Article ID: 1003-6326(2005)05-1103-05
Nanoparticles α-Al2O3 prepared by
combustion synthesis method
CHEN Kai-jun(陈凯军)1, ZHAI Xiu-jing(翟秀静)1,
CHU Gang(储 刚)1,2, FU Yan(符 岩)1,
ZHANG Xiao-shun(张晓顺)1, BAI Bin(白 斌)1
(1. School of Material and Metallurgy, Northeastern University, Shenyang 110004, China;
2. Department of Applied Chemistry, Fushun Petroleum Institute, Fushun 113001, China)
Abstract: Nanometer α-Al2O3 powders were synthesized by the method of low-temperature combustion synthesis (LCS) with aluminum nitrate nonahydrate and urea as raw materials. The prepared powders were studied by XRD, TG-DTA, FT-IR and TEM. It is found that the average size of particles is 60-80nm. The optimal synthetic conditions are obtained, i.e., the suitable fuel is urea; the molar ratio of oxidizer to fuel is 1∶2 and the igniting temperature is 700℃. The results show that the size of particles is governed by synthesizing temperature, the fuel and the molar ratio of oxidizer to fuel. TEM image of the particles collected shows that the crystal habits of particles have a spheric structure and particles are polycrystal.
Key words: low-temperature combustion synthesis; nanoparticle; α-Al2O3 powder CLC number: TF123.7+2
Document code: A
1 INTRODUCTION
Recent progress in the preparation and characterization of materials on the nanometer size scale has introduced a new point of view for physics and chemistry in the world about materials with grain sizes(d) less than 100nm because of the expectation of their unique properties.
The nanometer alumina (Al2O3) powders are very important materials because of their interesting properties and many technological applications[1-3], such as in ceramics materials, optical materials, catalyst materials and structure materials. There are many manufacturing technologies to be used to produce nanometer alumina, such as gel-sol method, vapor deposition method and hydrothermal process[4-6]. Low-temperature combustion synthesis (LCS) method[7] utilized the exothermicity of the reaction between an oxidizer and a fuel to directly synthesize the oxide phase from the molecular mixture of the precursor solution. The combustion reaction was initiated by bring the exoergic materials up to the ignition temperature whereupon combustion occurred simultaneously throughout the sample in a thermal explosion[8, 9]. Advantages of LCS method included: 1) higher purity of products; 2) low energy requirements; 3) relative simplicity and high efficiency of the process; and 4) cheaper raw materials[10].
In this work, the nano-structured alumina powder has been synthesized from aluminum nitrate nonahydrate by LCS using urea as fuel.
2 EXPERIMENTAL
In order to choose the suitable fuel, different chemicals such as urea, citric acid and ammonium oxalate were used as fuels for the combustion processes.
Further study was carried out to investigate the reaction using aluminum nitrate nonahydrate and urea as starting materials. Stoichiometric contents of aluminum nitrate nonahydrate and urea were dissolved in distilled water in a crucible. The crucible containing the resultant solution was placed in a muffle furnace which was pre-heated to the given temperature. During the heating, the solution in the crucible was evaporated and got boiling, releasing large amount of gases. Upon continued heating, the solution suddenly foamed up and caught fire, leaving a finely powdered mass in the form of a very low density sponge. The grain size, morphology and microstructure of the product were studied for different molar ratios of Al(NO3)3・9H2O to CO(NH2)2 and igniting temperatures.
The prepared powders were characterized by XRD(D/max-RB), TG-DTA(Universal V2.6D TA Instruments), FT-IR(Perkin-Ecmer2000) and TEM (PHILIPS EM420) techniques.
3 RESULTS AND DISCUSSION
3.1 Choice of fuel
Alumina has been previously synthesized by combustion synthesis process using various fuels, such as urea, citric acid, glycine and glycol[11]. It is noted that the choice of fuel alters the exothermicity of the combustion reaction, which has a strong influence on the properties of the product[12].
Firstly, citric acid is used as fuel. Various molar ratios of Al (NO3)3・9H2O to C6H8O7・H2O (6∶5(E1), 6∶3(E2), and 6∶1(E3)) are used for reactions at 600℃. Fig.1 indicates that all the powders synthesized with citric acid as fuel are amorphous alumina.

Fig.1 XRD patterns of powders synthesized using citric acid as fuel
Similar experiments that use ammonium oxalate as fuel are carried out. The reactions are ignited at 600℃ with Al (NO3)3・9H2O/(NH4)2C2O4・H2O molar ratios of 8∶15(E4), 4∶3(E5) and 16∶3(E6). The powders prepared using ammonium oxalate as fuel are amorphous alumina, as shown in Fig.2.
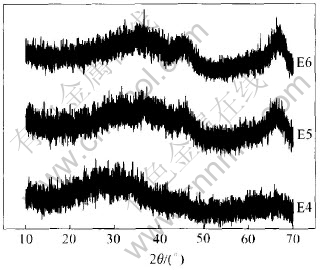
Fig.2 XRD patterns of powders synthesized using ammonium oxalate as fuel
Finally, using urea as fuel, α-Al2O3 powders are obtained. Fig.3 shows the XRD pattern of the powder prepared with molar ratio of Al (NO3)3・9H2O to CO(NH2)2 of 1∶2.5. In comparison with the XRD data of α-Al2O3 obtained from PDF card (No. 81-1667), all the XRD peaks are consistent with peak position of α-Al2O3 phase.

Fig.3 XRD pattern of powders synthesized using urea as fuel
As we know, α-Al2O3 synthesized by sintering of Al(OH)3 requires temperature above 1200℃. The energy generated is not enough in combustion reactions using citric acid or ammonium oxalate as fuel. Therefore, urea is selected as the suitable fuel, for the higher temperature (about 1400℃) is achieved in the reaction[13, 14].
3.2 Effect of molar ratios of Al(NO3)3・9H2O to CO(NH2)2
The molar ratio of aluminum nitrate nonahydrate to urea plays an important role in controlling the morphology and gain size of the synthesized powder. Given the exothermic redox reaction yields N2, H2O and CO2, the stoichiometric composition is calculated to be 1∶1.25 on the basis of the equivalent ratio theory. However, this excludes the possibility of formation of NO or NO2 in the process[15]. Therefore various oxidizers to fuel molar ratios (1∶1, 1∶1.5, 1∶2, 1∶2.5, 1∶3 and 1∶3.5) are used for reactions at 750℃. The XRD analysis of the as-prepared powder shows that the generation of yellowy amorphous alumina powders is found when ratios are 1∶1 and 1∶1.5 (Figs.4(a) and (b)), whereas if ratios are 1∶3 and 1∶3.5 (Figs.4(e) and (f)) gray nano-crystalline alumina powders are yielded. Only the ratio is 1∶2 or 1∶2.5 (Figs.4(c) and (d)), white foamy nano-crystalline alumina powders can be obtained. Fig.4 shows the XRD pattern of the prepared alumina powders. Because of the relatively smaller particle size, 1∶2 is the preferred molar ratio of the staring materials.
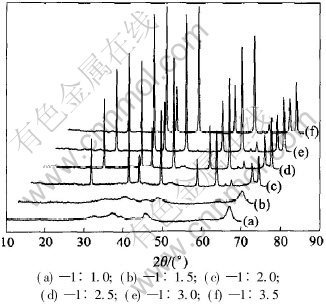
Fig.4 XRD patterns of prepared alumina powders prepared with different Al(NO3)3・9H2O/CO(NH2)2 molar ratios
3.3 Effect of different igniting temperatures
Igniting temperature is another important factor to the reaction. Elevating igniting temperature from 400 to 850℃ with the optimal molar ratio is used to investigate the influence of the temperature.
Fig.5 shows that amorphous alumina powders are obtained when the igniting temperature is 400℃.Only when the temperature is above 400℃ nano-crystalline alumina powders can be obtained. Particle size calculated by the Scherrer formula is listed in Table 1. Because of the relative smaller particle size and high speed of the reaction, 700℃ is the optimal igniting temperature.
Table 1 Particle size of powders prepared with different igniting temperatures
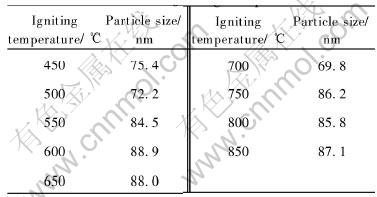
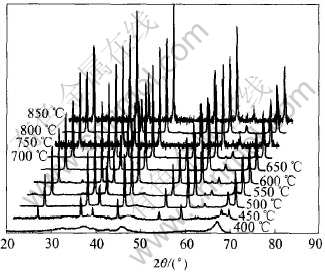
Fig.5 XRD patterns of alumina powders prepared at different igniting temperatures
3.4 Thermal investigation
Fig.6 shows the TG-DTA curves of the mixture of Al(NO3)3・9H2O and CO(NH2)2. The measurements are performed at the rate of 5℃/ min in air.
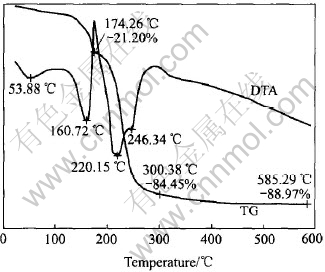
Fig.6 TG-DTA curves for mixture of Al(NO3)3・9H2O and CO(NH2)2
The TGA curve shows that a rapid mass loss occurs between 150℃ and 300℃, which is caused by the large amount of gas generated during the decomposition of the mixture. And 21.20 % mass loss at 174.26℃ is due to the decomposition of urea.
In the TG curve, the endothermic peak at 53.88℃ is attributed to the melting and decomposition reaction of Al(NO3)3・9H2O:
Al(NO3)3・9H2O→
Al(NO3)3・6H2O+3H2O
Subsequently, basic nitrate (Al(NO3)3・3Al(OH)3・5/2H2O) is formed at 160.72℃.
The decomposition of CO(NH2)2 is found to be an exothermic course and its reaction temperature is about 180℃. On the further heating process, (HNO)3 is formed at 220.15℃ and 246.34℃[16].
3.5 FT-IR analysis
Fig.7 illustrates the infrared spectrum of the prepared powders ignited at 350, 450, 550, 650 and 750℃ respectively. When ignited at 350℃, two absorption peaks are observed at 3300cm-1 and 1500cm-1. The former is attributed to O―H bending band and the latter to the O―H stretching band. When ignited at 550℃, 650℃ and 750℃, absorption peaks appearing at 400, 600 and 800cm-1 are assigned to the Al―O peaks of α-Al2O3. However, Al―O peaks observed at 400, 600 and 800cm-1 when ignited at 450℃ are not very sharp.
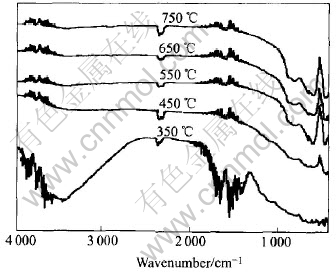
Fig.7 FT-IR spectra of as-prepared powdersignited at different temperatures
From the investigation of infrared spectra, we can come to the conclusion that Al2O3 powders with dramatic Al―O peaks can be synthesized by setting the igniting temperature above 650℃. This result is consistent with that of XRD measurements[17].
3.6 TEM images and electron diffraction patterns of samples
The morphology and particle size of α-Al2O3 nanocrystalline are observed by TEM. Fig.8(a) shows the TEM image of α-Al2O3 nanocrystalline powder. It is clearly seen that the particles are nearly spherical in shape and the average particle size of the sample is about 60-80nm. This agrees very well with the results of XRD measurements. Fig.8(b) shows a selective area electron diffraction pattern. The diffraction rings indicate that the sample is polycrystal.
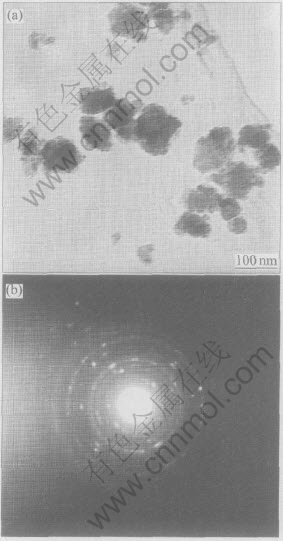
Fig.8 TEM image(a) and corresponding electron diffraction pattern(b) of α-Al2O3 powders
4 CONCLUSIONS
1) The optimal reaction conditions for synthesizing α-Al2O3 nanocrystalline powders by low-temperature combustion synthesis method are: using urea as fuel, oxidizer to fuel molar ratio of 1∶2, and igniting temperature of 700℃.
2) The polycrystal α-Al2O3 powders have an average gain size of about 60-80nm and are approximately spherical in shape.
REFERENCES
[1]Ngai Tungwai Leo, CHEN Wei-ping, XIAO Zhi-yu, et al. Warm compaction of Al2O3 particulate reinforced powder metallurgy iron-base composite [J]. Trans Nonferrous Met Soc China, 2002,12(5): 886-889.
[2]YU Zhong-qing, ZHAO Qing-sheng, ZHANG Qi-xiu. Preparation of ultrafine alumina powders by aluminum isopropoxide [J]. Trans Nonferrous Met Soc China,1994, 4(2): 21-24.
[3]WU Gao-hui, MA Sen-lin, ZHAO Yong-chun, et al. Suppression effect of fine Al2O3 particulates on aging kinetics in a 6061 matrix composite material [J]. Trans Nonferrous Met Soc China,1999, 9(4): 818-821.
[4]Deng S G, Lin Y S. Granulation of sol-gel-derived nanostructured alumina [J]. AIChE Journal, 1997, 43(2): 505-514.
[5]Xomeritakis G, Lin Yue-Sheng. CVD synthesis and gas permeation properties of thin palladium/alumina membranes [J]. AIChE Journal, 1998, 44(11): 174-183.
[6]Akporiaye D E, Dahl I M, Karlsson A, et al. Combinatorial approach to the hydrothermal synthesis of zeolites [J]. Angewandte Chemie International Edition, 1998, 37(5): 609-611.
[7]WANG Zhi-qiang, MA Tie-Cheng, CAI Ying-ji, et al. Study on α-Al2O3 synthesized by low temperature combustion process and its sintering character [J] . Bulletin of the Chinese Ceramic Society, 2000, (5): 28-31.(in Chinses)
[8]Birch H J, Michael K. Combustion Synthesis Method and Products [P] . US 5198188, 1993.
[9]WU Yi-quan, ZHANG Yu-feng, HUANG Xiao-xian, et al. Preparation of platelike nano alpha alumina particles [J] . Ceramics International, 2001, 27(3): 265-268.
[10]KUANG Jia-cai, ZHANG Chang-rui, ZHOU Xin-gui, et al. Synthesis and characterization of nanocrystalline aluminum nitride from a low temperature combustion precursor [J] . Journal of Materials Science, 2004, 39(9): 3167 - 3169.
[11]Pathak L C, Singh T B, Das S, et al. Effect of pH on the combustion synthesis of nano-crystalline alumina powder [J] . Materials Letters, 2002, 57(2): 380-385.
[12]LUO Shao-hua, TANG Zi-long, YAO Wei-hua, et al. Low temperature combustion synthesis and its dielectric property of barium titanate [J]. Journal of the Chinese Ceramic Society, 2003, 31(6): 561-565.(in Chinese)
[13]LI Da-bing, HU Jian-dong, LIAN Jian-she, et al. Synthesis of nanometer powders by sol-gel low temperature combustion [J] . Journal of the Chinese Ceramic Society, 2001, 29(4): 340-343.(in Chinese)
[14]Kazumasa Takatori, Takao Tani, Naoyoshi Watanabe, et al. Preparation and characterization of nano-structured ceramic powders synthesized by emulsion combustion method [J] . Journal of Nanoparticle Research, 1999, 197(204): 197-204.
[15]LI Wen-xia, YIN Sheng, WANG Hui. Micro powders of multiphase PSZ by low temperature combustion synthesis [J] . Journal of the Chinese Ceramic Society, 2000, 28(6): 534-537.(in Chinese)
[16]YEN Sheng. Combustion Synthesis [M] . Beijing: Metallurgical Industry Press, 1999.(in Chinese)
[17]ZHANG Chang-shuan, ZAO Feng, ZHANG Ji-jun, et al. Infrared spectroscopic studies of alumina on a nanometer scale [J] . Acta Chimica Sinica, 1999 , 57(3): 275-280.(in Chinese)
Received date: 2004-08-09; Accepted date:2005-05-17
Correspondence: ZHAI Xiu-jing, Professor, PhD; Tel: +86-24-83687729; E-mail: xjzhai@mail.neu.edu.cn
(Edited by YANG Bing)