����Bi2O3/ZnFe2O4���������Ʊ�������������л�������
��Դ�ڿ����й���ɫ����ѧ��2021���5��
�������ߣ������� ������ ���� ������ ������ ���
����ҳ�룺1320 - 1330
�ؼ��ʣ����ʽBi2O3/ZnFe2O4����绯ѧ������
Key words��heterojunction; Bi2O3/ZnFe2O4; photoelectrochemical; degradation
ժ Ҫ�����û�ѧˮԡ���Ʊ�Bi2O3��Ĥ����ͨ���������Ϻ��˻���������渺��ZnFe2O4���и��ԣ�ͨ��XRD��SEM��Raman��XPS��UV-vis�ȶ�Bi2O3��Ʒ��Bi2O3/ZnFe2O4���ϱ�Ĥ���б��������Ʊ��ı�Ĥ��Ϊ��缫���о����绯ѧ���ܣ����״α������ڹ��������л���Ⱦ���е�Ӧ�á����������ZnFe2O4�ĸ����ؿ�Bi2O3�����ⷶΧ����߹�����ϵ�������ƹ�绯ѧ��Ӧ�����й��������ӵĸ��ϣ��Ӷ�ʹBi2O3/ZnFe2O4���ϱ�Ĥ��Na2SO4��Һ�еĹ���Ӧ�����ܶ��Ǵ�����ϵ�4�����ң�AM1.5�����µĹ�罵���Ǽ������������Bi2O3��������ߡ�
Abstract: Bi2O3 thin film was prepared by chemical bath deposition, and ZnFe2O4 was prepared by electrodeposition and post-annealing. The Bi2O3 and Bi2O3/ZnFe2O4 composite films were characterized by XRD, SEM, Raman, XPS and UV-vis. The photoelectrochemical performance of the prepared film as a photoelectrode and its application in photoelectrocatalytic degradation of organic pollutants was studied for the first time. The results show that the load of ZnFe2O4 enhances the absorption range of Bi2O3, increases the light absorption coefficient and inhibits the recombination of carriers at the interface between semiconductor and solution. The photocurrent density of Bi2O3/ZnFe2O4 composite film is 4 times of pure phase material in the Na2SO4 solution. The performance of methylene blue photoelectric degradation under AM1.5 illumination is significantly improved compared with pure Bi2O3.
DOI: 10.11817/j.ysxb.1004.0609.2021-36436
�����ƣ������� ���������ˣ������壬�� ��
(���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083)
ժ Ҫ�����û�ѧˮԡ���Ʊ�Bi2O3��Ĥ����ͨ���������Ϻ��˻���������渺��ZnFe2O4���и��ԣ�ͨ��XRD��SEM��Raman��XPS��UV-vis�ȶ�Bi2O3��Ʒ��Bi2O3/ZnFe2O4���ϱ�Ĥ���б��������Ʊ��ı�Ĥ��Ϊ��缫���о����绯ѧ���ܣ����״α������ڹ��������л���Ⱦ���е�Ӧ�á����������ZnFe2O4�ĸ����ؿ�Bi2O3�����ⷶΧ����߹�����ϵ�������ƹ�绯ѧ��Ӧ�����й��������ӵĸ��ϣ��Ӷ�ʹBi2O3/ZnFe2O4���ϱ�Ĥ��Na2SO4��Һ�еĹ���Ӧ�����ܶ��Ǵ�����ϵ�4�����ң�AM1.5�����µĹ�罵���Ǽ������������Bi2O3��������ߡ�
�ؼ��ʣ����ʽBi2O3/ZnFe2O4����绯ѧ������
���±�ţ�1004-0609(2021)-05-1320-10���� ��ͼ����ţ�TF80���� ���ױ�־�룺A
���ĸ�ʽ��������, ������, �� ��, ��. ����Bi2O3/ZnFe2O4���������Ʊ�������������л�������[J]. �й���ɫ����ѧ��, 2021, 31(5): 1320-1329. DOI: 10.11817/j.ysxb.1004.0609.2021-36436
WANG Zi-jin, LIU Fang-yang, JIA Ming, et al. Preparation and photoelectrochemical property of novel Bi2O3/ZnFe2O4 photoanode[J]. The Chinese Journal of Nonferrous Metals, 2021, 31(5): 1320-1329. DOI: 10.11817/j.ysxb.1004.0609.2021-36436
�������ķ�չ����ԴΣ���ͻ�����Ⱦ����Խ��Խ���أ�Ⱦ�ϡ�ɱ���������ۡ��л��ܼ��ȹ�ҵ��ˮ�е��л���Ⱦ���ձ���ж��Ժ��°��ԣ���������������Σ��[1-3]����θ�Ч�����������ܵ�ȥ����Щ�л����Ϊ��ע�Ľ��㡣�ڸ��ִ��������У��뵼�������������С������Ѻá��Լ���Ч�º͵��ص�����㷺�о���������̫���ֽܷ�ˮ�������������л���Ⱦ��ɶԻ����º͵Ķ�����̼��ˮ[4-6]�����ѿ������ڶ�뵼������У���ϵ����������Bi 6s��O 2p������ӻ�ЧӦʹ�������TiO2��ZnO�ȴ�ͳ�뵼����϶Կɼ�����Ӧ����[7]��Ȼ�����ڹ������ӿ�Ѩ�Ե����ظ��ϣ�����������Ĺ���Ч�ʽϵ͡�Ϊ�˽����һ���⣬�о�����ͨ������ϡ�н�����Ϊ�������ķ�ʽ�������ϵĹ�������[8-9]�����Ǵ�������(Pt��Au)�dz������������ʵͣ����Ѱ��һ�־��и߿ɼ�����Ӧ�������ܴ��ṹ�İ뵼�������Bi2O3�γ����ʽṹ�ͱ����Ϊ�ؼ�������������뵼������������������С���Թ�����÷�Χ���ؿ����ɼ��������ҳɱ��������ȶ��Ըߣ��Ի����Ѻã������Ĵ���ʹ����ڻ��յ��ص㣬�ڹ�������չ�ֳ������õ�Ӧ��ǰ��[10-15]��Ȼ����ĿǰΪֹ�й�Bi2O3/ZnFe2O4��Ĥ��缫���о���δ��������
����ͨ����ѧˮԡ���ϳ�Bi2O3���ף����õ������Ϻ��˻����Ʊ�����Bi2O3/ZnFe2O4��缫��ͨ���Ա�Bi2O3��Ʒ��Bi2O3/ZnFe2O4���ϵ缫�Ľṹ����̬����绯ѧ���ܷ��֣�ZnFe2O4�ĸ�����Ч�����˱�Ĥ������������ɼ�����Ӧ������Ϊ��������Bi2O3�γ������ʽṹ���������������ڲ�������ͱ���ĸ��ϣ�����˹������ӿ�Ѩ�Եķ���Ч�ʣ�ʵ���˲��Ϲ�绯ѧ���ܵ�������
1 ʵ��
1.1 �Լ�
ʵ�������Լ�������Bi(NO3)2��5H2O��ŨHNO3(��ҩ����ҩҵ�ɷ�����˾����)��Zn(CH3COO)2��2H2O��FeSO4��7H2O�����Ҵ���(TEOA)��NaOH(�Ϻ������������Ƽ��ɷ�����˾����)�������Լ���Ϊ��������ʵ����ˮΪȥ����ˮ��
1.1.1 Bi2O3�缫���Ʊ�
���ȶ�FTO��������ϴ����ͪ������ϴ15 min����ˮ������ϴ15 min����ˮ�Ҵ�������ϴ15 min��ȥ����ˮ(18.2 M��/cm)������ϴ15 min����������ɱ��á���ȡһ������Bi(NO3)2��5H2O�ܽ���ȥ����ˮ�У����Bi(NO3)2��Һ������������TEOA���������NaOH������Һ���ڷ�Ӧ��ϵ��pH��13���ң�������ྻ��FTO����������Һ�У�����60 ����ˮԡ��Ӧ120 min����Ʒ��ȥ����ˮ��ϴ�ɾ����ڿ�����350 �����˻�1 h����������Ϊ5 ��/min��Ȼ����Ȼ��ȴ�����¡�
1.1.2 Bi2O3/ZnFe2O4���ϵ缫���Ʊ�
��ȡһ������FeSO4�ܽ���ȥ����ˮ�У����FeSO4��Һ����Ԥ���Ʊ���Bi2O3Ϊ������ʯī�缫Ϊ���������ʹ��缫Ϊ�αȵ缫��FeSO4��Һ�г��������1 h�����������ڳ��������½��У�������ѹΪ1 V������20 mL 1 mol/L������п��Һ��ȡ0.5 mL����п��Һ�ε�������ı�Ĥ�ϣ�400 ���¿���1 h����������Ϊ5 ��/min����Ȼ��ȴ�����¡���Ĥ�˻�����1 mol/L��NaOH��Һ��1 h����ȥ��Ĥ��������ZnO�����Ĥ��ϴ�ɾ������ɱ��á�
1.2 ���ϵı�������⼼��
ͨ��X����������(XRD, Rigaku-TTRIII, Cu K�� (1.54056 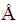 ), 250 mA, 40 kV)������Ʒ�ľ���ṹ��ͨ��������ɨ���������(SEM, Zeiss, MERLIN compact, 10 kV)������Ʒ����ò�ͽṹ��ͨ������-�ɼ��ֹ��ȼ�(UV-vis, Shimadzu)������Ʒ���������ܲ�ͨ��̨����(Veeco, Dektak150 Surface Profiler)���Ա�Ĥ�ĺ�Ȳ�������Ʒ�Ĺ�����ϵ�����������ȡ�ͨ��X���߹����������(XPS��Thermo fisher Scientific��K-Alpha+)������Ʒ��Ԫ����ɼ���Ϸ�ʽ���������̼�(��)�绯ѧ���ܲ��Ծ���PARSTAT4000�绯ѧ����վ�н��С�
), 250 mA, 40 kV)������Ʒ�ľ���ṹ��ͨ��������ɨ���������(SEM, Zeiss, MERLIN compact, 10 kV)������Ʒ����ò�ͽṹ��ͨ������-�ɼ��ֹ��ȼ�(UV-vis, Shimadzu)������Ʒ���������ܲ�ͨ��̨����(Veeco, Dektak150 Surface Profiler)���Ա�Ĥ�ĺ�Ȳ�������Ʒ�Ĺ�����ϵ�����������ȡ�ͨ��X���߹����������(XPS��Thermo fisher Scientific��K-Alpha+)������Ʒ��Ԫ����ɼ���Ϸ�ʽ���������̼�(��)�绯ѧ���ܲ��Ծ���PARSTAT4000�绯ѧ����վ�н��С�
1.3 Bi2O3��Bi2O3/ZnFe2O4�缫���������Ǽ���
��ȡһ�������Ǽ����ܽ���ȥ����ˮ�У�����Ǽ�����Һ����ȡһ������Na2SO4�ܽ�����Һ����Ϊ����ʡ��Ǽ�����ҺԤ���ڰ�̬�½���30 min�ﵽ���Ѹ�ƽ�⡣��Bi2O3�缫��Bi2O3/ZnFe2O4�缫Ϊ������ʯī�缫Ϊ���������ʹ��缫Ϊ�αȵ缫����AM1.5�����½����Ǽ����Ĺ�罵��ʵ�飬���ƫѹΪ1 V����Ӧÿ1 hȡһ������������ɼ��ֹ��ȼ��ϲⶨ627 nm�µ�����ֵ������Ч�������¹�ʽ��ʾ��
 (1)
(1)
ʽ�У���Ϊ�Ǽ���ȥ���ʣ�A0Ϊ�Ǽ�����Һ�ij�ʼ����ȣ�AtΪ��Ӧʱ��Ϊtʱ�Ǽ�����Һ������ȡ�
2 ���������
2.1 ��Ʒ����
2.1.1 XRD����
ͼ1��ʾΪBi2O3��Ĥ��Bi2O3/ZnFe2O4���ϱ�Ĥ��XRD�ס�����XRD��֪��Bi2O3����Ҫ�������PDF����Ƭ(JCPDS No. 65-1209)һ�¡�������2��Ϊ27.945�㡢31.754�㡢32.687�㡢46.215�㡢46.900��������ֱ��ӦBi2O3�����(201)��(002)��(220)��(222)��(400)���棬����ʵ��ɹ��ϳ��˴����ķ��ͦ�-Bi2O3[16-17]�����ϸ��Ժ������2��Ϊ29.919�㡢35.264�㡢42.844�㡢65.339������µ�����壬�ͱ���Ƭ(JCPDS No. 22-1012)��ƥ�䣬�ֱ��ӦZnFe2O4�����(220)��(311)��(400)��(531)����[18-19]��֤���������Ϻ��˻��ճɹ��ϳ���Bi2O3/ZnFe2O4���ϲ��ϣ����Ҹ��Ϻ�Bi2O3�������ǿ���������Ӽ���˵�����˻���ʹBi2O3���Ľᾧ�����ӡ�
ͼ1 Bi2O3��Ĥ��Bi2O3/ZnFe2O4���ϱ�Ĥ��XRD��
Fig. 1 XRD patterns of Bi2O3 film and Bi2O3/ZnFe2O4 composite film
2.1.2 XPS����
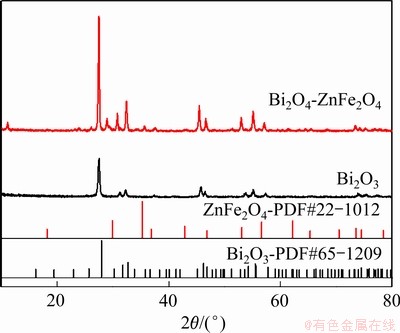
ͼ2��ʾΪBi2O3/ZnFe2O4���ϱ�Ĥ��XPSȫ��ͼ��Bi��Zn��Fe��O�ķַ���Ͻ������XPSȫ��(��ͼ2(a))��֪��Ʒ������Ҫ��Bi��O��Fe��Zn������Ԫ��(CΪ����У��Ԫ��)��ɡ�ͼ2(b)��ʾΪBi 4f�ĸ߷ֱ�XPSͼ�ף����ӽ����Ϊ164.1eV ��159.1eV �ķ��ӦBi 4f5/2��Bi 4f7/2�ĵ��ӣ�˵��Bi��Ҫ��+3�۵���ʽ����[8, 16, 20-21]��ͼ2(c)��ʾΪZn 2p�ĸ߷ֱ�XPSͼ�ף����ӽ����Ϊ1044.1 eV��1021.5 eV�ķ��ӦZn 2p1/2��Zn 2p3/2�ĵ��ӣ�˵��Zn��Ҫ��+2�۵���ʽ����[11]��ͼ2(d)��ʾΪFe 2p �ĸ߷ֱ�XPSͼ�ף�ͨ����Ͽ�֪�����ӽ������724.4 eV��710.6 eV���ķ���������ZnFe2O4��Fe 2p1/2��Fe 2p3/2һ�£����⣬732.5 eV��717.7 eV�����Ƿ�ȷ����Fe�ڸ��ϱ�Ĥ��Ϊ+3��[22-24]��ͼ2(e)ΪO 1s�ĸ߷ֱ�XPS�ף�Bi2O3/ZnFe2O4����O 1s�ķ�ϸ��ӣ����ַ���ϵ�����ֵ�ֱ�Ϊ531.5 eV��529.6 eV������531.5 eV�ķ�����ϱ����ȱ�ݡ���Ⱦ�����������йأ���������������������[25-26]��529.6 eV�ķ��Bi2O3��ZnFe2O4��O2-�ķ��Ӧ[27-29]�����У�Bi 4f5/2��Bi 4f7/2�ĵ��ӽ������NIST���ݿ���Bi2O3 (CAS Registry No: 1304-76-3)��Bi����Ⱦ����ָ�ƫ�ƣ�����������Bi2O3��ZnFe2O4֮�仯ѧ����ʹ��������������У�ʹBi�Ļ�ѧ̬�����˱仯����Թ����������ڽ���Ĵ�����ʮ��������[12]��
ͼ2 Bi2O3/ZnFe2O4��XPS��
Fig. 2 XPS spectra of Bi2O3/ZnFe2O4
2.1.3 SEM����
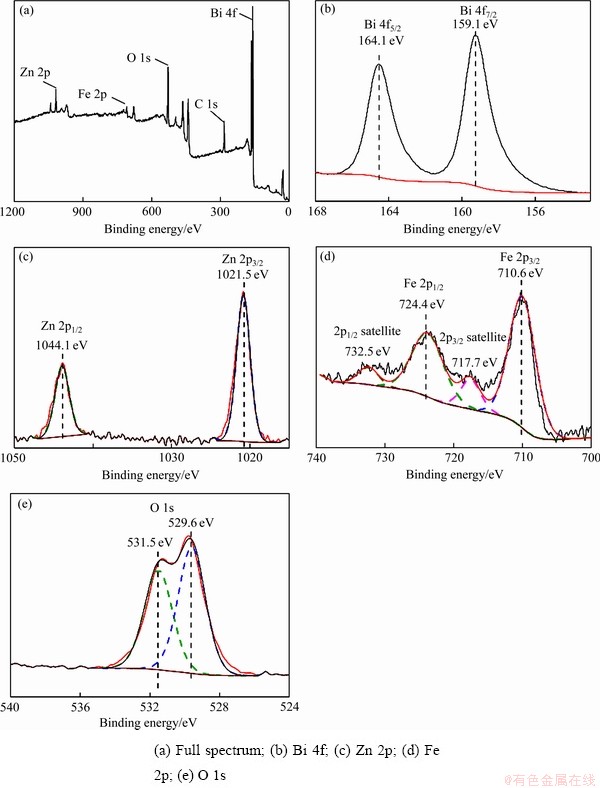
ͼ3(a)��(b)��ʾΪBi2O3��Bi2O3/ZnFe2O4���ϱ�Ĥ�ı�����òͼ��Bi2O3��Ƭ״������Լ2 ��m������ȽϹ⻬��Bi2O3/ZnFe2O4��Ĥ�Ƚϴֲڲ��ڱ��淢�ִ�������״���ʣ�ͼ3(c)~(e)��ʾΪBi2O3- ZnFe2O4��ĤBi��Fe��Zn����ɨ��ͼ�ף���Ĥ�ϼ���Fe��Zn�ķֲ������֮ǰXRD��XPS�IJ��Խ������ȷ��ZnFe2O4�������ȸ�����Bi2O3Ƭ�ϣ�ZnFe2O4�ĸ���ʹBi2O3�����ֲڣ��Ӷ�������Ĥ�ıȱ���������ϲ��ϵĴ�ȱ����Ϊ������Ӧ�ṩ�˸���Ļ���λ�㣬���̹������ӿ�Ѩ��Ǩ�ƾ��룬�Ӷ����Ʋ��ϵĹ����� ��[30-31]��
2.2 ���ϱ�Ĥ�Ĺ�������
2.2.1 ��������
ͼ3 Bi2O3��Ĥ��Bi2O3/ZnFe2O4���ϱ�Ĥ�ı�����ò�Լ�Bi��Fe��Z����ɨ��Ԫ�طֲ�
Fig. 3 Surface morphologies of Bi2O3 film(a) and Bi2O3/ZnFe2O4 composite film(b), and Bi(c), Fe(d), Zn(e) surface scan elements distribution of Fig. 3(b)
ͨ����������-�ɼ��ֹ���ױ���Bi2O3/ ZnFe2O4���ϱ�Ĥ�������ܣ���ͼ4(a)��ʾ��ZnFe2O4�ĸ���ʹ��Ĥ�Ĺ��������Խ��ͣ������˲��ϵĹ���������������˱�Ĥ�Ĺ�����ϵ�����ٽ��˲��϶Կɼ���IJ�����Kubelka�CMunk��ʽ����Bi2O3��ZnFe2O4�Ľ������ȣ�
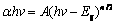 (2)
(2)
ʽ�У�hΪ���ʿ˳�����vΪ���ӵ�Ƶ�ʣ�Hz��EgΪ�뵼��Ľ������ȣ�eV��nΪ��������ͼ4(d)��ʾ��Bi2O3��ZnFe2O4��Ϊֱ�Ӵ�϶�뵼�壬Bi2O3�Ľ�������Ϊ2.29 eV��ZnFe2O4�Ľ�������Ϊ1.92 eV��������[33-34]������һ�¡�
2.2.2 ��绯ѧ����
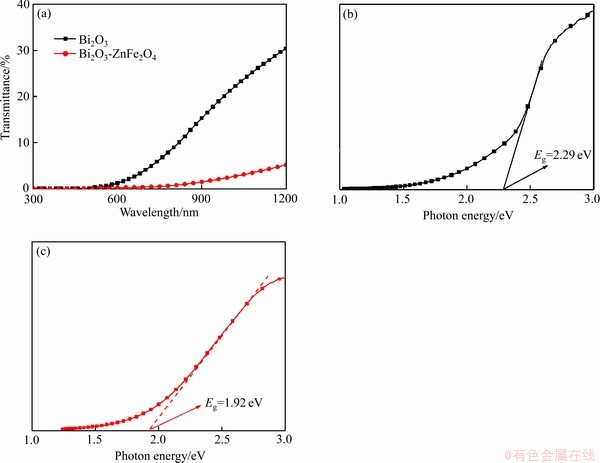
�����缫��ϵ�в��Բ��ϵĹ�绯ѧ���ܣ������Ϊ1 mol/L��Na2SO4��Һ�����Թ����У������ӱ�Ĥһ�����䡣ͼ5(a)��ʾΪBi2O3��Bi2O3/ ZnFe2O4��ն������µ�I-V���ߡ���AM1.5��Դ�������£�Bi2O3��Bi2O3/ZnFe2O4���й�����źš�����Bi2O3/ZnFe2O4���ϱ�Ĥ�������Լ4 ��A/cm2 (1.2V (vs RHE))����Bi2O3��4�����ҡ���Bi2O3��ȣ�Bi2O3/ZnFe2O4���ϱ�Ĥ�ġ���塱�����������ͣ�����塱���������ڹ�������-��Ѩ�Ը��ϻ������������ֵĸ���Ӧ�����[32]����˵��ZnFe2O4�ĸ��ؼ����˹��������ӵĸ��ϡ�ͨ������Bi2O3��Bi2O3/ZnFe2O4�ĵ绯ѧ�迹��(��ͼ5(b))���֣�Bi2O3/ZnFe2O4���ϱ�Ĥ���и�С��NyquistԲ���뾶��˵�������������ڴ���Ĺ������迹��С[2, 19, 35]���Ӳ���֤ʵ��Bi2O3��ZnFe2O4֮�����ʽ���γɡ����Ϻ�Ĥ�İ�����������С��˵����Ĥ���ȶ���������ߣ���XRD��֪��������븴�ϱ�Ĥ��Bi2O3�ᾧ�Ը����йء�ͨ������Bi2O3��ZnFe2O4��Ī��-Ф�ػ�����(�� ͼ5(c)��(d))��֪��Bi2O3��ZnFe2O4��ΪN�Ͱ뵼��[36-37]��ͨ����ֱ�߲������ӵ�X����ȡ�ؾ��Bi2O3��ƽ����λΪ0.58 V (vs RHE)������ZnFe2O4 (0.25 V (vs RHE))�����������У������N�Ͱ뵼��ĵ�����λԼ��ƽ����λ����0.2 V[38-40]��Bi2O3��ZnFe2O4�ĵ�����λԼΪ0.38 V��0.05 V (vs RHE)�����UV-vis����Ľ������ȣ�Bi2O3��ZnFe2O4�ļ۴���λ����Ϊ2.67 V��1.97 V (vs RHE)��Bi2O3�ĵ����ͼ۴�λ�þ�����ZnFe2O4�ĵ����ͼ۴�λ�ã�����type-�������ʽṹ���γ�����[41-43]�������£�����-��Ѩ�Ա����������Խ��糡�����������෴�����ƶ����ɹ������˹��������ӵĸ��ϣ��Ӷ�ʹ���ϱ�Ĥ�Ĺ����������ߡ�
ͼ4 Bi2O3��Bi2O3/ZnFe2O4��UV-vis�������Լ�Bi2O3��ZnFe2O4�Ĵ�϶������
Fig. 4 UV-vis transmittance spectra of Bi2O3 and Bi2O3/ZnFe2O4(a), band gap energy curves of Bi2O3(b) and ZnFe2O4 (c)
ͼ5 Bi2O3��Bi2O3/ZnFe2O4�Ĺ����Ӧ���ߡ�Bi2O3��Bi2O3/ZnFe2O4�Ľ����迹ͼ�Լ�Bi2O3��ZnFe2O4��Ī��-Ф�ػ�����
Fig. 5 Photoelectric response curves of Bi2O3 and Bi2O3/ZnFe2O4(a), AC impedance diagram of Bi2O3 and Bi2O3/ZnFe2O4(b) and Mott-schottky curve of Bi2O3(c) and ZnFe2O4(d)
2.2.3 ���������Ǽ���
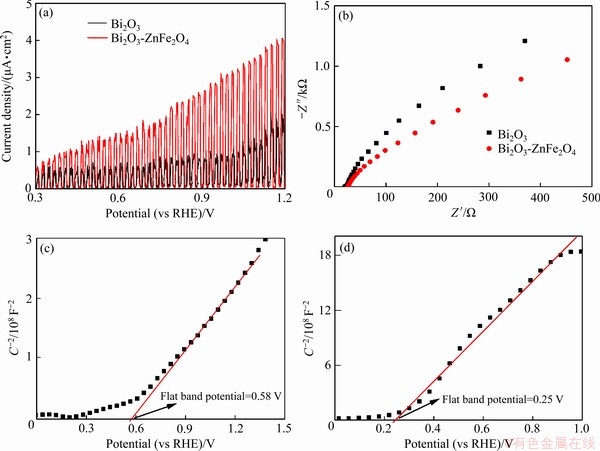
ʵ�����Ǽ���ΪĿ�꽵�����Bi2O3/ ZnFe2O4�缫�Ĺ����������ܣ�ͼ6(a)��(b)��ʾ�ֱ�ΪBi2O3(a)��Bi2O3/ZnFe2O4(b)�缫��罵��������Ǽ���������-�ɼ����չ��ף�627 nm�����շ�Ϊ�Ǽ����������壬595 nm���ļ��Ϊ�Ǽ����Ķ�����[44]������Bi2O3��Bi2O3/ ZnFe2O4�缫��罵���Ǽ�����ȥ������ʱ��仯����(��ͼ6(d))��֪��Bi2O3�缫����4 h���Ǽ���ȥ����Ϊ30.4%����Һ���Ǽ����ܶ�����ƽ�ȣ����϶��Ǽ����Ľ���Ч���ﵽ���͡�Bi2O3/ ZnFe2O4�缫��4 h���Ǽ���ȥ���ʴﵽ42.9%����Bi2O3�缫������������ͬʱ�Ǽ�����Ũ�������Ա����½����ƣ���ˣ��Ż���绯ѧ�صĽṹ����ַ��ӹ�绯ѧ�ص������Ǻ����о���Ҫ��������⡣�ɹ�罵�������Bi2O3��Bi2O3/ZnFe2O4�缫�ĵ���-ʱ������(��ͼ6(c))��֪����������������У�Bi2O3/ZnFe2O4�缫���Bi2O3���и� ��Ĺ������������������������ܺ�绯ѧ����һ�£���ζ������и�ǿ�Ĺ�罵����������罵��ʵ�����ٴ�֤����ZnFe2O4�ĸ��ϸ�����Bi2O3�Ĺ��ת��Ч�ʣ������˲��ϵĹ������ܡ�
ͼ6 �Ǽ�������-�ɼ����չ�����Bi2O3��Bi2O3/ZnFe2O4�缫��罵������ʱ��ı仯���ߡ���罵�������Bi2O3��Bi2O3/ZnFe2O4�缫�ĵ���-ʱ�������Լ�Bi2O3��Bi2O3/ZnFe2O4�缫��罵���Ǽ�����ȥ������ʱ��仯����
Fig. 6 UV-visible absorption spectra of methylene blue during photodegradation of Bi2O3(a) and Bi2O3/ZnFe2O4(b) electrodes, current-time curve of Bi2O3 and Bi2O3/ZnFe2O4 electrodes during photodegradation(c) and removal rate of methylene blue photodegradation by Bi2O3 and Bi2O3/ZnFe2O4 electrodes varies with time(d)
3 ����
1) ���û�ѧˮԡ���ϳ���Bi2O3���ף�Ȼ��ͨ���������Ϻ��˻����Ʊ�Bi2O3/ZnFe2O4���ϱ�Ĥ��ͨ��UV-vis��EIS��M-S�ȱ����ֶ�֤��ZnFe2O4�ĸ��ϳɹ�������Bi2O3��Ĥ�Ĺ������ܡ�Bi2O3/ZnFe2O4�缫��AM1.5��Դ������4 h���Ǽ���ȥ���ʴﵽ42.9%����罵�����ܽ�Bi2O3�缫���������������Ǽ���Ũ�ȱ����½����ơ�
2) խ��϶ZnFe2O4�ĸ��شٽ���Bi2O3�Կɼ���IJ���ʹ���ϵ����մ��ߺ��ƣ�����˱�Ĥ�Ĺ�����Ч�ʡ�
3) Bi2O3��ZnFe2O4�ĸ����γ���type-�������ʽṹ�������˹������ӿ�Ѩ�Եĸ��ϣ�ʵ���˸��ܶȹ����������ɷ��롣��Щ���ڹ��������л���Ⱦ��Ч�ʵ���߾�����Ҫ���塣
REFERENCES
[1] LUO Ju-hua, YAN Zhu, LIU Rong-qi, et al. Synthesis and excellent visible light photocatalysis performance of magnetic reduced graphene oxide/ZnO/ZnFe2O4 composites[J]. RSC Adv, 2017, 7: 23246-23254.
[2] YANG Jin, CHEN Dai-mei, ZHU Yi, et al. 3D-3D porous Bi2WO6/graphene hydrogel composite with excellent synergistic effect of adsorption-enrichment and photocatalytic degradation[J]. Applied Catalysis B: Environmental, 2017, 205: 228-237.
[3] ZHANG Peng, YANG Xiao-yan, ZHAO Zong-bin, et al. One-step synthesis of flowerlike C/Fe2O3 nanosheet assembly with superior adsorption capacity and visible light photocatalytic performance for dye removal[J]. Carbon, 2017, 116: 59-67.
YANG Heng-yan, WANG Ping, WANG Ding, et al. New understanding on photocatalytic mechanism of nitrogen- doped graphene quantum dots-decorated BiVO4 nanojunction photocatalysts[J]. ACS Omega, 2017, 2: 3766-3773.
[4] LEE K T, CHUAH X F, CHENG Y C, et al. Pt coupled ZnFe2O4 nanocrystals as a breakthrough photocatalyst for Fenton-like processes-photodegradation treatments from hours to seconds[J]. J Mater Chem A, 2015, 3: 18578-18585.
[5] YAO Yun-jin, CAI Yun-mu, LU Fang, et al. Magnetic ZnFe2O4-C3N4 hybrid for photocatalytic degradation of aqueous organic pollutants by visible light[J]. Industrial & Engineering Chemistry Research, 2014, 53: 17294-17302.
[6] SAISON T, CHEMIN N, CHANEAC C, et al. Bi2O3, BiVO4, and Bi2WO 6: Impact of surface properties on photocatalytic activity under visible light[J]. The Journal of Physical Chemistry C, 2011, 115: 5657-5666.
[7] HSIEH S H, LEE G J, CHEN C Y, et al. Synthesis of Pt doped Bi2O3/RuO2 photocatalysts for hydrogen production from water splitting using visible light[J]. Journal of Nanoscience and Nanotechnology, 2012, 12: 5930-5936.
[8] MONIZ S J A, BHACHU D, BLACKMAN C S, et al. A novel route to Pt-Bi2O3 composite thin films and their application in photo-reduction of water[J]. Inorganica Chimica Acta, 2012, 380: 328-335.
[9] UPRETI A R, LI Y, KHADGI N, et al. Efficient visible light photocatalytic degradation of 17��-ethinyl estradiol by a multifunctional Ag-AgCl/ZnFe2O4 magnetic nanocomposite[J]. RSC Adv, 2016, 6: 32761-32769.
[10] NGUYEN T B, DOONG R A. Fabrication of highly visible-light-responsive ZnFe2O4/TiO2 heterostructures for the enhanced photocatalytic degradation of organic dyes[J]. RSC Adv, 2016, 6: 103428-103437.
[11] ZHANG Fei, LI Xin-yong, ZHAO Qi-dong, et al. Rational design of ZnFe2O4/In2O3 nanoheterostructures: Efficient photocatalyst for gaseous 1,2-dichlorobenzene degradation and mechanistic insight[J]. ACS Sustainable Chemistry & Engineering, 2016, 4: 4554-4562.
[12] DHAL J P, MISHRA B G, HOTA G. Hydrothermal synthesis and enhanced photocatalytic activity of ternary Fe2O3/ ZnFe2O4/ZnO nanocomposite through cascade electron transfer[J]. RSC Adv, 2015, 5: 58072-58083.
[13] KIM T W, CHOI K S. Improving stability and photoelectrochemical performance of BiVO4 photoanodes in basic media by adding a ZnFe2O4 layer[J]. The Journal of Physical Chemistry Letters, 2016, 7: 447-451.
[14] GUO Pei-zhi, CUI Li-jun, WANG Yi-qian, et al. Facile synthesis of ZnFe2O4 nanoparticles with tunable magnetic and sensing properties[J]. Langmuir, 2013, 29: 8997-9003.
[15] BREZESINSKI K, OSTERMANN R, HARTMANN P, et al. Exceptional photocatalytic activity of ordered mesoporous ��-Bi2O3 thin films and electrospun nanofiber mats[J]. Chemistry of Materials, 2010, 22: 3079-3085.
[16] XIAO Xin, HU Rui-ping, LIU Chao, et al. Facile large-scale synthesis of ��-Bi2O3 nanospheres as a highly efficient photocatalyst for the degradation of acetaminophen under visible light irradiation[J]. Applied Catalysis B: Environmental, 2013, 140/141: 433-443.
[17] HAN Li-jun, ZHOU Xue, WAN Li-na, et al. Synthesis of ZnFe2O4 nanoplates by succinic acid-assisted hydrothermal route and their photocatalytic degradation of rhodamine B under visible light[J]. Journal of Environmental Chemical Engineering, 2014, 2: 123-130.
[18] HELAILI N, MITRAN G, POPESCU I, et al. Photoelectrochemical properties of AFe2O4 (A=Co,Cu,Zn) ferrospinels for water photo-reduction[J]. Journal of Electroanalytical Chemistry, 2015, 742: 47-53.
[19] GE Ming, LI Ya-fei, LIU Lu, et al. Bi2O3-Bi2WO6 composite microspheres: Hydrothermal synthesis and photocatalytic performances[J]. The Journal of Physical Chemistry C, 2011, 115: 5220-5225.
[20] BALACHANDRAN S, SWAMINATHAN M. Facile fabrication of heterostructured Bi2O3-ZnO photocatalyst and its enhanced photocatalytic activity[J]. The Journal of Physical Chemistry C, 2012, 116: 26306-26312.
[21] ZHAO Di, XIAO Ying, WANG Xia, et al. Ultra-high lithium storage capacity achieved by porous ZnFe2O4/��-Fe2O3 micro-octahedrons[J]. Nano Energy, 2014, 7: 124-133.
[22] SHARMA Y, SHARMA N, RAO G. V S, et al. Li-storage and cyclability of urea combustion derived ZnFe2O4 as anode for Li-ion batteries[J]. Electrochimica Acta, 2008, 53: 2380-2385.
[23] HOU Yang, LI Xin-yong, ZHAO Qi-dong, et al. Electrochemical method for synthesis of a ZnFe2O4/TiO2 composite nanotube array modified electrode with enhanced photoelectrochemical activity[J]. Advanced Functional Materials, 2010, 20: 2165-2174.
[24] WANG Yan, FENG Cai-xia, ZHANG Min, et al. Enhanced visible light photocatalytic activity of N-doped TiO2 in relation to single-electron-trapped oxygen vacancy and doped-nitrogen[J]. Applied Catalysis B: Environmental, 2010, 100: 84-90.
[25] ZHAO Xin, YANG Qiu-hua, CUI Jin-jin. XPS study of surface absorbed oxygen of ABO3 mixed oxides[J]. Journal of Rare Earths, 2008, 26: 511-514.
[26] MCINTYRE N S, COOK M G. X-ray photoelectron studies on some oxides and hydroxides of cobalt, nickel, and copper[J]. Analytical Chemistry, 2002, 47: 2208-2213.
[27] GAO Ying-jun, YIN Ling-hong, KIM Su Jae, et al. Enhanced lithium storage by ZnFe2O4 nanofibers as anode materials for lithium-ion battery[J]. Electrochimica Acta, 2019, 296: 565-574.
[28] UCHIDA K A A. Dynamic XPS measurements on bismuth molybdate surfaces[J]. Surface Science, 1996, 357: 170-175.
[29] SONG Hui, ZHU Li-ping, LI Ya-guang, et al. Preparation of ZnFe2O4 nanostructures and highly efficient visible-light- driven hydrogen generation with the assistance of nanoheterostructures[J]. J Mater Chem A, 2015, 3: 8353-8360.
[30] CAO Jun-yu, XING Juan-juan, ZHANG Yuan-jian, et al. Photoelectrochemical properties of nanomultiple CaFe2O4/ZnFe2O4 pn junction photoelectrodes[J]. Langmuir, 2013, 29: 3116-3124.
[31] BERGLUND S P, ABDI F F, BOGDANOFF P, et al. Comprehensive evaluation of CuBi2O4 as a photocathode material for photoelectrochemical water splitting[J]. Chemistry of Materials, 2016, 28: 4231-4242.
[32] XIE Dong, SU Qing-mei, ZHANG Jun, et al. Graphite oxide-assisted sonochemical preparation of ��-Bi2O3 nanosheets and their high-efficiency visible light photocatalytic activity[J]. Journal of Materials Science, 2013, 49: 218-224.
[33] ZOU Lei, WANG Hao-ran, YUAN Guo-liang, et al. Magnetically separable CdS/ZnFe2O4 composites with highly efficient photocatalytic activity and photostability under visible light[J]. ACS Applied Nano Materials, 2018, 1: 831-838.
[34] HE Ron-gan, CHENG Kai-yu, WEI Zhe-ying, et al. Room-temperature in situ fabrication and enhanced photocatalytic activity of direct Z-scheme BiOI/g-C3N4 photocatalyst[J]. Applied Surface Science, 2019, 465: 964-972.
[35] KANDI D, MARTHA S, THIRUMURUGAN A, et al. Modification of BiOI microplates with CdS QDs for enhancing stability, optical property, electronic behavior toward rhodamine B decolorization, and photocatalytic hydrogen evolution[J]. The Journal of Physical Chemistry C, 2017, 121: 4834-4849.
[36] VELDURTHI N K, ESWAR N K, SINGH S A, et al. Heterojunction ZnWO4/ZnFe2O4 composites with concerted effects and integrated properties for enhanced photocatalytic hydrogen evolution[J]. Catalysis Science & Technology, 2018, 8: 1083-1093.
[37] REDDY D A, PARK H, HONG S, et al. Hydrazine-assisted formation of ultrathin MoS2 nanosheets for enhancing their co-catalytic activity in photocatalytic hydrogen evolution[J]. Journal of Materials Chemistry A, 2017, 5: 6981-6991.
[38] LIU Xin, JIN Ai-ling, JIA Yu-shuai, et al. Synergy of adsorption and visible-light photocatalytic degradation of methylene blue by a bifunctional Z-scheme heterojunction of WO3/g-C3N4[J]. Applied Surface Science, 2017, 405: 359-371.
[39] ZENG Chao, HUANG Hong-wei, ZHANG Tie-rui, et al. Fabrication of heterogeneous-phase solid-solution promoting band structure and charge separation for enhancing photocatalytic CO2 reduction: A case of ZnxCa1-xIn2S4[J], ACS Appl Mater Interfaces, 2017, 9: 27773-27783.
[40] HUANG Zhen-feng, ZOU Ji-jun, PAN Lun, et al. Synergetic promotion on photoactivity and stability of W18O49/TiO2 hybrid[J], Applied Catalysis B: Environmental, 2014, 147: 167-174.
[41] HUANG Zhen-feng, SONG Jia-jia, WANG Xin, et al. Switching charge transfer of C3N4/W18O49 from type-�� to Z-scheme by interfacial band bending for highly efficient photocatalytic hydrogen evolution[J]. Nano Energy, 2017, 40: 308-316.
[42] PAN Lun, WANG Song-bo, XIE Jia-wei, et al. Constructing TiO2 p-n homojunction for photoelectrochemical and photocatalytic hydrogen generation[J]. Nano Energy, 2016, 28: 296-303.
[43] DEPUCCIO D P, BOTELLA P, O'ROURKE B, et al. Degradation of methylene blue using porous WO3, SiO2-WO3, and their Au-loaded analogs: Adsorption and photocatalytic studies[J]. ACS Appl Mater Interfaces, 2015, 7: 1987-1996.
WANG Zi-jin, LIU Fang-yang, JIA Ming, JIANG Liang-xing, LAI Yan-qing, LI Jie
(School of Metallurgy and Environment, Central South University, Changsha 410083, China)
Abstract: Bi2O3 thin film was prepared by chemical bath deposition, and ZnFe2O4 was prepared by electrodeposition and post-annealing. The Bi2O3 and Bi2O3/ZnFe2O4 composite films were characterized by XRD, SEM, Raman, XPS and UV-vis. The photoelectrochemical performance of the prepared film as a photoelectrode and its application in photoelectrocatalytic degradation of organic pollutants was studied for the first time. The results show that the load of ZnFe2O4 enhances the absorption range of Bi2O3, increases the light absorption coefficient and inhibits the recombination of carriers at the interface between semiconductor and solution. The photocurrent density of Bi2O3/ZnFe2O4 composite film is 4 times of pure phase material in the Na2SO4 solution. The performance of methylene blue photoelectric degradation under AM1.5 illumination is significantly improved compared with pure Bi2O3.
Key words: heterojunction; Bi2O3/ZnFe2O4; photoelectrochemical; degradation
Foundation item: Project(51674298) supported by the National Natural Science Foundation of China; Project (2017JJ3384) supported by the Natural Science Foundation of Hunan Province, China; Project(2018M630910) supported by the 63rd Batch of China Postdoctoral Science Foundation
Received date: 2020-06-25; Accepted date: 2021-03-31
Corresponding author: JIANG Liang-xing; Tel: +86-731-88830649; E-mail: lxjiang@csu.edu.cn
(�༭ �� ��)
������Ŀ��������Ȼ��ѧ����������Ŀ(51674298)������ʡ��Ȼ��ѧ����������Ŀ(2017JJ3384)����63���й���ʿ���ѧ����������Ŀ(2018M630910)
�ո����ڣ�2020-96-25�������ڣ�2021-03-31
ͨ�����ߣ������ˣ����ڣ���ʿ���绰��0731-88830649��E-mail��lxjiang@csu.edu.cn

