Article ID: 1003-6326(2005)05-1166-06
Surface tension of molten tin investigated with sessile drop method
LI Jing(李 晶), YUAN Zhang-fu(袁章福),
FAN Jian-feng(范建峰), KE Jia-jun(柯家骏)
(Multi-phase Reaction Laboratory, Institute of Process Engineering, Chinese Academy of Sciences, Beijing 100080, China)
Abstract: The surface tension of molten tin was determined by a set of self-developed digital equipment with sessile drop method at oxygen partial pressure of 1.0×10-6MPa under different temperatures, and the dependence of surface tension of molten tin on temperature was also discussed. The emphasis was placed on the comparison of surface tension of the same molten tin sample measured by using different equipments with sessile drop method. Results of the comparison indicate that the measurement results with sessile drop method under the approximate experimental conditions are coincident, and the self-developed digital equipment for surface tension measurement has higher stability and accuracy. The relationships of surface tension of molten tin and its temperature coefficient with temperature and oxygen partial pressure were also elucidated from the thermodynamic equilibrium analysis.
Key words: molten tin; surface tension; sessile drop method; oxygen partial pressure; temperature coefficient CLC number: TG111
Document code: A
1 INTRODUCTION
Surface tension is one of the most important thermophysical parameters of molten metals, which greatly influences the processes of crystallization, phase transition and crystal growth of molten metals, and plays a key role in the processes of metallurgy, casting and welding. Measurement of surface tension and its temperature coefficient is important to the research of metal thermophysical properties[1-3]. Tin and its alloys are applied widely in the solder industries and the manufacture of tin coated steel sheet etc. The reliable data of surface tension are significant to the design and optimization of tin alloys. Especially, space science develops rapidly at present, and surface tension and its temperature coefficient are the key parameters for preparation of high performance metal materials in space. Under the condition of micrograviation, temperature is one of the main factors affecting Marangoni convection driven by surface tension gradient, hence the surface tension of molten tin is also significant for the research of Marangoni convection of molten tin and the development of space material science[4-7].
The surface tension of molten tin is measured by self-developed digital equipment with sessile drop method under different temperatures in this paper. The dependence of surface tension on temperature and oxygen partial pressure is also discussed. Although some investigations on the surface tension of molten tin have been done at home and abroad, there are deviations among the obtained data[1, 4, 8-10]. Based on the summarized relationships of surface tension of molten tin with temperature and oxygen partial pressure reported in literatures, the reasons for the difference in those reported data have been analyzed. The surface tensions of molten tin measured by using the same sample with the same method have been emphasized on for comparison. The relationships of surface tension of molten tin and its temperature coefficient with temperature at different oxygen partial pressures are also elucidated from the thermodynamic equilibrium analysis.
2 EXPERIMENTAL
The cylindrical samples of high-purity tin used in present work (d 3.5mm×3.5mm, about 0.3g, purity 99.999%, mass fraction) were provided by the National Space Development Agency of Japan, and the schematic diagram of self-developed digital equipment with sessile drop method used in present experiment is shown in Fig.1. The equipment was composed of a heating and melting system, an imaging system, and a picture manipulation and calculation system. The photos of molten tin can be directly transmitted to computer system for image manipulation and calculation, and the procedures of film developing, picture scanning and point obtaining can be omitted. The present experiment realized the digitization,and reduced the human factors and got more objective experimental results.
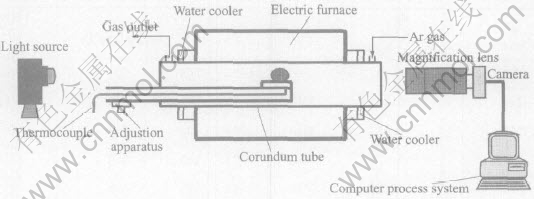
Fig.1 Schematic diagram of experimental equipment
The magnification of the imaging system was firstly determined by a standard steel ball, then experimental sample placed on a highly pure alumina substrate was put into the middle place of corundum pipe in an electric furnace, and the sample was adjusted horizontally. The droplet images of molten tin samples were taken when the furnace reached the scheduled temperature and held at constant temperature for 15min. The light source was adjusted to the best brightness to ensure the distinct images. Taken images were transmitted directly to computer system for manipulation and calculation, and the surface tension was obtained at scheduled temperature. Argon gas (φ(Ar)%>99.99%, p(O2)=10-6MPa) is used as protecting gas during the whole experimental process.
3 RESULTS AND DISCUSSION
3.1 Summary of surface tension of molten tin reported in literatures
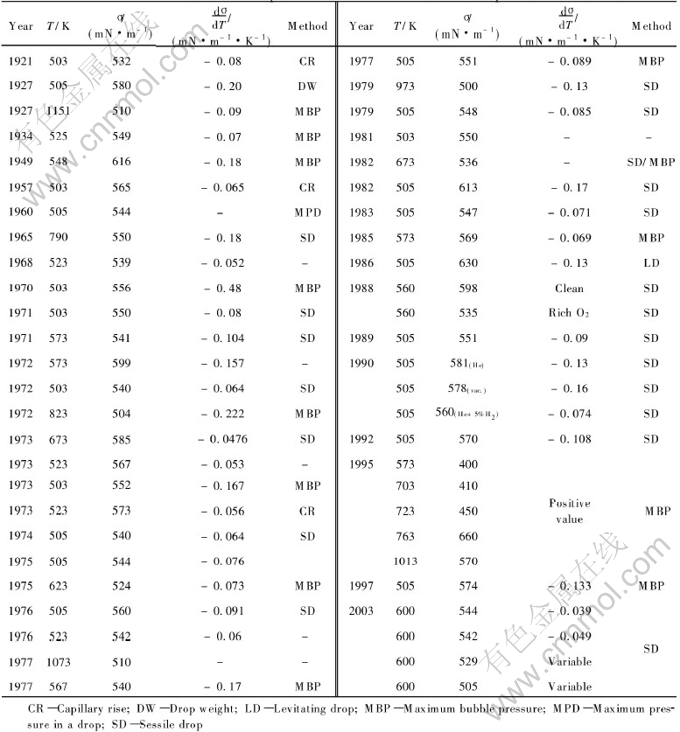
The surface tension of molten tin and its dependence on the factors including temperature and controlling atmosphere have been measured and investigated by domestic and foreign researchers[1, 4, 8-10], and the measured surface tension and its temperature coefficient reported in literatures have been summarized in Table 1.
Table 1 shows that the sessile drop method(SD) and the maximum bubble pressure method(MPD) are the most common methods for measurement of surface tension of molten tin, and the sessile drop method is more reliable and stable[1, 11, 12]. But there are obvious differences among reported values, in which the greater differences in earlier reported data maybe result from the limitation of experimental conditions and equipments at that time, and the different measurement methods lead to some different results as well. Even measuring with the same method, the results obtained could not be identical due to the systematical error caused by the differences of samples, experimental atmospheres and measurement equipments used.
3.2 Comparison of surface tension of same molten tin sample by using different equipments with sessile drop method
The surface tension of the same molten tin sample has been measured by authors with sessile drop method under different oxygen partial pressures (p(O2)) by using different equipments in the National Space Development Agency of Japan (equipment shown as Fig.2(a))[13-15], in Kyushu
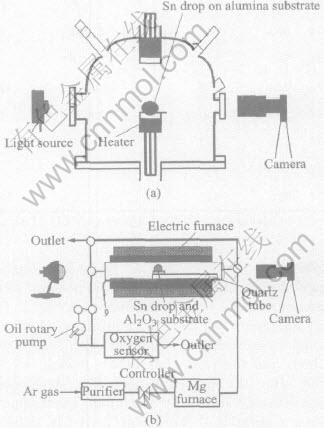
Fig.2 Schematic diagrams of experimental apparatus in National Space Developemnt
Agency of Jajpan(a) and in Kyushu Institute of Technology in Japan(b)
Table 1 Surface tension and temperature coefficient of molten tin reported in literatures
Institute of Technology in Japan (equipment shown as Fig.2(b))[4, 14, 15] and in present work. The experimental results are shown in Fig.3. The two equipments in Figs.2(a) and (b) are both traditional measurement equipments with sessile drop method, and the values of surface tension are obtained after the procedures of film developing, photo scanning and point obtaining. Furthermore, the characteristics of equipment(b) in Fig.2(b) are that the atmosphere can be controlled and the oxygen partial pressure can be measured.
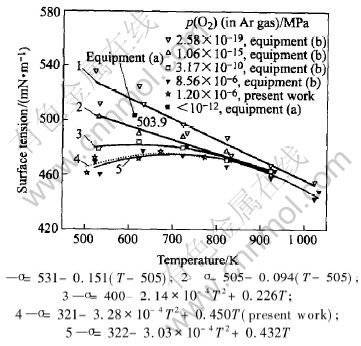
Fig.3 Surface tension of molten tin under different oxygen partial pressures with
different experimental equipments
In present experiments, the measurement values of surface tension of molten tin are 461.1mN/m, 472.2mN/m, 471.6mN/m, 477.2mN/m, and 472.9mN/m, under p(O2)=1.0×10-6MPa at 505K, 523K, 623K, 673K and 773K respectively. According to those experimental data, the regression relationship between surface tension of molten tin and temperature can be expressed as σ=321-3.28×10-4 T2+0.450T. It can be seen from the change tendency of regression curve in Fig.3 that the temperature coefficient of surface tension varies with temperature. Firstly, the surface tension increases gradually with the increase of temperature, and the temperature coefficient is positive. When the surface tension reaches the maximum, the surface tension decreases with the increase of temperature, and the temperature coefficient is negative.
It can be also seen from Fig.3 that the values measured in the three experiments above are coincident in general. The surface tension of molten tin is 503.9mN/m at 613K under the atmosphere condition of p(O2)〈10-12MPa in the National Space Development Agency of Japan. And the surface tension of molten tin is 494.8mN/m at the same temperature under the condition of similar oxygen partial pressure (p(O2)=1.06×10-15MPa) in Kyushu Institute of Technology, Japan. The absolute deviation of those two values is 9.1mN/m. Furthermore, the results measured under the condition of p(O2)=1.0×10-6MPa in present work are coincident with those measured under the condition of higher oxygen partial pressure (p(O2)=8.56×10-6MPa) in Kyushu Institute of Technology, Japan. And the absolute deviation of curvature (temperature coefficient) of corresponding point at the two curves is less than 0.05mN・m-1・K-1. The differences among all the values of surface tension measured at the same temperature are lower than ±2%, which suggests that the sessile drop method is more stable and accurate for measurement of surface tension of molten tin at high temperature.
3.3 Thermodynamic equilibrium analysis for dependence of surface tension of molten tin on temperature and oxygen partial pressure
Surface tension decreases with the increase of temperature generally, and its temperature coefficient is negative. But the positive values of temperature coefficient of surface tension also exist in experiments, and the correlative experimental results are summarized in Fig.4[4, 8-10].
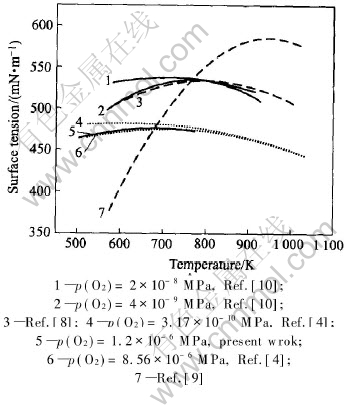
Fig.4 Surface tension of molten tin with positive temperature coefficient reported
in literatures and present work
Tin is active at high temperature and its activity is very high. Therefore, tin is easy to oxidize by oxygen in the experimental atmosphere. And oxygen is a kind of surface-active element, so the influence of oxygen partial pressure is one of the key factors on the measurement of surface tension of molten tin. The values of surface tension are smaller when the oxygen partial pressure is higher at the same temperature. Under different oxygen partial pressures, the relationships between surface tension of molten tin and temperature are different as well. Under the condition of low oxygen partial pressure, surface tension of molten tin decreases with the increase of temperature. Under the condition of higher oxygen partial pressure, the temperature coefficient of surface tension of molten tin varies with the increase of temperature.
Generally, the oxidization of tin proceeds in the atmosphere containing oxygen as the following reactions:
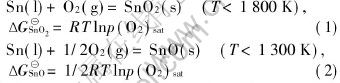
And the vapour equilibriums of Sn and SnO are also included in the system:
Sn(l)=Sn(g)(3)
SnO(l)=SnO(g)(4)
The 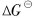 of equations (1) and (2) can be obtained within a range of temperature[16, 17], and if the activity values of Sn and SnO2 are both set as 1, p(O2)sat (the equilibrium oxygen partial pressure) values are gained by calculation. According to equation (3), a curve for vapor pressure of molten tin with temperature can be achieved[18]. According to Fiori et al[10], the oxygen surrounding molten metal has a limit value of the oxygen partial pressure, called effective oxidation pressure, p(O2)Es, discriminating surface oxidation and surface deoxidation (or oxide removal). For Sn-O system, if the quantity of Sn is in excess of that of O, Sn is only oxidized to SnO, and p(O2)Es can be described as follows[10]:
of equations (1) and (2) can be obtained within a range of temperature[16, 17], and if the activity values of Sn and SnO2 are both set as 1, p(O2)sat (the equilibrium oxygen partial pressure) values are gained by calculation. According to equation (3), a curve for vapor pressure of molten tin with temperature can be achieved[18]. According to Fiori et al[10], the oxygen surrounding molten metal has a limit value of the oxygen partial pressure, called effective oxidation pressure, p(O2)Es, discriminating surface oxidation and surface deoxidation (or oxide removal). For Sn-O system, if the quantity of Sn is in excess of that of O, Sn is only oxidized to SnO, and p(O2)Es can be described as follows[10]:
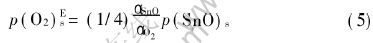
In equation (5), αSnO and αO2 are the condensation coefficient of the evaporating species, but those data are not available in literatures. If only SnO is considered, and saturated vapor pressure of molten SnO can be considered the effective oxidation pressure for molten tin approximately[10, 18], described as equation (6):
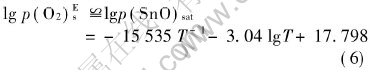
All the equilibrium curves for Sn-O system are shown in Fig.5. There are three main regions concerned to reactions of Sn-O system: surface oxidation region, effective surface deoxidation region and deoxidation region. The curves of p(O2)Es―T and p(Sn)―T divide the oxidation region and effective deoxidation region, and constitute the effective deoxidation along with the p(O2)sat―T curve, while the p(O2)sat―T curve delimits the effective deoxidation region and deoxidation region.
When p(O2)>p(O2)Es (or p(Sn)), the system is located in the oxidation region, in which the surface of tin is easy to oxidize. Oxygen as tensioactive element can decrease the surface tension of molten tin. When the oxygen partial pressure strides the p(O2)Es (or p(Sn)) curve, the system is located in the effective deoxidation region, in which the tensioactive effect of oxygen is also concerned, but not necessarily concerning the formation of oxides on the surface, and the effect of oxygen partial pressure on surface tension is slight. When p(O2)〈p(O2)sat, the system is located in the deoxidation region, where a “clean” surface can be related to it and oxygen partial pressure hardly affects the surface tension of molten tin.
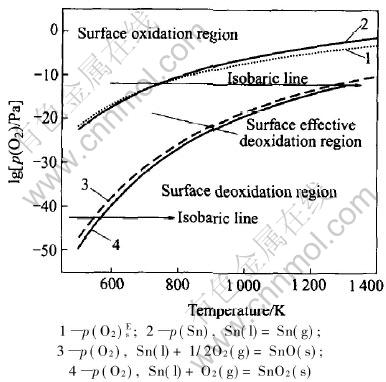
Fig.5 Equilibrium for tin-oxygen system
When p(O2) in experiments is high, molten tin is firstly located in the oxygen region; with the increase of temperature, it goes across to the effective deoxidation region, finally reaches the deoxidation region. When p(O2) is extremely low, molten tin almost lies in the deoxidation region all along, and is hardly influenced by the oxygen. When the experimental system is situated in the effective deoxidation region, molten tin is affected by oxygen, yet not forming the surface oxides, and in this region the surface tension increases very slowly with the increase of temperature until reaching a “platform”, and then the tendency changes from increase to decrease. The influence of oxygen in experimental atmosphere on surface tension of molten tin can be elucidated reasonably by thermodynamic equilibrium analysis, which is coincident with the experimental results.
4 CONCLUSIONS
1) The surface tension of molten tin and its temperature coefficient are obtained at p(O2)=1.2×10-6MPa under different temperatures using self-developed digital equipment with sessile drop method. The experimental results are coincident with the values reported in references under the approximate conditions.
2) By comparing surface tension measured using different equipments with sessile drop method, it is suggested that sessile drop method is a steady and accurate method for surface tension measurement of molten tin at high-temperature and the experimental error is lower than ±2%. The measurement procedure of digital equipment for surface tension measurement used in present work is simpler and more reliable.
3) The thermodynamic analysis has been processed for Sn-O system, which elucidates the dependence of surface tension of molten tin on temperature under different oxygen partial pressures. Under the condition of extremely low oxygen partial pressure, the surface tension of molten tin decreases with the increase of temperature. While at higher oxygen partial pressure, the surface tension of molten tin firstly increases to a maximum with the increase of temperature, and then decreases with the increase of temperature. The experimental results are coincident with the results of thermodynamic analysis.
REFERENCES
[1]Keene B J. Review of data for the surface tension of pure metals [J]. International Materials Reviews, 1993, 38(4): 157-192.
[2]Keene B J. Review of data for the surface tension of iron and its binary alloys [J]. International Materials Reviews, 1988, 33(1): 1-37.
[3]QIAO Zhi-ya, YAN Li-jun, CAO Zha-min, et al. Surface tension prediction of high-temperature melts [J]. Journal of Alloys and Compounds, 2001, 325: 180-189.
[4]YUAN Zhang-fu, Mukai K, Takagi K, et al. Surface tension and its temperature coefficient of molten tin determined with the sessile drop method at different oxygen partial pressures [J]. Journal of Colloid and Interface Science, 2002, 254(2): 338-345.
[5]WANG Tian-min. Eco-materials [M]. Tianjin: Tianjin University Press, 2002. 83-87.(in Chinese)
[6]JIANG Long. Conspectuses of Colloid Chemistry [M]. Beijing: Science Press, 2002. 23-25.(in Chinese)
[7]Lee J H, Lee D N. Use of the thermodynamic data to calculate surface tension and viscosity of Sn-based soldering alloy systems [J]. Journal of Electronic Materials, 2001, 30(9): 1112-1119.
[8]Friedrichs H A, Ronkow L W. Processing and editing of kinetic data of substances for the data and program base system Kindas [J]. Steel Research, 1995, 66(3): 110-112.
[9]Friedrichs H A, Ronkow L W, Vermot P, et al. New method for simultaneous measurement of viscosity, density and surface tension of metallic melts at high temperatures [J]. Steel Research, 1995, 66(12): 509-515.
[10]Fiori L, Ricci E, Arato E. Dynamic surface tension measurement on molten metal-oxygen system: model validation on molten tin [J]. Acta Materialia, 2003, 51: 2873-2890.
[11]WANG Chang-zhen. Research Technique of Metallurgic Physical Chemistry, 3rd edition [M]. Beijing: Metallurgical Industry Press, 2002. 278-309.(in Chinese)
[12]FAN Jian-feng, YUAN Zhang-fu, KE Jia-jun. Development in measuring surface tension of high temperature molten liquid [J]. Chemistry, 2004, 67(11): 802-808.(in Chinese)
[13]Takagi K, Otaka M, Natsui H, et al. Experimental study on transition to oscillatory thermocapillary flow in a low Prandtl number liquid bridge [J]. Journal of Crystal Growth, 2001, 233: 399-407.
[14]Rotenberg Y, Roruvka L, Neumann A W. Determination of surface tension and contact angle from the shapes of axisymmetric fluid interfaces [J]. Journal of Colloid and Interface Science, 1983, 93(1): 169-183.
[15]Kawai Y, Shiraishi Y. Handbook of Physico-chemical Properties at High Temperatures [M]. Tokyo: The Iron and Steel Institute of Japan, 1988, 2-3.
[16]Barin I. CHENG Nai-liang, ZHU Si-tong, XU Gui-ying, et al. Tr. Thermochemical Data of Pure Substances [M]. Beijing: Science Press, 2003. 1538-1550.(in Chinese)
[17]Kubaschewski O, Alcock C B. Metallurgical Thermochemistry, 5th edition [M]. New York: Pergamon, 1979. 383.
[18]Knacke O, Kubaschewski O, Hesselmann K. Thermochemical Properties of Inorganic Substances, 2nd edition [M]. Heidelberg, Berlin, Springer-Verlag, 1991. 1868-1893.
Foundation item: Project(50474043) supported by the National Natural Science Foundation of China
Received date: 2005-01-07; Accepted date:2005-05-10
Correspondence: YUAN Zhang-fu, Professor, PhD; Tel: +86-10-62527440; E-mail: yuanzhf@home.ipe.ac.cn
(Edited by YANG Bing)