
Thermodynamic analysis of growth of ternary III-V semiconductor materials by molecular-beam epitaxy
YE Zhi-cheng, SHU Yong-chun, CAO Xue, GONG Liang, PI Biao,
YAO Jiang-hong, XING Xiao-dong, XU Jing-jun
Key Laboratory of Advanced Technique and Fabrication for Weak-light Nonlinear Photonics Materials of
Ministry of Education, Nankai University, Tianjin 300457, China
Received 17 June 2010; accepted 15 August 2010
Abstract: Thermodynamic models for molecular-beam epitaxy (MBE) growth of ternary III-V semiconductor materials are proposed. These models are in agreement with our experimental materials InGaP/GaAs and InGaAs/InP, and reported GaAsP/GaAs and InAsP/InP in thermodynamic growth. The lattice strain energy ΔG and thermal decomposition sensitive to growth temperature are demonstrated in the models simultaneously. ΔG is the function of the alloy composition, which is affected by flux ratio and growth temperature directly. The calculation results reveal that flux ratio and growth temperature mainly influence the growth process. Thermodynamic model of quaternary InGaAsP/GaAs semiconductor material is discussed also.
Key words: semiconductor materials; III-V compounds; growth; thermodynamics
1 Introduction
Ternary III-V semiconductor materials have been used extensively in the fabrication of optoelectronic devices, optical detectors and modulators. Among plenty of ways to grow the specific materials, liquid phase epitaxy, metal organic chemical vapor deposition (MOCVD) and molecular beam epitaxy (MBE) are three of the most widely used methods. However, the thermodynamic growth of the compounds is a complicated problem during this procedure. There is no direct linear dependence of composition on flux ratio of ternary compounds in practice. Although the same thermodynamic growth models have been proposed[1-2], it is difficult to explain the experimental results correctly[3]. For instance, the previous models predict that the alloy composition should be dependent on V/III beam-flux ratio[4-5], which is inconsistent with reported data in Refs.[6-7].
In thermodynamic models reported previously for MBE[2, 8-10], the growth processes were analyzed by equations of chemical reactions, chemical potentials, reevaporated group V elements and the lattice strain energy. However, the lattice strain energy and thermal
decomposition were not discussed in the theoretical system simultaneously; a clear numeric project for the setup parameters was not either. Thus, an adequate thermodynamic model for MBE of multi-component compounds is still an open problem.
In this work, for the III-V compounds with two group III elements, the correlation among flux ratio, growth temperature and alloy composition is investigated. The lattice strain energy and thermal decomposition sensitive to growth temperature are demonstrated in the models simultaneously.
2 Theory analysis
Based on the thermodynamics principle, EGOROV et al[8] evaluated the relationship between the flux ratio of group Ⅴ element and composition in the growth of AⅢCⅤyDⅤ1-y semiconductor materials:
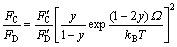 (1)
(1)
where kB is the Boltzmann constant; F is the corresponding flux ratio of the element; and Ω is an interaction parameter between AC and AD compounds in the ternary solution.
For the III-V compounds with two groups of III elements AⅢx+BⅢ1-x+CⅤ= AⅢxBⅢ1-xCⅤ, if the lattice strain energy and thermal decomposition sensitive to growth temperature are considered simultaneously, the model is modified as follows:
 (2)
(2)
In the thermodynamics growth model, the lattice strain energy is given by ΔG=αM′(Δa)2, where α is the figure factor, M′ is the per unit length elastic modulus, and Δa is the amount of lattice mismatch.
For the III-V compounds with two volatile groups of V elements[11], AⅢ+CyⅤ+DⅤ1-y=AⅢCyⅤDⅤ1-y , there is
 (3)
(3)
The efficient flux F′ is a linear function of the alloy composition in the case of equilibrium. F′A=nAx, F′B=nB(1-x), where ni is incorporation factor, and assuming it is a constant.
If  , then
, then 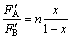 (4)
(4)
By combing functions (4) with (2), we get
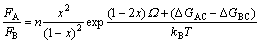 (5)
(5)
The relationship between steam pressure and temperature is: lnpv=A+B/T, where A and B are constants independent of temperature in the same compound.
Taking into account the effect on the absorption and desorption by the change of temperature, the F′A in Eq.(4) can be modified as
 (6)
(6)
According to previous investigations[12-13], efficient incorporation coefficient constant without the desorption n is modified as
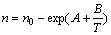 (7)
(7)
By combining Eqs.(5) with (7), we get

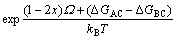 (8)
(8)
The lattice strain energy is given by ΔG=αM′(Δa)2.
Assuming that the strain is an elastic strain of formula crystal, figure factor α=1, and the film lattice matches to substrate, x=x0, the difference of their lattice strain energies is
ΔG=ΔGAC-ΔGBC=M(x-x0)2 (9)
where M=M′(aAC-aBC)2 (a and aBC are lattice constants of AC and BC, respectively).
By combining Eqs.(9) with (8), we get

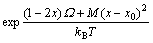 (10)
(10)
For the III-V compounds, AⅢCyⅤDⅤ1-y, with two volatile groups of V elements, we have
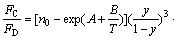
 (11)
(11)
where aAC, aAD and a0 are the lattice constants of AⅢCⅤ, AⅢDⅤ and substrate, respectively.
3 Experimental validation
3.1 Thermodynamic analysis for InGaP/GaAs
For InxGa1-xP/GaAs, the film lattice matches to the substrate at x≈0.485.
InGaP/GaAs compounds are hetero-epitaxial materials which are grown on SC-GaAs (100) substrates by a Riber Compact 21T MBE system equipped with a Riber KPC250 phosphorus valve cracker cell. By inputting the experiment results into Eq.(10), the values of n0, A, B, M and Ω can be obtained after optimization: n0=7, A=1.2, B=353 K, M=±21.56×10-20 J (x≤0.485, M=-21.56×10-20 J; other x>0.485, M=21.56×10-20 J) and Ω=3.2×10-20 J.
The function of In/Ga flux ratio F, growth temperature and composition is transformed into

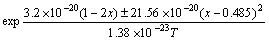 (12)
(12)
The results of calculations for Eq.(12) and the experimental data are shown in Figs.1 and 2, respectively. Fig.1 shows indium composition as a function of the In/Ga flux ratio at temperature of 480 °C. Fig.2 shows indium composition as a function of growth temperature when the In/Ga flux ratio is equal to 1.78. It can be seen that the calculated data are fitted well with the experimental results.

Fig.1 Dependence of In/Ga flux ratio on indium composition at 480 °C

Fig.2 Dependence of growth temperature on indium composition with In/Ga flux ratio of 1.78
By using Eq.(12) to fit the results of HASENOHRL et al[14], the conditions of InGaP/GaAs material growth are as follows: using metal organic vapor phase epitaxy (MOVPE) as the growth technique, high V/III flux ratio (250) and high growth temperature (640 °C). Then the function is transformed into
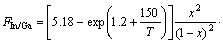
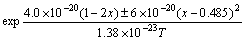 (13)
(13)
By plotting the results (Fig.3), a good agreement is obtained. By comparing this modified Eq.(13) with Eq.(12), which are suitable to the growth in MBE system (Riber 21T), it can been seen that when the efficient incorporation coefficient n decreases from 7 to 5.18 and indium composition ratio becomes high when the material is growing; interaction parameter Ω is also increased from 3.2×10-20 J to 4.0×10-20 J, which can be mainly attributed to high phosphorous flux favorable to In+P→InP. It is also found that the decomposition related parameter B decreases from 353 K to 150 K, probably because the beam equivalent pressure of phosphorous flux is so high (250) that a reverse reaction In+P→InP is introduced.
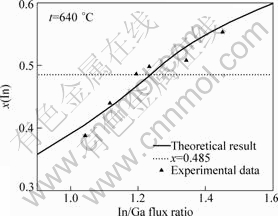
Fig.3 Dependence of In/Ga flux ratio on indium composition at 640 °C (according to results of HASENOHRL S et al[14])
3.2 Thermodynamic analysis for InGaAs/InP
SC-InP (100) substrate is used for the experimental study of InGaAs/InP growth. The thickness of the film is 130-160 nm by involuntary doping growth. The temperature of phosphorus cracking furnace is 880 °C. The equivalent flux pressure range of (p2) is 399.966×10-6-1 279.891×10-6 Pa, and p2 is auto controlled by the needle controller. Equivalent flux pressure range of indium is 533.288×10-7-799.932×10-7 Pa, and gallium 293.31×10-7-466.63×10-7 Pa. The growth temperature is 480 °C. V/III flux ratio is 7:1, and In/Ga flux ratio is (1.6-2.6):1.
For InxGa1-xAs/InP, the film lattice matches the substrate at x0≈0.532.
The values of n0, A, B, M and Ω are calculated using Eq.(10) based on the experiment results. n0=5.38, A=1, B=200 K, M=±19.16×10-20 J (x≤0.532, M=-19.16× 10-20 J; other x>0.532, M=19.16×10-20 J) and Ω=3.0× 10-20 J are obtained after optimizing the values.
Hence, the function of In/Ga flux ratio F, growth temperature and composition is transformed into

 (14)
(14)
The results of calculations and experimental data are shown in Fig.4.
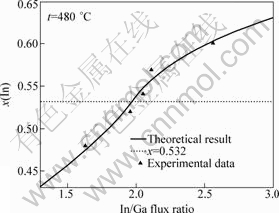
Fig.4 Dependence of In/Ga flux ratio on indium composition of InxGa1-xAs
3.3 Thermodynamic analysis for GaAsP/GaAs
According to the experimental data in Ref.[8], those conditions of the growth of GaAsyP1-y on GaAs substrate, a0 of the substrate GaAs of 5.653 25 ?, the temperature of the substrate of 500 °C and the stationary growth temperature of 500 °C are employed for this study. Due to the function only depending on growth temperature, effective adhesive factor n is regarded as a constant. Putting experimental data into Eq.(11), we have n= 1.864 9, Ω=3.578 2×10-20 J, M=-4.593 4×10-20 J.
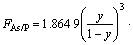
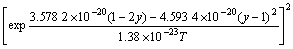 (15)
(15)
The results of calculations and experimental data are shown in Fig.5.
3.4 Thermodynamic analysis for InAsP/InP
During the growth of InAsyP1-y on InP substrate, temperature of substrate is taken as 520 °C based on the experimental data of HAO et al[15].
The values of n, Ω and M are obtained through inputting experimental data into Eq.(11) as follows: n=0.013 8, Ω=2.876 9×10-20 J, M=1.670 2×10-20 J.
The function of As/P flux ratio F and arsenic composition y is transformed into

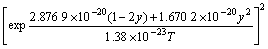 (16)
(16)
The results of calculations and experimental data are shown in Fig.6.
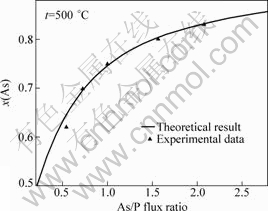
Fig.5 Dependence of As/P flux ratio on arsenic composition (according to experimental data of EGOROV et al[8])
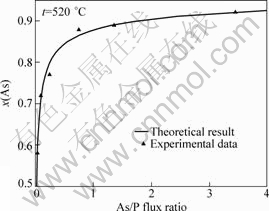
Fig.6 Dependence of As/P flux ratio on arsenic composition (according to experimental data of HAO et al[15])
3.5 Thermodynamic analysis for quaternary compounds
For the quaternary compounds, growth of GaxIn1-xAsyP1-y on GaAs substrate by solid source MBE had been reported[16-17]. The compound formed during the growth of InxGa1-xAsyP1-y on GaAs substrate can be considered a mixture of two groups of ternary compounds InGaP and GaAsP through the following reaction:
InGaP(or InGaAs)+InAsP(or GaAsP)→InGaAsP (17)
The thermodynamic model for growth of InxGa1-xP on GaAs substrate with group V elements fixed as the function of group III elements In/Ga flux ratio F, growth temperature and composition can be expressed as
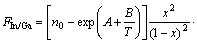
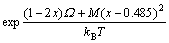 (18)
(18)
While the thermodynamic model for the growth of GaAsyP1-y on GaAs substrate with group III elements composition fixed as the function of group V elements As/P flux ratio, growth temperature and composition can be expressed as
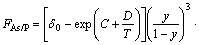
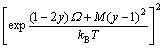 (19)
(19)
where C and D are constants independent of temperature in the same compound.
Therefore, the above equations (18) and (19) can be used to predict the growth of InGaAsP on GaAs substrate and the control of InGaAsP quaternary compound.
4 Conclusions
1) The thermodynamic growth models for InGaP/GaAs, InGaAs/InP, GaAsP/GaAs and InAsP/InP are developed and evaluated.
2) As two very sensitive parameters are added into function simultaneously for the first time, the lattice strain energy ΔG and thermal decomposition sensitive to growth temperatures.
3) In the growth of III-V compound semiconductor materials by MBE, the calculated results reveal the interactional relations of the main parameters: flux ratio, growth temperature and the alloy composition. The calculation results fit very well the experimental and reported data.
References
[1] DONG Jian-rong, WANG Zhan-guo, LIU Xiang-lin, LU Da-cheng, WANG Du, WANG Xiao-hui. Photoluminescence of ordered Ga0.5In0.5P grown by metalorganic vapor phase epitaxy [J]. Applied Physics Letters, 1995, 67(11): 1573-1575.
[2] KANGAWA Y, ITO T, KUMAGAI Y, KOUKITU A. Thermodynamic study on compositional instability of InGaN/GaN and InGaN/InN during MBE [J]. Applied Surface Science, 2003, 216(1-4): 453-457.
[3] LIANG B W, TU C W. A kinetic model for As and P incorporation behaviors in GaAsP grown by gas-source molecular beam epitaxy [J]. Journal of Applied Physics, 1993, 74(1): 255-259.
[4] HOU H Q, LIANG B W, CHIN T P, TU C W. In situ determination of phosphorus composition in GaAs1-xPx grown by gas-source molecular beam epitaxy [J]. Applied Physics Letters, 1991, 59(3): 292-294.
[5] IVANOV S V, KOP`EV P S, LEDENTSOV N N. Thermodynamic analysis of segregation effects in molecular beam epitaxy [J]. Journal of Crystal Growth, 1990, 104(2): 345-354.
[6] KUMAGAI Y, KIKUCHI J, MATSUO Y, KANGAWA Y, TANAKA K, KOUKITU A. Thermodynamic analysis of InN and InxGa1-xN MOVPE using various nitrogen sources [J]. Journal of Crystal Growth, 2004, 272(1-4): 341-347.
[7] KOUKITU A, KIKUCHI J, KANGAWA Y, KUMAGAI Y. Thermodynamic analysis of AlGaN HVPE growth [J]. Journal of Crystal Growth, 2005, 281(1): 47-54.
[8] EGOROV A Y, KOVSH A R, USTINOV V M, ZHUKOV A E, KOP’EV P S, TU C W. A thermodynamic analysis of the growth of III-V compounds with two volatile group V elements by molecular-beam epitaxy [J]. Journal of Crystal Growth, 1998, 188(1-4): 69-74.
[9] LI Y, KOUKITU A, SEKI H. Thermodynamic analysis of InxGa1-xN growth conditions in molecular beam epitaxy [J]. Journal of Applied Physics, 2000, 88(1): 571-575.
[10] WANG Y Q, WANG Z L, BROWN T, BROWN A, MAY G. Thermodynamic analysis of anion exchange during heteroepitaxy [J]. Journal of Crystal Growth, 2002, 242(1-2): 5-14.
[11] PI Biao. The growth and physics of Ⅲ-Ⅴcompound semiconductors containing phosphorus [D]. Tianjin: School of Applied Physics, Nankai University, 2007: 73-83. (in Chinese)
[12] IIEGEMS M, PANISH M B, ARTHUR J R. Phase equilibria and vapor pressures in the Ga+P system [J]. The Journal of Chemical Thermodynamics, 1974, 6(2): 157-177.
[13] PANISH M B, ARTHUR J R. Phase equilibria and vapor pressures of the system In+P [J]. The Journal of Chemical Thermodynamics, 1970, 2(3): 299-318.
[14] HASENOHRL S, KUDELA R, NOVAK J, TUOMI T O, KNUUTTILA L. Anisotropic surface structure in ordered strained InGaP [J]. Materials Science and Engineering B, 2002, 88(2-3): 134-138.
[15] HAO Zhi-biao, REN Zai-yuan, GUO Wen-ping, LUO Yi. Studies on incorporation of As2 and As4 in III-V compound semiconductors with two group V elements grown by molecular beam epitaxy [J]. Journal of Crystal Growth, 2001, 224(3-4): 224-229.
[16] WANG X Z, ZHANG D H, ZHENG H Q, YOON S F, KAM C H, SHI W, RAMAN A. Effects of phosphorous beam equivalent pressure on GaInAsP/GaAs grown by solid source molecular beam epitaxy with a valve phosphorous cracker cell [J]. Journal of Crystal Growth, 2000, 210(4): 458-462.
[17] HAO Zhi-biao, LU Jing-hui, REN Zai-yuan, LUO Yi. Solid-source molecular beam epitaxial growth of InP and InGaAsP [J]. Chinese Journal of Semiconductors, 2000, 21(12): 1193-1197. (in Chinese)
III-V族三元化合物半导体材料分子束外延的生长热力学
叶志成,舒永春,曹 雪,龚 亮,皮 彪,姚江宏,邢晓东,许京军
南开大学 弱光非线性光子学教育部重点实验室,天津 300457
摘 要:建立III-V族三元化合物半导体材料的分子束外延生长热力学模型。该模型与实验材料InGaP/GaAs, InGaAs/InP 及已发表的GaAsP/GaAs, InAsP/InP 的数据吻合得很好。将晶格应变能ΔG及脱附对温度敏感这两个因素同时纳入热力学模型中,束流和生长温度直接影响合金组分,晶格应变能是合金组分的函数。热力学模型计算结果反映了束流和生长温度是生长过程中最主要的影响因素。讨论和分析了四元半导体材料InGaAsP/GaAs的热力学生长模型。
关键词:半导体材料;III-V 族化合物;生长;热力学
(Edited by YANG Hua)
Foundation item: Projects (06YFJZJC01100, 08JCYBJC14800) supported by Applied Basic Study Foundation of Tianjin, China; Project (2006AA03Z413) supported by the Hi-tech Research and Development Program of China
Corresponding author: SHU Yong-chun; Tel +86-22-66229609; E-mail: shuyc@nankai.edu.cn
DOI: 10.1016/S1003-6326(11)60691-2