���±�ţ�1004-0609(2014)03-0779-08
H2SO4����������Ȼʯī�ĵ绯ѧ���ܼ�Ƕ��﮶���ѧ
�컪��1, 2��³����2��������2���� ��1���� ��2���ű���2��������1
(1. ���ϴ�ѧ ұ���뻷��ѧԺ����ɳ 410083��
2. ��ɳ������ѧ ��������ӿ�ѧѧԺ����ɳ 410004)
ժ Ҫ��������Ϊ�������μ������ý��շ�����Ȼʯī���б������θ��ԡ�����Ҷ�任���������(FTIR)�����������������������ε���Ȼʯī���桪OH��C=C��ʧ����COO-�������ࡣ�绯ѧ���ܲ��Խ��������������������ε���Ȼʯī��ѭ�����ܺͱ������ܾ��õ���ߡ���3 mol/L H2SO4����12 h����Ȼʯī(NGS3)��0.5C��20��ѭ�����������Ϊ320.5 mA��h/g����δ���������ε���Ȼʯī(NG)����ͬ�����µ����������Ϊ299.9 mA��h/g�����ý����迹��ʯī����Ƕ��﮶���ѧ�о��������ʾ��������������ε���ȻʯīĤ����(RSEI)�͵��ת�Ƶ���(Rct)����С��Ĥ����(CSEI)��˫������(CCPE)���ӣ����ѭ����RSEI�����ȶ���NG�Ļ��(Ea)Ϊ87.7 kJ/mol��NGS3��EaΪ77.2 kJ/mol������ H2SO4���������������ȥ�ܼ�����������ߣ����������γ��ȶ��Ĺ������ʽ���(Solid electrolyte interface��SEI)��
�ؼ��ʣ���Ȼʯī���������Σ��������������ʽ��棻�����迹�����
��ͼ����ţ�TQ035���� ���ױ�־�룺A
Electrochemical performance and kinetics on lithium ion insertion/deinsertion of sulfuric acid surface-modified natural graphite
ZHU Hua-li1, 2, LU Pan-pan2, CHEN Zhao-yong2, LI Jie1, CHEN Fen2, DU Bing-lin2, LAI Yan-qing1
(1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. School of Physics and Electronic Science, Changsha University of Science and Technology, Changsha 410004, China)
Abstract: The natural graphite was modified by impregnation method with sulfuric acid as surface modification agent. FTIR results show that ��OH and C=C bonds on the surface of natural graphite disappear while the number of ��COO- increases after sulfuric acid surface-modification. The electrochemical performance reveals that the cycle performance and ratio performance are improved. The lithium-deinsertion capacities of natural graphite impregnated by 3 mol/L H2SO4 for 12 h (NGS3) and unmodified natural graphite (NG) are respectively 320.5 and 299.9 mA��h/g after 20 cycles at 0.5C. AC impedance was used to investigate kinetics on lithium ion insertion/ deinsertion. It is found that the membrane resistance (RSEI) and charge transfer resistance (Rct) decrease, while the membrane capacitance (CSEI) and extracted capacitance (CCPE) increase. Moreover, RSEI keeps constant after several cycles. The activation energies of NG and NGS3 are 87.7 and 77.2 kJ/mol, respectively, which indicates that sulfuric acid treatment is beneficial to the improvement of the desolvation of lithium ion from solvent molecules and the formation of a stable solid electrolyte interface (SEI).
Key word: natural graphite; surface-modification; anode; solid electrolyte interface; alternating impedance; activation energy
��Ȼʯī������Դ�ḻ���۸�������ŵ㣬�����������ӵ�ظ������ϡ�Ȼ������Ȼʯī�γɹ����д��ڵIJ��־���ȱ�ݺ�Ƕ﮹����е��ܼ����ӹ�Ƕ������ѭ�������з���ʯīƬ���������ѭ�����������½�[1]�����⣬����ʯīͬ��ԲƬ��ѻ��ṹ[2]��������������������롣��ˣ���Ȼʯī�뾭�����Դ����ſ���������ӵ�ظ������ϵ�Ҫ��
�������δ���һ�����������ʯī�����ȱ��[3]������ʯī����Ļ��Ե㣬�Ӷ���������ӵ����ģ���һ���������һЩ������ͨ��[4]�����Ӵ��λ��ͬʱ���������ԡ�COO-���ţ��γ����ܵĹ������ʽ���(SEI)�ۻ�Ĥ[5]���Ӷ���ֹ�ܼ��Ĺ�Ƕ�ͷֽ�[6]�����Ͳ��������������ѭ����������������ʯī�������ϵĵ绯ѧ����[7]��Ŀǰ����Ȼʯī�ı������η�����Ϊ���෨��Һ�෨�����෨������������PELED��[8]�״�����ģ���ѡ����������μ��п�����������CO2��[9-10]���������෨��Ҫ�о�����ʧ���������¶Ⱥ�����ʱ��Ե绯ѧ���ܡ���������ż�SEIĤ�γɵ�Ӱ�졣Һ�෨���õ����μ���HNO3��H2O2��CeSO4��(NH4)2S2O8��H2SO4�ȣ�����[11-15]��Ҫ�о������μ��Ա�������š��ṹ���绯ѧ���ܵ�Ӱ�졣
�������߲���H2SO4����Ȼʯī���б������Σ��Ż��˷�ӦŨ�Ⱥͷ�Ӧ�¶ȵȸ������������ý����迹���о�Ƕ��﮹����г����Ԫ��(CPE)[16]������˫������(CCPE)[17]��Ĥ����(CSEI)[18]�Ķ���ѧ�仯������ǰ��Ļ��(Ea)�仯����H2SO4�������ζ���Ȼʯī�绯ѧ���ܵ�Ӱ�졣
1 ʵ��
1.1 ��Ʒ�Ʊ�
ȡ25 g������Ȼʯī���ڴ�����������װ�õ�������ƿ�У��ֱ����100 mL 1��2��3 mol/L��H2SO4��Һ���ڲ�ͬ���¶�(0��60��100 ��)��ˮԡ12 h����ˣ����ò��ᆳȥ����ˮ����ϴ������ҺΪ���ԣ����˺������100 ����ո���12 h���õ���Ʒ��H2SO4����������Ȼʯī�ķ�Ӧ��������1��
1.2 ���ϵı���
����Nicolet Magna��˾������AVATAR-360�ͺŸ���Ҷ�任���������(FTIR)������Ʒ����Ļ�ѧ��֣�ɨ�跶ΧΪ4000~600 cm-1���ֱ���Ϊ4 cm-1��
1.3 ��ص���װ���绯ѧ���ܲ���
��1 H2SO4����������Ȼʯī�ķ�Ӧ����
Table1 Reaction conditions of natural graphite surface- modification with H2SO4

��������94:2.5:3.5��ȡ��Ȼʯī��Ʒ�������(��Ȳ��)��ճ���(PVDF)������������N-��������ͪ(NMP)��ֻ���Ƴɽ��ϣ���������2 h��Ϳ���ں��Ϊ10 ��m��ͭ���ϣ���120 ����ո���4 h���ѹ��ֱ��Ϊ10 mm��ԲƬ��Ϊ�����缫���缫���Ϊ0.785 cm2���Խ����ƬΪ�Ե缫��1 mol/L LiPF6/(EC+DMC+EMC) (������Ϊ1:1:1)Ϊ���Һ��Celgard 2400Ϊ��Ĥ���ڸߴ�Ar������������(�������й�����˾MIKROUNA Super(1220/750)��)����װ��CR2025�Ϳ�ʽ��ء�
����CT2001A�������ز����ǶԵ�ؽ��г�ŵ���ԡ����Ե�ѹ��ΧΪ0~2 V����ŵ籶�ʷֱ�Ϊ0.1C��0.2C��0.5C�������¶�Ϊ25 �档
�����Ϻ�������˾������CHI660B�͵绯ѧ����վ���н����迹���ԡ������迹�IJ����������£�ɨ���Ƶ�ʷ�Χ0.01~100000 Hz�����Ϊ0.005 V��Ϊ�˼����ܣ���ģ�������ڸߵ���ʵ����(�Ϻ�³��GDW/LX-100)�У��ֱ���0��15��25��35 ���½��н����迹���ԡ�
2 ���������
2.1 FTIR����
ͼ1��ʾΪ��H2SO4��������ǰ (NG)��(NGS3) ��Ȼʯī�ĺ�����ס�3516��1643 cm-1�������շ�ֱ��ӦNG������ǻ�(��OH)[19]��̿̿˫��(C=C)��[20]��1697��1732 cm-1�����շ�ֱ��ӦNG��NGS3������Ȼ�(��COO-)��[19, 21]����ͼ1��֪�� NGS3��3500��1640 cm-1�����ķ���ʧ����COO-����ǿ����˵��H2SO4��������ʹ��COO-���ŵ���������[22]��
2.2 �绯ѧ����

ͼ1 ��H2SO4��������ǰ(NG)��(NGS3)��Ȼʯī�ĸ���Ҷת���������
Fig. 1 FTIR spectra of natural graphite samples before (NG) and after (NGS3) H2SO4 surface-modification
��H2SO4��������ǰ����Ȼʯī���״�Ƕ������ܼ���2���ɱ�2��֪��H2SO4���������������Ȼʯī���״ο����������״ο���������345.6 mA��h/g���ӵ�365.0 mA��h/g���������������״�Ч��û�����Եı仯��
ͼ2��ʾΪ��Ȼʯī�;�H2SO4�������ε���Ȼʯī�ڲ�ͬ�����µ�ѭ���������ߡ��ɼ�����H2SO4����������Ȼʯī��ѭ��������H2SO4Ũ�ȵ����Ӻͷ�Ӧ�¶ȵĽ��Ͷ���ߡ���H2SO4�������εõ�����Ȼʯī��ƷNGS1��NGS3��NGS3-1����0.5C������ѭ��20�κ����ﮱ������ֱ�Ϊ313.1��320.5��316.8 mA��h/g�����������ʷֱ�Ϊ90.4%��87.8%��88.6%����δ���������ε���ȻʯīNG��0.5C������ѭ��20�κ����ﮱ�������Ϊ299.9 mA��h/g������������Ϊ86.8%���Ҿ�H2SO4����������Ȼʯī�ı�������Ҳ�õ���ߣ�NGS3��0.2C��0.5C�µ���ﮱ������ֱ�Ϊ339.5��333.8 mA��h/g����Ӧ�����������ʷֱ�Ϊ93.0%��91.5%����NG����ͬ�����µ���ﮱ������ֱ�Ϊ311.9��294.6 mA��h/g����Ӧ�����������ʷֱ�Ϊ90.2%��85.2%��
H2SO4�������������������Ȼʯī�ĵ绯ѧ���ܣ�����������H2SO4�������κ����ɴ�����RO��COO-[23]��RO��COO-��Li+��Ӧ����RO��COOLi[24-25]����ΪSEIĤ����Ҫ�ɷ֣��ܹ��ٽ�SEIĤ�Ŀ����γɣ��Ӷ�������Ȼʯī��ѭ�����ܺͱ������ܡ�
2.3 �����迹����
��2 H2SO4��������ǰ����Ȼʯī���״�Ƕ�������(0.1C)
Table 2 First insertion/deinsertion performance of natural graphite before and after H2SO4 surface-modification (0.1C)


ͼ2 ��ͬ�����±������κ���Ȼʯī��Ʒ��ѭ���ͱ�����������
Fig. 2 Cycle performance and rate capacity of natural graphite surface-modified under different conditions

ͼ3 ��������ǰ(NG)��(NGS3)��Ȼʯī��Ʒ�ڲ�ͬ��ѹ��(0.7��0.5��0.2��0.1 V)�״�ѭ���Ľ����迹������Ӧ���������
Fig. 3 Electrochemical impedance spectroscopy Nyquist plots and fitted curves of natural graphite samples before and after surface-modification at various potentials (0.7, 0.5, 0.2, 0.1 V) for the first cycle
SEIĤ���״�Ƕ﮹������γ�[26]��Ϊ���о�H2SO4�������ζ���Ȼʯī����SEIĤ�γɹ��̼��γɺ��ȶ��Ե�Ӱ�죬������Ʒ�ڲ�ͬǶ��﮵�ѹ���״κͶ��ѭ����Ľ����迹�ױ仯��ͼ3��ʾΪ��Ȼʯī��������ǰ����Ʒ�Ľ����迹����Ӧ��������ߡ�ͼ4��ʾΪ��ϲ��õĽ����迹��·ͼ��ͼ4�У�Re�������Һ�迹��CSEI����Ĥ���ݣ�RSEI����Ĥ�迹��CPE���������Ԫ����Rct�������ת�Ƶ��裬Wo������ɢ�迹������Re��CSEI��RSEI�����ڸ�Ƶ����CPE��Rct��������Ƶ����Wo�����ڵ�Ƶ��[27-29]����ͼ3��֪��������ߺ�ʵ���������غϣ�˵����ϵĽ����迹��·ͼ�Ǻ����ġ��ڴ�����о���ֻ�ص�������RSEI��Rct�ı仯��������CSEI��CPE[30-31]��CPE�Ǹ��ϸ��ӵ�Ԫ������Щ�о���Ϊ�˼���Cdl��ȡ��CPE[32]���������߳��о�RSEI��Rct�⣬����CSEI��CPE���������ۣ���CPE����ȡ����ݲ��֣���ȡ�ĵ��ݲ���CCPE���㹫ʽ���£�
 (1)
(1)
ʽ�У�QΪ����������ʾ�缫����ļ����̶ȣ�Ϊ0~1֮��������ٳ��������迹ͼ�����õ�������ȡ������HSU�����[17, 33]��
��Ȼʯī��������ǰ��Ƕ��﮹����в�ͬ��ѹ�µĽ����迹�����仯��ͼ5����ͼ5(a)��(b)��֪��NG��Ƕ﮹�����RSEI���������ƣ�����2.050 ��������3.361 ����Rct�ʼ�С���ƣ�����11.570 ����С��7.484��������﮹�����RSEI��Rct�������������ƣ���RSEI��2.544 ��������3.918 ����Rct��14.610 ��������22.440 ������NGS3��Ƕ��﮵Ĺ����е�RSEI��Rct�����㶨���䣬RSEIά����1.468 �����ң�Rctά����5.783�����ҡ�����ͬ��Ƕ��﮵ı仯�����У�NGS3��RSEI��Rct��С��NG�ģ�����Ƕ�ʱ��0.5 V��ѹ�£�NG��Ӧ��RSEI��Rct�ֱ�Ϊ2.635��8.355 ������NGS3��Ӧ��RSEI��Rct�ֱ�Ϊ1.623��6.631 ����˵����H2SO4�������ε���Ȼʯī��Ƕ��﮹������ܿ����γ��ȶ���SEIĤ��

ͼ4 �����迹����ϵ�·ͼ
Fig. 4 Equivalent circuit used to fit AC impedance Nyquist plots

ͼ5 ��ͬ��ѹ�±�������ǰ(NG)��(NGS3)��Ȼʯī��Ʒ�״�Ƕ��﮹����еĽ����迹����
Fig. 5 AC impedance parameters of natural graphite samples before (NG) and after (NGS3) surface-modification at different voltages during first lithium insertion/de-insertion process
�����������ɵ�������ṹ�й�[34]�����ݵĴ�С�ɷ�ӳ�洢﮵��������ṹ��������ͼ5(c)��(d)�ɿ�����NG��Ƕ﮹�����CSEI�ʼ�С���ƣ�����5.335 ��F��С��3.449 ��F��CCPE���������ƣ�����21.05 ��F������28.57 ��F������﮹�����CSEI��CCPE���ʼ�С���ƣ���CSEI��2.745 ��F��С��1.745 ��F��CCPE��8.581 ��F��С��4.252 ��F��NGS3��Ƕ﮹�����CSEI��CCPE�����������ƣ���CSEI��7.502 ��F������8.242 ��F��CCPE��34.25 ��F������41.67 ��F������﮹�����CSEI��CCPE�ʼ�С���ƣ���CSEI��9.059 ��F��С��4.447 ��F��CCPE��38.38 ��F��С��24.40 ��F����ͬ����Ƕ��ﮱ仯�����У�NGS3��CSEI��CCPE������NG�ģ�˵����H2SO4�������ε���Ȼʯī����ṹ���ӣ�����������ӵĴ洢��Ƕ����������ӣ���ͼ2�е绯ѧ����һ�¡�
�����������ǰ����Ȼʯī�����ѭ���Ľ����迹��RSEI�ı仯��ͼ6��ʾ����ͼ6��֪����ѭ���Ľ��У�NGS3��RSEI����ά����2.548 �����Ҳ��䣬��NG��RSEI�����Եı仯�Ҽ��䲻�ȶ�����4.323 ��ͻ����7.342 ����Ȼ���С��5.848 ������һ��˵��H2SO4����������Ȼʯī���γ��ȶ���SEIĤ�������ڵ绯ѧ���ܵ���ߡ�
2.4 ��ܷ���

ͼ6 ��������ǰ(NG)��(NGS3)��Ȼʯī��Ʒ�����ѭ����Ľ����迹��RSEI
Fig. 6 Electrochemical impedance spectra and RSEI changes of natural graphite samples before (NG) and after (NGS3) surface-modification after manifold cycles
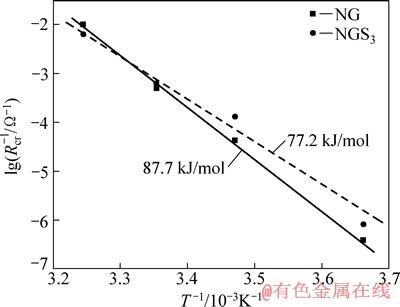
ͼ7 ��Ȼʯī��LiPF6/(EC+DMC+EMC)(������Ϊ1:1:1)�ĵ��Һ�н��浼���ʺ��¶ȵĹ�ϵ
Fig. 7 Temperature dependence of natural graphite on interfacial conductivity (1/Rct) on in LiPF6/(EC+DMC+EMC) (mass ratio of 1:1:1) electrolytes
��ܿɷ�ӳ�����������Ȼʯī�缫�͵��Һ���洫��Ķ���ѧ[35]������Ӵ��ܼ�������ȥ�ܼ������������Խ�ͱ���ȥ�ܼ�������Խ��[32]��Ϊ���о�H2SO4�������ζ���Ȼʯī��ܵ�Ӱ�죬�����˱�������ǰ����Ȼʯī��Ʒ��Ƕ﮵�ѹΪ2.0 V����ͬ�¶�(0��15��25��35 ��)�½����迹�ı仯��ͼ7��ʾΪ��Ȼʯī��LiPF6/(EC+DMC+EMC)(������Ϊ1:1:1)�ĵ��Һ�н��浼�������¶ȵĹ�ϵ�����洫������ѭ��������˹����ʽ[36]��
 (2)
(2)
ʽ�У�A��Ea��R��T�ֱ����Ƶ�����ӡ����ۻ�ܡ�Ħ�����峣��������ѧ�¶ȡ�����ʽ(2)�����֪��NG��EaΪ87.7 kJ/mol��NGS3��EaΪ77.2 kJ/mol��YAMADA��[32]��ABE��[35]��õ�ʯī/���Һ����Ļ����50 kJ/mol���һ���ߣ����о�������������õĽ������������ɻ�ܽ����֪��H2SO4�������ν����˽���Ļ�ܣ��Ӷ����������ӵ�ȥ�ܼ����������ǵ绯ѧ������ߵ�ԭ��֮һ��
3 ����
1) H2SO4���������������Ȼʯī�ı���������ѭ�����ܣ���0.2C�µ���ﮱ�������311.9������339.5 mA��h/g��������������90.2%������93.0%����0.5C�µ���ﮱ�������294.6������333.8 mA��h/g��������������85.2%������91.5%������0.5C��20��ѭ��������������299.9����320.5 mA��h/g��
2) ��H2SO4���κ����Ȼʯī���������˴�����COO-�������ڿ����γ��ȶ���SEIĤ������RSEI��Rct��H2SO4��������������SEIĤ����Ŀ��ʹCSEI��CCPE����������Ȼʯī�Ĵ��������
3) NG��NGS3��Ʒ��Ea�ֱ�Ϊ87.7��77.2 kJ/mol������H2SO4���������������������Ӵ��ܼ������е�ȥ�ܼ�������������SEIĤ���ȶ��ԣ��Ӷ������Ȼʯī�ĵ绯ѧ���ܡ�
REFERENCES
[1] �ƿ���, ������, ������. ����ӵ��ԭ����ؼ�����[M]. ����: ��ѧ��ҵ������, 2008.
HUANG Ke-long, WANG Zhao-xiang, LIU Su-qing. Principle and key technology of lithium ion battery[M]. Beijing: Chemistry Industry Press, 2008.
[2] YOSHIO M, WANG H Y, FUKUDA K, UMENO T, ABE T, OGUMI Z. Improvement of natural graphite as a lithium-ion battery anode material: From raw flake to carbon-coated sphere[J]. Journal of Materials Chemistry, 2004, 14: 1754-1758.
[3] WU Y P, JIANG C Y, WAN C R, TSUCHIDA E. Composite anode material for lithium ion battery with low sensitivity to water[J]. Electrochemistry Communications 2000, 2(9): 626-629.
[4] WU Y P, JIANG C Y, WAN C R, TSUCHIDA E. Effects of catalytic oxidation on the electrochemical performance of common natural graphite as an anode material for lithium ion batteries[J]. Electrochemistry Communications, 2000, 2(4): 272-275.
[5] PELED E. The electrochemical behavior of alkali and alkaline earth metals in nonaqueous battery systems��Solid electrolyte interphase model[J]. Journal of Electrochemical Society, 1979, 126(12): 2047-2051.
[6] ����ƽ, Ԭ����, �� ��, �μ�Ԩ. ����ӵ�ء���Ӧ����ʵ��[M]. 2��. ����: ��ѧ��ҵ������, 2012: 59.
WU Yu-ping, YUAN Xiang-yun, DONG Chao, DUAN Ji-yuan. Lithium ion battery����Application and practice[M]. 2nd ed. Beijing: Chemistry Industry Press, 2012: 59.
[7] WU Y P, JIANG C Y, WAN C R, HOLZE R. Mild preparation of anode materials by a salt-free green method[J]. Electrochemistry Communications, 2002, 4(6): 483-487.
[8] PELED E, MENACHEM C, BAR-TOW D, MELMAN A. Improved graphite anode for lithium-ion batteries chemically bonded solid electrolyte interface and nanochannel formation[J]. Journal of Electrochemical Society, 1996, 143(1): L4-L7.
[9] MENACHEM C, PELED E, BURSTEIN L, ROSENBERG Y. Characterization of modified NG7 graphite as an improved anode for lithium-ion batteries[J]. Journal of Power Sources, 1997, 68(2): 277-282.
[10] BUQA H, GOLOB P, WINTER M, BESENHARD J O. Modified carbons for improved anodes in lithium ion cells[J]. Journal of Power Sources, 2001, 97/98: 122-125.
[11] WU Y P, JIANG C Y, WAN C R, TSUCHIDA E. A green method for the preparation of anode materials for lithium ion batteries[J]. Journal of Materials Chemistry, 2001, 11: 1233-1236.
[12] WU Y P, JIANG C Y, WAN C R, HOLZE R. Modified natural graphite as anode material for lithium ion batteries[J]. Journal of Power Sources, 2002, 111(2): 329-334.
[13] ZHAO Hai-peng, REN Jian-guo, HE Xiang-ming, LI Jian-jun, JIANG Chang-yin, WAN Chun-rong. Modification of natural graphite for lithium ion batteries[J]. Solid State Sciences, 2008, 10(5): 612-617.
[14] WU Y P, JIANG C Y, WAN C R, HOLZE R. Anode materials for lithium ion batteries by oxidative treatment of common natural graphite[J]. Solid State Ionics, 2003, 156(3/4): 283-290.
[15] ���ij�, �� ��, ���, �మˮ. ��ȩ��֬����������Ȼʯī��Ϊ����ӵ�ظ�������[J]. ������ѧѧ��, 2011, 27(9): 2129-2134.
GAO Wen-chao, HUANG Tao, SHEN Yu-dong, YU Ai-shui. Phenolic resin coated natural graphite oxide as an anode material for lithium ion batteries[J]. Acta Physico-Chimica Sinica, 2011, 27(9): 2129-2134.
[16] HIRSCHORN B, ORAZEM E M, TRIBOLLET B, VIVIER V, FRATEUR I, MUSIANI M. Constant-phase-element behavior caused by resistivity distributions in films[J]. Journal of Electrochemical Society, 2010, 157(12): C452-C457.
[17] HIRSCHORN B, ORAZEM E M, TRIBOLLET B, VIVIER V, FRATEUR I, MUSIANI M. Constant-phase-element behavior caused by resistivity distributions in films[J]. Journal of Electrochemical Society, 2010, 157 (12): C458-C463.
[18] ZHANG S S, XU K, JOW T R. EIS study on the formation of solid electrolyte interface in Li-ion battery[J]. Electrochimica Acta, 2006, 51(8/9): 1636-1640.
[19] ����ʯ, �ܿ���, ������. �Ҵ������ܻ�ʯīϩ���Ʊ������[J]. ��ѧѧ��, 2011, 69(12): 1463-1468.
MA Wen-shi, ZHOU Jun-wen, LIN Xiao-dan. Preparation and characterization of functionalized graphene with ethanolamine[J]. Acta Chimica Sinica, 2011, 69(12): 1463-1468.
[20] ������, ������, �� ��, ŷ����, �����, �� ��, ��˴��. ����Ҷ�任��������ģʽʶ���ټ���ʳ���͵���α[J]. ��ѧѧ��, 2012, 70(8): 995-1000.
LIU Lin-lin, WU Yan-wen, ZHANG Xu, OUYANG Jie, LI Bing-ning, HOU Ming, CHEN Shun-cong. Application of Fourier transform infrared spectroscopy combined with pattern recognition method for rapid authentication of edible oil[J]. Acta Chimica Sinica, 2012, 70(8): 995-1000.
[21] ����, �� �, ���鲨, �����. �۱���/����ʯī�ĺϳɼ�����DNAʶ���ϵ�Ӧ��[J]. ����̿����, 2005, 20(4): 360-364.
ZOU Yan-hong, WU Jing, LIU Hong-bo, CHEN Zong-zhang. Preparation of polyaniline-intercalated graphite oxide composite and its application in detecting DNA[J]. New Carbon Materials, 2005, 20(4): 360-364.
[22] ФԾ��, �� �, �� ��, �� ��, ������, �˴�Ծ. �����������������﮵Ŀ��ٶ�������[J]. ������ѧѧ��, 2012, 28(3): 357-360.
XIAO Yue-long, DAI Xuan, ZHANG Yi, XU Hang, LIU Hong-bo, PAN Chun-yue. Rapid quantitative analysis of lithium hexafluorophosphate by infrared spectroscopy[J]. Journal of Analytical Science, 2012, 28(3): 357-360.
[23] WU Y P, JIANG C, WAN C, HOLZE R. Effects of pretreatment of natural graphite by oxidative solutions on its electrochemical performance as anode material[J]. Electrochim Acta, 2003, 48: 867-874.
[24] AURBACH D. Review of selected electrode-solution interactions which determine the performance of Li and Li ion batteries[J]. Journal of Power Sources, 2000, 89(2): 206-218.
[25] LU M, CHENG H, YANG Y. A comparison of solid electrolyte interphase (SEI) on the artificial graphite anode of the aged and cycled commercial lithium ion cells[J]. Electrochimica Acta, 2008, 53(9): 3539-3546.
[26] GUO Kun-kun, PAN Qin-min, FANG Shi-bi. Poly (acrylonitrile) encapsulated graphite as anode materials for lithium ion batteries[J]. Journal of Power Sources, 2002, 111(2): 350-356.
[27] LIU Ping, WU Hao-qing. Construction and destruction of passivating layer on LixC6 in organic electrolytes: an impedance study[J]. Journal of Power Sources, 1995, 56(1): 81-85.
[28] ANDREA D, MEILER M, STEINER K, WIMMER CH, SOCZKA-GUTHA T, SAUER D U. Characterization of high-power lithium-ion batteries by electrochemical impedance spectroscopy. I: Experimental investigation[J]. Journal of Power Sources, 2011, 196(12): 5334-5341.
[29] MOMMAT, MATSUNAGA M, MUKOYAMA D, OSAKA T. AC impedance analysis of lithium ion battery under temperature control[J]. Journal of Power Sources, 2012, 216: 304-307.
[30] ITAGAKI M, KOBARIN, YOTSUDA S, WATANABE K, KINOSHITA S, UE M. In-situ electrochemical impedance spectroscopy to investigate negative electrode of lithium-ion rechargeable batteries[J]. Journal of Power Sources, 2004, 135(1/2): 255-261.
[31] GUO J C, SUN A, CHEN X L, WANG C S, MANIVANNAN A. Cyclability study of silicon-carbon composite anodes for lithium-ion batteries using electrochemical impedance spectroscopy[J]. Electrochimica Acta, 2011, 56(11): 3981-3987.
[32] YAMADA Y, IRIYAMA Y, ABE T, OGUMI Z. Kinetics of lithium ion transfer at the interface between graphite and liquid electrolytes: Effects of solvent and surface film[J]. Langmuir 2009, 25(21): 12766-12770.
[33] HSU H C, MANSFELD F. Technical note: Concerning the conversion of the constant phase element parameter Y0 into a capacitance[J]. Corrosion, 2001, 57(9): 747-748.
[34] SHUKLA A K, SAMPATH S, VIJAYAMOHANAN K. Electrochemical supercapacitors: Energy storage beyond batteries[J]. Current Science, 2000, 79(12): 1656-1661.
[35] ABE T, FUKUDA H, IRIYAMA Y, OGUMI Z. Solvated Li-ion transfer at interface between graphite and electrolyte[J]. Journal of Electrochemical Society, 2004, 151(8): A1120-A1123.
[36] YAMADA Y, IRIYAMA Y, ABE T, OGUMI Z. Kinetics of electrochemical insertion and extraction of lithium ion at SiO[J]. Journal of Electrochemical Society, 2010, 157(1): A26-A30.
(�༭ ����Ƽ)
������Ŀ����������ѧ�ع���Ա������������������Ŀ([2011]1139)������ʡ�о������д�����Ŀ(CX2012B370)����ѧ���о���ѧϰ�봴����ʵ��ƻ���Ŀ(201210536049)
�ո����ڣ�2013-03-25�������ڣ�2013-10-26
ͨ�����ߣ������£����ڣ���ʿ���绰��0731-85258224��E-mail��chenzhaoyongcioc@126.com