Synergistic coagulation of bauxite residue-basedpolyaluminum ferric chloride for dyeing wastewater treatment
��Դ�ڿ������ϴ�ѧѧ��(Ӣ�İ�)2019���2��
�������ߣ����� ���� �߽��� �߷��� ��� ���˻�
����ҳ�룺449 - 457
Key words��bauxite residue; polyaluminum ferric chloride (PAFC); synergy coagulation; printing and dyeing wastewater
Abstract: Considering that Fe, Al elements in bauxite residue are active components for water purification, an effective polyaluminum ferric chloride (PAFC) coagulant derived from bauxite residue, with Fe2O3 content > 5.1%, Al2O3 % > 6.5%, basicity > 65%, was successfully prepared. The effect of as-prepared PAFC on the zeta potential for printing and dyeing wastewater was investigated. Comparing with polyferric chloride (PFC) and polyferric sulfate (PFS) for printing and dyeing wastewater treatment, prepared bauxite residue-based PAFC exhibited the optimal performance in the aspects of chromaticity and chemical oxygen demand (COD) removal rate. Furthermore, the combination of bauxite residue-based PAFC and PFS for synergy coagulation of such wastewater demonstrated an obvious positive effect. With the proportion between as-prepared PAFC and PFS to be 2.5:1, the COD of treated wastewater could be further reduced to meet the national level A standard of China, providing a promising route to solve the problem of substandard printing and dyeing sewage outfall by a simple coagulation strategy.
Cite this article as: GAO Jian-yang, GAO Fang-zhou, ZHU Feng, LUO Xing-hua, JIANG Jun, FENG Li. Synergistic coagulation of bauxite residue-based PAFC for dyeing wastewater treatment [J]. Journal of Central South University, 2019, 26(2): 449�C457. DOI: https://doi.org/10.1007/s11771-019-4017-7.

ARTICLE
J. Cent. South Univ. (2019) 26: 449-457
DOI: https://doi.org/10.1007/s11771-019-4017-7
GAO Jian-yang(�߽���)1, 2, GAO Fang-zhou(�߷���)3, ZHU Feng(���)4,LUO Xing-hua(���˻�)4, JIANG Jun(����)4, FENG Li(����)1
1. Collage of Chemical Engineering, China University of Mining and Technology, Xuzhou 221116, China;
2. Research Institute of Chalco Shandong, Zibo 255061, China;
3. School of Precision Instruments and Optoelectronic Engineering, Tianjin University,Tianjin 300072, China;
4. School of Metallurgy and Environment, Central South University, Changsha 410083, China
 Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Central South University Press and Springer-Verlag GmbH Germany, part of Springer Nature 2019
Abstract: Considering that Fe, Al elements in bauxite residue are active components for water purification, an effective polyaluminum ferric chloride (PAFC) coagulant derived from bauxite residue, with Fe2O3 content > 5.1%, Al2O3 % > 6.5%, basicity > 65%, was successfully prepared. The effect of as-prepared PAFC on the zeta potential for printing and dyeing wastewater was investigated. Comparing with polyferric chloride (PFC) and polyferric sulfate (PFS) for printing and dyeing wastewater treatment, prepared bauxite residue-based PAFC exhibited the optimal performance in the aspects of chromaticity and chemical oxygen demand (COD) removal rate. Furthermore, the combination of bauxite residue-based PAFC and PFS for synergy coagulation of such wastewater demonstrated an obvious positive effect. With the proportion between as-prepared PAFC and PFS to be 2.5:1, the COD of treated wastewater could be further reduced to meet the national level A standard of China, providing a promising route to solve the problem of substandard printing and dyeing sewage outfall by a simple coagulation strategy.
Key words: bauxite residue; polyaluminum ferric chloride (PAFC); synergy coagulation; printing and dyeing wastewater
Cite this article as: GAO Jian-yang, GAO Fang-zhou, ZHU Feng, LUO Xing-hua, JIANG Jun, FENG Li. Synergistic coagulation of bauxite residue-based PAFC for dyeing wastewater treatment [J]. Journal of Central South University, 2019, 26(2): 449�C457. DOI: https://doi.org/10.1007/s11771-019-4017-7.
1 Introduction
Bauxite residue, a highly alkaline solid residue originated from the caustic digestion of bauxite ores to produce aluminum, has exerted a significant adverse impact on the sustainable development of aluminium industry [1�C4]. This was caused by the alkaline nature, as well as the chemical and mineralogical species presented in bauxite residue, which remained the proper disposal of bauxite residue a huge challenge [5, 6]. Up to date, disposal methods to bauxite residue could be categorized into two main groups: harmless treatment and resource treatment. For the resource treatment, from the aspect of disposal scale, it is important to explore the applicability of bauxite residue as resources to reuse in a large scale. According to current disposal progress, the change of bauxite residue into soil-like resources is the mainstream of strategies to deal with bauxite residue in large scale. XUE et al [7�C10] reported that these bauxite residue disposal areas (BRDD) could enable pioneer plant production after long-term natural weathering process, due to the amelioration on the aspects of alkalinity regulation, formation of the agglomerates [11�C13] and screening of tolerant microorganisms and plants [14�C16]. In consideration of the low additional value of soil as compared to other function materials derived from bauxite residue, to explore a facile method to convert bauxite residue into a functional material of practical use is extremely urgent. Especially, this bauxite residue- based material could be consumed with large demand. Considering the high content of aluminum, iron and other valuable components in the bauxite residue, a coagulant derived from bauxite residue might process promising potential application for waste water treatment.
Coagulant, usually a metallic salt, is added into the water to neutralize the repulsive charge and ��destabilize�� the particles in a colloidal suspension, promoting the increase of zeta potential of colloidal particles. When zeta potential is approximately equal to zero, sewage system exhibits an unstable state and begins to flocculate each other [17, 18]. In the case of bauxite residue-based coagulant, the colloidal particles are negatively charged, and Al elements from bauxite residue served as a coagulant to create positively charged ions. Once the repulsive charges have been neutralized, the van der Waals force will cause the particles to agglomerate into micro-floc. So, the type of coagulant used, its dose and mass were important factors to affect its performance. Currently, WANG et al [19] reported that with the assistance of MgCl2, bauxite residue could remove more than 98% of the coloring material at a dosage of 25 g RM/L dye solution. NI et al [20] also verified that a novel composite bauxite residue/ polyaluminum chloride coagulant was prepared for effective removal of phosphate from aqueous solution. Also, bauxite residue could beused for other waster water treatment applications [21�C26]. On the basis of above reports, the coagulant produced from the bauxite residue has a few advantages compared with the commercial Fe and Al salts [27]: 1) unnecessary coagulant aid or any other chemical during coagulation/flocculation process; 2) negligible pH variations of the treated solution; 3) simultaneous removal of the cationic and anionic species from the wastewater; 4) long-term repeated treatment cycles; 5) negligible leaching of heavy metals from the waste mud under normally environmental conditions; 6) safe transport, storage and handling. However, due to the complicated components in the bauxite residue system, how to convert bauxite residue into efficient coagulant with high Fe/Al content via a facile strategy is still challenging.
The present paper is aimed to develop a kind of economical method to prepare effective polyaluminum ferric chloride (PAFC) coagulant derived from bauxite residue. To evaluate its coagulation effect, this coagulant was used to treat the dyeing wastewater as compared to commercial polyacrylamide (PAM) and polymeric ferric sulphate (PFS) under the same conditions. Results demonstrated that bauxite residue-based PAFC exhibited a great influence on zeta potential of dyeing wastewater and the removal rate of chemical oxygen demand (COD) under the conditions of small addition amount, indicating that bauxite residue-based PAFC was obviously superior to similar commercial materials with higher removal rate of chromaticity and COD. Moreover, the utilization of a continuous reactor (SBR) process of chemical coagulation for disperse dyes and reactive dye wastewater, used continuous coagulation technology in coagulation stage and the COD of sewage, which could be further reduced to reach the national first-class A standard of China when prepared bauxite residue-based PAFC and PFS, was synergistic ally used.
2 Materials and methods
2.1 Materials and experimental instrument
Bayer bauxite residue was collected from Shandong Chalco Aluminum Refinery (China), alkalizing aid, HCl solution (6 mol/L), JS94F microelectrophoresis, 722 spectrophotometer, PHS-3C acidity meter were purchased from Sinopharm Chemical Reagent Co., China. Industrial water glass (modulus 3.21, w(SiO2)=26.1%), caustic soda, hydrochloric acid, 0.025 mol/L CuSO4 standard solution, 0.025 mol/L EDTA standard solution and triethanolamine were purchased from Huiwen Co., China. All materials were used without further purification. Electronic constant temperature water bath (DZKW-4), high pressure reactor (ZBCX-50), precision meter (PHS-3), circulating water type multipurpose vacuum pump (SHZ-C), spectrophotometer (VIS-723), program- controlled flocculation experimental stirrer (TA6-4), zeta potentiometer (Nano-ZS90), microwave sealed digestion COD speed meter were used in the current study.
2.2 Synthesis and characterization of bauxite residue-based PAFC
The principle for PAFC synthesis is shown in Eqs. (1)�C(3):
Al2O3+6HCl+9H2O��2[Al(H2O)6]Cl3 (1)
2AlCl3+9H2O��2[Al(H2O)6]Cl3 (2)
2FeCl3+9H2O��2[Fe(H2O)6]Cl3 (3)
Preparation method: 100 g of bauxite residue and 500 mL of hydrochloric acid (25%) were added into the reaction vessel, by controlling the reaction temperature at 98 ��C. These mixtures reacted for 2 h to get a solution of 450 mL. Then, with a mass of 70% to obtain the basicity, alkalify agents (230 mL) were added into the system repeatedly, by controlling the reaction temperature at 60 ��C. These mixtures were polymerized for 2 h to obtain the clear PAFC products. Finally, these products were cured at room temperature for 24 h to carry out follow-up experiments. Also, basicity, pH, Al2O3, Fe2O3 were measured and the coagulation performance was evaluated. The synthesized PAFC components are shown in Table 1.
Table 1 Chemical composition of bauxite residue-based PAFC

Table 1 shows that the total content of Al and Fe in bauxite residue-based PAFC reached to 11.8% and the basicity was 71.24%. Figure 1 presents the ferron complexation timed spectrophotometric spectrum of bauxite residue-based PAFC, Fe 6 and Fe 4 are the main peaks in the system. Fe 4 exhibits the largest domain value, indicating that the content of Fe in the current PAFC system was close to the highest peak in the PAC system. These results demonstrated that the as-prepared bauxite residue- based PAFC possessed the flocculation properties similar with that of PAC and PFC.
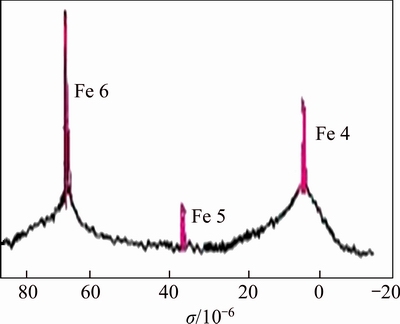
Figure 1 Ferron complexation timed spectrophotometric spectrum of bauxite residue-based PAFC
2.3 Coagulation experiment of PAFC on dyeing wastewater
Dyeing wastewater is discharged from printing and dyeing plants, which mainly contains cotton, hemp, chemical fiber and blended products. According to previous report [38], the common characteristics of dyeing wastewater included large-scale emissions, complex composition, high concentration, high content of organic matter, wide fluctuation of water quality, mutability in water temperature, water volume and pH and difficult treatment. The process of coagulation experiment on dyeing sewage using bauxite residue-based PAFC, PFS and PAM as the basic agent is shown in Figure 2. The water output index of second settling tank is shown in Table 2.
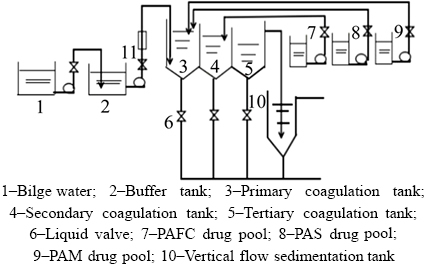
Figure 2 Flow chart of coagulation experiment:
Table 2 Sewage index of second settling sink

In detail, the sewage from secondary settling tank was added into coagulation tank through the buffer tank. Then, by controlling the flow rate at 0.5 m3/h, bauxite residue-based PAFC and PFS were added in order, then PAM (0.01 wt%) was added. Afterwards, setting the stirrer rotation speed to 150 r/min and stirring for 3 min, then remaining the liquid volume residence time for 25 min, the upper coagulation solution entered into vertical flow sedimentation tank. Finally, the supernatant liquid went into the large process final settling tank and the bottom sludge entered the process sludge pressure filtration. The collected samples were analyzed.
3 Results and discussion
3.1 Effect of PAFC with different Al/Fe ratios on COD removal rate
It has been well-established that Al/Fe ratio in coagulant is an important operating factor influencing its coagulation performance. In the current work, the as-prepared bauxite residue-based PAFC samples with different Al/Fe ratios were selected for coagulation experiments to investigate their effect on the COD removal rate for wastewater, with the dosage of PAFC 0.2% and the addition amount of PAM 0.1%. Figure 3 shows the COD removal rate based on different Al/Fe ratios. It was obviously found that the COD removal rate reached 85% with the Al/Fe ratio 1:1. By decreasing the Al/Fe ratio to 0.6 and 0.4, the corresponding COD removal rate decreased to 81% and 80.5%. Also, a higher Al/Fe ratio would result in a lower COD removal rate. In detail, the values of COD removal rate were 82%, 80%, 79%, when the Al/Fe ratio was set to be 1.5, 2.5, 3.5, respectively. The decreasing of COD removal rate with increasing Al/Fe ratio was related to the specific composition of dyeing wastewater. From the above, the bauxite residue-based PAFC with Al/Fe ratio 1 was selected as the optimized value to carry out the next experiments.
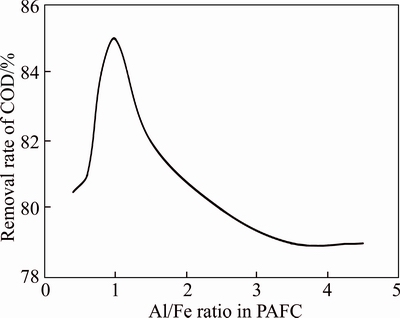
Figure 3 Effect of PAFC with different Al/Fe ratio on COD removal rate
3.2 Effect of PAFC and PFS dosage on zeta potential
In order to compare the coagulation performance of as-prepared bauxite residue-based PAFC with commercial PFS, the dosage of coagulant was selected to investigate its effect on the zeta potential and COD removal efficiency of dyeing wastewater. Figure 4(a) shows the zeta potential related to the dosage of coagulants. With the addition of commercial PFS, the zeta potential increased significantly and the value achieved 0 with 2.5 g/L PFS. In the case of bauxite residue- based PAFC, the increase rate of zeta potential was slower than that of the PFS with the addition of bauxite residue-based PAFC, indicating that PFS and PAFC possess different intrinsic potential zeros for dyeing wastewater. It was interesting to find that bauxite residue-based PAFC reached the same potential as PFS while the dosage was less than that of PFS, indicating that the hydrolysis form of bauxite residue-based PAFC was more conducive to removing COD of dyeing wastewater than that of PFS. Meanwhile, the COD removal rate for the dyeing wastewater originated from the different dosage of bauxite residue-based PFCA and PFS was investigated. As can be seen from Figure 4(b), the COD removal rate increased quickly with the addition of PFS during the initial period as the removal rate of COD was increased from 35% to 75% with an added dosage of 1 g/L, and the final value maintained at 77.5% even a further dosage of 3 g/L added. For the bauxite residue-based PAFC, with the addition of dosage, the COD removal rate was rising slowly, which only reached 56 with 1 g/L of bauxite residue-based PAFC added. But the final COD removal ratio could get 85% with a dosage of 2 g/L. These results demonstrated that the COD removal rate of bauxite residue-based PAFC was lagging behind that of PFS, while the effect was obviously better than that of PFS by the removal rate increased about 8%. In addition, the COD of effluent decreased by 20% and chromaticity reduced by 40%. Therefore, the effect of secondary coagulation technology to treat dyeing wastewater was significant.
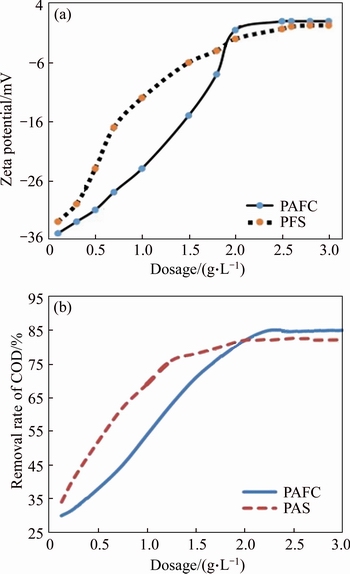
Figure 4 Comparison of influence of coagulant dosage on zeta potential (a), and effect of coagulant dosage on COD removal rate (b)
3.3 Effect of synergistic coagulation on COD removal rate
According to the above experiments, the advantages and disadvantages of various coagulants were complementary. Thus, the effect of synergistic coagulation between bauxite residue-based PAFC and commercial PFS was further investigated. Firstly, on the basis of PFS primary coagulation, additional secondary bauxite residue-based PAFC synergistic coagulation experiment was tested. The PFS-treated water sample was used in the experiment, and the sequence of bauxite residue- based PAFC was added to conduct the experiment to investigate the effect of COD removal rate. The effect of PFS to bauxite residue-based PAFC ratio on COD removal rate is investigated with the total addition amount controlled to 3 g/L. Figure 5 illustrates that the ratio of PFS to bauxite residue-based PAFC exhibited a great influence on the COD removal rate. The lower the removal rate, the smaller the ratio, and the removal rate dropped rapidly. The removal rate could reach the maximum of 90%�C93% at 3:1�C4:1, and further increasing the ratio, the COD removal rate began to decrease.
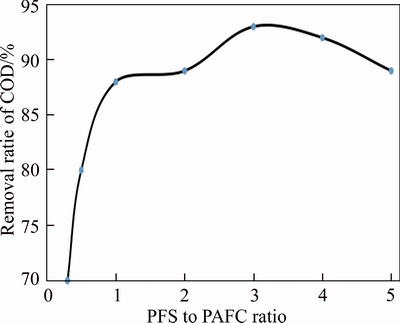
Figure 5 Different dosages between PFS and bauxite residue-based PAFC on COD removal rate
On the contrary, effect of COD removal rate was investigated by adding bauxite residue-based PAFC firstly and adding PFS subsequently. Figure 6 shows the addition ratio of PFS to bauxite residue- based PAFC exhibited a great influence on the COD removal rate, which reached a maximum value of 83 at around 5:1, indicating a poor synergistic coagulation performance. According to positive coagulation with PFS to bauxite residue-based PAFC ratio of 2.5:1�C3.5:1, the COD removal rate could be greater than 90%.
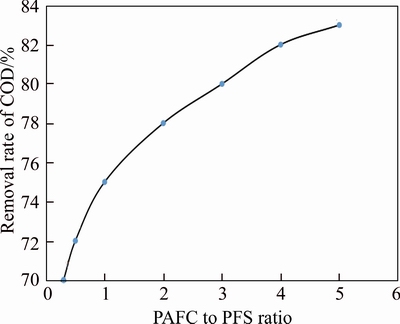
Figure 6 Reverse synergistic effect of PFS to PAFC ratio on COD removal rate
Furthermore, the effect of positive synergistic coagulation derived from the bauxite residue-based PAFC and PAS on the chromaticity of dyeing wastewater was investigated. In the current work, the influence of time on the chromaticity of dyeing wastewater was tested after the coagulation of PFS, bauxite residue-based PAFC and PFS + bauxite residue-based PAFC in accordance with the unified additive amount. The data changes are shown in Figure 7. It can be seen that the chromaticity values of pure PFS and bauxite residue-based PAFC were not as low as the chromaticity of synergistic coagulation under the same addition amount. With the addition of coagulants, all the chromaticity of dyeing wastewater decreased quickly. After 4 h, the slope of curve is becoming smaller, indicating that the dyeing wastewater with low chromaticity became harder to remove. Finally, after 30 h, the values for PFS, bauxite residue-based PAFC and PFS + bauxite residue-based PAFC were 30, 13 and 11, respectively. Thus, the chromaticity removal effect of bauxite residue-based PAFC was better than that of PFS and only the positive synergistic coagulation eventually reached 11.
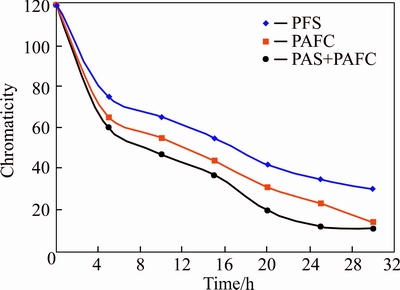
Figure 7 Effect of positive synergistic coagulation time on chromaticity
3.4 Two-stage positive synergistic coagulation continuous experiment
Continuous experiment of samples from industrial sites was carried out under secondary synergistic coagulant technical conditions. Control conditions were set as follows: the effluent of the aerobic tank was stirred and added with 0.25% PFS. After 3 min, 0.1% bauxite residue-based PAFC with Al/Fe=1 was added. After 0.5 min, 0.05% PAM was added, the mixture was stirred for another 15 min, and the supernatant was measured. The final sinking tank is shown in the inset picture in Figure 8, from which we can judge that the final sinking water was clear and transparent with low chromaticity and less pollutants, reaching the standard of efflux. The measurement results are shown in Figure 8. It was obvious to find that the results were better reproduced. Using a secondary synergistic coagulation technique by adding 0.25% PFS and 0.1% bauxite residue-based PAFC, the three replicates achieved a COD removal rate less than 50% and a minimum of 42.8% with the average of 43.23%, which meet the national first- class A standard. Compared with the data of a single coagulation of PFS and bauxite residue- based PAFC in the field, the technical effect was obviously advantageous.
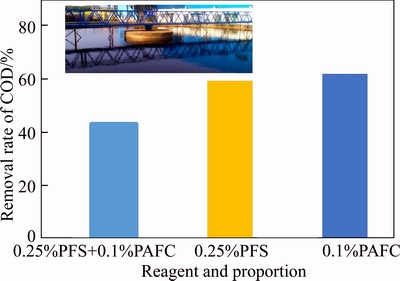
Figure 8 Effect of different industrial two-stage coagulation on COD removal rate
Considering that for the printing and dyeing sewage of Wuxi Water Co., Ltd. from the printing and dyeing industrial park, all kinds of sewage were complex and often fluctuate greatly, we carried out experiments to investigate the COD fluctuation in the following tests. As shown in Figure 9(a), it can be seen that the COD fluctuated greatly within 10 d. In accordance with such fluctuations, coordinated coagulation was conducted, and the changes of bauxite residue-based PAFC and PFS synergistic coagulation effects were tested respectively. Under the conditions of bauxite residue-based PAFC to PFS ratio 3:1, it can be seen that the effluent COD still fluctuates with the fluctuation of COD, but the range is not large. The tracking was under 50 mg/L for 10 d. These results indicated that cooperative coagulation exhibited a strong adaptability to the COD change of printing and dyeing sewage. In addition, the cost of the coagulant is another important index parameter to be considered. In the current work, the treatment cost mainly included raw materials, freight, process modification, and labor costs. The detail cost was calculated as follows: according to the market dry powder PAFC price, the factory price was 1400 RMB/t. The cost of the dry powder prepared into a solution of 1:3 was (1100+300)/3=467 RMB/t. A chemical tank was added which did not need to modify the process and was introduced directly in the middle with the labor cost of 100000 RMB/year, and the incremental cost was 10/1.3=7.7 RMB/t. So, only an increased total cost of 0.49 RMB/t is needed to meet the national first-class A standard. Thus, two- stage positive synergistic coagulation continuous experiment with the dosing bauxite residue-based PAFC and PFS displayed advantages in the aspects of universality and quality, and its cost was also attractive.
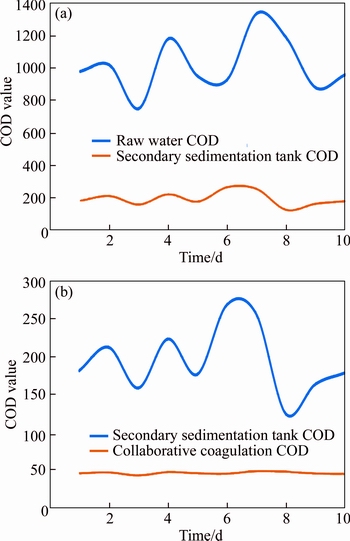
Figure 9 Effects of water quality changes on collaborative coagulation of bauxite residue-based PAFC and PFS
3.5 Mechanism analysis of synergetic coagulation
According to the industrial application test, bauxite residue-based PAFC is composed of polynuclear iron, polyaluminum and chlorine, while PFS is composed of polynuclear iron and sulfate. In coordination with each other, they are complex inorganic polymeric coagulants of polynuclear iron, polyaluminum, chlorine and sulfate, presenting excellent properties of polyaluminum and iron. Synergetic polymerization coagulants mechanism is as follows:
Fe3++H2O��[Fe(OH)]2++H+
or
Al3++H2O��(AlOH)2++H+
Synergistic coagulation of bauxite residue- based PAFS and PFS can be regarded as the formation of PAFCS with the effective Fe/Al content (A12O3+Fe2O3) greater than 10% and basicity>45% for composite products. The results show that PAFCS has better effect on drinking water and sewage treatment when the effective aluminum and iron content of PAFC is higher than PFS, and the decolorization ability of PAFCS is better than that of bauxite residue-based PAFC or PFS for printing and dyeing wastewater. The effluent rate is high, so it is suitable for industrial application and popularization. Compared with the traditional inorganic coagulants, the raw materials of bauxite residue-based PAFC are bauxite residue and hydrochloric acid, which have wider sources and simpler production process. The copolymer of aluminium salt and ferric salt is a coagulants which synthesizes the advantages of PAC and FeC13 more effectively.
4 Conclusions
1) The total amount of Al and Fe in bauxite residue-based PAFC synthesized by bauxite residue reached more than 11.8% and the basicity was 71.24%, which possessed the flocculation properties of PAC and PFC. Moreover, the Fe content in the large system of PAC was close to the highest peak.
2) The PAFC synthesized by bauxite residue had different potential characteristics from PFS, which caused the COD removal rate of PAFC lagged behind PFS. When the effect was obviously better than PFS with the removal rate reaches about 85%, the effluent COD was reduced by 20% and the chromaticity value was reduced by 40%.
3) The bauxite residue-based PAFC with Al/Fe=1 was added to final sinking water treated by PFS coagulation for positive synergistic coagulation could further reduce COD according to (2.5�C3.5):1 and the COD through industrial re-certification reached 42.23, which meets the national first-class A standard of China.
4) The second synergistic coagulation technology did not need to be greatly modified in the process and the cost per ton of water was increased by 0.49 RMB, which could improve the upgrading of dyeing wastewater treatment.
5) The synergetic polymerization coagulation mechanism derived from the bauxite residue-based PAFC and PFS could release even more polyaluminum and iron-based active constituents to enhance their synergistic coagulation on the dyeing wastewater treatment.
References
[1] LI Xiao-fei, YE Yu-zhen, XUE Sheng-guo, JIANG Jun, WU Chuan, KONG Xiang-feng, HARTLEY W, LI Yi-wei. Leaching optimization and dissolution behavior of alkaline anions in bauxite residue [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(6): 1248�C1255. DOI: 10.1016/S1003-6326(18)64763-6.
[2] LIU Gui-hua, WU Guo-yu, JIANG Hai-lang, QI Tian-gui, PENG Zhi-hong, ZHOU Qiu-sheng, LI Xiao-bin. Safe utilization of chromium-bearing vanadate residue by recovery of vanadium and chromium based on calcium circulation [J]. Journal of Central South University, 2018, 25: 2349�C2359. DOI: org/10.1007/s11771-018-3919-0.
[3] LIAO Jia-xin, JIANG Jun, XUE Sheng-guo, CHENG Qing- yu, WU Hao, RAJENDRAN M, HARTLEY W, HUANG Long-bin. A novel acid-producing fungus isolated from bauxite residue: The potential to reduce the alkalinity [J]. Geomicrobiology Journal, 2018, 35(10): 840�C847. DOI: 10.1080/01490451.2018.1479807.
[4] CRAMER A J, COLE J M. Removal or storage of environmental pollutants and alternative fuel sources with inorganic adsorbents via host-guest encapsulation [J]. Journal of Materials Chemistry A, 2017, 5(22): 10746�C10771. DOI: 10.1039/C7TA02401K.
[5] KONG Xiang-feng, JIANG Xing-xing, XUE Sheng-guo, HUANG Ling, HARTLEY W, WU Chuan, LI Xiao-fei. Migration and distribution of saline ions in bauxite residue during water leaching [J]. Transactions of Nonferrous Metals Society of China, 2018, 28(3): 534�C541. DOI: 10.1016/ S1003-6326(18)64686-2.
[6] MEHMET K, ORHAN T C, MAHMUT B. Treatment of textile wastewaters by electrocoagulation using iron and aluminum electrodes J]. Journal of Hazardous Materials, 2003, 100: 163�C178. DOI: org/10.1016/S0304- 3894(03)00102-X.
[7] XUE Sheng-guo, LI Meng, JIANG Jun, MILLAR G J, LI Chu-xuan, KONG Xiang-feng. Phosphogypsum stabilization of bauxite residue: Conversion of its alkaline characteristics [J]. Journal of Environmental Sciences, 2019, 77: 1�C10. DOI: 10.1016/j.jes.2018.05.016.
[8] WANG Xin-ke, ZHANG Yi-he, LU Rong-rong, ZHOU Feng-shan, AN Qi, MENG Zi-lin, FEI Bin, LV Feng-zhu. Novel multiple coagulant from Bayer red mud for oily sewage treatment, Desalination and Water Treatment [J]. Journal of Water Process Engineering, 2015, 6: 158�C165. DOI: org/10.1016/j.jwpe.2015.04.003.
[9] KONG Xiang-Feng, TIAN Tao, XUE Sheng-guo, HARTLEY W, HUANG Long-bin, WU Chuan, LI Chu-xuan. Development of alkaline electrochemical characteristics demonstrates soil formation in bauxite residue undergoing natural rehabilitation [J]. Land Degradation and Development, 2018, 29(1): 58�C67. DOI: 10.1002/ldr.2836.
[10] XUE Sheng-guo, YE Yu-zhen, ZHU Feng, WANG Qiong-li, JIANG Jun, HARTLEY W. Changes in distribution and microstructure of bauxite residue aggregates following amendments addition [J]. Journal of Environmental Sciences, 2019, 78: 276�C286. DOI: 10.1016/j.jes.2018.10.010.
[11] ZHU Feng, CHENG Qing-yu, XUE Sheng-guo, LI Chu-xuan, HARTLEY W, WU Chuan, TIAN Tao. Influence of natural regeneration on fractal features of residue microaggregates in bauxite residue disposal areas [J]. Land Degradation and Development, 2018, 29(1): 138�C149. DOI: 10.1002/ ldr.2848.
[12] ZHU Feng, LIAO Jia-xin, XUE Sheng-guo, HARTLEY W, ZOU Qi, WU Hao. Evaluation of aggregate microstructures following natural regeneration in bauxite residue as characterized by synchrotron-based X-ray micro-computed tomography [J]. Science of the Total Environment, 2016, 573: 155�C163. DOI: 10.1016/j.scitotenv.2016.08.108.
[13] ZHU Feng, LI Yu-bing, XUE Sheng-guo, HARTLEY W, WU Hao. Effects of iron-aluminium oxides and organic carbon on aggregate stability of bauxite residues [J]. Environmental Science and Pollution Research, 2016, 23(9): 9073�C9081. DOI: 10.1007/s11356-016-6172-9.
[14] WU Chuan, SHI Li-zheng, XUE Sheng-guo, LI Wai-chin, JIANG Xing-xing, RAJENDRAN M, QIAN Zi-yan. Effect of sulfur-iron modified biochar on the available cadmium and bacterial community structure in contaminated soils [J]. Science of the Total Environment, 2019, 647: 1158�C1168. DOI: 10.1016/j.scitotenv.2018.08.087.
[15] WANG Jun, CHENG Qing-yu, XUE Sheng-guo, RAJENDRAN M, WU Chuan, LIAO Jia-xin. Pollution characteristics of surface runoff under different restoration types in manganese tailing wasteland [J]. Environmental Science and Pollution Research, 2018, 10: 9998�C10005. DOI: 10.1007/s11356-018-1338-2.
[16] XUE Sheng-guo, WU Yu-jun, LI Yi-wei, KONG Xiang-feng, ZHU Feng, HARTLEY W, LI Xiao-fei, YE Yu-zhen. Industrial waste applications for alkalinity regulation in bauxite residue: A comprehensive review [J]. Journal of Central South University, 2019, 26(2): 268�C288.
[17] ZHOU Lyu, ZHOU Hong-jie, YANG Xiao-yu. Preparation and performance of a novel starch-based inorganic/organic composite coagulant for textile wastewater treatment [J]. Separation and Purification Technology, 2019, 210: 93�C99. DOI: org/10.1016/j.seppur.2018.07.089.
[18] LIANG Yan-ling, KRAUS T E C, SILVA L C R, BACHAND P A M, BACHAND S M, DOANE T A, HORWATHA W R. Effects of ferric sulfate and polyaluminum chloride coagulation enhanced treatment wetlands on Typha growth, soil and water chemistry [J]. Science of the Total Environment, 2019, 648: 116�C124. DOI: org/10.1016/j.scitotenv.2018.07.341.
[19] WANG Qi, LUAN Zhao-kun, WEI Ning, LI Jin, LIU Cheng-xi. The color removal of dye wastewater by magnesium chloride/red mud (MRM) from aqueous solution [J]. Journal of Hazardous Materials, 2009, 170: 690�C698. DOI: org/10.1016/j.jhazmat.2009.05.011.
[20] NI Fan, HE Jin-song, WANG Yan-bin, LUAN Zhao-kun, Preparation and characterization of a cost-effective red mud/polyaluminum chloride composite coagulant for enhanced phosphate removal from aqueous solutions [J]. Journal of Water Process Engineering, 2015, 6: 158�C165. DOI: org/10.1016/j.jwpe.2015.04.003.
[21] EL-GOHARY F, TAWFIK A. Decolorization and COD reduction of disperse and reactive dyes wastewater using chemical-coagulation followed by sequential batch reactor (SBR) process [J]. Desalination, 2009, 249(3): 1159�C1164. DOI: org/10.1016/j.desal.2009.05.010.
[22] KOBYA M, ONCEL M S, DEMIRBA E,  IKB E, AKYOL A, INCE M. The application of electrocoagulation process for treatment of the red mud dam wastewater from Bayer��s process [J]. Journal of Environmental Chemical Engineering, 2014, 2(4): 2211�C2220. DOI: org/10.1016/j.jece.2014.09. 008.
IKB E, AKYOL A, INCE M. The application of electrocoagulation process for treatment of the red mud dam wastewater from Bayer��s process [J]. Journal of Environmental Chemical Engineering, 2014, 2(4): 2211�C2220. DOI: org/10.1016/j.jece.2014.09. 008.
[23] OLIVEIRAA A A S, TEIXEIRAA I F, CHRISTOFANIA T A I S, TRIST OB J C, GUIMAR
OB J C, GUIMAR ESC I R, MOURA F C C. Biphasic oxidation reactions promoted by amphiphilic catalysts based on red mud residue [J]. Applied Catalysis B: Environmental, 2014, 144: 144�C151. DOI: org/10.1016/ j.apcatb.2013.07.015.
ESC I R, MOURA F C C. Biphasic oxidation reactions promoted by amphiphilic catalysts based on red mud residue [J]. Applied Catalysis B: Environmental, 2014, 144: 144�C151. DOI: org/10.1016/ j.apcatb.2013.07.015.
[24] BENTOA N I, SANTOSA P S C, de SOUZAB T E, OLIVEIRAB L C A, CASTRO C S. Composites based on PET and red mud residues as catalyst for organic removal from water [J]. Journal of Hazardous Materials, 2016, 314: 304�C311. DOI: org/10.1016/j.jhazmat.2016.04.066.
[25] CRINI G. Non-conventional low-cost adsorbents for dye removal: A review [J]. Bioresource Technology, 2006, 97(9): 1061�C1085. DOI: org/10.1016/ j.biortech.2005.05.001.
[26] KAZAK O, TOR A, AKINB I, ARSLANC G. Preparation and characterization of novel polysulfone-red mud composite capsules for the removal of fluoride from aqueous solutions [J]. RSC Advances, 2016, 6(89): 86673�C86681. DOI: 10.1039/C6RA12055E.
[27] WANG Shao-bin, ANG T H M, TADE M O. Novel applications of red mud as coagulant, adsorbent and catalyst for environmentally benign processes [J]. Chemosphere, 2008, 72: 1621�C1635. DOI: org/10.1016/j.chemosphere.2008. 05.013.
[28] YANG Cong-ren, JIAO Fen, QIN Wen-qing. Leaching of chalcopyrite: An emphasis on effect of copper and iron ions [J]. Journal of Central South University, 2018, 25: 2380�C2386. DOI: org/10.1007/s11771-018-3922-5.
(Edited by YANG Hua)
���ĵ���
�����PAFCЭͬ����ӡȾ��ˮ�������о�
ժҪ�����ó��������е�Fe��Al�Ⱦ��о�ˮ���õ�Ԫ�أ��Ʊ���Fe2O3����>5.1%��Al2O3����>6.5%�����>65%�ij����PAFC�������˳����PAFC��ӡȾ��ˮzeta��λ��Ӱ�졣�Գ�����ۺ��Ȼ�������PAFC�����ۺ��Ȼ�����PAC�����ۺ��Ȼ�����PFC���;ۺ���������PFS����ӡȾ��ˮ�����н��л����Աȵ��о������������PAFC��ɫ�Ⱥ�CODȥ���ʷ�����������ͬ���Ʒ�� ��һ���Ա��о�PAFC��PFSЭͬ������Ч�����Ż��������PAFC����Эͬ����PFS������2.5:1��Ͷ�ϱȣ��ɽ�һ������ӡȾ��ˮCODȥ���ʣ��ﵽ����1��A�ı����ܺõؽ����ӡȾ��ˮ���Ų��������⡣
�ؼ��ʣ����ࣻ�ۺ��Ȼ�������PAFC����Эͬ������ӡȾ��ˮ
Foundation item: Project(BE2015628) supported by Jiangsu Province Science and Technology Support Program, China
Received date: 2018-11-10; Accepted date: 2018-12-13
Corresponding author: JIANG Jun, PhD; Tel: +86-13618496840; E-mail: junjiang@csu.edu.cn; FENG Li, PhD; Tel: +86-13852488050; E-mail: cumthgfl@163.com; ORCID: 0000-0002-4288-2908

