Article ID: 1003-6326(2005)03-0636-06
Microwave synthesis of LiCoO2 cathode materials
YU Yong-li(������)1, ZHAI Xiu-jing(���㾲)2,
FU Yan(�� ��)2, YAO Guang-chun(Ҧ�㴺)2
(1. School of Science, Northeastern University, Shenyang 110004, China;
2. School of Materials and Metallurgy, Northeastern University,
Shenyang 110004, China)
Abstract: LiCoO2 powder used as cathode material for lithium ion battery was synthesized by microwave heating with Co3O4 and LiOH��H2O as starting materials. The selection of output power and heating time of microwave can markedly affect the purity, morphology and electrochemical behaviors of the samples. X-ray diffraction (XRD) patterns display that the samples synthesized at 360W for 10min are pure layered LiCoO2, and SEM photos show that the powders are crystalline with well-defined facets whose sizes are about 5��m. The performance of Co3O4 and starting materials by microwave heating and conventional heating was investigated. It is indicated that Co3O4 decomposes into CoO in microwave field at 750�� and the mechanism of preparing LiCoO2 by microwave heating is different from that by conventional heating. The electrochemical behaviors of samples were tested. As a result, the highest specific discharge capacity is 134.3mAh/g and the coulomb efficiency is 92.56%.
Key words: microwave synthesis; lithium-cobalt-oxygen compound; cathode materials; lithium ion battery CLC
number: TM910.1 Document code: A
1 INTRODUCTION
LiCoO2 is a commercially used cathode material for lithium ion battery. Compared with other cathode materials, such as LiNiO2, LiMn2O4 and their doped compounds, LiCoO2 has the advantages of high potential, excellent reversibility, long cycle life and reliable performance[1-4]. There are mainly several methods for synthesis of LiCoO2, such as conventional ceramic method[5, 6], sol-gel synthesis[7], and low-temperature solution combustion[8]. Among these methods, the conventional ceramic method has been applied to industrial production, but it asks high temperature and long heating.
Microwave heating synthesis has been applied to prepare ceramic materials, nanometer sized powder, superconductors[9-11], etc, since it was invented in 1986. Recently microwave heating has been employed for the preparation of cathode materials for lithium ion battery[12-15]. Unlike the conventional ceramic method microwave heating is selective. The process of material being heated is associated closely with the polarization in molecules of the material, and the polarization in molecules leads to expending of current density in electrical field. The advantages of microwave synthesis are quick, clean, efficient and energy saving. Yan et al[14] have synthesized LiCoO2 with LiCH3COO��2H2O and Co(CH3COO)2��4H2O by microwave heating. Elumalap et al[15] mixed Co(NO3)2 and LiOH2��H2O with water to prepare LiCoO2. Compared with organics, Co3O4 is cheap and can absorb microwave effectively. In this paper, we use Co3O4 and LiOH��H2O as starting materials to synthesize LiCoO2, and then do some feasible research on industrial production of the compounds by microwave energy. Additionally the preparing mechanism of LiCoO2 in microwave field was revealed.
2 EXPERIMENTAL
2.1 Preparation of LiCoO2
Co3O4 and LiOH��H2O (AR grade) were weighed accurately with a mole ratio of Co/Li=1��1 and mixed in an agate mortar thoroughly. The sample was put into a porcelain crucible, and then the crucible was placed into a microwave oven with 2.45GHz. At a proper output power the crucible was radiated for some time. After reaction, the product was cooled to room temperature in the oven. The as-prepared sample was poured into an agate mortar and ground. The final product was characterized by D/max-RB powder X-ray diffractometer and Shimadzu SSX-550 scanning electron microscope.
During the reaction, if the temperature measurement was needed, turned off the microwave oven and put a thermocouple into the sample as soon as the crucible was taken out. Temperature reading was done by a data display temperature meter.
The preparation of LiCoO2 by conventional ceramic method was carried out in a box resistant furnace.
2.2 Constant current charge-discharge test
The electrochemical characteristics of samples were studied using a two-electrode cell. LiCoO2, carbon black and PVDF with a mass ratio of 85��10��5 were mixed with N-Methyl-2-ketopyrrolidine to form syrupy mixture. The mixture was repeatedly spread on to a nickel grid so as to obtain a uniform coating and finally dried at 120�� for 8h under vacuum. The electrodes thus prepared were compacted at 30MPa pressure and then transferred into an argon filled glove box. The cell was assembled in the box with a lithium metal sheet as the counter electrode and a polypropylene film (Celgard) as the inter-electrode separator. The electrolyte consisted of 1mol/L LiPF6 in an ethylene carbonate (EC) and dimethyl carbonate (DMC) mixture (volume ratio 1��1). The constant current charge-discharge experiment was carried out with a battery-testing instrument and the current is 0.5mA/cm2.
3 RESULTS AND DISCUSSION
3.1 Preparation of LiCoO2
Firstly, the performance of Co3O4 and LiOH��H2O mixture in microwave field was examined. At different output powers the mixture was irradiated for different times and the temperature was detected. The results are shown in Fig.1. It is seen that the temperature increases sharply at the beginning of heating, and then the curves are flat. At different output powers, the temperature increases at different rates. The higher the output power is, the higher the rate is. Because microwave selectively heats the starting materials, Co3O4 can absorb microwave efficiently as cobalt is transition metal, but LiOH��H2O can not. So the temperature elevation of starting materials is chiefly due to Co3O4 , and LiOH��H2O is heated indirectly. Generally LiCoO2 is synthesized at 750-850��. As seen in Fig.1, the proper synthesizing time for LiCoO2 at different power can be determined. At power 720, 540 and 360W, the proper time is about 2, 6 and 10min respectively. At power 180W, even the heating time is 16min, the temperature is only about 300��. This indicates that the power is too low to synthesize LiCoO2.
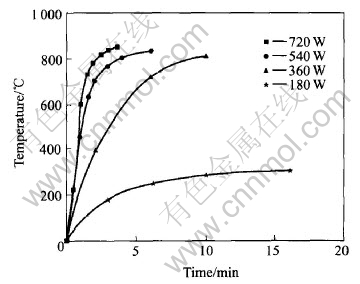
Fig.1 Temperature elevation of starting materials in microwave field
The performance of LiCoO2 in microwave field was studied too. LiCoO2 can absorb microwave efficiently, but the temperature does not increase as fast as the starting materials. The trend of temperature elevation curves is similar to that of Co3O4 and LiOH��H2O mixture.
Samples prepared at different powers for different heating times were characterized by X-ray diffractometer, the results are shown in Fig.2. It is observed that at 720W there are impurity peaks at 2�ȡ�42�� and 61�� in the pattern, and at 540W and 360W the patterns are identical with reference standard��s. This shows that the samples prepared at 540W and 360W are pure layered LiCoO2.
LiCoO2 can be synthesized with Co3O4 and
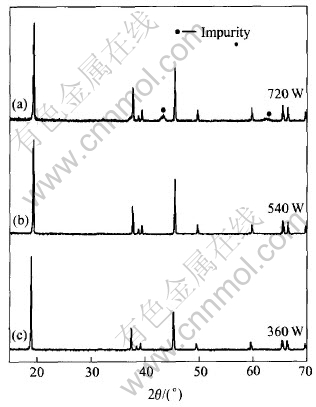
Fig.2 XRD patterns of samples prepared at different power
LiOH��H2O at proper temperature for enough reaction time. Although the temperature of starting materials after being heated for 2min at 720W is about 800��, at which LiCoO2 can be prepared, 2min reaction time is too short to complete the synthesis of LiCoO2 , which leads to the impurity in the sample. Slight sinter of samples is observed for longer heating time at such power, thus a lower power is suggested.
At 360W power the starting materials were irradiated for 4, 6, 8, 10min respectively and the as-prepared samples were characterized by XRD. The XRD patterns are shown in Fig.3. For the heating time 4 or 6min, there are impurity peaks, which are attributed to low reaction temperature. For heating time of 8 and 10min the samples are pure layered LiCoO2.
The scanning electron microscopy (SEM) photos of the above four samples are shown in Fig.4. The particles become larger as the heating time increases. For heating time of 8 and 10min the particles have well-defined facets.
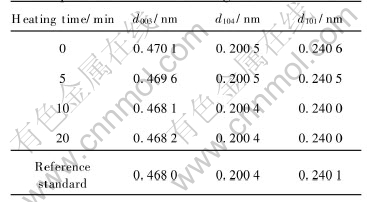
Additionally, the crystallinity of the four samples increases with the heating time increasing, as shown in Table 1. The three strongest peaks of LiCoO2 are (003), (104) and (101) peaks. The relative crystallinity of a sample is calculated as
R=h003w003+h104w104+h101w101(1)

Fig.3 XRD patterns of samples prepared with different microwave heating times

Fig.4 SEM photos of samples prepared with different heating times
Table 1 Relative crystallinity of samples prepared with different heating times

where h is the height and w is the half-peak width of the peak, and R is relative crystallinity. The high crystallinity represents a well developed crystalline, which is beneficial to good electrochemical behaviors of LiCoO2.
After the starting materials react completely, it is necessary to keep samples at proper temperature for some time in order to obtain well developed crystallinity. It is seen in Fig.1 that, the temperature may become not suitable to synthesize LiCoO2 if the heating time is too long at 720, 540 and 360W. At 180W the samples can absorb microwave but the temperature can retain stable. In the experiment, the synthesized LiCoO2 are heated for some time at 180W to improve their crystallinity.
The mixture of starting materials was heated for 10min at power 360W and irradiated for another 5min at 180W, then it was cooled to room temperature naturally. Using the above method the other two samples were prepared but the irradiating time at 180W was 10 and 20min instead. The as-prepared samples were characterized by XRD. The XRD patterns show that all samples are pure layered LiCoO2, and the d values of (003), (104) and (101) peaks are listed in Table 2. It is seen that the d values decrease for heating time from 0min to 10min and tend to reference standard��s, and the d values for heating 20min are the same as those for heating 10min.
Table 2 Comparison of d values of samples for different heating times at 180W
3.2 Mechanism of synthesizing LiCoO2 by microwave
Co3O4 was heated by microwave at 360W for 4min and the detected temperature is about 750��. It was observed that the color of the sample was light brown. This suggests that Co3O4 has decomposed as the color of Co3O4 is black. The sample was characterized by XRD, as shown in Fig.5(a). There are two other peaks at 2��=42.37�� and 61.47�� which do not belong to Co3O4. These two peaks are indexed to peaks of CoO. This indicates that by microwave heating Co3O4 decomposes into CoO at 750��. By conventional heating no change of Co3O4 is observed until 900��, and its XRD pattern is identical to the reference standard of Co3O4(as seen in Fig.5(b)). It is suggested that the performance of Co3O4 in microwave field is different from that by conventional heating.
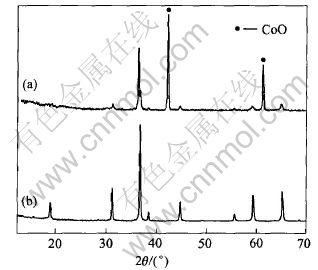
Fig.5 XRD patterns of Co3O4 by microwave heating at 750��(a) and conventional heating at 900��(b)
At 360W the starting materials were heated for 4, 6, 8 and 10min respectively. The color of the first two samples was light brown and the last two samples were blue-grey. The XRD patterns of the four samples are shown in Fig.3. Certainly the impurity peaks belong to CoO. This indicates that during synthesizing LiCoO2 the following reactions must occur:
2Co3O4��6CoO+O2
4CoO+O2+4LiOH��4LiCoO2+2H2O
By conventional heating the starting materials were heated at 350, 400, 450, 500, 700, 750�� respectively, and the XRD patterns are shown in Fig.6. It is seen that there are only peaks of starting materials and LiCoO2 but no CoO peaks. By conventional heating the synthesis reaction is: 4Co3O4+O2+12LiOH[FY]12LiCoO2+6H2O. This indicates that the mechanism of synthesizing LiCoO2 by conventional heating is different from that by microwave heating. By the conventional heating both of the samples prepared at 700�� and 750�� are LiCoO2, but the first one is spinel-like structure(LT-LiCoO2) and the other is ideal layered structure(HT-LiCoO2).
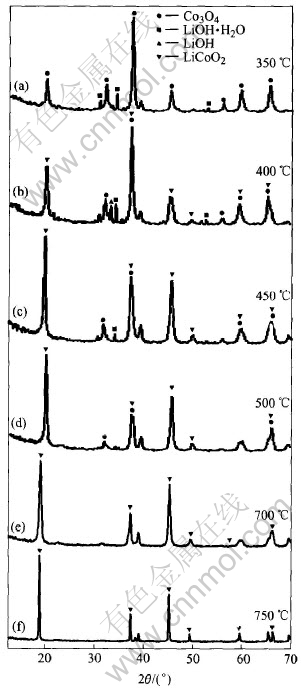
Fig.6 XRD patterns of samples prepared by conventional heating
The temperature elevation mechanism of material by conventional heating is different from that in microwave field. In microwave field the material is heated from inner of molecules that are polarized by microwave. Besides temperature elevation the material could obtain microwave effect, which is different from that by conventional heating. It is a very good example of microwave effect that Co3O4 decomposes at 750�� in microwave field and has no change until 900�� by conventional heating.
3.3 Electrochemical behaviors of LiCoO2
The electrochemical characterization of the four samples prepared at different powers for different heating times was carried out at 3.0-4.2V, as seen in Fig.7. The discharge capacity of the sample for profile a is as high as 134.3mAh/g because the crystallinity of the sample has developed well and the charge-discharge efficiency is 92.56%. The discharge capacity of the sample for profile d is the lowest. Thus it is very important to control proper output power and heating time of microwave so as to synthesize LiCoO2 with good electrochemical behaviors. After 10 cycles the discharge capacity of the sample for profile a is 118mAh/g. There is a 12% capacity decrement at the end of 10 cycles.
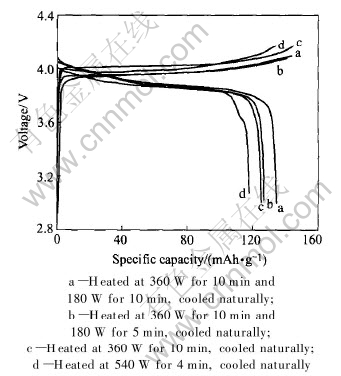
Fig.7 Charge and discharge curves of prepared samples
4 CONCLUSION
By microwave heating the pure layered LiCoO2 was synthesized with Co3O4 and LiOH��H2O as the starting materials and the samples were characterized by X-ray diffractometer and scanning electron microscope. It is indicated that the output power and heating time of microwave have great effect on the purity, morphology and electrochemical behaviors of samples. The performance of Co3O4 and the starting materials by microwave heating and conventional heating was examined. It is found that Co3O4 decomposes into CoO in microwave field at 750�� and no change occurs by conventional heating until 900��, and the synthesis of LiCoO2 in microwave field is two-step reaction. The electrochemical behaviors of samples were tested. The sample prepared at 360W for 10min and 180W for another 10min and then cooled naturally has the highest discharge capacity of 134.3mAh/g and the charge-discharge efficiency of 92.56%. After 10 cycles there is a 12% discharge capacity decrement. It is shown that synthesizing LiCoO2 by microwave heating is a clear, quick and energy saving method.
REFERENCES
[1]Ritchie A G. Recent developments and future prospects for lithium rechargeable batteries[J]. Journal of Power Sources, 2001, 96(1): 1-4.
[2]XU C Q, TIAN Y W, ZHAI Y C, et al. Synthesis and character of spinel LiMn2O4[J]. Trans Nonferrous Met Soc China, 2004, 14(3): 470-474.
[3]YING J R, JIANG C R, WAN C R. Preparation and characterization of high-density spherical LiCoO2 cathode material for lithium ion batteries[J]. Journal of Power Sources, 2004, 129(2): 264-269.
[4]Wang M J, Navrotsky A. Enthalpy of formation of LiNiO2, LiCoO2 and their solid solution, LiNi1-x-CoxO2[J]. Solid State Ionics, 2004, 166(1-2): 167-173.
[5]WANG J, QI L, KE K, et al. Structure and electrochemical performance studies of LiCoO2 Cathode material prepared by new mathod[J]. Chinese J Inorg Chem, 2004, 20(6): 635-640. (in Chinese)
[6]Imanishi N, Fujji M, Hirano A, et al. Synthesis and characterization of nonstoichiometric LiCoO2[J]. Journal of Power Sources, 2001, 97-98(6): 287-289.
[7]Nieto-Ramos S, Tomar M S. Synthesis of lithium intercalation materials for rechargeable battery[J]. International Journal of Hydrogen Energy, 2001, 26(2): 159-163.
[8]Lu C H, Yeh P Y. Microstructural development and electrochemical characteristics of lithium cobalt oxide powders prepared by the water-in-oil emulsion process[J]. Journal of the European Ceramic Society, 2002, 22(5): 673-679.
[9]Ganesh I, Srinivas B, Johnson R, et al. Microwave assisted solid state reaction synthesis of MgAl2O4 spinel powders[J]. Journal of European Ceramic Society, 2004, 24(2): 201-207.
[10]Cherian M, Raoa M S, Manoharan S S, et al. Influence of the fuel used in the microwave synthesis of Cr2O3[J]. Topics in Catalysis, 2002,18(3/4): 225-230.
[11]Serrano D P, Uguina M A, Sanz R, et al. Synthesis and crystallization mechanism of zeolite TS-2 by microwave and conventional heating[J]. Microporous and Mesoporous Materials, 2004, 69(3): 197-208.
[12]YANG S T, JIA J H, DING L, et al. Studies of structure and cycleability of LiMn2O4 and LiNd0.01Mn1.99O4 as cathode for Li-ion batteries[J]. Electrochimica Acta, 2003,48(5): 569-573.
[13]Whitfield P S, Davidson I J. Microwave synthesis of Li1.025Mn1.975O4 and Li1+xO4-yFy[J]. J Electrochem Soc, 2000, 147(12): 4476-4484.
[14]YAN H W, HUANG X J, LU Z H, et al. Microwave synthesis of LiCoO2 cathode materials[J]. Journal of Power Sources, 1997, 68(2): 530-532.
[15]Elumalai P, Vasan H N, Munichandraiah N. Microwave synthesis and electrochemical properties of LiCo1-xMxO2(M=Al and Mg) cathodes for Li-ion rechargeable batteries[J]. Journal of Power Sources, 2004, 125(1): 77-84.
(Edited by YUAN Sai-qian)
Foundation item: Project(50174016) supported by the National Natural Science Foundation of China
Received date: 2004-10-10; Accepted date: 2005-03-04
Correspondence: ZHAI Xiu-jing, Professor, PhD; Tel: +86-24-83678296; E-mail: yongli200004@163.com