Mg alloy surface alloying layer fabricated through evaporative pattern casting technology
来源期刊:中国有色金属学报(英文版)2010年第12期
论文作者:陈东风 董选普 张雄 樊自田
文章页码:2240 - 2245
Key words:Mg alloy; evaporative pattern casting; surface alloying layer; micro-hardness
Abstract: The influencing factors of surface alloying layer by evaporative pattern casting technology were investigated. A certain thickness alloying layer was formed on the surface of Mg-alloy matrix when the pouring temperature was 780 °C with different vacuum degree and alloying powder size. The surface layer microstructure, micro area composition of the new phase formed on the matrix and the composition characteristics on the surface layer were examined by SEM and element scanning. The results show that the content of aluminum increases greatly on the surface layer. The micro-hardness of alloyed layer has a more obvious increase compared with that of the matrix. The size of alloying element and the vacuum degree are the key factors influencing the alloying layer, with the increase of element powder size from 0.074 to 0.15 mm and vacuum degree from 0.04 to 0.06 MPa, the surface alloying effect becomes better.

CHEN Dong-feng(陈东风), DONG Xuan-pu(董选普), ZHANG Xiong(张 雄), FAN Zi-tian(樊自田)
State Key Laboratory of Material Processing and Die and Mold Technology,
Huazhong University of Science and Technology, Wuhan 430074, China
Received 25 December 2009; accepted 19 April 2010
Abstract: The influencing factors of surface alloying layer by evaporative pattern casting technology were investigated. A certain thickness alloying layer was formed on the surface of Mg-alloy matrix when the pouring temperature was 780 °C with different vacuum degree and alloying powder size. The surface layer microstructure, micro area composition of the new phase formed on the matrix and the composition characteristics on the surface layer were examined by SEM and element scanning. The results show that the content of aluminum increases greatly on the surface layer. The micro-hardness of alloyed layer has a more obvious increase compared with that of the matrix. The size of alloying element and the vacuum degree are the key factors influencing the alloying layer, with the increase of element powder size from 0.074 to 0.15 mm and vacuum degree from 0.04 to 0.06 MPa, the surface alloying effect becomes better.
Key words: Mg alloy; evaporative pattern casting; surface alloying layer; micro-hardness
1 Introduction
Magnesium alloys have advantageous properties such as low density, high speci?c strength, good castability, excellent machinability and good weldability, hence, these alloys are widely used in the automotive, aircraft, aerospace and 3C industries. However, the research and application of Mg-alloys are not perfect compared with aluminum alloys because of their poor resistance to wear and corrosion, which limits their applications[1-6].
Surface alloying process is an efficient and reliable technique to improve the surface properties by changing the microstructure. Various surface treatment technologies were developed, such as laser remelting surface alloying[7], electron beam alloying[8-9], ion implantation[10] and plasma surface alloying[11]. These conventional methods have disadvantages of high cost and low efficiency to an extent. Casting surface alloying is a practical and economic technology, especially with the development of magnesium alloys evaporative pattern casting (EPC) technology. The alloying coating containing aluminum powder is paved on foam pattern surface where the properties need to be improved. Alloying layer is formed on the magnesium alloy substrate surface, a large volume fraction of second phase and inter metallic compound can not only increase the hardness, but also improve the corrosion resistance of magnesium alloys[12], so surface alloying can modify the alloy surface properties by EPC process. At present, the most difficult problem of magnesium alloys surface alloying by EPC is low pouring temperature and thermal capacity. Related researches mainly concentrate on the cast iron and steel, however, research on magnesium alloy surface alloying by EPC has a significant theory meaning.
In this work, based on the related previous surface alloying investigation[13-15], aluminum powder was chosen as the alloying element. The research was conducted under different process parameters, the factors and process of surface alloying layer were analyzed by establishing the EPC surface alloying model.
2 Experimental
2.1 Material
AZ91D magnesium alloy was used as the substrate material in the experiment. The chemical composition (mass fraction) is: 8.5%-9.5%Al, 0.45%-0.90%Zn,0.17%-0.4%Mn, ≤0.05%Si, ≤0.025%Cu, ≤0.001%Ni, ≤0.004%Fe, and balance Mg. The density of polystyrene foam pattern was 0.02 kg/cm3. Aluminum powder with different size (0.074 and 0.15 mm) was served as the alloying element. The alloying coating was made of the metal powder and the polyvinyl alcohol water solution.
2.2 Method
The polystyrene foam pattern was prepared with certain foaming technology parameters. The coating was composed of alloying element (aluminum powder) and adhesive according to desired proportion.
First, 5% (mass fraction) adhesive of polyvinyl alcohol water solution was prepared. Then aluminum powder with different size was weighted, put into the adhesive and stirred for 10-15 min. The coating on the surface of foam pattern was paved. In this work, the bottom of foam pattern was heated to pave EPC coating and then dried at 50 °C. The molten magnesium alloy was poured at 780 °C with vacuum degree of 0.04, 0.06 and 0.08 MPa, respectively. The foam pattern was decomposed and vaporized when it met the high temperature metal melt, then, the metal melt infiltrated into the capillary of the alloying coating under the effect of vacuum degree and penetrability. The alloying coating was melted gradually because the heat from the matrix acted with substrate, forming an inter-metallic compound layer with a certain thickness on the substrate surface layer. The surface alloying principle by EPC technique was shown in Fig.1.
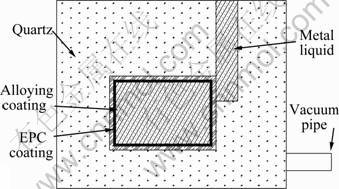
Fig.1 Schematic diagram of surface alloying principle
The specimens were cut along the direction from the surface alloyed layer to substrate, ground with No.600, 800, 1 500 emery papers consequently, then polished using standard metallographic techniques, degreased with ethanol and acetone, and finally etched with 4% nitric acid in ethanol solution. The microstructure and composition distribution of the samples were analyzed by SEM.
The micro-hardness of the sample surface layer was examined on an HV-1000 hardness tester. Appropriate test load and holding time were selected during the test. At least three points were measured in each experiment, and the average value was regarded as the micro-hardness.
3 Results and discussion
3.1 Microstructure of surface alloying layer
Fig.2 shows the microstructures of the Mg-alloy substrate surface layer after alloying treatment, in which the pouring temperature and vacuum degree were 780 °C and 0.04 MPa, respectively. The alloying layer was formed when two kinds of aluminum powder were used. But the alloying layer with the aluminum powder size of 0.15 mm becomes denser and the thickness increases compared with that of 0.074 mm, as shown in Figs.2(a) and (b). With increasing aluminum powder size, the second phase takes up a larger volume fraction, the alloying layer becomes denser and the thickness increases prominently. The grain growth of the new forming second phase is formed by mutual connection and covers the substrates surface during surface alloying process. The alloying process can be explained as follows: polystyrene foam pattern is decomposed because of the heat from substrate when Mg-alloy is poured, the pyrolysis products give off from sand mould via EPC coating under the action of vacuum degree. Then the metal melt contacts with alloying coating and infiltrates into the coating gradually. The alloying elements melted and diffused to the substrate. The diffusion effect would be more significant if the metal particles melt sufficiently and have a longer holding time in high temperature status. Finally, the molten alloying element acts with the substrate and forms the second phase before solidification. Besides, a number of pores are found in the alloying layer. The analysis indicates that the adhesive burns out and gives off gas. The pores come out if those gases are not exhausted from the coating completely. Hence, in order to avoid the formation of pores, the vacuum degree should be increased as high as possible.

Fig.2 SEM images of alloying layer with different aluminum powder size at pouring temperature 780 °C and vacuum degree of 0.04 MPa: (a) 0.074 mm; (b) 0.15 mm
The influence of vacuum degree on the surface alloying effect was investigated. Fig.3 shows the alloying layer microstructure obtained under different vacuum degree. The process parameters are as follows: the pouring temperature was 780 °C, and the size of aluminum powder was 0.15 mm. The results show that the microstructure of surface alloying layer changes with the increase of vacuum degree from 0.04 to 0.08 MPa. It is found that the alloying layer covers the substrate surface in the form of network. The microstructure of surface layer becomes compact and well-distributed when the vacuum degree reaches 0.06 MPa, as shown in Figs.3(a) and (b). There is evident transition region between the alloying layer and the substrate as shown in Fig.3(d). However, the thickness of the alloying layer turns uneven and the microstructure becomes incompact when the vacuum degree reaches 0.08 MPa, as shown in Fig.3(c). A conclusion is drawn that the casting infiltration driving force of metal liquid increases remarkably with the increase of vacuum degree, so the casting infiltration depth is different in the whole alloying coating layer when the vacuum degree changes, which causes an uneven alloying layer. Hence, the vacuum degree plays an important role during surface alloying process.
The model of surface alloying process is illustrated in Fig.4. It can be seen that the foam pattern is decomposed when the magnesium alloy is poured, and the pyrolysis products of pattern is removed from EPC coating. The mould filling velocity is higher than the infiltration velocity of pyrolysis products in the coating layer, hence the infiltration process of pyrolysis products continues after the mould filling, and the metal liquid also penetrates into the alloying coating capillary porocity. In order to analyze the surface alloying driving force, the model of interface between metal liquid and alloying coating is simplified. On the assumption that the alloying coating capillary porocity is a regular cylinder, its radius is R and the distance from the cast gate is h, the pressure of metal liquid infiltration into the alloying coating is
![]() (1)
(1)
where p is the surface alloying driver force, N/m2; pair is the atmospheric pressure, N/m2; p1 is the negative pressure, N/m2; p2 is the capillary porocity pressure, N/m2; p3 is the pyrolysis products pressure, N/m2; p4 is the internal pressure of slag, N/m2; h is the distance from cast gate to capillary pore; ρ is the density of substrate liquid; and R is the radius of capillary porocity. From the drive force p, it is obvious that vacuum degree plays an important role during surface alloying process. The metal liquid penetrates into alloying coating in such a short time owing to the capillary porocity, which makes the coating melt and react with the substrate. Surface alloying layer having a good metallurgical combination with the substrate can be attained. Based on Ref.[16], the infiltration capacity can be calculated as
![]()
![]() (2)
(2)
where Q is the infiltration capacity; R is the radius of the capillary pore; ![]() is the filling velocity of metal liquid; η is the kinetic viscosity; l is the penetration depth of metal liquid in the alloying coating; σ is the interfacial tension between metal melt and slag; θ is the wetting angle. From Eq.(2), the infiltration capacity of substrate liquid is proportional to the pore diameter formed by the alloying element. The capillary porosity diameter becomes big with the increase of metal particles, so the metal liquid can penetrate into the alloying coating easily, and enough energy is conducted to the particles, which makes them melt and react with molten substrate. The result is in accordance with the alloyed layer thickness as shown in Figs.2 and 3.
is the filling velocity of metal liquid; η is the kinetic viscosity; l is the penetration depth of metal liquid in the alloying coating; σ is the interfacial tension between metal melt and slag; θ is the wetting angle. From Eq.(2), the infiltration capacity of substrate liquid is proportional to the pore diameter formed by the alloying element. The capillary porosity diameter becomes big with the increase of metal particles, so the metal liquid can penetrate into the alloying coating easily, and enough energy is conducted to the particles, which makes them melt and react with molten substrate. The result is in accordance with the alloyed layer thickness as shown in Figs.2 and 3.
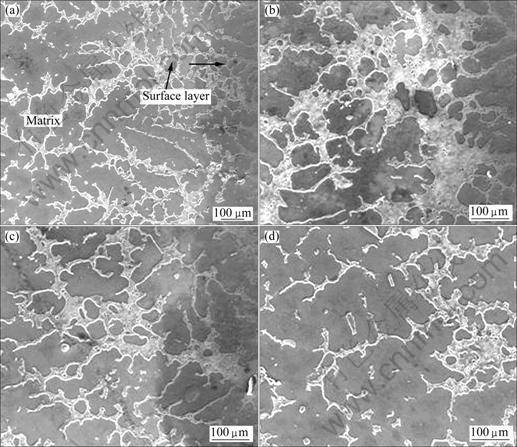
Fig.3 SEM images of surface alloying layer with Al powder size of 0.15 mm at pouring temperature of 780 °C under different vacuum degree: (a) 0.04 MPa; (b) 0.06 MPa; (c) 0.08 MPa; (d) Transition region
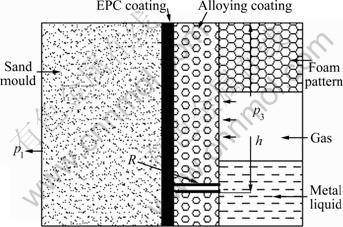
Fig.4 Model of surface alloying through EPC
3.2 Chemical composition and properties of alloying layer
The element distribution was analyzed by means of line composition scan from surface alloying layer to substrate as shown in Fig.5. It indicates that aluminum content in alloying layer is higher than that in the substrate and declines from the surface layer to the substrate gradually; magnesium content in the surface layer is slightly lower than that in the substrate. This is because the alloying coating is mainly composed of aluminum powder. The aluminum powders melt when they contact with the substrate liquid and diffuse along the direction from surface to substrate, which makes the aluminum content increase. So, the inter-metallic compounds take up a high volume fraction, but the content of oxygen and zinc element changes slightly.

Fig.5 Elements line scan of surface alloying layer
The phase composition in the substrate and new phases in the surface alloying layer was observed, as shown in Fig.6. β-Mg17Al12 is divorced eutectic phase existing along grain boundaries, and the new phase formed during alloying process distributes on the substrate surface with a chrysanthemum shape. The energy spectrum analysis show that the content of Al and Mg element in the new phase is equivalent with that in the β-Mg17Al12, so it can be predicted that the new phase in the alloying layer is mainly β-Mg17Al12. The different microstructure from the β-phase in the substrate may be because beside the solid solution of part of aluminum element the form of α-Mg, most of it acts with substrate and forms intermetallic compounds β phase at grain boundaries, then the adjacent β phases contact and grow up, and the flake shape β phase covers the substrate surface with the solidification of substrate liquid.
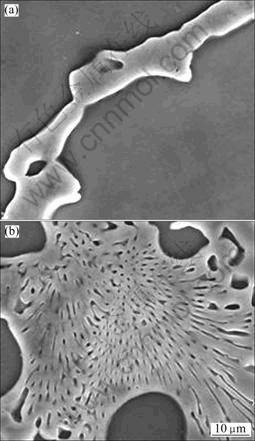
Fig.6 SEM images of β phase and new phase: (a) β-phase in matrix; (b) New β phase in alloying layer
The influence of β-Al12Mg17 on the corrosion resistance of magnesium alloy was investigated[6, 17], and the results indicated that β phase has a good stability under general corrosion environment because of its high potential. For AZ91D magnesium alloy, the corrosion mainly takes place on α-Mg, however, β phase plays completely different roles in corrosion process[18]. On one hand, β phase is the cathode of corrosion cell, which accelerates the corrosion rate; on the other hand, β phase becomes an impediment of anodic reaction to restrain corrosion. At the beginning of alloy corrosion process, α-Mg is first etched and β phase remains as a framework. β phase has a big volume fraction on the substrate surface and distributes with a shape of network by mutual connection, so the protective layer is composed of β phase, corrosion products and intermetallic compound to prevent corrosion development. Hence, the alloying layer on the magnesium alloy surface can modify the corrosion performance through EPC surface alloying technology. But the prerequisite is that the intermetallic compound must be compact in the surface layer. As a result, the process parameters need to be further optimized in order to prepare a compact intermetallic compound layer.
From the above analysis, network microstructure is formed on the magnesium alloy surface layer. The alloying microstructure changes gradually with the increasing distance from the surface alloying layer to substrate (r), and has a metallurgical combination with the substrate. The microstructure of the sample exhibits excellent comprehensive properties. As shown in Fig.7, when the pouring temperature is 780 °C and the vacuum degree is 0.06 MPa, the highest hardness value reaches HV 210 on the surface of alloying layer, it declines gradually to substrate, which improves the wear resistance of substrate. To some extent, the hardness is in good agreement with the microstructure distribution on the substrate surface layer.
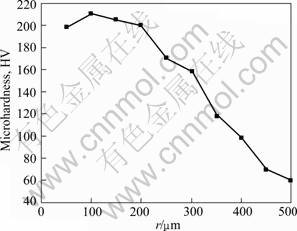
Fig.7 Hardness profile of sample from surface to matrix
4 Conclusions
1) Alloying layer on the substrate is attained through EPC technology. The effect of capillary pores is remarkable when the size of aluminum powder varies from 0.074 to 0.15 mm. The thickness of alloying layer increases and forms a metallurgical combination with Substrate.
2) Microstructure of alloying layer is compact when the pouring temperature is 780 °C and the vacuum degree is 0.06 MPa. The new second phase grows up by mutual connection and covers the substrate surface with a thickness of 300-500 μm, which provides protection for the substrate and enhances the surface properties, but the alloying layer is not densified when the vacuum degree reaches 0.08 MPa.
3) The surface layer hardness is improved prominently, and the highest value comes up to HV 210, which is almost four times that of the substrate. The hardness declines gradually from surface layer to substrate.
References
[1] GRAY J E, LUAN B. Protective coatings on magnesium and its alloys―A critical review [J]. Journal of Alloys and Compounds, 2002, 336(1/2): 88-113.
[2] HARA N, KOBAYSHI Y, KAGAYA D, AKAO N. Formation and breakdown of surface films on magnesium and its alloys in aqueous solutions [J]. Corrosion and Science, 2007, 49(1): 166-175.
[3] ZHANG Tao, SHAO Ya-wei, MENG Guo-zhe, WANG Fu-hui. Electrochemical noise analysis of the corrosion of AZ91D magnesium alloy in alkaline chloride solution [J]. Electrochimica Acta, 2007, 53(2): 561-568.
[4] MAKAR G L, KRUGER J. Corrosion of magnesium [J]. International Materials Reviews, 1993, 38(3): 138-153.
[5] AGHION E, BRONFIN B, FRIEDRICH H, RUBINOVICH Z. The environmental impact of new magnesium alloys on the transportation industry [C]//Magnesium Technology 2004. TMS, 2004: 167-172.
[6] SONG G, ATRENS A. Corrosion mechanisms of magnesium alloys [J]. Advanced Engineering Materials, 1999 (1): 11-13.
[7] WANG Y, LI C G, TIAN W, YANG Y. Laser surface remelting of plasma sprayed nanostructured Al2O3-13wt%TiO2 coatings on titanium alloy [J]. Applied Surface Science, 2009, 255(20): 8603-8610.
[8] IWATA A. Possibility of electron beam alloying of magnesium alloys [J]. Journal of the Japan Society of Precision Engineering, 2002, 28(11): 1465-1469.
[9] YE H, YAN Z L. Electron beam surface alloying of a magnesium alloy with Al [J]. Surface engineering, 2008, 373/374: 326-329.
[10] CORLU B, ?RGEN M, Surface modification of iron containing aluminum alloys by treatment with copper plasma produced with cathodic arc [J]. Surface and Coatings Technology, 2009, 204(6/7): 872-877.
[11] WAN Y Z, XIONG G Y, LUO H L, HE F, HUANG Y, WANG Y L. Influence of zinc ion implantation on surface nanomechanical performance and corrosion resistance of biomedical magnesium-calcium alloys [J]. Applied Surface Science, 2008, 254(17): 5514-5516.
[12] MA You-ping, WEN Wei-xin, HE Xi-peng, LIU Peng-fei. The effect of solid diffusion surface alloying on properties of ZM5 magnesium alloy [J]. Surface and Coatings Technology, 2005, 190(2/3): 165-170.
[13] YUAN Zhong-yue, ZHANG Zhong-ming, LI Chao-sheng, LIN You-dong. Microstructures of surface alloying layer prepared by evaporable pattern casting process [J]. Ordnance Material Science and Engineering, 2002, 25(4): 36-39. (in Chinese)
[14] YANG Gui-rong, SONG Wen-ming, MA Ying. Microstructure and bending deformation about the surface composite materials on copper substrate [J]. Chinese Journal of Materials Research, 2006, 20(6): 631-636. (in Chinese)
[15] LI Zu-lai, JIANG Ye-hua, ZHOU Rong, LU De-hong, ZHOU Rong-feng. Dry three-body abrasive wear behavior of WC reinforced iron matrix surface composites produced by V-EPC infiltration casting process [J]. Wear, 2007, 262(5/6): 649-654.
[16] JI Zhao-hui, ZHANG Cheng-jun. Study on mechanism of casting infiltration for surface alloying of steel castings in EPC process [J]. Foundry, 2000, 49(3): 130-133. (in Chinese)
[17] CHEN Dong-feng, DONG Xuan-pu, FAN Zi-tian, XU Qian, ZHANG Xiong. Study on microstructure and properties of mg-alloy surface alloying layer fabricated by EPC [J]. China Foundry, 2010, 17(1): 13-18.
[18] AMBAT R, AUNG N, ZHOU W. Evaluation of microstructural effects on corrosion behaviour of AZ91D magnesium alloy [J]. Corrosion Science, 2000, 42(8): 1433-1455.
Foundation item:Project(50775085) supported by the National Natural Science Foundation of China
Corresponding author: DONG Xuan-pu; Tel/Fax: +86-27-87558252; E-mail: dongxp@mail.hust.edu.cn
DOI: 10.1016/S1003-6326(10)60635-8

