���Ͳ��ռ������³������뽺��ķ�ѡ������
��Դ�ڿ����й���ɫ����ѧ��(Ӣ�İ�)2013���2��
�������ߣ���٩Ƽ ������ ������
����ҳ�룺501 - 507
�ؼ��ʣ�����������ѡ���룻���ף����ײ��ռ�
Key words��hematite; collophane; reverse flotation separation; dephosphorization; dephosphorization collector
ժ Ҫ���������Ƶķ���ѡ���ײ��ռ�(RFP-138)��ϵ�г�����ͽ���Ŀ�ѡ�Լ����û��������о���������ͽ���ĵ����P�˹���Ͽ�ķ���ѡ�������������RFP-138�dz�����ͽ�������ò��ռ����ڼ��������²��ú��ʵ����Ƽ����ܹ���Ч��ʵ�����ֿ���ķ�ѡ����P�������˹���Ͽ��е�1.05%����0.12%����TFe�Ļ����ʴﵽ91.30%����RFP-138��Һ�Ļ�ѧ��Ϊ����������з�����̽��RFP-138�����ֿ���ѡ���Բ��յĻ�����
Abstract: The reverse flotation separation performance and mechanism of hematite and cellophane in the presence of RFP-138, a newly synthesized dephosphorization collector, were studied. Reverse flotation tests on monominerals and artificially mixed minerals of hematite and collophanite show that, this anionic collector performs excellently in reducing the phosphorus content in hematite. It can decrease the content of P in artificially mixed minerals from 1.05% to 0.12% and achieve the TFe recovery rate of 91.30%. The chemical behavior of solution and infrared spectra of RFP-138 were investigated to explore the selective collecting mechanism of RFP-138 to these two minerals.
Trans. Nonferrous Met. Soc. China 23(2013) 501-507
Kan-ping YU 1,2, Yong-fu YU 1,2,3, Xiang-yang XU1
1. School of Minerals Processing and Bioengineering, Central South University, Changsha 410083, China;
2. Changsha Research Institute of Mining and Metallurgy Co., Ltd., Changsha 410012, China;
3. School of Resources and Environmental Engineering, Wuhan University of Technology, Wuhan 430070, China
Received 15 May 2012; accepted 17 December 2012
Abstract: The reverse flotation separation performance and mechanism of hematite and cellophane in the presence of RFP-138, a newly synthesized dephosphorization collector, were studied. Reverse flotation tests on monominerals and artificially mixed minerals of hematite and collophanite show that, this anionic collector performs excellently in reducing the phosphorus content in hematite. It can decrease the content of P in artificially mixed minerals from 1.05% to 0.12% and achieve the TFe recovery rate of 91.30%. The chemical behavior of solution and infrared spectra of RFP-138 were investigated to explore the selective collecting mechanism of RFP-138 to these two minerals.
Key words: hematite; collophane; reverse flotation separation; dephosphorization; dephosphorization collector
1 Introduction
Due to the high speed development of modern industry, there is an increasing demand for the iron resources, and the die-away process for rich and easy-to-recover iron ore resources has accelerated dramatically in recent years, therefore, it is essential to carry out a beneficiation study on complex ultrafine iron ore with high content of impurities such as phosphoric and sulfur [1,2]. The high-phosphorus iron mines are abundant worldwide, while these mines in China account for 14.86% of the total domestic iron mines, namely, 7.45 billion tons. Unfortunately, these iron mines are generally acknowledged refractory ones as the occurrence state of these phosphate minerals in high-phosphorus iron ores are mostly in the form of collophane rock associated complicatedly with hematite (limonite) [3,4]. Abundant researches have been conducted on dephosphorization technology of high- phosphorus iron ores and some progresses have been achieved, yet, the dephosphorization efficiency is still comparatively low while the iron loss is considerably high [5-10]. To actualize eventually a rational exploitation and utilization of this kind of resource, it is crucial to achieve an effective separation of hematite and collophane.
A novel high-efficient reverse flotation collector, RFP-138, was developed and studied [11], with which flotation tests on monomineral and artificially mixed ore were carried out and the floatability differences among two minerals, collophane and hematite, were discussed, while the influence of some commonly used depressants on the dephosphorization effect was investigated. Then, the mechanism to actualize the effective separation of these two minerals was studied based on the discussion of solution chemistry and the analysis on the Fourier transform infrared spectroscopy (FTIR) spectra characterizating the reaction between minerals and RFP-138.
2 Experimental
2.1 Materials
Preparation procedure of hematite monomineral ore is as follows: firstly, rich lump ore of Qidashan hematite was artificially selected, rushed with a jaw crusher and double-roll crusher, and ground to a fineness less than 0.074 mm fraction weighing 90%. Thereafter, magnetite in the sample was removed by a low intensity magnetic separation, while other non-magnetic minerals such as quartz were removed by, repeatedly, high intensity magnetic separation. Then, shaking table was repeatedly used to remove some weakly magnetic impurities like hornblende and chlorite as the specific gravities for these minerals are small, and the concentrated powder was washed with distilled water, dried below 50 ��C and finally screened to obtain a hematite monomineral ore with a fineness less than 0.043 mm.
According to the chemical analysis, the content of total iron(TFe) of this ore is 68.76%. And, it can be concluded from the corresponding X-ray powder diffraction (XRD) pattern of the sample shown in Fig. 1 that, the ore prepared is of hematite crystal structure, while no diffraction peak of other impurities can be detected.
Fig. 1 XRD pattern of hematite
Collophane monomineral ore was prepared following the procedure described as follows. Collophane-rich lump ore was artificially collected from the Yichang Phosphorite Mine of Hubei Province, China. The bulk ore was then crushed with a double-roll crusher and ground with a porcelain ball mill till the fineness was less than 0.198 mm completely. Then, the samples were repeatedly cleaned using a shaking table and washed with distilled water. After being dried at a temperature below 50 ��C and screened, the classified product was stored in a glass dryer.
According to the chemical analysis, the purity of collophane is higher than 92%, and the P2O5 content reached 38.09%, which can as well be testified by XRD pattern shown in Fig. 2, as the intensity of apatite diffraction peaks is quite strong, and there appear only some diffraction peaks of quartz, indicating that quartz is a main impurity of this sample.
Artificially mixed ore is composed of 29.8 g hematite and 2.0 g collophane. Multi-element analysis of this mixed ore is shown in Table 1, and among the chemical elements, the content of phosphorus is 1.05%.

Fig. 2 XRD pattern of cellophane
Table 1 Chemical compositions of mixed ore (mass fraction, %)

A lab-synthesized anionic collector, RFP-138, was used in this study. Analytically pure H2SO4 and Na2CO3 were used mainly to adjust pH value of the pulp, while water glass, starch, sodium humate, sodium carboxymethyl cellulose are of industrial grade.
2.2 Methods and equipment
Under room temperature, flotation tests were carried out using SFG, a hanging cell flotation machine with a 30 mL cell and with a spindle speed of 1900 r/min, and the single-factor method was adopted. For each test, 2.0 g sample was transferred to the flotation cell; after adding distilled water to a certain pulp volume, the mixture was agitated for 1 min at a fixed rotation speed; then, the pulp was regulated to a desired pH value using H2SO4 or Na2CO3. And after another agitating for 1min, depressant was added and after agitating for another 1 min, collector was added; after agitating for 2 min minerals were floated for 3 min at an aeration rate of 0.6 mL/min. The froth product and the material left in the cell were collected, filtered, dried and weighed, and the recovery was calculated. As for the monomineral ore, the recovery can be named as the yield of material left in the cell, while the recovery of the artificially mixed ore will be calculated after the contents of minerals were measured.
The flotation flowsheet for monomineral ore and artificially mixed minerals flotation are shown in Fig. 3.
The adsorption of reagents on mineral surface was studied using an ultraviolet-visible spectrometer UV-260. For a single measurement, 1 g sample with the size less than 0.045 mm was added into 20 mL distilled water or clarified liquid and NaOH or H2SO4 was used to regulate the pH value. Observing the reagent adding procedure, after being agitated for 2 min for each added reagent, collector was added and agitated for 8 min. The well-mixed solution was then transferred into a centrifugal tube and centrifuged for 10 min, and the supernatant liquid was used to measure the absorbance with an ultraviolet-visible spectrometer, therewith the adsorption of reagent onto mineral surface can be calculated.

Fig. 3 Flowsheet of flotation experiment
FTIR spectra obtained with 6700 (Nigaoli Nicolet Instrument Company, USA) were investigated to determine the surface groups of minerals. Pulps containing particles which were ground to a fineness less than 2 ��m were agitated with reagents, and the pulps were agitated for 30 min after the collector was added. The mixture was centrifuged for 5 min at a rotation speed of 3000 r/min, and the sediment was washed 3 times with deionized water. The collected precipitate was dried at a temperature of below 50 ��C, then the samples were stored in a drying oven for further FTIR analysis.
3 Results and discussion
3.1 Mineral flotation tests
When no adjustment agent was added, flotation tests were carried out with the concentration of RFP-138 as 100 g/t at 25 ��C, and the influence of pulp pH on the floatability of collophane and hematite was studied and the results are shown in Fig. 4
It can be seen from Fig. 4 that under acidic conditions, the floatability of hematite and collophane increases gradually along with the increase of pH value. When the pH value is 6, the highest recovery rate of hematite, 85.32%, can be reached. Then, with the further increase of pulp basicity, the recovery begins to decline rapidly until it becomes unfloatable. After a gradual ascent along with the increase of pH value, the recovery rate of collophane attains the maximum value of about 83.74% at pH 9, yet, compared with hematite, different tendency can be observed with the further increase of pulp pH, as the recovery rate maintains its high value, although a slight decline can be observed for a high alkaline pulp (pH��10). When the pH value is 12, flotation rate of collophane is nearly 60% higher than that of hematite. It can be concluded that RFP-138 possesses strong capacity to collect both minerals correspondingly at appropriate pH ranges, and this capacity shows difference under varied pH conditions, which testified that RFP-138 can actualize the flotation selectivity of these two minerals. So, the minerals can be effectively separated in a pulp with an appropriately regulated pH value.
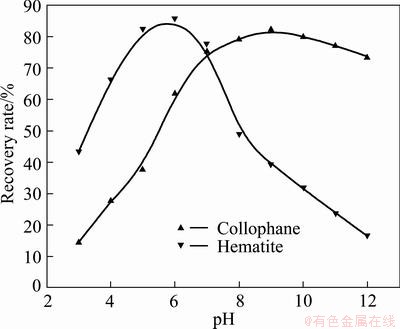
Fig. 4 Effect of pH value on floatability of minerals (RFP-138: 100 g/t)
The collector dosage test was conducted with the pulp pH value around 8.5, and the influence of RFP-138 dosage on the floatability of collophane and hematite can be illustrated in Fig. 5.
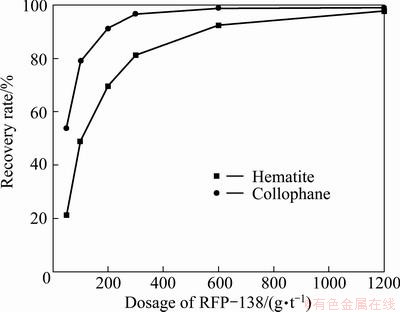
Fig. 5 Effects of RFP-138 dosage on floatability of minerals (pH=8.5)
From Fig. 5 it can be concluded that, when the pH value is 8.5, anion collector RFP-138 shows a stronger capacity in collecting collophane than hematite, although for both minerals, the recovery rate climbs along with the increase of collector dosage, yet, when the concentration of RFP-138 amounts 300 g/t, almost 100% of collophane is floated, while, for hematite flotation, RFP-138 dosage should be over 1000 g/t to attain the same recovery.
3.2 Adsorption test
Residual concentration method was used in adsorption test, and, with the adsorption amount measured by UV-Vis spectrometer as the apparent amount adsorbed, corresponding concentration can be obtained on RFP-138 standard curve, and the adsorption amount on the mineral surface can be calculated as shown in Fig. 6.
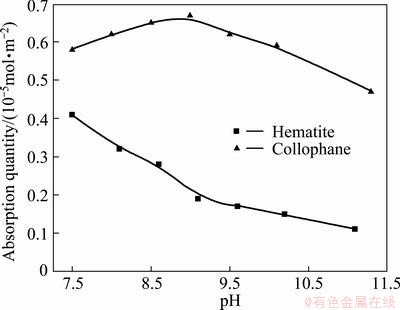
Fig. 6 Effects of pH value on adsorption of hematite and collophane (RFP-138: 100 g/t)
It can be seen from Fig. 6 that, when pulp pH is in the range of 7.5-11.5, adsorption amount of RFP-138 on cellophane is obviously higher than that on hematite, and, with the increase of pH, RFP-138 adsorption on hematite keeps declining constantly, while the adsorption on collophane increases to the maximum when pH is 8.5, and then declines, which is corresponding to the data in the pH test showing the influence of collectors on the flotability of single minerals. As a fatty acid collector, RFP-138 can react with calcium ions on the surface of collophane and form precipitate of fatty acid calcium. The adsorption of medicament was reduced by the precipitate attached on collophane, which is the reason for the decrease of RFP-138 adsorption on collophane.
3.3 Influence of regulator on hematite and collophane floatability
Hydrolysis products of Na2CO3, a strong base-weak acid salt including OH-, 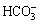 and
and 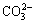 , and hydrolysis products of water glass including OH-,
, and hydrolysis products of water glass including OH-,  and
and  can crowd out the hydrolytic anions of a long chain carboxylic acid soap collector. But hydrolysis products of Na2CO3 adsorb preferentially on carbonate and collophane, which can minimize the inhibition of silicic acid radical ions thereon, meanwhile, the selective effect of water glass can therewith be improved [12-14]. In addition, Na2CO3 can soften water, reducing Ca2 +, Mg2 + and other unavoidable ions in the pulp. Na2CO3 solution has somewhat buffer action, which may ensure the pH value of the solution maintaining at around 10, and hydrolytic effective components of long chain carboxylic acid soap collector can be increased and the flotation effect can be strengthened. Therefore, Na2CO3 was adopted as a pH regulator in this study and the influence of Na2CO3 on the recovery of collophane and hematite is shown in Fig. 7.
can crowd out the hydrolytic anions of a long chain carboxylic acid soap collector. But hydrolysis products of Na2CO3 adsorb preferentially on carbonate and collophane, which can minimize the inhibition of silicic acid radical ions thereon, meanwhile, the selective effect of water glass can therewith be improved [12-14]. In addition, Na2CO3 can soften water, reducing Ca2 +, Mg2 + and other unavoidable ions in the pulp. Na2CO3 solution has somewhat buffer action, which may ensure the pH value of the solution maintaining at around 10, and hydrolytic effective components of long chain carboxylic acid soap collector can be increased and the flotation effect can be strengthened. Therefore, Na2CO3 was adopted as a pH regulator in this study and the influence of Na2CO3 on the recovery of collophane and hematite is shown in Fig. 7.
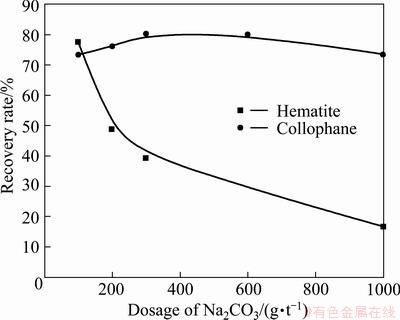
Fig. 7 Effects of Na2CO3 dosage on floatability of minerals (RFP-138: 100 g/t)
From Fig. 7, with the increase of Na2CO3 dosage, the difference between the floatability of hematite and collophane increases gradually. When the dosage of Na2CO3 is 600 g/t, the pH value of the slurry is about 9, while, when the dosage of Na2CO3, is 1000 g/t, the pH value is close to 12. It can be concluded that, when Na2CO3 is used as a regulator of RFP-138 flotation system, the separation of these two minerals can be realized.
3.4 Influence of commonly used hematite depressants on collophane flotation
With RFP-138 as the collector, the depressing effect of caustic starch, water glass, sodium humate and sodium carboxymethyl cellulose, four commonly used hematite depressants, on the floatability of collophane was investigated, and the result is shown in Fig. 8.
As can be seen in Fig. 8, the influence of these four depressants on the floatability of collophane varied and their depressing effects are in the following sequence: humic acid sodium carboxymethyl cellulose>starch>> water glass, and some center ions absorbed on the surface of collophane are Ca2+, PO43�C and other ions containing a basic group as well as an acidic group. In alkaline conditions, these icons interact with various depressants, and the water glass performs somewhat a weaker depressing effect than in acid condition. In addition, in alkaline environment, as a result of competitive adsorption, 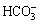 and
and 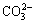 in the solution and hydrolytic products of water glass such as OH-,
in the solution and hydrolytic products of water glass such as OH-,  and
and  , the adsorption of water glass on the surface of collophane is hindered, which weakened strongly its depressing function on collophane. Meanwhile, the floatability of collophane decreases quickly with the increase of starch dosage.
, the adsorption of water glass on the surface of collophane is hindered, which weakened strongly its depressing function on collophane. Meanwhile, the floatability of collophane decreases quickly with the increase of starch dosage.
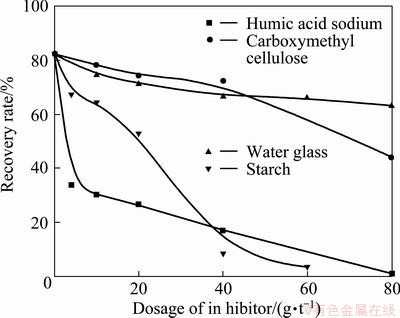
Fig. 8 Influence of common hematite depressants on flotability of collophane (pH=8.5)
3.5 Artificially mixed minerals reverse flotation test
With the aim to reduce phosphorus content, reverse flotation separation test of artificial mixed ore was conducted using Na2CO3 with a dosage of 600 g/t as the regulator and water glass with a dosage of 60 g/t as the depressant, and the influence of the dosage of collector RFP-138 on the flotation efficiency was investigated, while the test results are shown in Table 2.
Table 2 Effects of RFP-138 dosages on floatation separation of mixed ore

It can be seen from Table 2 that, with the increase of RFP-138, TFe of the concentrate is gradually increased, while the phosphorus content decreases. When the dosage of RFP-138 is 160 g/t, the phosphorus content decreases from 1.05% for the raw mixed ore to 0.12%, and an excellent dephosphorization effect is attained.
4 Mechanism analyses
Differences in flotation behavior of different minerals are always related closely with their crystal structures. Subjected to external mechanical forces, bonding between material points in mineral crystals was partially destroyed amid the crushing and milling processes, and newly cleaved surface of mineral crystal can be obtained, and the metal ion species and their contents and distribution on this fractured surface impact directly the floatability of minerals [13].
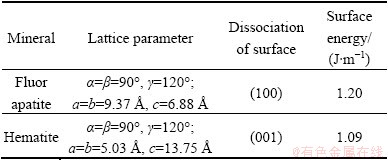
The data of lattice parameters and surface energy of apatite and hematite are listed in Table 3 [15].
Table 3 Fluorapatite and hematite lattice parameters and surface energy
On the cleavage plane(001), a large number of Fe��O bonds were ruptured during the crushing and milling processes of hematite, with free iron and oxygen ions on fractured surfaces, and there exist on the cleavage plane(100) of collophane Ca2+, Mg2+ and other anions with similar diameter such as  and
and 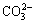 , or even some partially substituted anions such as
, or even some partially substituted anions such as 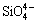 ,
, 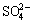 and
and 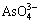 . The composition and structure of unsaturated bonds on the fractured surface are varied and complicated, and the surface properties for these two minerals are therefore quite different. Besides, as the surface energy of apatite is 0.11 J/m higher than that of hematite, it possesses stronger surface actively than the latter. This may be as well referred as a circumstantial evidence to testify that. Compared with hematite, collophane can be more easily collected by RFP-138. The composition and distribution of dissolved components in the pulp of different pH values are criteria to evaluate the interaction of collector and minerals. Through the pulp solution equilibrium calculation, the solubility logarithmic diagram of dissolved components can be drawn to judge the interfacial properties of mineral particles in the pulp. The dissolving logarithmic graphs of hematite and collophane calculated by dissolution equilibrium equation are shown in Figs. 9 and 10, respectively.
. The composition and structure of unsaturated bonds on the fractured surface are varied and complicated, and the surface properties for these two minerals are therefore quite different. Besides, as the surface energy of apatite is 0.11 J/m higher than that of hematite, it possesses stronger surface actively than the latter. This may be as well referred as a circumstantial evidence to testify that. Compared with hematite, collophane can be more easily collected by RFP-138. The composition and distribution of dissolved components in the pulp of different pH values are criteria to evaluate the interaction of collector and minerals. Through the pulp solution equilibrium calculation, the solubility logarithmic diagram of dissolved components can be drawn to judge the interfacial properties of mineral particles in the pulp. The dissolving logarithmic graphs of hematite and collophane calculated by dissolution equilibrium equation are shown in Figs. 9 and 10, respectively.

Fig. 9 Hematite dissolved logarithmic diagram

Fig. 10 Phosphate dissolved logarithmic diagram
Figure 9 shows that, when the pH value is over 6, there is no Fe3+ dissolved from hematite. While, for a pulp with a pH less than 6, there is dissolved Fe3+ in the solution and its concentration increases with the increase of solution acidity. In the RFP-138 flotation system, the optimal pH range of separating hematite and collophane is 8.5-9.0. From Fig. 9, it can be seen that there are only Fe(OH)2+ and  interacting with hematite cleavage plane through hydration in this solution, so only some degree of hydrogen bonding reaction occurs between collector and hematite.
interacting with hematite cleavage plane through hydration in this solution, so only some degree of hydrogen bonding reaction occurs between collector and hematite.
According to Fig. 10, Ca2+, Ca (OH) +, 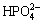 and
and  are localization ions of collophane, so the composition of ions in the solution is complicated. The anionic collector RFP-138 may react chemically with Ca2+ and Ca (OH) + on the surface of collophane.
are localization ions of collophane, so the composition of ions in the solution is complicated. The anionic collector RFP-138 may react chemically with Ca2+ and Ca (OH) + on the surface of collophane.
In order to confirm the inference about the reaction mechanism of RFP-138 and minerals, surface groups on raw minerals and minerals reacting with the collector were characterized by infrared spectra. For both collophane and hematite, the FTIR spectra of raw minerals and the samples reacting with RFP-138 are shown in Figs. 11 and 12.

Fig. 11 FTIR spectra of raw collophane (a) and collophane reacting with RFP-138 (b)

Fig. 12 FTIR spectra of raw hematite (a) and hematite reacting with RFP-138 (b)
A comparison of Fig. 11 and Fig. 12 shows that after the reaction with RFP-138, a C��H stretching vibration peak at 3000 cm-1 hydrated ion near the hydrogen bonds can be observed for both collophane and hematite, while, for collophane reacting with RFP-138, a new characteristic peak centered obviously at around 2031.24 cm-1 which may be attributed to different reaction mechanism for the collector with these two minerals. Thus, it confirms the separation performance of new collector to two kinds of minerals and it is consistent with the previous result of monomineral flotation tests.
5 Conclusions
1) With the aim to reduce phosphorus content in iron ore, a novel collector, RFP-138, was introduced to the reverse flotation process. In the presence of RFP-138, the floatabilities for both hematite and collophane are quite good, and the capacity of RFP-138 in collecting these two minerals varies at different pH values, showing a selectivity of floatability.
2) The recovery rate of iron reaches 91.30%, and the phosphorus content can be decreased from 1.05% to 0.12% using optimal dosage of 600 g/t Na2CO3, 60 g/t water glass and 160 g/t RFP-138 in a reverse flotation process of artificially mixed ore, respectively.
3) According to the solution chemistry and the analyses on FTIR spectra of raw minerals and minerals reacting with RFP-138, RFP-138 can strongly adhere to the surface of collophane in the form of hydrogen bonding and there exists as well a strong chemical adsorption between them, while there is only hydrogen bonding between RFP-138 and hematite. Thus, this study provides a new approach for the rational exploitation of high-phosphorus iron mine resources.
References
[1] YU Yong-fu, QI Chao-ying, MAI Xiao-yu, TANG Xue-feng. Outstanding influence of technical progress in iron ore mineral processing on energy-saving, emission-reduction and efficiency- beneficiation of iron-smelting [J]. Mining and Metallurgical Engineering, 2010(4): 27-32, 35. (in Chinese)
[2] YU Yong-fu. Development trends of iron mines and status and problems of beneficiation technology in China [J]. Mining and Metallurgical Engineering, 2006(1): 21-25. (in Chinese)
[3] YUAN Zhi-tao, GAO Tai, YIN Wan-zhong, HAN Yue-xin. Refractory status and development direction of iron ore resource utilization in China [J]. Metal Mines, 2007(1): 1-6. (in Chinese)
[4] HAO Xian-yao, DAI Hui-xin, ZHAO Zhi-qiang. State quo of phosphorous reduction of high phosphorus iron ore and discussion on its problems [J]. Metal Mines, 2007(1): 7-11. (in Chinese)
[5] RAO Peng. Yichang high phosphorus hematite reverse flotation iron dephosphorization process and mechanism study [D]. Wuhan: Wuhan University of Technology, 2008: 12. (in Chinese)
[6] LIN Xiang-hui, LUO Ren-mei, LIU Jing , LUO Ai-ping , LIN Yuan, HUANG Jun. Recent development of mineral processing methods and flotation reagents for refractory iron ores in the west of hubei [J]. Mining and Metallurgical Engineering, 2007(3): 28-29. (in Chinese)
[7] DONG Yi-bin, QIANG Min, DUAN Zheng-yi, XU Jun-jun, WANG Xin. Effects of reverse flotation with QD series collector on high phosphorus oolitic hematite ores [J]. Metal Mines, 2010(2): 62-65. (in Chinese)
[8] GULMARACS R C, ARAUJO A C, PERES A E A. Reagents in igneous phosphate ores flotation [J]. Minerals Engineering, 2005, 2(8): 181-185.
[9] PI Ke-wu, GONG Wen-qi, LI Yu-biao. Experimental study on leaching dephosphorization of high-phosphorus iron ore in western Hubei [J]. China Mining Magazine, 2010(9): 78-81. (in Chinese)
[10] XIA Wen-tang, LI Yan-hong. High phosphorus iron ore selective removal technology research [J]. Wet Metallurgy, 2011(2): 163-165. (in Chinese)
[11] XIAO Guo-guang, YU Kan-ping, XIAO Yan, YANG Guo-chao. Iron ore flotation anionic/cationic collector agent and its preparation method: China, CN101912822A [P]. 2010-12-15. (in Chinese)
[12] ABOUZEID A Z M, NEGM A T, ELGILLANI D A. Upgrading of calcareous phosphate ores by flotation: Effect of ore characteristics [J]. International Journal of Mineral Processing, 2009, 90(1): 81-89.
[13] ILER R K. The colloid chemistry of silica and silicates [M]. Ithaca, N. Y: Cornell Univ Press. 1955.
[14] XIAO Li-ping, CHEN Jin. Solution chemistry of salt-type mineral flotation [J]. Transactions of Nonferrous Metals Society of China, 1992, 2(3): 19-24.
[15] LUODETING A J, GU Zheng-xing. Crystal chemical characteristics of apatite of violarite flotability influence [J]. Metallic Ore Dressing Abroad, 1994(2): 25-33, 42. (in Chinese).
��٩Ƽ1, 2��������1, 2, 3��������1
1. ���ϴ�ѧ ��Դ�ӹ������﹤��ѧԺ����ɳ 410083��
2. ��ɳ��ұ�о�Ժ�������ι�˾����ɳ 410012��
3. �人������ѧ ��Դ�뻷������ѧԺ���人 430070
ժ Ҫ���������Ƶķ���ѡ���ײ��ռ�(RFP-138)��ϵ�г�����ͽ���Ŀ�ѡ�Լ����û��������о���������ͽ���ĵ����P�˹���Ͽ�ķ���ѡ�������������RFP-138�dz�����ͽ�������ò��ռ����ڼ��������²��ú��ʵ����Ƽ����ܹ���Ч��ʵ�����ֿ���ķ�ѡ����P�������˹���Ͽ��е�1.05%����0.12%����TFe�Ļ����ʴﵽ91.30%����RFP-138��Һ�Ļ�ѧ��Ϊ����������з�����̽��RFP-138�����ֿ���ѡ���Բ��յĻ�����
�ؼ��ʣ�����������ѡ���룻���ף����ײ��ռ�
(Edited by Xiang-qun LI)
Foundation item: Project (2010FJ3069) supported by Science and Technology Project of Hunan Province, China
Corresponding author: Kan-ping YU; Tel: +86-731-88657139; E-mail: yukpp@126.com
DOI: 10.1016/S1003-6326(13)62491-7

