Using sensitive surface plasmon resonance to detect binding of peptide molecules and immobilized vancomycin
来源期刊:中南大学学报(英文版)2011年第4期
论文作者:何永辉 刘斌 詹艳峰 刘又年 向娟
文章页码:1024 - 1028
Key words:flow injection; surface plasmon resonance; vancomycin; Lys-Lys-Gly-D-Ala-D-Ala; binding; sensitivity
Abstract:
To perform the mechanism study of special association for vancomycin and D-Ala-D-Ala-containing peptides on the interface of solution and self-assemble monolayer, the binding between vancomycin and pentapeptide (Lys-Lys-Gly-D-Ala-D-Ala) was investigated by flow injection surface plasmon resonance (FI-SPR) and flow injection quartz crystal microbalance (FI-QCM). To facilitate the formation of a compact vancomycin adsorbates layer with a uniform surface orientation, vancomycin molecules were attached onto a preformed alkanethiol self-assembled monolayer. By optimizing the conditions for the binding between Lys-Lys-Gly-D-Ala-D-Ala and vancomycin on the assembled chip, the detecting limit of Lys-Lys-Gly-D-Ala-D-Ala was greatly improved (reaching 0.5×10-6 mol/L or 7.5×10-12 mol). The equilibrium constant of the association of Lys-Lys-Gly-D-Ala-D-Ala with vancomycin was also obtained (KAds=5.0×104 L/mol).
J. Cent. South Univ. Technol. (2011) 18: 1024-1028
DOI: 10.1007/s11771-011-0798-z![]()
HE Yong-hui(何永辉), LIU Bin(刘斌), ZHAN Yan-feng(詹艳峰), LIU You-nian(刘又年), XIANG Juan(向娟)
School of Chemistry and Chemical Engineering, Central South University, Changsha 410083, China
? Central South University Press and Springer-Verlag Berlin Heidelberg 2011
Abstract: To perform the mechanism study of special association for vancomycin and D-Ala-D-Ala-containing peptides on the interface of solution and self-assemble monolayer, the binding between vancomycin and pentapeptide (Lys-Lys-Gly-D-Ala-D-Ala) was investigated by flow injection surface plasmon resonance (FI-SPR) and flow injection quartz crystal microbalance (FI-QCM). To facilitate the formation of a compact vancomycin adsorbates layer with a uniform surface orientation, vancomycin molecules were attached onto a preformed alkanethiol self-assembled monolayer. By optimizing the conditions for the binding between Lys-Lys-Gly-D-Ala-D-Ala and vancomycin on the assembled chip, the detecting limit of Lys-Lys-Gly-D-Ala-D-Ala was greatly improved (reaching 0.5×10-6 mol/L or 7.5×10-12 mol). The equilibrium constant of the association of Lys-Lys-Gly-D-Ala-D-Ala with vancomycin was also obtained (KAds=5.0×104 L/mol).
Key words: flow injection; surface plasmon resonance; vancomycin; Lys-Lys-Gly-D-Ala-D-Ala; binding; sensitivity
1 Introduction
Vancomycin group antibiotic, as an antibiotic of glycopeptide, performs a last line of defense against the serious clinical pathogen MRAS (methicillin resistant staphylococcus) [1]. Vancomycin group antibiotics can inhibit the cell wall biosynthesis of growing bacterial by specifically binding to cell wall peptidoglycans that terminate in D-Ala-D-Ala [2], resulting in bacterial death [3]. Therefore, the kinetics and mechanism investigation of special association for vancomycin and D-Ala-D-Ala- containing peptides have become important. Till now, many traditional methods, such as thin paper chromato- graphy and affinity capillary electrophoresis [4], X-ray analysis [5], nuclear magnetic resonance (NMR) [6], absorption spectrum [7], fluorescence spectrum [8] and surface plasmon resonance (SPR) [9-10], have been used in the research of the interactions of vancomycin and D-Ala-D-Ala-containing peptides. Among these methods, SPR shows favorable advantage for lower detection limit attributing to its high sensitivity.
SPR, as a highly sensitive real-time technique, has been extensively applied for detection of adsorbates in recent years, especially biomacromolecular ones, at extremely low quantities [11-12]. Because the SPR phenomenon is extremely sensitive to the refractive index of the medium (or solution composition) next to the metal film, adsorption of a trace amount of adsorbates at the surface or infinitesimal conformational/ orientation changes in the adsorbed molecules can be detected. Moreover, due to the needlessness of fluorescent tags or radioactive labels [13], SPR can provide more accurate data close to the physiological process.
However, in reported SPR works, vancomycin molecules are usually linked onto the chip via carboxymethylated dextran, which induces inevitable nonspecific binding and high cost [14-15]. Moreover, because dextran film is thick and non-uniform, it could cause the vancomycin incorporation with peptide antibody to be measured at distances further away from the metal film where the evanescent field is weaker. Consequently, the limit of detection could be higher.
In this work, in order to overcome the above problems, we combined self-assembled monolayers (SAMs) and ethanolamine to optimize the binding condition of Lys-Lys-Gly-D-Ala-D-Ala and vancomycin. Lower peptide ligand detection level than that obtained by recent similar study [9] was achieved. Moreover, the equilibrium constant of association of Lys-Lys-Gly-D- Ala-D-Ala with vancomycin was obtained.
2 Experimental
2.1 Materials and reagent
All materials and reagents were used as received. Vancomycin, N-hydroxysuccinimide (NHS), ethylene dichloride (EDC), 11-mercaptoundecanoic acid (MUA), and ethanolamine hydrochloride (AE) were all obtained from Sigma-Aldrich (St. Louis, MO). The pentapeptide (Lys-Lys-Gly-D-Ala-D-Ala) was purchased from Shanghai Bootech Bioscience & Technology Co., Ltd. (China). K2HPO4, KH2PO4, NaCl, and H2SO4 were all of AR grade and acquired from Sigma-Aldrich. Phosphate-buttered saline (PBS: 1.0×10-2 mol/L phosphate, 0.138 mol/L NaCl, pH=7.4) and the stock solution of AE (1.0 mol/L, pH=8.5) were prepared in distilled deionized water, respectively. Vancomycin and pentapeptide were dissolved in PBS, respectively. Solutions of NHS (0.1 mol/L) and EDC (0.4 mol/L) were prepared in distilled deionized water immediately before use. The stock solution of MUA (4.0×10-3 mol/L) was prepared in ethanol. All experiments were performed at room temperature.
2.2 Procedure
2.2.1 Immobilization of vancomycin
The gold film was prepared as described in Ref.[11]. Prior to use, the gold film was briefly annealed under a hydrogen flame to reduce surface contamination and to generate a smooth Au surface, then was immediately immerged in a 4×10-3 mol/L MUA solution overnight, followed by rinsing with water and drying with N2. The gold film was then covered with a mixture of 0.4 mol/L EDC and 0.1 mol/L NHS (3.0×10-5 L each, pH=7.0) for 30 min, followed by dripping down 3.0×10-5 L vancomycin (1.0×10-3 mol/L, pH=7.0) stored in a humidified chamber for 4 h. Another way of binding the vancomycin on the assembled chip is flow-inject- assembling, which means injecting vancomycin solution on the surface of Au film following flowing through the surface in 10 min.
After the vancomycin attachment step, the surface was rinsed with PBS buffer and water and dried with N2. To block the unreacted sites, 5.0×10-5 L of AE solution (1.0 mol/L) was spread onto the surface. The resultant film was either used immediately or stored at 4 °C for future use.
2.2.2 Flow injection surface plasmon resonance (FI-SPR)
The FI-SPR setup was the same as described in Ref.[16]. A BK7 planocylindrical lens (Melles Griot, Carlsbad, CA) was used as the SPR prism. A BK7 glass slide (Fisher Scientific, Tustin, CA, USA), coated with a 5.0×10-9 m-thick gold film and a 2.0×10-9 m-thick Cr underlayer using a sputter coater (Cressington model 108SE, Ted Pella, Inc. Redding, CA, USA), was placed onto the prism with an index-matching fluid (Cargille Laboratories, Cedar Grove, NJ, USA). A diode laser with emission wavelength at 6.35×10-7 m was controlled by a LDC 500 laser driver (Thorlabs, Newton, NJ, USA). The reflected light was detected with two photodetectors (A and B) of a bicell detector (Hamamatsu Corp., Model S272102). The SPR dip shift was proportional to the ratio of (A-B)/(A+B) [17]. The SPR signals were recorded via a data acquisition board (PCI-1731, Advantech Co., Inc.) controlled by a Labview program. Conversion of the (A-B)/(A+B) values to SPR angle changes follows the procedure in Refs.[18-20]. The homemade single-channel acrylic flow cell with an internal volume of ~4.0×10-6 L was used for all FI-SPR experiments. For each experiment, the sample was preloaded into a 1.0×10-4 L loop with a microsyringe (Fisher). Upon injection, the sample solution was introduced into the flowing buffer solution and subsequently delivered to the flow cell by a syringe pump (Harvard Apparatus).
2.2.3 Flow injection quartz crystal microbalance (FI- QCM)
FI-QCM measurements were conducted with a CHI 440 electrochemical workstation (Chenhua Instr. Co., Shanghai). The 9.0×106 Hz AT-cut quartz wafers were purchased from Maxtek (St. Beaverton, OR). The curve of frequency shift versus time was recorded in real time.
3 Results and discussion
3.1 In-situ monitoring immobilization process of Vancomycin
Figure 1 illustrates the detection process of pentapeptide ligand on the modified Au film. Vancomycin was first immobilized covalently to the Au film using EDC/NHS method. Then, before detecting the binding between pentapeptide and vancomycin, AE was used to block the unwanted NHS ester and the non-special absorption. An in-situ monitoring of the immobilization process was recorded by FI-SPR, as shown in Fig.2. The results show a large SPR response after injecting the solution of EDC/NHS, for its refractive index is different from the PBS carry solution, and accompany the active effect of EDC/NHS to the carboxyl of MUA binding onto the surface of gold film. The result of active effect is SPR angle change of 0.028 6°. Then, the solution of vancomycin is injected on the surface of the gold film, which bands on the surface of gold film by the association reaction of the vancomycin amidogen and NHS ester. It brings that the change of SPR angle is 0.031 5°. This shows the vancomycin has been immobilized onto the SAM. In the interest of blocking out the unwanted NHS ester after activation, the solution of AE (1 mol/L) is injected. Because the refractive index of the solution of AE is quite different from PBS, the SPR signal is very large. However, the final SPR dip shift is only 0.002°-0.003°. The infinitesimal SPR dip shift implies that most NHS ester reacted with vancomycin and little free NHS ester was left to react with AE. The feasibility of binding the vancomycin on the Au film was clearly demonstrated.
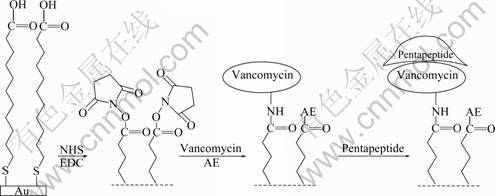
Fig.1 Schematic representation of interaction of pentapeptide ligand and vancomycin on surface-modified Au film
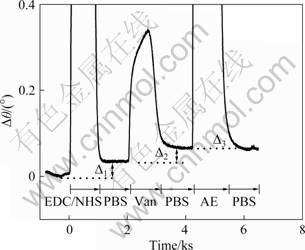
Fig.2 SPR dip shift changes corresponding to injection in turns of EDC/NHS, 1.0×10-3 mol/L Vancomycin, and 1 mol/L AE onto MUA modified Au film surface (Carrier solution: PBS buffer solution; flow rate: 0.5×10-3 L/h)
3.2 Ascertaining methods for self-assembled monolayers
Surface density of vancomycin is an important factor that directly determines the detection limit of antibody. To evaluate the surface density of vancomycin, a QCM technique was used. Following the same immobilization procedure of vancomycin, the in-situ frequency changes can be obtained, as shown in Fig.3. According to the Saurebrey formula [21-22], the value of mass change can be calculated and the surface density is about 2.437×10-11 mol/cm2, which is much larger than the previously reported value [9].
The pentapeptide Lys-Lys-Gly-D-Ala-D-Ala has two amidogens in each lysine residue, which makes it easy to have non-special absorption with surface-modified Au film. Eliminating this non-special absorption is important for the sensitivity of our FI-SPR setup. In this work, AE is utilized to block the unwanted EHS ester. As shown in Fig.4, curve (c) represents the SPR dip shifts of injecting the pentapeptide on the AE-blocked Au film. The observed slight SPR dip shifts suggest that there is no significant association. Therefore, it is confirmed that the non-special association of pentapeptide and modified gold film can be effectively eliminated by AE.
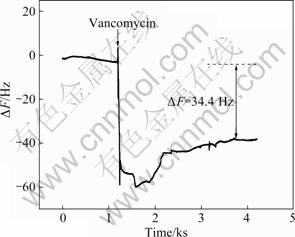
Fig.3 Frequency changes of MUA and EDC/NHS modified Au film after injecting of 1.0×10-3 mol/L vancomycin
Curves (a) and (b) in Fig.4 are two typical SPR sensorgrams. The obvious SPR dip shift changes before and after the injection of the pentapeptide solution indicate that the interaction of vancomycin and pentapeptide has happened and can be detected by SPR. Two ways for association of reagent onto the surface of Au film has always been used: one is dip-in-assembled (i.e. dip in the vancomycin onto the surface of Au film), and the other is flow-inject-assembled (i.e. inject the vancomycin onto the surface of Au film following flowing through the surface in 10 min). In this work, we have ascertained the better one for SPR detection. As shown in curves (a) and (b) in Fig.4, the signal from the dip-in-assembled is obviously more intense than that of the other. It is mainly caused by the fact that the contact extent of sample and gold film in a flow system is not enough. Thus, our bind method is the dip-in-assembled way.
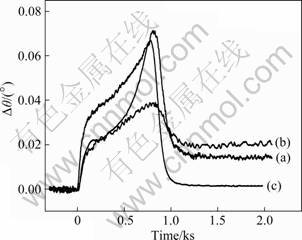
Fig.4 SPR dip shifts corresponding to injection of 1.0×10-3 mol/L Lys-Lys-Gly-D-Ala-D-Ala pentapeptide to flow-inject- assembled vancomycin (a) and dip-in-assembled vancomycin (b) on AE-blocked Au film, and SPR response of pentapeptide directly on AE-blocked Au film (c) (Carry solution: PBS buffer; flow rate: 0.5×10-3 L/h)
3.3 Binding of peptide molecules to immobilized vancomycin
Following the above defined optimal conditions for the binding of pentapeptide and vancomycin, different pentapeptide solutions of different concentrations have been injected, so the pentapeptide could interact with vancomycin binding on the assembled chip each other. The SPR sensor can detect this interaction in real-time, so the SPR images for different concentrations of pentapeptide have been obtained.
Thus, from these SPR images, the dependence of the SPR dip shifts on the concentration of pentapeptide has been gained, as shown in Fig.5. Obviously, the SPR response in Fig.5 contains two regions, viz., from 0.5×10-6 to 2.5×10-5 mol/L (region 1) and from 2.5×10-5 to 5.0×10-4 mol/L (region 2). Linear regression analysis of region 1 yields the following equation:
Δθ=0.000 294 08×C
where Δθ is the SPR dip shift; C is the concentration of pentapeptide. Region 2 shows little variety, suggesting that saturation state (Δθmax=0.014 4°) has been reached for the interaction of pentapeptide in the flow cell with vancomycin associated on the gold film. So, the appropriate concentration range for SPR detection is 0.5×10-6-2.5×10-5 mol/L. The lowest detection concentration for pentapeptide by SPR is 0.5×10-6 mol/L (7.5×10-12 mol, 4.087×10-9 g), which is approximately 500 times lower than that reported in Ref.[9].
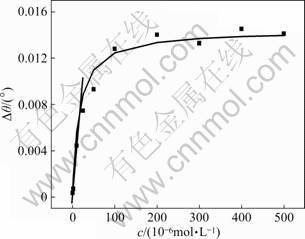
Fig.5 Dependence of SPR dip shifts on concentration of pentapeptide ligand (Calibration curve is non-linear fitting result following Langmuir adsorption isotherm; beeline is linear fitting curve between 0.5×10-6 mol/L and 2.5×10-5 mol/L)
The equilibrium adsorption coefficient, KAds (KAds=Ka/Kd), which represents the binding affinity between pentapeptide and vancomycin binding on the assembled chip, can also be obtained according to the dependence of the SPR dip shifts on the concentration of pentapeptide
Γ=KAds×C/(1+KAds×C) (1)
where Γ represents the coverage ratio of pentapeptide on immobilized vancomycin, which can be estimated using Γ=Δθ/Δθmax. Δθ is the SPR dip shift corresponding to the concentration of pentapeptide C, and Δθmax is the SPR dip shift at saturation state. So, with the non-linear fitting method, the equilibrium constant of association of Lys-Lys-Gly-D-Ala-D-Ala with vancomycin was determined to be 5.0×104 L/mol, which is similar to the previously reported results [24-25].
Thus, the SPR-based biosensor screening methodology described here has been established for sensitive detection of D-Ala-D-Ala-containing peptides and vancomycin immobilized on the surface. Due to high sensitivity, lower detect limit has been obtained, which means less sample consumption.
4 Conclusions
Vancomycin molecules are cross-linked onto preformed self-assembled monolayers and the sensor chips are treated with AE. Then, the pentapeptide solution is injected in the flow cell, and the interaction of pentapeptide and vancomycin is detected by the SPR-based biosensor. The results demonstrate that the interaction of vancomycin and the pentapeptide ligand in the flow cell can be readily detected by highly sensitive FI-SPR. Due to the formation of compact vancomycin film, a lower concentration detection level (0.5×10-6 mol/L) for pentapeptide is obtained. The equilibrium constant of association of pentapeptide (Lys-Lys-Gly-D- Ala-D-Ala) with vancomycin binding on the assembled chip is also determined to be 5.0×104 L/mol.
References
[1] GERACI J E, HERMANS P E. Vancomycin [J]. Mayo Clin Proc, 1983, 58(2): 88-91.
[2] FOLDES M, MUNRO R, SOMELL T C, SHANKAR S. In-vitro effects of vancomycin, rifampicin, and fusidic acid, alone and in combination, against methicillin-resistant Staphylococcus aureus [J]. J Antimicroh Chemother, 1983, 11(1): 21-26.
[3] REYNOLDS P E. Studies on the mode of action of vancomycin [J]. Biochim Biophys Acta, 1961, 52(2): 403-405.
[4] CHU Yen-ho, WHITESIDES G M. Affinity capillary electrophoresis can simultaneously measure binding constants of multiple peptides to vancomycin [J]. J Org Chem, 1992, 57: 3524-3525.
[5] WILLIAMS D H, BUTCHER D W. Binding site of the antibiotic vancomycin for a cell-wall peptide analog [J]. J Am Chem Soc, 1981, 103(19): 5697-5700.
[6] DANCER R J, TRY A C, SHARMAN G J, WILLIAMS D H. Binding of a vancomycin group antibiotic to a cell wall analogue from vancomycin-resistant bacteria [J]. Chem Commun, 1996: 1445-1446.
[7] POPIENIEK P H, PRATT R F. A fluorescent ligand for binding studies with glycopeptide antibiotics of the vancomycin class [J]. Anal Biochem, 1987, 165(1): 108-113.
[8] POPIENIEK P H, PRATT R F. Kinetics and mechanism of binding of specific peptides to vancomycin and other glycopeptide antibiotics [J]. J Am Chem Soc, 1991, 113(6): 2264-2270.
[9] TSENG Ming-chung, CHU Yen-ho. Using surface plasmon resonance to directly identify molecules in a tripeptide library that bind tightly to a vancomycin chip [J]. Anal Biochem, 2005, 336: 172-177.
[10] WEGNER G J, WARK A W, LEE H J, CODNER E, SAEKI T, FANG Shi-ping, CORN R M. Real-time surface plasmon resonance imaging measurements for the multiplexed determination of protein adsorption/desorption kinetics and surface enzymatic reactions on peptide microarrys [J]. Anal Chem, 2004, 76(19): 5677-5684.
[11] ZHANG Yin-tang, XU Mao-tian, WANG Yan-jun, TOLEDO F, ZHOU Fei-meng. Studies of metal ion binding by apo-metallothionein attached onto preformed self-assembled monolayer using a highly sensitive surface plasmon resonance spectrometer [J]. Sens Actuators B: Chem, 2006, 123(2): 784-792.
[12] YUE Li-duo, YE Fei, XU Xiao-ying, SHEN Jian-hua, CHEN Kai-xian, SHEN Xu, JIANG Hua-liang. The conserved residue Phe273(282) of PPAR alpha(gamma), beyond the ligand-binding site, functions in binding affinity through solvation effect [J]. Biochimie, 2005, 87(6): 539-550.
[13] RICH R L, MYSZKA D G. A survey of the year 2002 commercial optical biosensor literature [J]. J Mol Recognit, 2003, 16(6): 351-382.
[14] MACKENZIE C R, HIRAMA T, DENG Su-jun, BUNDLE D R, NARANG S A, YOUNG N M. Analysis by surface plasmon resonance of the influence of valence on the ligand binding affinity and kinetics of an anti-carbohydrate antibody [J]. J Biol Chem, 1996, 271(3): 1527-1533.
[15] NIEBA L, KREBBER A, PLUCKTHRN A. Competition BIAcore for measuring true affinities: Large differences from values determined from binding kinetics [J]. Anal Biochem, 1996, 234(3): 155-165.
[16] RICH R L, MYSZKA D G. Survey of the year 2001 commercial optical biosensor literature [J]. J Mol Recognit, 2002, 15(6): 352-376.
[17] TAO Nong-jian , BOUSSAAD S, HUANG W L, ARECHABALETA R A, DAGNESE J. High resolution surface plasmon resonance spectroscopy [J]. Rev Sci Instrum, 1999, 70(12): 4656-4660.
[18] XIN Yin-ling, GAO Yu, GUO Jun, CHEN Qian, XIANG Juan, ZHOU Fei-meng. Real-time detection of Cu2+ sequestration and release by immobilized apo-metallothioneins using SECM combined with SPR [J]. Biosens Bioelectron, 2008, 24(3): 369-375.
[19] XIANG Juan, GUO Jun, ZHOU Fei-meng. Scanning electrochemical microscopy combined with surface plasmon resonance: Studies of localized film thickness variations and molecular conformation changes [J]. Anal Chem, 2006, 78(5): 1418-1424.
[20] ZHAI Pei-min, GUO Jun, XIANG Juan, ZHOU Fei-meng. Electrochemical surface plasmon resonance spectroscopy at bilayered silver/gold films [J]. J Phys Chem C, 2007, 111(2): 981- 986.
[21] SAUERBREY G. Measurement of very small cumplitude vibration of plate by lightbeam modulation [J]. Z Phys, 1964, 178: 457-471. (in Germen)
[22] SAUERBREY G. Use of crystal oscillators for weighing thin films and for microweighing [J]. Z Phys, 1959, 155: 206-222. (in Germen)
[23] ADAMSON A W, GAST A P. Physical Chemistry of Surfaces [M]. 6ed. New York: John Wiley &Sons Inc., 1999: 603-615.
[24] LAHIRI J, LSAACS L, TIEN J, WHITESIDES G M. A strategy for the generation of surfaces presenting ligands for studies of binding based on an active ester as a common reactive intermediate: A surface plasmon resonance study [J]. Anal Chem, 1999, 71(4): 777-790.
[25] RAO Jiang-hong, YAN Lin, XU Bing, WHITESIDES G M. Using surface plasmon resonance to study the binding of vancomycin and its dimer to self-assembled monolayers presenting D-Ala-D-Ala [J]. J Am Chem Soc, 1999, 121(11): 2629-2630.
(Edited by PENG Chao-qun)
Foundation item: Projects(20773165, 20876179) supported by the National Natural Science Foundation of China; Project(09JJ1002) supported by the Hunan Provincial Natural Science Foundation, China; Project(NCET-07-0865) for New Century Excellent Talents in Chinese University; Project(2007AA022006) supported by the National High Technology Research and Development Program of China
Received date: 2010-03-26; Accepted date: 2010-05-19
Corresponding author: XIANG Juan, Professor, PhD; Tel: +86-731-88836954; E-mail: xiangj@mail.csu.edu.cn

