intense band near 1 650 cm-1 indicates the presence of an amide group (amide I band). The band at 1 540 cm-1 is attributed to NH bending of the secondary amide group ―CONH (amide II band). The bands near 1 450 cm-1 and 1 230 cm-1 are attributed to ―CH3 bending and ―CH3 wagging respectively. The band at 1 050 cm-1 is attributed to ―CH3 rocking and ―CH2 wagging modes. Comparing the spectrum of A. ferrooxidans with A. caldus, it was illustrated that the two type bacterium had similar surface chemical compositions.
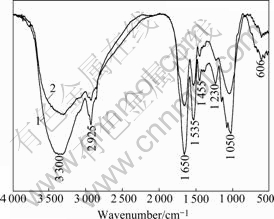
Fig.5 FTIR spectra of chalcopyrite-adapted A. ferrooxidans and A. caldus: 1―Chalcopyrite adapted A. f; 2―Chalcopyrite adapted A. c
Spectra of chalcopyrite interacted with A. ferrooxidans and A. caldus are shown in Fig.6. The changes on chalcopyrite interacted with A. ferrooxidans were more observable than those on chalcopyrite interacted with A.caldus. From the adsorption, A. ferrooxidans had stronger ability to adhere to chalcopyrite surface than A. caldus. Compared with the pure chalcopyrite spectra, some peaks become less intensive and some new small characteristic peaks from bacterial spectra appeared. This proved that the cells adsorbed on chalcopyrite surface.
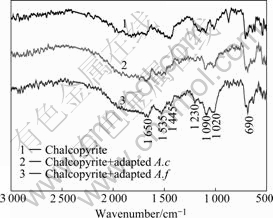
Fig.6 FTIR spectra of chalcopyrite interacted with adapted A. ferrooxidans and A. caldus
3.4 Contact angle measurements
Fig.7 shows the change of chalcopyrite contact angle interacted with different cells. The contact angle and surface hydrophobicity of chalcopyrite surface increased at initial bioleaching stage. This may be caused by the formation of elemental sulfur and intermediate copper sulphides on chalcopyrite surface (Reaction (1)). With the interaction time increasing, the contact angle of chalcopyrite surface decreased, which inferred the oxidation of elemental sulfur by the sulfur-oxidizing microorganisms (Reaction (4)). Previously published work also reported the initial chalcopyrite oxidation led to the formation of a passive layer of growing thickness, possibly elemental sulfur and bornite, which was further oxidized to nonstoichiometric copper sulphides[15-18] (Reactions (1), (3) and (4)).
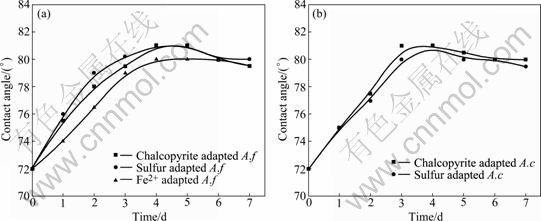
Fig.7 Changes of contact angle of chalcopyrite leached by A. ferrooxidans (a) and A. caldus (b) under different bioleaching conditions
The change of chalcopyrite contact angle implies that direct action at the initial stage is more important when bioleaching chalcopyrite with A. ferrooxidans and A. caldus.
3.5 Chalcopyrite bioleaching
Comparisons of chalcopyrite leaching by A. ferrooxidans and A. caldus grown on different energy sources are shown in Figs.8-9.
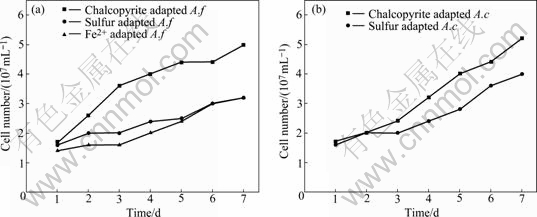
Fig.8 Growth curves of A. ferrooxidans (a) and A. caldus (b) in different bioleaching time
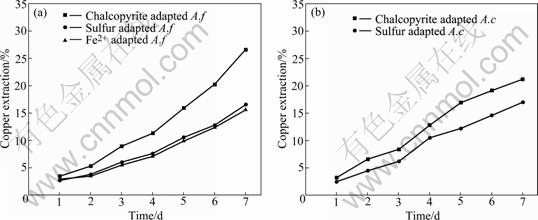
Fig.9 Copper extraction of chalcopyrite leached by A. ferrooxidans (a) and A. caldus (b)
The growth rate of A. ferrooxidans and A.caldus cells was increased during the leaching time, and both bacterium adapted by chalcopyrite grew faster than unadapted bacterium. After 7 d, the copper extraction by A. ferrooxidans and A. caldus on chalcopyrite reached 26.6% and 21.1%, respectively. The reason that A. ferrooxidans could oxidize iron and sulfur and the A. caldus just had the ability to oxidize sulfur could explain this phenomenon. However, at the initial bioleaching stage, the increasing trend of chalcopyrite leaching rate by A. ferrooxidan was nearly the same as that by A. caldus. This indicates that bacteria with the ability to oxidize sulfur play a dominant action. Linking to the results of surface properties, our studies support that the direct action is dominant at the initial stage of bioleaching.
4 Conclusions
1) The IEP of cells grown on solid substrate is generally higher for EPS and proteins with higher amount. Conditioning with bacterial cells, the IEP of chalcopyrite moves towards the IEP of pure cells indicating the adsorption of cells on chalcopyrite surface.
2) The changes on chalcopyrite FT-IR spectra conditioning with A. ferrooxidans are more observable than those with A. caldus. However, the higher amount of adsorbed A. ferrooxidans cells on chalcopyrite surface than A. caldus cells can explain this phenomenon.
3) Due to the formation of elemental sulfur and intermediate copper sulphides on chalcopyrite surface, the contact angle and surface hydrophobicity of chalcopyrite surface increase at the initial bioleaching stage.
4) The mesophilic A. ferrooxidans has the ability to oxidize ferrous ion and elemental sulfur has stronger leaching ability for chalcopyrite than moderately thermophilic A. caldus only oxidizing sulfur.
5) The direct action is dominant at the initial stage of bioleaching chalcopyrite. As the oxidation of sulfur by bacteria, both direct and indirect actions enhanced the metal dissolution in bioleaching process.
References
[1] Brierley C L. Microbiological mining [J]. Scientific American, 1982, 247(2): 42-53.
[2] Pradhan N, Nathasharma K C, Rao K S, Sukla L B, Mishra B K. Heap bioleaching of chalcopyrite: A review [J]. Minerals Engineering, 2008, 21(5): 355-365.
[3] Qiu Mu-qing, Xiong Shui-ying, Zhang Wei-min. Efficacy of chalcopyrite bioleaching using a pure and a mixed bacterium [J]. Journal of University of Science and Technology Beijing, 2006, 13(1): 7-10.
[4] Preston Devasia, Natarajan K A, Sathyanarayana D N, Ramananda Rao G. Surface chemistry of Thiobacillus ferrooxidans relevant to adhesion on mineral surfaces [J]. Applied and Environmental Microbiology, 1993, 59(12): 4051-4055.
[5] Sharma P K, Das A, Hanumantha Rao K, Forssberg K S E. Surface characterization of Acidithiobacillus ferrooxidans cells grown under different conditions [J]. Hydrometallurgy, 2003, 71(8): 285-292.
[6] Edwards K J, Rutenberg A D. Microbial response to surface microtopography: The role of metabolism in localized mineral dissolution [J]. Chemical Geology, 2001, 180(4): 19-32.
[7] Natarajan K A, Das A. Surface chemical studies on Acidithiobacillus group of bacteria with reference to mineral flocculation [J]. International Journal of Mineral Processing, 2003, 72(1): 189-198.
[8] Chandraprabha M N, Natarajan K A, Modak J M. Selective separation of pyrite and chalcopyrite by biomodulation [J]. Colloids and Surfaces B: Biointerfaces, 2004, 37(3): 93-100.
[9] Chandraprabha M N, Natarajan K A, Somasundaran P. Selective separation of arsenopyrite from pyrite by biomodulation in the presence of Acidithiobacillus ferrooxidans [J]. Journal of Colloid and Interface Science, 2004, 276(2): 323-332.
[10] Ohmura N, Kitamura K, Saild H. Mechanism of microbial flotation using Thiobacillus ferrooxidans for pyrite suppression [J]. Biotechnol Bioeng, 1993, 41(6): 671-676.
[11] Attia Y A. Feasibility of selective biomodification of pyrite floatability in coal desulfurization by froth flotation [J]. Resources Conservation and Recycling, 1990, 3(2): 169-175.
[12] Yelloji Rao M K, Natarajan K A, Somasundaran P. Effect of biotreatment with Thiobacillus ferrooxidans on the flotability of sphalerite and galena [J]. Miner Metallurg Processing, 1992, 9(4): 95-100.
[13] WATLING H R. The bioleaching of sulphide minerals with emphasis on copper sulphides―A review [J]. Hydrometallurgy, 2006, 84(1): 81-108.
[14] Sharma P K, Hanumantha Rao K. Adhension of Paenibacillus polymyxa on chalcopyrite and pyrite: Surface thermodynamics and extended DLVO theory [J]. Colloids and Surfaces B: Biointerfaces, 2003, 29(1): 21-38.
[15] Rodriguez Y, Ballester A, Blazquez M L, Gonzalez F. New information on the chalcopyrite bioleaching mechanism at low and high temperature [J]. Hydrometallurgy, 2003, 71(1): 47-56.
[16] LU Z Y, JEFFREY M I, LAWSON F. An electrochemical study of the effect of chloride ions on the dissolution of chalcopyrite in acidic solutions [J]. Hydrometallurgy, 2000, 56(2): 145-155.
[17] LU Yi-ping, JIANG Xiao-hui, FENG Qi-ming, OU Le-ming, ZHANG Guo-fan. Electrochemical behavior of chalcopyrite at normal temperature in acidic solution [J]. Trans Nonferrous Met Soc China, 2007, 17(3): 465-470.
[18] Schippers A, Sand W. Bacterial leaching of metal sulfides proceeds by the indirect mechanisms via thiosulfate or via polysulfides and sulfur [J]. Appl Environ Microbiol, 1999, 65(1): 319-321.
Foundation item: Project(2004CB619204) supported by the National Basic Research Program of China; Projects(50621063; 50674102) supported by the National Natural Science Foundation of China
Corresponding author: GU Guo-hua; Tel: +86-731-8830545; E-mail: guguohua@126.com
(Edited by LI Xiang-qun)