�ж���������������Ӷ����ͭ������Ż�
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2014���4��
�������ߣ������ ������ ��ΰ�� ����� ������ ���»� �ܺ鲨
����ҳ�룺1161 - 1170
�ؼ��ʣ��ж��������������������Ӷ����ͭ����ͭ�������Ż�
Key words��moderate thermophiles; bioleaching; complex Cu-polymetallic concentrate; copper extraction; optimization
ժ Ҫ���о����ж���������������Ӷ����ͭ������г�ʼpH���¶ȡ�װҺ����ת�١�ԭ���ЧӦ(���������)�Ϳ�Ũ�ȵ�Ӱ�졣�����������ʼpHΪ1.5��ͭ�����ʷֱ��dz�ʼpHΪ1.0��2.0��ʵ��������ʵ�1.5����1.4�������¶�Ϊ45 ��Cʱͭ�Ľ����ʱ��¶�Ϊ50 ��Cʱ�ĸ߳�1236.8%������ת�ٵ����ߺ�װҺ���Ľ��ͣ�ͭ�Ľ�����������ߡ���������ı�������ʱ��ͭ�Ľ�����Ҳ����ߣ����ǵ��������������20.0%��ͭ�����ʲ������ӡ��¶Ⱥ�pH���¶Ⱥͻ��������֮����������Ľ������á�
Abstract: Effects of initial pH, temperature, liquid volume, rotation speed, galvanic interaction (pyrite ratio) and pulp density on bioleaching of complex Cu-polymetallic concentrate were investigated. The results indicated that the copper extraction at pH 1.5 was 1.5 and 1.4 times that at pH 1.0 and pH 2.0 respectively. The copper extraction obtained at 45 ��C was 1236.8% higher than that at 50 ��C. With the increase of rotation speed or the decrease of liquid volume, copper extraction was improved obviously. Copper extraction was improved gradually with the increase of pyrite ratio. However, when the ratio was higher than 20.0%, no further increase in copper extraction was observed. And the statistically significant interactive effects on copper extraction were found between temperature and pH, and temperature and pyrite ratio.
Trans. Nonferrous Met. Soc. China 24(2014) 1161-1170
Yu-guang WANG1, Li-jun SU2, Wei-min ZENG1,3, Guan-zhou QIU1,3, Li-li WAN1, Xin-hua CHEN4, Hong-bo ZHOU1,3
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Department of Clinic Laboratory, Changsha Medical University, Changsha 410219, China;
3. Key Laboratory of Biometallurgy of Ministry of Education, Central South University, Changsha 410083, China;
4. Key Laboratory of Marine Biogenetic Resources, State Oceanic Administration, Xiamen 361005, China
Received 13 May 2013; accepted 30 September 2013
Abstract: Effects of initial pH, temperature, liquid volume, rotation speed, galvanic interaction (pyrite ratio) and pulp density on bioleaching of complex Cu-polymetallic concentrate were investigated. The results indicated that the copper extraction at pH 1.5 was 1.5 and 1.4 times that at pH 1.0 and pH 2.0 respectively. The copper extraction obtained at 45 ��C was 1236.8% higher than that at 50 ��C. With the increase of rotation speed or the decrease of liquid volume, copper extraction was improved obviously. Copper extraction was improved gradually with the increase of pyrite ratio. However, when the ratio was higher than 20.0%, no further increase in copper extraction was observed. And the statistically significant interactive effects on copper extraction were found between temperature and pH, and temperature and pyrite ratio.
Key words: moderate thermophiles; bioleaching; complex Cu-polymetallic concentrate; copper extraction; optimization
1 Introduction
Bioleaching has been developed into a frontier technology for extraction of gold, copper, cobalt, nickel, zinc and uranium from minerals in the 21st century [1]. In the case of complex Cu-polymetallic concentrate, high copper recovery is first priority due to its high grade. Nowadays, the focus has been more and more shifted to moderate thermophiles in bioleaching. Most existing commercial and planned plants use moderate thermophiles to recover metal. More than 90% of copper was extracted during bioleaching of copper sulphides by moderate thermophiles [2,3]. However, copper sulphides used in the most previous studies contained a lot of secondary copper sulfides, which are easily oxidized during bioleaching.
In addition, complex Cu-polymetallic concentrate contains more complicated components such as chalcopyrite, galena, sphalerite, pyrrhotite and high silver grade. Bioleaching of chalcopyrite is a complex process affected by multi-factors [4]. The proper choice of the value of these parameters is necessary if an economic complex Cu-polymetallic concentrate bioleaching process is to be developed. There is an extensive report on bioleaching of chalcopyrite but an answer to the question of what is the optimum level of these parameters cannot be reliably found in these reported studies. Only one study indicated that a statistically significant interactive effect is present between pH and temperature during bioleaching of sulfide minerals [5]. However, previous publications did not provide too many clues whether interactive effects among different factors exist or not during bioleaching of complex Cu-polymetallic concentrate [6-8]. It can provide large amounts of useful data and give conclusions applicable to the larger scale if the interactive effects were considered.
The aim of the present work was to identify and quantify interactive effects between important physicochemical parameters in bioleaching of complex Cu-polymetallic concentrate by moderate thermophiles.
The factors that affect dissolution kinetics, such as initial pH, pulp density, temperature, liquid volume, rotation speed and galvanic interaction, were investigated by five series of experiments. Furthermore, response surface methodology (RSM) was employed to determine the interactive effects among significant factors for further improvement of copper extraction. The main focus of the present work was on optimization and improvement of copper extraction. Although there is no doubt that the recovery of zinc, lead and silver is also important, it is not the focus of the present work.
2 Experimental
2.1 Enrichment and adaptation of moderately thermophilic culture
Acid mine drainage (AMD), coal mine waste water and thermal spring samples were collected from different sites in China. The samples were mixed and then inoculated into the culture medium at 45 ��C for enrichment of moderately thermophilic culture. The culture medium contained the following compounds: 3 g/L (NH4)2SO4; 0.1 g/L KCl; 0.5 g/L K2HPO4; 0.5 g/L MgSO4��7H2O; 0.01 g/L Ca(NO3)2. FeSO4��7H2O and S, or complex Cu-polymetallic concentrate were added as energy source. Finally, cultures growing on different substrates were pooled equally, resulting in a moderately thermophilic culture. Denaturing gradient gel electrophoresis analysis indicated that the major phylogenetic groups of the mixed culture belong to Acidithiobacillus caldus, Leptospirillum ferriphilum, Sulfobacillus sp. and Ferroplasma sp. (unpublished data).
2.2 Minerals components
The complex Cu-polymetallic concentrate was obtained from the Dongshengmiao Copper Mine in Inner Mongolia, China. X-ray diffraction (XRD) analysis shows chalcopyrite (60%, w/w), sphalerite (15%) and pyrrhotite (10%) as the major components and galena (5%) and quartz (3%) as the minor ones (Fig. 1(a)). The main chemical composition of the concentrate was (w/w) 18.97% Cu, 24.20% Fe, 28.17% S, 4.03% Pb, 5% Zn and 0.34% Ag. Pyrite used in this study was obtained from the Yunfu Mine in Guangdong, China. The pyrite contains 94% pyrite and 6% quartz (Fig. 1(b)). The main chemical composition of the mineral was 44.46% Fe and 47.78% S.
2.3 Optimization of complex Cu-polymetallic concentrate bioleaching
In the first series of experiments, initial pH values were 1.0, 1.5 and 2.0, respectively. In the second series of experiments, the pulp densities were 1%, 2%, 4% and 6% (w/v), respectively. In the third series of experiments, the temperatures were set to 40 ��C, 45 ��C and 50 ��C, respectively. The liquid volumes were 30%, 40% and 50% (v/v) based on every specific rotation speed (150, 180 and 200 r/min) in the fourth series of experiments. Effect of galvanic interaction between chalcopyrite and pyrite on dissolution was investigated in the final series of experiments. The pyrite ratios were 33.3%, 20.0%, 14.3% and 11.1% (w/w), respectively. The experiments were carried out in 250 mL batch shake flask containing 100 mL medium, each series of experiments were carried out under optimal conditions according to previous experiments. Samples were withdrawn at regular intervals to determine the pH, redox potential, cell density and concentrations of copper and iron. All experiments were carried out in triplicate.
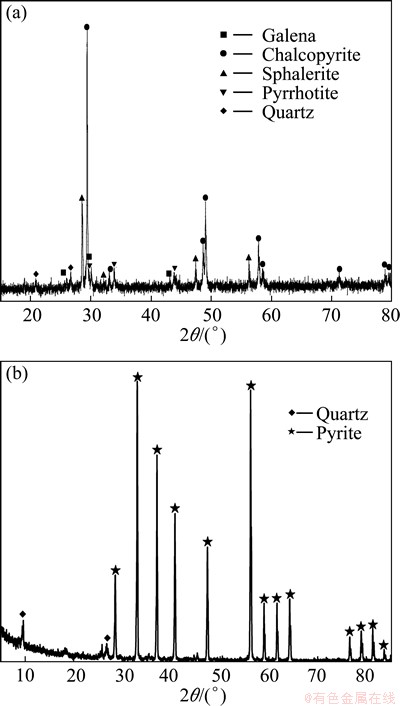
Fig. 1 XRD patterns of complex Cu-polymetallic concentrate (a) and pyrite (b) used
2.4 Experimental design for RSM
Based on results obtained in the section 2.3, a central composite design (CCD), a standard RSM, was employed to optimize bioleaching of complex Cu-polymetallic concentrate. Three factors and five levels experimental blocks were constructed by the principle of RSM using Design-expert 8.0 (DX) and the quality of analysis model was evaluated based on an analysis of variance (ANOVA).
3 Results and discussion
3.1 Optimal pH for bioleaching of complex Cu- polymetallic concentrate
As can be seen from Table 1, the initial pH has important effect on copper extraction. The copper extraction was up to 90.9% at the end of bioleaching in the experiment of initial pH 1.5. The copper extractions were only 58.8% and 64.2% when initial pH values were 1.0 and 2.0, respectively.
Table 1 Copper extractions during bioleaching of complex Cu-polymetallic concentrate with moderately thermophilic culture
In the experiment of initial pH 1.0, pH value was nearly below 1.0 during the whole bioleaching process. To the contrary, when initial pH was 2.0, pH value was above 1.7 all the time, even up to 2.6 on the 3rd day (Fig. 2(a)). Such pH inhibited the growth of microorganisms of the culture, which could be confirmed by variation of cell density as shown in Fig. 2(b). Cell density at initial pH 1.5 was almost three times that at initial pH 2.0 on the 15th day. Thus, only S-oxidizer such as A.caldus, whose optimal pH for growth is about 2.0, could get a competive advantage when initial pH was 2.0. But A.caldus, only utilizing reduced inorganic sulfur compounds (RISCs) as energy, cannot dissolve minerals directly. Therefore, the copper extractions were 43.9% and 29.4%, which were lower than that when initial pH was 1.5 on the 9th day and the 15th day. According to previous study [9], only Ferroplasma spp. can grow well when the pH was below 1.0, but its oxidization capability of ferrous iron was very weak. Thus, although cell density reached 9��107 cell/mL when initial pH was 1.0, the copper extraction was far lower than that at initial pH 1.5.
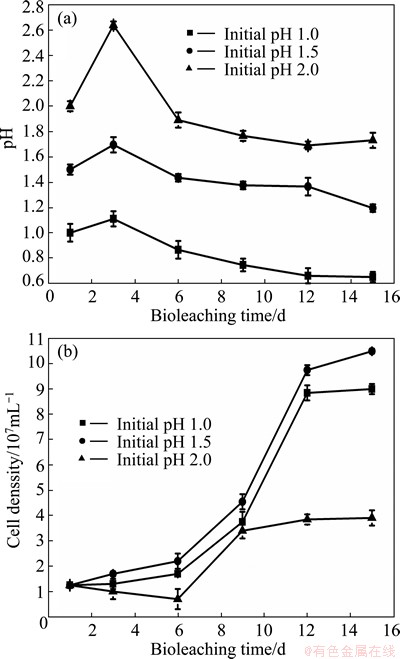
Fig. 2 Variations of pH (a) and cell density (b) during bioleaching of complex Cu-polymetallic concentrate at different initial pH values
Since lower pH benefits chalcopyrite dissolution, the copper extraction at initial pH 1.0 was 28.2% higher than that at initial pH 2.0 on the 9th day. However, the scenario was opposite on the 15th day as shown in Table 1. XRD analyses indicated that elemental sulfur content in the leached residues at initial pH 2.0 was lower than that in the experiment of initial pH 1.0 at the end of bioleaching (Fig. 3). Previous report has confirmed sulfur as one of main passivation layers during bioleaching of chalcopyrite, especially in the final stage, which causes obstructions between minerals and microorganisms [10]. But S-oxidizers can alleviate this negative effect by oxidizing sulfur, and many reports indicated that metal extraction was improved obviously when A.caldus or/and A.thiooxidans existed during bioleaching of sulfide minerals [11,12].
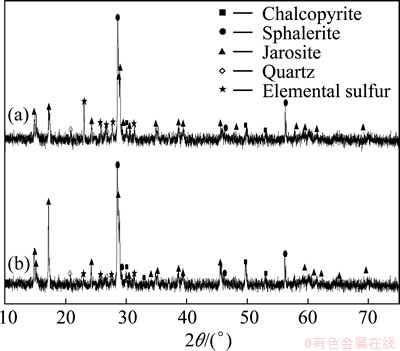
Fig. 3 XRD patterns of residues at end of bioleaching of complex Cu-polymetallic concentrate at initial pH 1.0 (a) and pH 2.0 (b)
3.2 Optimal pulp density for bioleaching of complex Cu-polymetallic concentrate
As can be seen from Fig. 4(a), the higher the pulp density was, the more obvious the pH value varied. The highest pH values were up to 1.6, 1.7, 2.3 and 2.6 at pulp densities of 1%, 2%, 4% and 6% in the initial stage, respectively. Moreover, cell density showed a downward trend with the increase of pulp density in the initial stage as shown in Fig. 4(b). Even more, the cell densities from the 3rd day to the 6th day were lower than the inoculums density when pulp density was 6%. The variations of pH and cell density demonstrated that high pulp density was adverse to growth of microorganisms, especially in the initial stage. As described in section 3.1, the high pH had a significant negative effect on the growth of microorganisms, which, in turn, could exacerbate harmful influence on pulp density and led to slow kinetics of copper extraction as shown in Table 1.
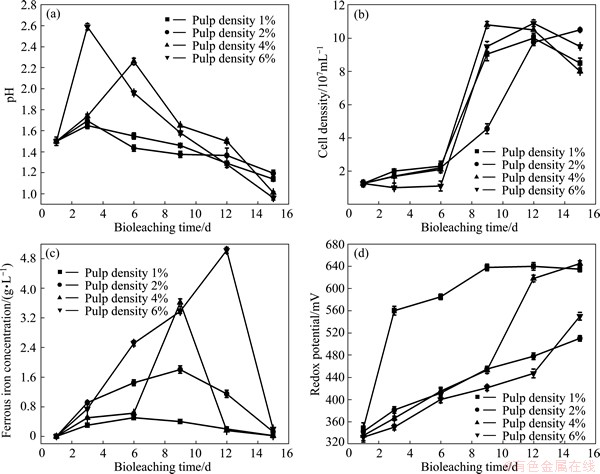
Fig. 4 Variations of pH (a), cell density (b), ferrous iron concentration (c) and redox potential (d) during bioleaching of complex Cu-polymetallic concentrate at different pulp densities
Although cell density did not show obvious difference after 6 d, high pulp density also showed negative influence on activities of microorganisms. This was consistent with high concentration of ferrous iron due to weak oxidization ability of ferrous iron at high pulp density (Fig. 4(c)). It is believed that high pulp density caused powerful shear force and limited oxygen and mass transfer; and also contained high concentration of flotation reagents and metal ions. Especially for silver, it has been shown to be the most toxic [13]. All of these factors and interactions among them will inhibit activities of microorganisms and reduce copper extraction greatly. As bioleaching progressed, this effect performed more and more obviously according to Table 1. In the first 9 d, copper extractions at pulp densities 4% and 6% were lower by 4.2% and 6.0% compared with pulp density 2%. But the gaps reached 6.4% and 8.6% after 6 d, respectively. Although the effect of pulp density was the weakest among all factors investigated in this study (The copper extraction did not decrease obviously when pulp density was higher than 2%), it can be found that high pulp density was harmful to growth of microorganisms and chalcopyrite dissolution to some extent.
The copper extraction at pulp density 1% was the lowest (75.6%) among all pulp densities, which seems to be contrary to above discussions. But the reason was closely related to the high level of redox potential during bioleaching as shown in Fig. 4(d). High redox potential value (>420 mV) is adverse to chalcopyrite dissolution [3].
3.3 Optimal temperature for bioleaching of complex Cu-polymetallic concentrate
To the mixed culture enriched in this study, temperature has a remarkable effect on copper extraction as shown in Table 1 and Fig. 5. At 50 ��C, the negative effect was fatal and only 6.8% of copper was extracted at the end of bioleaching. The highest copper extraction was obtained at 45 ��C, which was 1236.8% higher than that at 50 ��C. The phenomenon was probably related to the fact that growth of the mixed culture was seriously inhibited at 50 ��C (Fig. 5(a)). Cell density was always at very low level all the time, even lower than the inoculums density. Correspondingly, pH value increased continually during the whole bioleaching process (Fig. 5(b)), which indicated that RISCs were not oxidized to sulfuric acid efficiently by S-oxidizers. In addition, the redox potential value was very low compared with that at temperatures 40 ��C and 45 ��C (Fig. 5(c)). It has been proved that ferric iron is the major oxidization agent for chalcopyrite dissolution. Thus, extraction of copper was at a very slow rate, corresponding with weak capacity of ferric iron regeneration.
Since iron and sulfur cycles were disrupted, microorganisms hardly contributed chalcopyrite dissolution which would mainly depend on hydrogen ions attack existing in the medium. However, this contribution to chalcopyrite dissolution was very weak as shown in Table 1. And hydrogen ions contribution became less and less as time went on due to the decrease of its concentration. Only 0.2% of copper was extracted from the 9th day to the end of bioleaching. According to the views of FRANZMANN et al [14], temperature is major selective pressure for the microorganism during bioleaching. It is detrimental to the bioleaching process when temperature is above the maximal temperature which microorganisms can tolerate. And it is important that the temperature is maintained near optimal growth temperature of microorganism for improving bioleaching performance. It seems that 40-45 ��C and 50 ��C are the optimal temperature and maximal temperature for growth of the mixed culture used in this study. As a result of thermodynamic influence that low temperature brings about slow reaction kinetics, copper extraction at 40 ��C was 16.4% lower than that at 45 ��C, although there were no obvious differences between two experiments in the variations of pH, cell density, redox potential, etc.
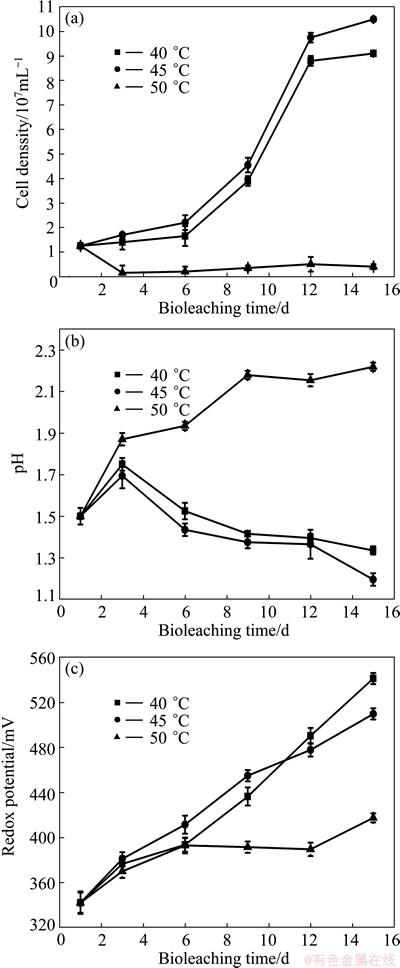
Fig. 5 Variations of cell density (a), pH (b) and redox potential (c) during bioleaching of complex Cu-polymetallic concentrate at different temperatures
3.4 Optimal liquid volume and rotation speed for bioleaching of complex Cu-polymetallic concentrate
Liquid volume and rotation speed mainly affect mass transfer efficiency [15,16], thus the experiments were designed to investigate the effects on copper extraction. It can be found that liquid volume and rotation speed have great effects on copper extraction (Table 1). Since moderate thermophiles have great ability to resist shear force, the highest copper extraction (92.7%) was obtained at liquid volume 30% (v/v) and rotation speed 200 r/min.
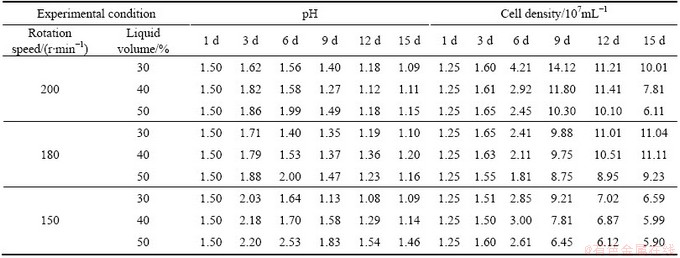
As can be seen in Table 2, pH value rose slowly in the initial stage, and after that dropped quickly with the increase of rotation speed. Furthermore, pH only increased by 0.12-0.36 in the initial stage at 200 r/min; while it ascended to 2.03-2.53 when rotation speed was 150 r/min. The effect of liquid volume was similar to the effect of rotation speed. It can be found from Table 1 that when rotation speed was fixed, the copper extraction decreased with the increase of liquid volume. When the liquid volume was up to 50%, the copper extraction decreased by 12.3% (200 r/min), 17.7% (180 r/min) and 5.3% (150 r/min) compared with that at liquid volume 30%, respectively. It was probably related to low efficiency of mass transfer in the experiments of rotation speed 150 r/min or liquid volume 50% [15,16]. It was inevitable, therefore, that oxidization capacities of ferrous iron and RISCs were suppressed to some extent compared with that at higher rotation speed and lower liquid volume.
However, there were no obvious differences in cell density between 200 r/min and 150 r/min before day 9. And cell density at 150 r/min even was higher than that at 180 r/min as shown in Table 2. It might be due to anaerobic growth of microorganisms to some degree. Some moderate thermophiles, such as Sulfobacillus sp. and Ferroplasma sp., can grow anaerobically at low dissolved oxygen concentration using ferric iron as sole electron acceptor. It was no doubt that dissolved oxygen concentration was low when rotation speed was 150 r/min due to low efficiency of oxygen and mass transfer [15,16]. This can be confirmed by the low level of redox potential during bioleaching process (data not shown), which indicated that oxidation of ferrous iron was poor at 150 r/min. However, the anaerobic growth of microorganisms has no direct contribution to chalcopyrite dissolution. Thus, it can be found from Table 1 that only 29.2 %, 30.6% and 25.0% of copper were extracted within 9 d when rotation speed was 150 r/min. Therefore, the negative effect of repressed iron cycle resulting from low dissolved oxygen concentration led to slow kinetics of chalcopyrite dissolution.
3.5 Optimal pyrite ratio for bioleaching of complex Cu-polymetallic concentrate
According to the results in section 3.4, the copper extractions did not show significant discrepancy between experiments at rotation speeds 200 r/min and 180 r/min. The scenario was also observed when liquid volumes were 40% and 30%. Thus, liquid volume and rotation speed were suggested to be set as 180 r/min and 40% in the following experiments.
Table 2 Variations of pH and cell density during bioleaching of complex Cu-polymetallic concentrate at different liquid volumes and rotation speeds
The rest potential of pyrite is higher than that of chalcopyrite in acidic medium. When pyrite and chalcopyrite are in contact with each other, galvanic interaction will occur. Pyrite acted as cathode and was protected, while chalcopyrite acted as anode and its oxidation was intensified [17]. If pyrite is absence, both cathodic reaction and anodic reaction will take place on the chalcopyrite surface with slow reaction rates, which is considered one of main reasons for slow rate of chalcopyrite dissolution [18]. It can be found from Table 1 that appropriate pyrite ratio can improve kinetics of copper extraction obviously. In this study, the highest copper extraction (96.0%) was obtained when the pyrite ratio was 20.0% (w/w).
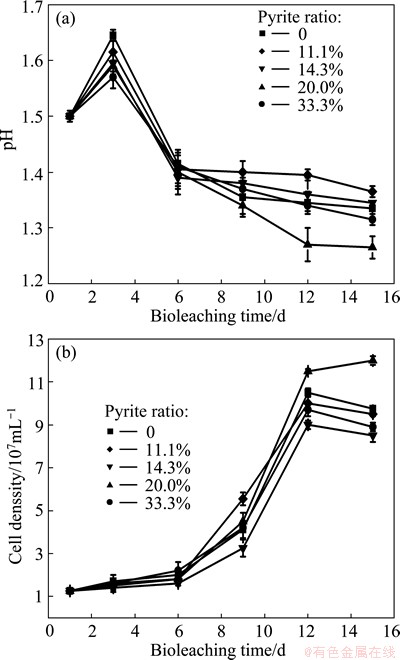
Fig. 6 Variations of pH (a) and cell density (b) during bioleaching of complex Cu-polymetallic concentrate at different pyrite ratios
Figure 6(a) shows the variations of pH in the leachate during bioleaching of complex Cu-polymetallic concentrate at different pyrite ratios. The pH at pyrite ratio 20% was the lowest after 6 d among all experiments. Moreover, it decreased to 1.26 at the end of bioleaching. The lower the pH of leachate was, the higher the corrosion current density was [18] and the faster the chalcopyrite dissolution rate was. It also can be found from Fig. 6(b) that appropriate pyrite ratio can promote growth of microorganisms. The highest cell density in other experiments was much lower than that at pyrite ratio 20.0% (1.2��108 mL-1). Ferrous iron released from chalcopyrite dissolution was oxidized by Fe-oxidizers (L. ferriphilum, Sulfobacillus sp. and Ferroplasma sp.), which enlarged gap of rest potential between pyrite and chalcopyrite and then enhanced galvanic interaction [18].
Many factors have significant effects on galvanic interaction, such as electric conductivity, corrosion current density and contact area. In this study, the copper extraction was improved gradually with the increase of pyrite ratio (Table 1). However, when the pyrite ratio was up to 33.3%, no further increase in copper extraction was observed, even more it decreased by 11.5% compared with that at pyrite ratio 20.0%. The results were in agreement with previous studies [17,19]. Thus, only appropriate pyrite ratio can improve kinetics of copper extraction obviously.
3.6 Further optimization of complex Cu-polymetallic concentrate bioleaching by RSM
According to the above results, three factors (pH, temperature and pyrite ratio) were selected for the CCD experiments. All experiments were carried out at liquid volume 40%, rotation speed 180 r/min and pulp density 2%. The copper extractions obtained under different experimental parameters are given in Table 3. Based on the calculated values of the regression coefficients, a polynomial regression model equation is proposed as follows:
y=-4825.35+1037.68x1+177.85x2+13.18x3-4.94x1x2-1.06x1x3+0.18x2x3-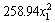 -
-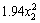 -
-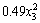
where y is the copper extraction; x1 is the pH; x2 is the temperature; x3 is the pyrite ratio.
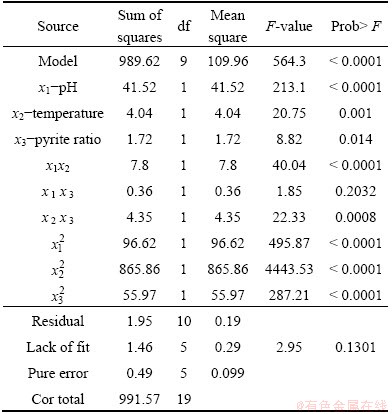
The values of R2 (0.9980) and Adj. R2 (0.9963) indicate a high degree of correlation between the observed and predicted values (Table 3). Statistical test of the model was done by ANOVA. The regression model demonstrates that the model is highly significant (P<0.0001). The model also shows statistically non-significant (P=0.1301) in lack of fit for the copper extraction as shown in Table 4, which indicated that the model is credible. The linear coefficients and quadratic term coefficients (x1x2, x2x3, 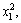
 and
and 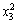 ) are significant. This means that the optimal level of temperature depended on the level of pH as well as the level of pyrite ratio during bioleaching of complex Cu-polymetallic concentrate by the culture enriched in this study. On the other hand, the optimum levels of pH and pyrite ratio depended on the level of temperature. Furthermore, the results shown in Table 4 show that the interactive effect between pH and pyrite ratio was statistically insignificant.
) are significant. This means that the optimal level of temperature depended on the level of pH as well as the level of pyrite ratio during bioleaching of complex Cu-polymetallic concentrate by the culture enriched in this study. On the other hand, the optimum levels of pH and pyrite ratio depended on the level of temperature. Furthermore, the results shown in Table 4 show that the interactive effect between pH and pyrite ratio was statistically insignificant.
Table 3 Central composite design arrangements and responses
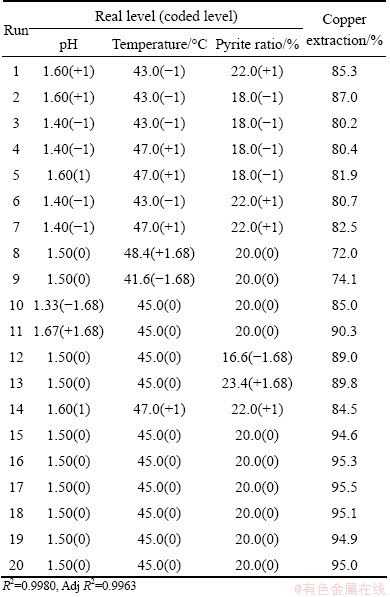
Table 4 Analysis of variance for predictive equation for copper extraction
It can be found from Fig. 7(a) that pH and temperature had a quadratic effect on the copper extraction. Moreover, this effect was more and more obvious as the pH decreased. Figure 7(b) shows that the interactive effect between temperature and pyrite ratio was more sharply when temperature was outside of 44-46 ��C, especially higher than 46 ��C and pyrite ratio was at relatively low level. As can be seen from Fig. 7, the copper extraction depended more on pH than on temperature and pyrite ratio. The optimal levels of the factors were obtained by analyzing the response surface contour plots. The model predicts that the maximal copper extraction (95.4%) appears at pH 1.53, temperature 44.9 ��C and pyrite ratio 20.1%. To validate the adequacy of the model, six additional experiments under the optimal conditions were performed. The mean value of copper extraction was 95.3%, which was in good agreement with the predicted value.
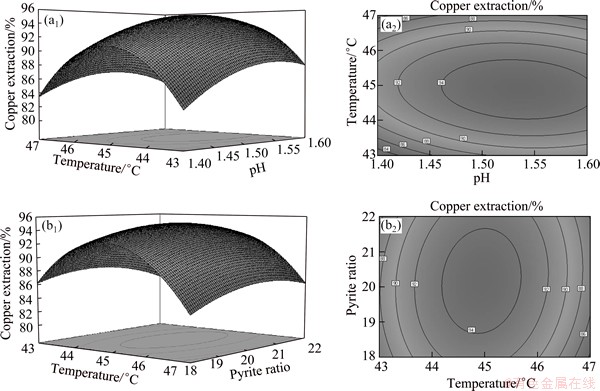
Fig. 7 3D response surface and contour plots of effects of temperature-pH (a1, a2) and temperature-pyrite ratio (b1, b2) and their interactive effects on copper extraction during bioleaching of complex Cu-polymetallic concentrate with moderately thermophilic culture
4 Conclusions
1) A moderately thermophilic culture is obtained, which shows good performance in bioleaching of the complex Cu-polymetallic concentrate after adaptation and optimization.
2) Initial pH, temperature, liquid volume, rotation speed and pyrite ratio are statistically significant factors to dissolution kinetics of the complex Cu-polymetallic concentrate bioleaching.
3) There are statistically significant interactive effects between temperature and pH, temperature and pyrite ratio.
4) The maximal copper extraction is improved by 20% compared with original process.
References
[1] WANG Z H, XIE X H, LIU J S. Experimental measurements of short-term adsorption of Acidithiobacillus ferrooxidans onto chalcopyrite [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(2): 442-446.
[2] AHMADI A, SCHAFFIE M, PETERSEN J, SCHIPPERS A, RANJBAR M. Conventional and electrochemical bioleaching of chalcopyrite concentrates by moderately thermophilic bacteria at high pulp density [J]. Hydrometallurgy, 2011, 106(1-2): 84-92.
[3] GERICKE M, GOVENDER Y, PINCHES A. Tank bioleaching of low-grade chalcopyrite concentrates using redox control [J]. Hydrometallurgy, 2010, 104(3-4): 414-419.
[4] ZHU W, XIA J L, PENG A A, NIE Z Y, QIU G Z. Characterization of apparent sulfur oxidation activity of thermophilic archaea in bioleaching of chalcopyrite [J]. Transactions of Nonferrous Metals Society of China, 2013, 23(8): 2383-2388.
[5] HAGHSHENAS D F, BONAKDARPOUR B, ALAMDARI E K, NASERNEJAD B. Optimization of physicochemical parameters for bioleaching of sphalerite by Acidithiobacillus ferrooxidans using shaking bioreactors [J]. Hydrometallurgy, 2012, 111-112(1): 22-28.
[6] GERICKE M, MULLER H H, VAN STADEN P J, PINCHES A. Development of a tank bioleaching process for the treatment of complex Cu-polymetallic concentrates [J]. Hydrometallurgy, 2008, 94(1-4): 23-28.
[7] LIU Z X, YIN Z L, HU H P, CHEN Q Y. Leaching kinetics of low-grade copper ore containing calcium-magnesium carbonate in ammonia-ammonium sulfate solution with persulfate [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(11): 2822-2830.
[8] GUO Z H, PAN F K, XIAO X Y, ZHANG L, JIANG K Q. Optimization of brine leaching of metals from hydrometallurgical residue [J]. Transactions of Nonferrous Metals Society of China, 2010, 20(10): 2000-2005.
[9] JOHNSON D. Biohydrometallurgy and the environment: intimate and important interplay [J]. Hydrometallurgy, 2006, 83(1-4): 153-166.
[10] SASAKI K, NAKAMUTA Y, HIRAJIMA T, TUOVINEN O. Raman characterization of secondary minerals formed during chalcopyrite leaching with Acidithiobacillus ferrooxidans [J]. Hydrometallurgy, 2009, 95(1-2): 153-158.
[11] ZHANG Y S, QIN W Q, WANG J, ZHEN S J, YANG C R, ZHANG J W, NAI S S, QIU G Z. Bioleaching of chalcopyrite by pure and mixed culture [J]. Transactions of Nonferrous Metals Society of China, 2008, 18(6): 1491-1496.
[12] XIA L, TANG L, XIA J, YIN C, CAI L, ZHAO X, NIE Z, LIU J, QIU G. Relationships among bioleaching performance, additional elemental sulfur, microbial population dynamics and its energy metabolism in bioleaching of chalcopyrite [J]. Transactions of Nonferrous Metals Society of China, 2012, 22(1): 192-198.
[13] DE G, OLIVER D, PESIC B. Effect of silver on the ferrous iron oxidizing ability of Thiobacillus ferrooxidans [J]. Hydrometallurgy, 1996, 41(2): 211-229.
[14] FRANZMANN P D, HADDAD C M, HAWKES R B, ROBERTSON W J, PLUMB J J. Effects of temperature on the rates of iron and sulfur oxidation by selected bioleaching bacteria and archaea: Application of the Ratkowsky equation [J]. Minerals Engineering, 2005, 18(13-14): 1304-1314.
[15] LOTTER S,  J. Utilization of specific power input measurements for optimization of culture conditions in shaking flasks [J]. Biochemical Engineering Journal, 2004, 17(3): 195-203.
J. Utilization of specific power input measurements for optimization of culture conditions in shaking flasks [J]. Biochemical Engineering Journal, 2004, 17(3): 195-203.
[16] MAIER U,  J. Characterisation of the gas�Cliquid mass transfer in shaking bioreactors [J]. Biochemical Engineering Journal, 2001, 7(2): 99-106.
J. Characterisation of the gas�Cliquid mass transfer in shaking bioreactors [J]. Biochemical Engineering Journal, 2001, 7(2): 99-106.
[17] KOLEINI S M J, AGHAZADEH V,  A. Acidic sulphate leaching of chalcopyrite concentrates in presence of pyrite [J]. Minerals Engineering, 2011, 24(5): 381-386.
A. Acidic sulphate leaching of chalcopyrite concentrates in presence of pyrite [J]. Minerals Engineering, 2011, 24(5): 381-386.
[18] LIU Q, LI H, ZHOU L. Study of galvanic interactions between pyrite and chalcopyrite in a flowing system: Implications for the environment [J]. Environmental Geollogy, 2007, 52(1): 11-18.
[19] SADOWSKI Z, JAZDZYK E, KARAS H. Bioleaching of copper ore flotation concentrates [J]. Minerals Engineering, 2003, 16(1): 51-53.
�����1, ������2, ��ΰ��1,3, �����1,3, ������1, ���»�4, �ܺ鲨1,3
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083��
2. ��ɳҽѧԺ ҽѧ����ϵ����ɳ 410219��
3. ���ϴ�ѧ ����ұ��������ص�ʵ���ң���ɳ 410083��
4. ���Һ���� ���������Ŵ���Դ�ص�ʵ���ң����� 361005
ժ Ҫ���о����ж���������������Ӷ����ͭ������г�ʼpH���¶ȡ�װҺ����ת�١�ԭ���ЧӦ(���������)�Ϳ�Ũ�ȵ�Ӱ�졣�����������ʼpHΪ1.5��ͭ�����ʷֱ��dz�ʼpHΪ1.0��2.0��ʵ��������ʵ�1.5����1.4�������¶�Ϊ45 ��Cʱͭ�Ľ����ʱ��¶�Ϊ50 ��Cʱ�ĸ߳�1236.8%������ת�ٵ����ߺ�װҺ���Ľ��ͣ�ͭ�Ľ�����������ߡ���������ı�������ʱ��ͭ�Ľ�����Ҳ����ߣ����ǵ��������������20.0%��ͭ�����ʲ������ӡ��¶Ⱥ�pH���¶Ⱥͻ��������֮����������Ľ������á�
�ؼ��ʣ��ж��������������������Ӷ����ͭ����ͭ�������Ż�
(Edited by Hua YANG)
Foundation item: Project (2012zzts026) supported by the Fundamental Research Funds for the Central Universities, China; Project (201205020) supported by Scientific Research Program of Marine Public Welfare Industry of China; Project (51074195) supported by the National Natural Science Foundation of China; Project (CX2012B123) supported by Research Innovation for Graduate Student of Hunan Province, China; Project (12C517) supported by Education Department of Hunan Province, China
Corresponding author: Hong-bo ZHOU; Tel: +86-731-88877216; E-mail: zhouhb@csu.edu.cn
DOI: 10.1016/S1003-6326(14)63175-7

