
Thermodynamic analysis of production of high purity titanium by thermal decomposition of titanium iodide
CHEN Xiao-hu(陈肖虎)1, 2, WANG Hua(王 华)1, LIU Yi-min(刘义敏)2, FANG Min(方 敏)2
1. Faculty of Materials and Metallurgy Engineering, Kunming University of Science and Technology,
Kunming 650093, China;
2. Materials and Metallurgy College, Guizhou University, Guiyang 550003, China
Received 17 June 2009; accepted 15 August 2009
Abstract: High purity titanium was prepared by thermal decomposition of titanium iodide. The feasible synthetic route and optimum decompositon temperaure were obtained by thermodynamic analysis in the process of thermal decomposition of titanium iodide and nucleation growth theory. The temperature for the formation of titanium iodide is in the range of 800-900 K, at which a large amount of titanium iodide vapour can be obtained. The decomposition temperature of titanium iodide is in the range of 1 300-1 500 K, at which a favourable decomposition rate can be achieved. The experiment results show that the purity of the produced titanium is more than 99.995%.
Key words: high purity titanium; titanium iodide; thermal decomposition
1 Introduction
The high purity titanium was currently used as sputtering target materials in the preparation of control electrode of large-scale integrated circuits, diffusion impervious layer and wiring materials due to its excellent specific strength, workability and corrosion resistance. The impurities in the titanium will result in performance reduction of the materials, so it is necessary to remove the impurities in the titanium used as sputtering target materials[1-4]. The preparation of high purity titanium has been deeply researched in USA and Japan[5-9], but there are few reports on this field in China. The methods to prepare high purity titanium involve Kroll process, iodization process and molten salt electrolysis process [10-13].
A conventional method for obtaining high purity titanium is the iodide thermal decomposition process (also known as the iodization process). In the process, the titanium tetra-iodide(TiI4) is synthesized firstly in a closed container at 500-700 K by using sponge titanium and iodine as raw materials, and then the synthesized TiI4 is heated to 1 600-1 800 K on the titanium wire at the center of the reactor to deposit high purity titanium.
The iodine produced as a by-product by the thermal decomposition reaction can be cyclically used in the synthesis of titanium iodide. The reactions in the process can be written as:
Synthesis reaction
Ti(crude)+2I2→TiI4;
Thermal decomposition reaction
TiI4→2I2+Ti (high purity)
During the synthesis of titanium iodide, there exist two kinds of compounds, TiI2 and TiI4. The synthesis of higher-valent iodide(TiI4) can be completed at lower temperature (500-700 K), but it must be decomposed at higher temperature (1 600-1 800 K). While the synthesis of lower-valent iodide(TiI2) must be performed at higher temperature(1 000-1 200 K), but it can be decomposed at relatively low temperature (1 300-1 500 K)[14-15]. Accordingly, the thermal decomposition of the metal impurities contained in the lower valent titanium iodides used as the gas source for depositing titanium is hindered, thereby eliminating the possibility that any metal impurities might be deposited on the titanium[16-18]. Under optimal process conditions, higher synthesis rate and decomposition rate of titanium iodides can be obtained, while that of impurities in the raw titanium can be controlled at a low level. So the impurities cannot deposit on the titanium and a high purity titanium is produced.
In this work, high purity titanium is obtained using sponge titanium and iodine as raw materials with the self-made apparatus. Based on the thermodynamic analysis on the process of thermal decomposition of titanium iodides and nucleation growth theory, the optimum conditions of preparing high purity titanium are obtained.
2 Thermodynamic analysis
2.1 Thermodynamic analysis of titanium iodide reactions
Assuming there are equilibriums in the iodization process and the thermal decomposition process, the relationships between Gibbs free energies and equilibrium constants of the reactions (1) and (2) can be determined based on the thermodynamic data of iodine, titanium and titanium iodide as shown in Table 1.
2I2(g)+Ti(s)?TiI4(g) (1)
TiI4(g)+Ti(s)?2TiI2(g) (2)
Table 1 Thermodynamic parameters of some matters[16]

The relationship between Gibbs free energy 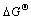 and reaction temperature T can be determined by the following equation:
and reaction temperature T can be determined by the following equation:
 =-415.36+0.391T-11.046×10-3TlnT+
=-415.36+0.391T-11.046×10-3TlnT+
5.69×10-6T2+1.061×102T-1 (3)
Also, the Gibbs free energy of the reactions (2) can be determined by the following equation approximately:
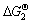 =170.43-0.384T+5.53×10-3TlnT+5.142×10-6T2-
=170.43-0.384T+5.53×10-3TlnT+5.142×10-6T2-
0.115×102T-1 (4)
According to the classical thermodynamic theory, the relationship between the equilibrium constant Kp and 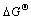 can be determined by the following equation:
can be determined by the following equation:
lgKp=-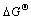 /2.303RT (5)
/2.303RT (5)
Based on Eq.(5), the reaction temperature T can be calculated with Kp and 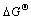 .
.
The reactions (1) and (2) are expected to proceed to right to form much amounts of iodides including TiI4 and TiI2 in the iodide source area. However, it is expected to deposit titanium as much as possible on the substrate in the deposition area, where a low concentration of TiI4 is needed. If the temperature of the iodide area is T2 and the temperature of the deposition area is T1, the difference ?T=|T2-T1| should be low enough to ensure the reversibility of the reactions (1) and (2) changing with the operating temperature, i.e. lg Kp≈0.
The temperature can be calculated according to Eq.(5) when lg Kp=0. Then, the temperature T2 in the case of lg Kp>0 is beneficial for synthesizing iodides in the iodide source area; in contrary, in the case of lg Kp<0, the decomposition reaction in the deposition area is preferential.
By plotting the equilibrium constants (Kp) against the reaction temperatures T, two curves are obtained as shown in Fig.1.

Fig.1 Relationship between lgKp and T
As shown in Fig.1, curve 1 and curve 2 cross at lg Kp=8 (marked by a dashed line). In the upside of the dashed line , Kp1>Kp2 (Kp1 and Kp2 are the equilibrium constants of reactions (1) and (2) respectively), the reaction (1) moves right to produce more TiI4 and thus more TiI2; in the downside of the dashed line, Kp1<Kp2, reaction (2) proceeds to the left, thus TiI2 decomposes to deposit titanium on the deposition substrate. So, the downside zone of the dashed line is the favorable deposition area, and the upside zone of the dashed line is the favorable iodide area.
2.2 Thermodynamic analysis of nucleation
The thermal decomposition process of iodides can be expressed by the following reactions:
2TiI2(g)→Ti(g)+TiI4(g) (6)
Ti(g, p)→Ti(s, p0) (7)
According to the classical nucleation theory, under the condition of homogeneous nucleation and spherical nucleus, the activation energy of nucleation process may be defined as:
 (8)
(8)
where V is the molar volume of titanium; γ is the surface tension of titanium; T is the temperature; p is the partial pressure of gaseous titanium; p0 is the vapour pressure of solid titanium; p/p0 is the vapour supersaturation[19-20]. So, under steady state, the nucleation speed and the critical nucleation diameter can be determined by the following equations:
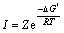 (9)
(9)
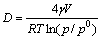 (10)
(10)
where Z and R are constants. As for titanium, the molar volume V is 10.64×10-6 m3/mol, and the surface tension γ is 1.588 J/m2. When the diameter of nucleation is smaller than the critical diameter, the newly formed nucleation is unstable which can be dissolved again; when the diameter of nucleation is larger than the critical diameter, the nucleation can grow.
For reaction (6), p can be calculated by the following equations:
?G=170.43-0.984T+5.53TlnT×10-3+5.142T2×10-6-
11.5T-1 (11)
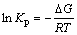 (12)
(12)
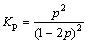 (13)
(13)
p0 can be calculated by the following equation:
lg(p0/Pa)=141.8-3.23×105T-1-0.0306T
(1 200-2 000K) (14)
Fig.2 shows the relationship between the nucleation speed I and the vapour supersaturation p/p0, and Fig.3 shows the relationship between critical nucleation diameter D and vapour supersaturation p/p0.
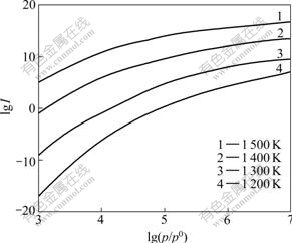
Fig.2 Relationship between nucleation speed and vapour supersaturation
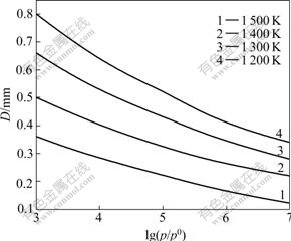
Fig.3 Relationship between critical nucleation diameter and vapour supersaturation
As shown in Fig.2 and Fig.3, in the temperature range of 1 200-1 500 K, the faster nucleation speed I and smaller critical nucleation diameter D can be obtained simultaneously at proper vapor supersaturation, which verifies that a fast deposition rate can be guaranteed under this operating condition.
3 Results and discussion
During the preparation process of high purity titanium, the reaction (1) of crude titanium with iodine to form TiI4 can begin at temperatures of 500-600 K in the iodide source area, and the reaction (2) of titanium with titanium tetra-iodide to form TiI2 can occur at 700-900 K. In the experiments, the temperatures of iodide source area was controlled at 800-900 K, at which the iodides can be kept in gaseous form. The reaction (6) of TiI2 thermal decomposition initiated at 1 300-1 500 K, by which high purity titanium deposited on the substrate in the deposition area. The iodine and iodide produced by the thermal decomposition reaction were then recycled for reaction with crude titanium[8-9].
Fig.4 shows the SEM images of sectional morphology of high purity titanium as-prepared. Fig.4(a) displays the junction between the deposition substrate and columnar crystals. There is a cluster of columnar crystals, which is the typical phenomenon in the crystallization of pure metals. These compact columnar crystals radially grow in the axial direction.

Fig.4 SEM images of sectional morphology of high purity titanium as prepared
Figs.4(b) and (c) show the gap between two columnar crystals. The impurities in the gap can be seen clearly, which is the main factor affecting the purity of titanium.
Fig.5 shows the chemical ingredients of the impurities in the gap between columnar crystals by EDS. And the analysis data of the impurities are listed in Table 2.
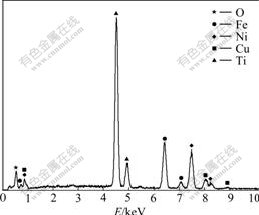
Fig.5 EDS spectrum of impurities in gap between columnar crystals
Table 2 Chemical composition of gap between columnar crystals analysed by EDS

As shown in Fig.5 and Table 2, there are elements of O, Fe, Ni, Cu and Ti existing in the gap. The high purity titanium produced by the thermal decomposition of titanium iodides is mainly composed of columnar crystals. In the columnar crystals, there is almost no impurities. When the crystal grains grow in different directions intersect, at the boundary there exist impurities, which mainly come from the crude titanium by iodide reaction of impurities. Table 3 lists the average chemical compositions of the produced high purity titanium by GDMS analyses. Table 4 lists the standard impurities contents of 4N5 titanium abroad.
Table 3 Chemical compositions of produced high purity titanium by GDMS analyses

Table 4 Standard impurity contents of 4N5 titanium abroad
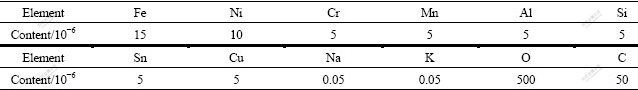
From Table 3, the total impurities content in the product is 42.2×10-6. Except for the elements of Fe, Ni and Mn, the rest are in accordance with the standard of 4N5 titanium abroad. These impurities of Fe, Ni and Mn mainly come from the etching of equipment, including the reactor and the vacuum pipeline, which can be readily eliminated by electron beam smelting in the following process.
4 Conclusions
1) Based on the analyses of the thermodynamics and nucleation theories, the optimum temperature ranges of synthesis and decomposition reaction for preparing highly pure titanium by iodization process were obtained.
2) The temperature for synthesizing titanium iodides should be controlled at higher level such as 800-900 K, which can ensure the iodide gas formation rate and thus accelerate the iodide process. The thermal decomposition of titanium iodides in the deposition area should be controlled at a lower temperature in order to get faster decomposition rates. The suitable temperature should be at 1 300-1 500 K. Under this temperature, a faster nuclei forming rates of titanium can be obtained.
3) The high purity titanium with a purity of 99.995% was obtained by the thermal decomposition process of titanium iodide.
References
[1] WU Q X, Preparation of high purity titanium [J]. Processing Technology, 1996(5): 14-16. (in Chinese)
[2] LI Z, GUO S M. The direction of development and preparation of high purity titanium [J]. Titanium Industry Progress, 1997(3): 20-24. (in Chinese)
[3] DENG G Z. Titanium metallurgy progress and the development discussion [J]. Precious Metals, 2002, 26(5): 391-396.
[4] SHI Y J. The production and application of high purity titanium [J]. Shanghai Metal (Nonferrous Fascicule), 1993, 14(6): 30-37. (in Chinese)
[5] MA W Y. The current situation of high purity metals in foreign countries [J]. Chinese Journal of Rare Metals, 1994, 18(1): 59. (in Chinese)
[6] ZHANG W M. The application and manufacturing technology of high purity titanium [J]. Journal of Foreign Metal Working, 2000(3): 25-28. (in Chinese)
[7] HARRY R, NIGEL W. Apparatus for producing titanium crystal and titanium: US6024847 [P]. 1998-02-02.
[8] EIJI N, MASAMI K. Method and apparatus for producing high purity titanium: US5336278 [P]. 1994-08-09.
[9] CUEVAS F, FERNANDEZ J F, SANCHEZ C. Kinetics of the iodide titanium process by the thermal decomposition of titanium tetraiodide [J]. Journal of the Electrochemical Society, 2000, 147(7): 2589-2596.
[10] LIU Z H, CHEN Z Q. The producing methods and application of high purity titanium [J]. Rare Metals Letters, 2008, 27(2): 1-8. (in Chinese)
[11] GAO X J. Preparation of high purity titanium by a novel iodide process [J]. Titanium Industry Progress, 1995(4): 19-20. (in Chinese)
[12] LI Y G. Refining high purity titanium by iodide [J]. World Non-ferrous Metals, 2003(12): 25-27. (in Chinese)
[13] YASUNORI Y, YASUHIDE I. Method for obtaining high purity titanium: US5232485 [P]. 1993-08-03.
[14] PAN W, HUANG Q, CHEN J. Mechanism of titanium deposition on A12O3 ceramic surface by molten salt reaction [J]. Mater Lett, 1997, 31: 317-320.
[15] KOLTHOFF M, THOMAS J. Polarography in cetonitrile of titanium tetrachloride and tetraiodide in various supporting electrolytes [J]. Electrochem Soc, 1964, 111(9): 1065-1074.
[16] LIANG Y J, CHE Y C. Thermodynamics data enchiridion of inorganic matters [M]. Shenyang: Northeastern University Press, 1993: 191, 372, 376-378. (in Chinese)
[17] ZHANG C R, LIU R J, CAO Y B. Effects of deposition temperature on the microstructures of SiC coatings by CVD [J]. Journal of Inorganic Materials, 2007, 22(1): 153-158. (in Chinese)
[18] LI Y G. Iodide refining with a high purity titanium [J]. World Non-ferrous Metals, 2003(12): 25-27. (in Chinese)
[19] LI H P. High purity titanium [J]. Shanghai Steel Research, 2003(1): 60-61. (in Chinese)
[20] LEONE O Q, KNUDSEN H, COUCH D E. High purity titanium electrowon from titanium tetrachloride [J]. JOM, 1967, 19(3): 18-23.
Foundation item: Project supported by Zunyi Titanium Co., Ltd.
Corresponding author: CHEN Xiao-hu; Tel: +86-13037825668; E-mail: cxhlxd@126.com
DOI: 10.1016/S1003-6326(08)60448-3
(Edited by YUAN Sai-qian)