
Effect of dopant content on microstructure and properties of
Si-Al-K doped molybdenum
LI Da-cheng(李大成)1, ZHOU Zhang-jian(周张健)2, PU Chun-yang(卜春阳)1
1. Technology Center, Jinduicheng Molybdenum Industry Co., Xi’an 710068, China;
2. Laboratory of Special Ceramic and Powder Metallurgy, University of Science and Technology Beijing,
Beijing 100083, China;
Received 15 July 2007; accepted 10 September 2007
Abstract: The effect of aluminum, potassium and silicon (AKS) content on the high-temperature properties and microstructure of molybdenum powder, sintering product and fabricated wires were studied. The molybdenum powder was produced firstly by reduction of molybdenum oxide which is doped with K2SiO3 and Al(NO3)3?9H2O. Then doped molybdenum powder was cold pressed into cylindrical compact with diameter of 17 mm, and sintered in induction furnace. The microstructure of molybdenum ingots was examined by transmission electron microscopy. The results show found that the doped elements form non-crystalline phase and distribute along the interface of Mo, thus inhibit the grain growth of Mo. Finally, the sintered Mo was drawn into wires with a diameter of 0.70 mm. The formed non-crystalline particles are changed into row of fine particle during the drawing process.
Key words: doped molybdenum; wire drawing; microstructure
1 Introduction
Mo is widely used in fabrication aircraft parts, electrical contacts, and filaments, due to its ability to withstand extreme temperatures without significantly expanding or softening. But the ductile of Mo is not very good at room temperature, and its DBTT (ductile-brittle transition temperature) is a little high, which limit its further industry application. It is found that when doped by some elements, the recrystallization temperature and high temperature strength of Mo will increase significantly. There have two kinds of doped elements, one is lanthanon, such as La, Ce, Y[1-3], the other is Si, Al and K, the so called AKS[4-6]. The AKS-doped Mo wire is produced in a similar manner to AKS-doped W wire, but lower processing temperatures are typically used for the production of Mo wire. Previous studies on AKS-doped W wire have shown that the dispersion which provides the interlocking grain structure in recrystallized tungsten wire is bubbles of elemental K; these enhance incandescent lamp filament life[7-8]. However, there is little previous work on the K-containing dispersion in AKS-doped Mo wire. The important features of the doped evolution are not well understood. In AKS-doped Mo, the dispersion can be either K bubbles, solid oxide particles or other phase, depending on the processing method.
The aim of the present work is to investigate the effect of K and Si content on the high-temperature properties and microstructure of molybdenum powder, sintering product and fabricated wires.
2 Experimental
Mo powder doped by AKS was prepared according to the following steps. First, an aqueous solution containing different volumes of K2SiO3 and Al(NO3)3?9H2O according to the designed content of doped elements was prepared, then the solution was vacuum sprayed into MoO2 powder to get AKS doped MoO2 powders. After drying the doped MoO2 powders under vacuum, Mo doped with AKS was produced by reduction of the fabricated AKS doped MoO2 in dry hydrogen.
The doped Mo was compacted by cold isostatically pressure to form a rod compact with diameter of 17 mm, the green compact was sintered in a induction furnace. The sintered AKS doped Mo rod was processed to Mo wire with diameter of 0.7 mm by drawing method.
The content of doped element was examined by atom spectrum. SEM was used to observe the microstructure of the AKS doped Mo wire. A Hitachi H-800 TEM was used to observe the morphology of the secondary particle in the AKS doped Mo.
3 Results and discussion
3.1 Yield of doped elements
Table 1 shows the change of K and Si content in the Mo powder between the designed content and content after reduction process. From Table 1, it is found that the content of K and Si are decreased obviously after reduction process. The yield ratio of K is lower than that of Si, this is because that the melting point of K2O is lower than that of SiO2, more K2O will be lost due to sublimation after reduction and sintering[9].
3.2 Effect of doped element content on morphology and size of Mo powder
Fig.1 shows the morphologies of Mo powders doped by different contents of AKS. It can be found that the particle size of Mo will be finer and less agglomeration when more contents of AKS are doped. This is because that the elements are doped in the MoO2 powder by solid-liquid blending method, the doped elements are homogeneous dispersed on the surface of MoO2 particle.
During the process of reduction, the doped element will inhibit the grain growth of Mo, and the more the content of AKS, the more the effective of grain growth inhibiting.
3.3 Effect of doped element content on microstructure of Mo rod and wire
Fig.2 shows the morphologies of fracture surface of Mo rod with different content of doped elements. It can be found that when more elements are doped in Mo, the grain size of sintered Mo will be smaller and more homogenous. The doped elements will be acted as grain growth inhibiter during sintering, thus more content of doped element will lead to smaller grain size.
Table 1 Yield ratio of K and Si in Mo powder

Fig.1 Morphologies of AKS-doped Mo powders: (a) Sample 1; (b) Sample 2; (c) Sample 3; (d) Sample 4

Fig.2 Fracture morphologies of AKS-doped Mo rod: (a) Sample 1; (b) Sample 2; (c) Sample 3; (d) Sample 4
Fig.3 shows the relationship between the content of doped elements and the hardness of sintered Mo. The hardness of Mo will increase when more elements were doped. It is because that, the more the content of doped elements the finer the grain size of Mo, and thus, the higher the hardness of Mo.
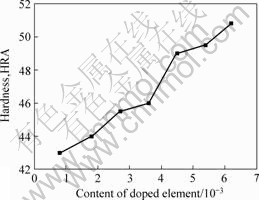
Fig.3 Relationship between hardness and AKS content
Fig.4 shows the TEM images of sintered Mo rod with different doped element contents. It can be found that two kinds of secondary particle exist, the smaller one is located in the Mo grain, and the larger one is located along the grain boundary of Mo.
When increasing the content of doped elements, there will be more secondary phase particle. EDX test shows that the composition (mass fraction) of the secondary particle (Fig.4(c)) is as follows: Al, 9.6%; Si, 35.8%; K, 2.3%; Mo, 52.3%. Electron diffraction (Fig.4(e)) shows that the secondary particle is composed by crystalline Mo and unknown non-crystalline phase, this is different from the previous study results[10].
Fig.5 shows the microstructures of Mo wires with different content of doped elements. The formed non-crystalline particles doped by AKS change into row of fine particle when Mo ingots were drawn into wires. When increasing the content of doped elements, the fibre of Mo wire will be finer, this will improve the properties of Mo wire.
4 Conclusions
1) Si, Al and K are doped in MoO2 powders by solid-liquid blending method. Only parts of designed contents of Si and K are retained after reduction and sintering. The yield of Si is higher than that of K.
2) The particle size of Mo powder will be finer when increasing the content of doped element.
3) The doped element will form non-crystalline phase and distribute along the interface of Mo, and thus inhibit the crystalline growth of Mo. The higher the volume fraction of the doped AKS is, the better the properties of Molybdenum wires are.
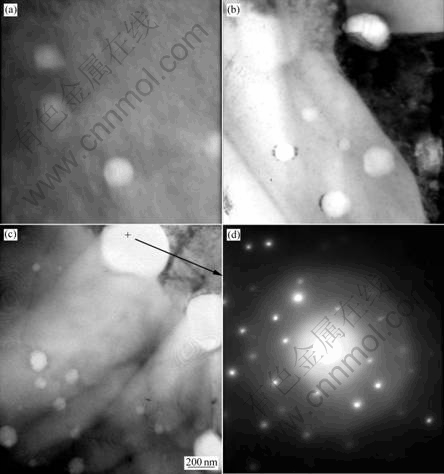
Fig.4 TEM micrographs of doped Mo rod: (a) Sample 1; (b) Sample 3; (c) Sample 4; (d) Electron diffraction

Fig.5 SEM images of doped Mo wires: (a) Sample 1; (b) Sample 2; (c) Sample 3; (d) Sample 4
References
[1] ZHANG Jiu-xing, ZHOU Mei-ling, ZHOU Wen-yuan, WANG Jin-shu, NIE Zuo-ren, ZUO Tie-yong. A study on mechanical and thermionic properties of rare earth oxide-doped molybdenum[J]. Chinese Journal of Rare Metals, 2002, 26: 124-128.
[2] WANG Jin-shu, ZHOU Mei-ling, ZUO Tie-yong, ZHANG Jiu-xing, NIE Zuo-ren, HU Yan-cao. Effect of La2O3 Nanoparticles on the properties of molybdenum powder[J]. Rare Metal Materials and Engineering, 2002, 31: 140-143. (in Chinese)
[3] ZHANG Jiu-xing, LIU Yan-qin, LIU Dan-min, ZHOU Mei-ling, ZUO Tie-yong. Toughness of La2O3-doped Mo alloy[J]. The Chinese Journal of Nonferrous Metals, 2004, 14(1): 13-17. (in Chinese)
[4] Yoo M K, Hiraoka Y, CHOI J. Recrystallization of molybdenum wire doped with potassium silicate[J]. Metall Mater Trans A, 1995, 26(4): 801-816.
[5] BEWLAY B P, LEWIS N, LOU K A. Observation on the evlution of potassium bubbles in tungsten ingots during sitering[J]. Metall Mater Trans A, 1992, 23(1): 121-133.
[6] IORIO L E, BEWLAY B P, LARSEN M. Analysis of AKS- and lanthana-doped molybdenum wire[J]. International Journal of Refractory Metals and Hard Materials, 2006, 24(4): 306-310.
[7] SCHADE P. Bubble evolution and effects during tungsten processing[J]. International Journal of Refractory Metals and Hard Materials, 2002, 20(4): 301-309.
[8] HORACSEK O, BARTHA L. Development of the bubble structure from selectively deforming potassium-pores in doped tungsten wires[J]. International Journal of Refractory Metals and Hard Materials, 2004, 22(1): 9-15.
[9] GR?GER A, VAΒEN R, MERTENS F. The evaporation of potassium during sintering of doped tungsten[J]. International Journal of Refractory Metals and Hard Materials, 1998, 16(1): 37-44.
[10] IORIO L E, BEWLAY B P, LARSEN M. Dopant particle characterization and bubble evolution in aluminum- potassium-silicon-doped Mo wire[J]. Metall Mater Trans A, 2002, 33 (11): 3349-3356.
Foundation item: Project(2006KJA21) supported by the Science and Technology Foundation of Aluminum Corporation of Limited, China
Corresponding author: LI Da-cheng; Tel: +86-29-88378322; E-mail: zhouzhj@mater.ustb.edu.cn
(Edited by YANG Hua)