三水铝石型铝土矿还原溶出赤泥中铁矿物的富集与分离
来源期刊:中国有色金属学报(英文版)2020年第7期
论文作者:李小斌 周兆宇 王一霖 周秋生 齐天贵 刘桂华 彭志宏
文章页码:1980 - 1990
关键词:三水铝石型铝土矿;赤泥;铁矿物;富集;矿物学特征
Key words:gibbsitic bauxite; red mud; iron minerals; enrichment; mineralogical characteristics
摘 要:研究3种高铁三水铝石型铝土矿的矿物学特征以及不同拜耳法溶出条件对铁矿物富集与分离的影响。结果表明,添加合适的有机物还原剂,例如甘油,可以促进伴生的一水硬铝石,一水软铝石和铝针铁矿的溶出,同时还可以促进针铁矿向赤铁矿转化。利用拜尔法还原溶出处理高铝硅比三水铝石型铝土矿(A/S=25.41)可直接产出合格铁精矿(TFe>56%),实现赤泥的零排放。对于中低A/S的矿石(7.82和3.35),通过重选获得的铁精矿中相应全铁含量分别为52.05%和50.16%,铁回收率分别为65.13%和79.13%,赤泥排放量减少约50%以上。
Abstract: The mineralogical characteristics of three kinds of gibbsitic high-iron bauxite and the effects of various digestion conditions on the enrichment and separation of iron minerals were investigated. The results show that adding an appropriate organic reductant such as glycerol can promote the digestion of concomitant diaspore, boehmite and alumogoethite as well as the conversion of goethite to hematite in the reductive Bayer digestion. Processing Bauxite A with A/S of 25.41 can directly produce qualified iron concentrates (TFe>56%) by the reductive Bayer digestion, and thus realize the zero red mud discharge. For Bauxite B and Bauxite C with A/S of 7.82 and 3.35, the iron recoveries of 65.13% and 79.13% can be achieved with the corresponding TFe of 52.05% and 50.16% in the iron concentrates by gravity separation, respectively, resulting in the red mud discharge reduction of ~50% or above.
Trans. Nonferrous Met. Soc. China 30(2020) 1980-1990
Xiao-bin LI1,2, Zhao-yu ZHOU1,2, Yi-lin WANG1,3, Qiu-sheng ZHOU1,2, Tian-gui QI1,2, Gui-hua LIU1,2, Zhi-hong PENG1,2
1. School of Metallurgy and Environment, Central South University, Changsha 410083, China;
2. National Engineering Laboratory for High Efficiency Recovery of Refractory Nonferrous Metals, Central South University, Changsha 410083, China;
3. Powder Metallurgy Research Institute, Central South University, Changsha 410083, China
Received 10 October 2019; accepted 6 May 2020
Abstract: The mineralogical characteristics of three kinds of gibbsitic high-iron bauxite and the effects of various digestion conditions on the enrichment and separation of iron minerals were investigated. The results show that adding an appropriate organic reductant such as glycerol can promote the digestion of concomitant diaspore, boehmite and alumogoethite as well as the conversion of goethite to hematite in the reductive Bayer digestion. Processing Bauxite A with A/S of 25.41 can directly produce qualified iron concentrates (TFe>56%) by the reductive Bayer digestion, and thus realize the zero red mud discharge. For Bauxite B and Bauxite C with A/S of 7.82 and 3.35, the iron recoveries of 65.13% and 79.13% can be achieved with the corresponding TFe of 52.05% and 50.16% in the iron concentrates by gravity separation, respectively, resulting in the red mud discharge reduction of ~50% or above.
Key words: gibbsitic bauxite; red mud; iron minerals; enrichment; mineralogical characteristics
1 Introduction
The gibbsitic bauxite residue (red mud), which is featured by high-alkali and ultra-fine grain size, presents a globally critical issue to the sustainable development of the alumina industry [1]. Significant efforts have been made in the treatment of red mud, and many methods were proposed including (1) recovery of valuable metals by direct separation [2,3], sintering [4-6], and acid leaching [7,8], etc, (2) utilization as a raw material for brick making [9,10], road construction [11,12], and cement [13], and (3) land reclamation for vegetational cover [14,15]. The researches on this topic have been well summarized in several previous reviews [16,17]. However, the majority of red muds can only be stored in containment dams nowadays due to the environmental risk, cost stress and/or limited consumption for those methods.
Iron minerals usually account for a high percentage of red mud, and the efficient iron-utilization incorporated to alumina extraction is meaningful to directly decrease the red mud discharge. Several alumina refineries for treating diasporic bauxite in China adopt high-gradient magnetic separators to recover iron concentrates. According to the difference in iron minerals distribution, the TFe in concentrate ranges from 50% to 56% with a relatively low iron recovery of <40% in practice. One reason for the low efficiency is that plenty of lime must be added and thus the iron minerals content degrades. Besides, lime introduced in the high temperature Bayer digestion (HTD) potentially reacts with other components to produce more than 20 Ca-bearing compounds [18], leading to the complex structure of red mud. Therefore, substituting an effective additive for lime in digestion may contribute to the enrichment of iron minerals and their dissociation with the desilication product (DSP) in red mud. For these objectives, reductive Bayer digestion (RBD) followed by magnetic separation was proposed to treat diasporic bauxite by adding iron powder (1-4 wt.% of bauxite) instead of lime (10-13 wt.% of bauxite). From the previous report [19], a qualified iron concentrate (TFe>56%) with the iron recovery of ~86% was obtained and over 70% of the red mud discharge could be decreased. To optimize the red mud discharge reduction, we believe that the following premises should be met: (1) To maximum enrich the iron minerals, full digestion of alumina (including gibbsite, boehmite and diaspore) [20] and the dehydration of goethite/ limonite [21] should be achieved simultaneously; (2) To effectively separate the iron minerals, the iron minerals particles should be dissociated with the DSP and be prevented from becoming smaller.
For treating the gibbsitic bauxite residue [22], the similar issues in iron recovery are also to enrich and dissociate the iron minerals. Gibbsitic bauxite usually contains concomitant impurities such as goethite, alumogoethite, boehmite and/or even diaspore. Under low-temperature Bayer digestion (LTD) conditions, the boehmite, diaspore and alumogoethite cannot be fully digested, and meanwhile goethite is rather difficult to dehydrate. Theoretically, the reductive Bayer digestion (RBD) process for treating diasporic bauxite is also suitable for treating gibbsitic bauxite. However, the reaction of the impurity minerals may be different from that in treating diasporic bauxite as the gibbsite can be rapidly digested and thus the caustic soda concentration decreases quickly in the initial stage. Furthermore, compared with the iron powder additive, some organic additives [23] are reasonable to decrease the red mud generation as they are soluble in the caustic soda solution. LI [24] reported that the addition of 20 g/L glucose together with 2 g/L lime in high-temperature digestion process can produce a red mud with large particle size and small specific surface area by converting the goethite to hematite and/or magnetite. Hence, adding appropriate organic chemicals such as cellulose, glycol and glycerol may play a similar role in the iron mineral conversion and thus benefits the following beneficiation.
In this work, three high-iron gibbsitic bauxites with various A/S (alumina to silica ratio) were adopted to investigate the effects of mineral characteristics and digestion conditions on the enrichment and separation of iron minerals in the red mud.
2 Experimental
2.1 Bauxites and reagents
Both the Bauxite A with an A/S of 25.41 and Bauxite B with an A/S of 7.82 were derived from boke region in Guinea. In 2018, the bauxite output in Guinea was ~59 million tons and accounted for >20% of the total output in the world, in which >65% of the bauxite were exported to China. Bauxite C with an A/S of 3.35 was sampled from Guangxi region (China) which is the main occurrence zone of gibbsitic bauxite in China. The main chemical and mineral compositions of the bauxites shown in Table 1 and Fig. 1 illustrate that they are mainly composed of gibbsite, hematite, goethite, anatase, etc. Furthermore, a characteristic peak of diaspore can be observed in the XRD pattern of Bauxite A, while it cannot be exactly affirmed as the peak intensity is relatively low. Therefore, TG-DTA analyses were conducted and the curves are drawn in Fig. 2. The endothermal peaks detected near 500 °C in Bauxite A and Bauxite B are in accordance with the dehydration peak of the monohydrate Al-minerals [25], which can further verify the existence of diaspore or boehmite in these two bauxites. In contrast, the results also indicate that there is no diaspore in Bauxite C. According to the XRD, TG-DTA and chemical analyses, the mineral compositions of these bauxites were estimated and the semi- quantitative results are listed in Table 2.
Table 1 Main chemical compositions of gibbsitic bauxites

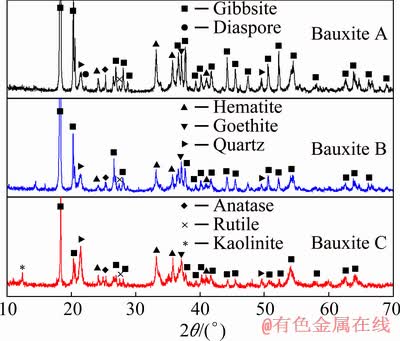
Fig. 1 XRD patterns of gibbsitic bauxites
To avoid the effect of iron powder addition on the total iron content in red mud, the glycerol (C3H8O3) with analytical purity (>99.5%) was adopted as a digestion additive to realize the reductive Bayer digestion process in this work. Lime was obtained by roasting calcium hydroxide at 850 °C for 60 min. Sodium aluminate solutions were prepared by dissolving industrial grade aluminum hydroxide and sodium hydroxide (purity >98.5%) in boiling water.
2.2 Methods
2.2.1 Bauxite digestion
The low-temperature (for LTD) and high- temperature (for HTD and RBD) processes were respectively carried out in a glycerol kettle (DY-8) and a molten mixed nitrate salt kettle (XYF-d44×6), which were purchased from the machinery plant affiliated with Central South University, China. The required dosages of gibbsitic bauxite and additives (glycerol or lime) were added to a 150 mL stainless steel bomb along with 100 mL sodium aluminate solution.
The sealed bomb was immersed and then rotated in the reaction kettle. 2×d15 mm and 4×d5 mm steel balls were placed in advance inside the bomb to aid the agitation. After a designated duration, the bomb was removed from the kettle and immediately placed in tap water to cool. The resulting slurry was filtered, and the cake was washed with hot water and then dried at 90 °C for 12 h before analyses. Pictorial view of stainless- steel bomb along with the mechanism of the reaction kettle are shown in Fig. 3.
The alumina recovery (η(Al2O3)) was calculated with Eq. (1):
 (1)
(1)
where m1 and m2 are the masses of bauxite and red mud; w1(Al2O3) and w2(Al2O3) are the mass fractions of alumina in bauxite and red mud, respectively.
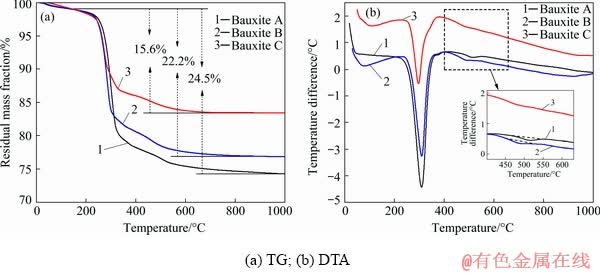
Fig. 2 TG-DTA curves of gibbsitic bauxites
Table 2 Estimated mineral compositions of gibbsitic bauxites (wt.%)
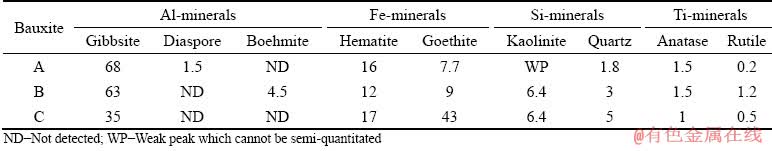

Fig. 3 Pictorial view of stainless-steel bomb along with mechanism of reaction kettle
2.2.2 Iron mineral separation
As hematite has the highest density of the bulk of minerals present in red mud, it can be separated using density fractions. The performance of gravity concentration for red mud was preliminarily evaluated using a settling tube (d3.5 cm × 32 cm). 5 g red mud and 200 mL water were fully mixed in the settling tube, and then the upper suspension was dumped after apparent stratifying of the slurry. Water was added to the underlying slurry to keep the total volume constant and the above operations were repeated 3 times. The underlying slurry (high density minerals, concentrate) and total upper suspensions (low density minerals) were filtered and dried separately for analyses. The schematic of iron mineral separation by gravity settling are described in Fig. 4. The iron recovery (η(Fe)) was calculated with Eq. (2):
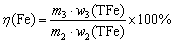 (2)
(2)
where m3 represents the mass of concentrate; w2(TFe) and w3(TFe) denote the iron contents in red mud and concentrate, respectively.

Fig. 4 Schematic of iron mineral separation by gravity settling
2.3 Characterization
The alumina, ferric oxide, and silica contents in samples were measured using Zn2+ titration, Cr6+ titration, and silicon-molybdenum blue colorimetry, respectively. The mineral phases were identified by XRD (TTR-III, Rigaku Corporation, Japan) using Cu Kα radiation at a scan rate of 5 (°)/min. To get a lower background base, the signals were recorded using a monochromator during the test. The TG-DTA of bauxite was identified by SDTQ600 thermal analyzer at the heating rate of 10 °C/min with a 100 mL/min flow rate of argon gas. Surface microscopic morphology and micro area composition analyses were conducted by SEM (Quanta-200, FEI, USA) and EDX (GENSIS60S, EDAX, USA). The procedures of preparing samples for SEM and EDX analyses are as follows. (1) Bauxite and red mud powders were mounted in an epoxy resin block polymerized by triethanolamine (mass ratio of epoxy to triethanolamine was 9:1). (2) Then, the solidification of the resin block was performed at 70 °C for 12 h. (3) Finally, the sample surface was mechanically polished using different grade silicon carbide papers successively (300, 600, 1000 and 3000 grit), in which the polish time was 10 min for each step.
3 Results and discussion
3.1 Minerals distribution of gibbsitic bauxite
The reactions of Al-, Fe-, Si- and Ti-bearing minerals in the Bayer digestion process are closely associated with their mineralogical characteristics in gibbsitic bauxite, and thus clarifying the process mineralogy is helpful to optimizing the digestion and separation conditions. The backscatter scanning electron microscopy (BSEM) and elemental map-scan images of polished gibbsitic bauxite samples are shown in Fig. 5.
From Fig. 5, most of the gibbsite and hematite in high A/S Guinea Bauxite A exist in the forms of dissociated particles, laying a good foundation for the separation of iron minerals from red mud. Besides, it can be observed from the EDS results that a small quantity of the Al/Fe minerals disseminate to each other in some particles, which may decrease the alumina recovery and the TFe in red mud. Ti/Si map-scan images further indicate that the Ti-bearing minerals present in some relatively-coarse particles, while the Si-bearing minerals mainly disseminate in the iron mineral particles. The occurrence of Fe-/Al-/Si-/Ti-bearing minerals in Bauxite B and Bauxite C is similar to that in Bauxite A.
Based on the mineral distribution characteristics, it can be predicted that: (1) Under the LTD conditions, the uncovered gibbsite can be easily digested, while the covered Al-bearing minerals are difficultly digested by caustic soda solution; (2) Under the HTD conditions, the dissolution-precipitation of Fe-bearing minerals in caustic soda solution will facilitate the digestion of the inner Al-bearing minerals; (3) The Ti-bearing minerals embedded in the other mineral particles may still inhibit the diaspore/boehmite digestion as they are exposed gradually with the digestion of gibbsite in the HTD; (4) The disseminated Si-bearing minerals will decrease the dissociation between iron minerals and DSP particles in red mud during the digestion process.
3.2 Bayer digestion and iron mineral enrichment
Generally, the LTD and HTD processes are adopted to treat the gibbsitic bauxite and the boehmitic/diasporic bauxite, respectively. However, the boundary between these processes has become increasingly blurred with the bauxite quality degrades. For example, the double digestion process was employed to treat gibbsitic/boehmite mixed bauxite [26], and some refineries directly treated the gibbsitic bauxite under HTD conditions. Therefore, to verify those predictions in Section 3.1, the Bayer digestions of gibbsitic bauxites were conducted under various conditions, e.g., temperature, time, caustic soda concentration and additive dosage. In this section, Bauxite A was taken as the raw material for a case study. The details of these experiments are listed in Table 3.
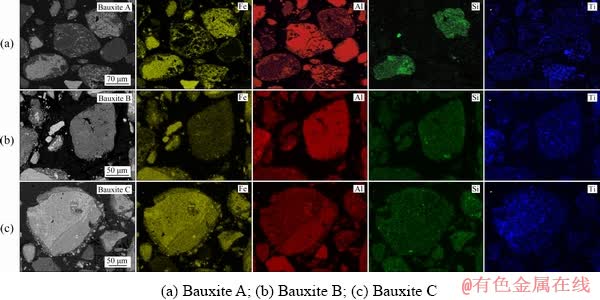
Fig. 5 BSEM and elemental map-scan images of gibbsitic bauxites
Table 3 Details for treating Bauxite A by LTD, HTD and RBD processes
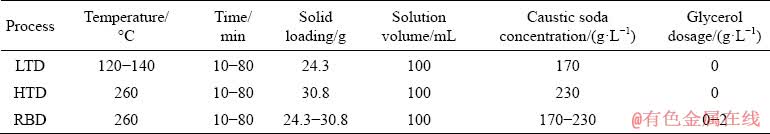

Fig. 6 Effects of digestion time and temperature on η(Al2O3) (a) and TFe in red mud (b)
Figures 6(a) and (b) show the effects of digestion temperature and time on the alumina recovery and TFe in red mud. Under the LTD conditions (140 °C), the alumina recovery is 90% after 10 min and TFe in the red mud is about 45%. Further increasing digestion time does not exert an influence on alumina recovery. According to the chemical analyses, the A/S of red mud produced at 140 °C for 80 min still reaches up to 6.29, meaning that ~10% of alumina cannot be digested. One reason for the alumina loss is that the disseminated distribution of Fe- and Al-bearing minerals inhibits the digestion of inner Al minerals as the Fe minerals are stable under LTD conditions (Section 3.1). Another reason is that some of the Al-bearing minerals are in the form of diaspore (Section 2.1). Under HTD conditions (260 °C), the alumina recovery increases to 94.46% and the A/S in red mud decreases to 2.55, upgrading the TFe in red mud to 47.50%. Since the diaspore is difficult to be digested without lime under the HTD conditions, it can be considered that the dissolution-precipitation of iron minerals contributes to the exposure of inner gibbsite and thus promotes its digestion. Furthermore, adding lime as an additive can slightly improve the alumina recovery (95.21% for 1% lime and 96.67% for 6% lime), whereas the TFe in red mud decreases to merely 44.46% and 37.78% due to the marked increase of red mud quantity. To avoid the increase of red mud generation caused by lime addition, a soluble additive (glycerol) was adopted in the RBD process to improve the digestion of alumina. The alumina recovery of the RBD process is close to that of the HTD process in the initial 30 min and gradually increases from 94.5% to 98.0% with the digestion time increases from 30 to 80 min. The increment of alumina recovery can be attributed to the fact that glycerol can eliminate the inhibition of sodium titanate on diaspore digestion. Because the liquid additive does not increase the residue mass, the TFe in red mud can be enriched to 56.83% in the RBD process.
Figures 7(a) and (b) show the effects of caustic soda concentration and glycerol dosage on the alumina recovery and TFe in red mud. A caustic soda solution with 170 g/L of Na2OK is commonly used for digesting gibbsitic bauxite in the industry. Nevertheless, only a slight increase of alumina recovery and TFe can be observed with the increase of glycerol dosage under the simulated conditions, indicating that the function of glycerol heavily depends on the caustic soda concentration. At a typical caustic soda concentration of 230 g/L for diasporic bauxite digestion, increasing the glycerol dosage promotes the digestion of diaspore and the enrichment of iron minerals. Being consistent with the results in Fig. 6, 54.37% and 56.83% of TFe in red mud can be obtained by adding 1.5 and 2.0 g/L glycerol, respectively.
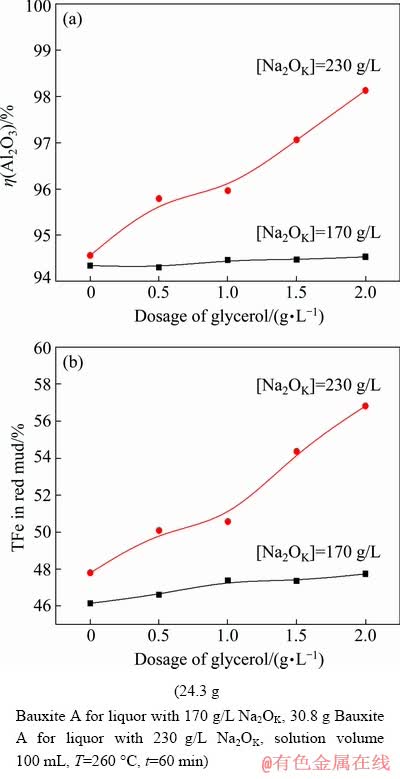
Fig. 7 Effects of caustic soda concentration and glycerol dosage on η(Al2O3) (a) and TFe in red mud (b)
3.3 Mineral phase evolution
To clarify the mineral evolution during various digestion processes, the typical red mud samples were characterized by XRD, and the results are shown in Fig. 8. For the sample from LTD process, the characteristic peaks of hematite, goethite, diaspore, quartz, rutile, anatase, pyrophyllite and boehmite can be detected in the XRD pattern. For the sample from HTD process, the characteristic peaks of boehmite, pyrophyllite and anatase disappear, but those of diaspore and goethite still exist. This result proves once again that the presences of undigested diaspore and unconverted goethite lead to the decrease of alumina recovery and TFe in red mud. The mineral composition of red mud from RBD process for treating Bauxite A is greatly simplified, and only characteristic peaks of quartz and hematite can be observed in the XRD pattern. Compare with the HTD results, it can be found that the goethite may convert to hematite and/or magnetite depending on the dosage of reductant [19]. Besides, the decomposition of Al/Fe-bearing minerals could expose more interfaces of rutile and thus accelerate the reaction between rutile and alkali liquor. According to the elemental analyses, the TFe in the RBD red mud is up to 56.83%, while the alumina content is only 3.00%. The XRF analysis results of trace elements (Table 4) further show that the contents of phosphorus, sulfur, magnesium and other harmful elements could meet the requirements of iron concentrates for blast furnace ironmaking. Therefore, it is feasible to directly produce qualified iron concentrate by treating Guinea high A/S Bauxite A with RBD process and thus to realize the zero red mud discharge in alumina production. For the Bauxite B and Bauxite C with A/S of 7.82 and 3.35, only hematite and zeolite (a kind of DSP) can be detected in the XRD patterns of red mud generated from RBD process. Zeolite generation degrades the iron concentrate; therefore, it is necessary to separate iron minerals from red mud by appropriate beneficiation measures in order to reduce the red mud discharge.
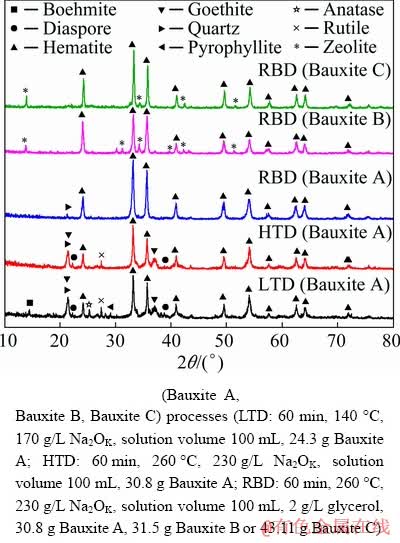
Fig. 8 XRD patterns of red mud derived from LTD (Bauxite A), HTD (Bauxite A) and RBD
Table 4 Chemical composition of red mud from RBD process of Bauxite A (wt.%)
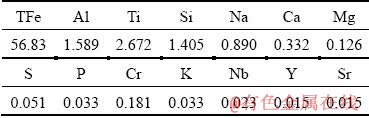
The above results can also be verified by the BSEM analyses, as shown in Fig. 9. Coarse Al-bearing minerals can be detected in both the red muds produced by LTD and HTD processes, while they are almost non-existent in the RBD red mud. Furthermore, elevating the digestion temperature markedly decreases the particle size of Fe-bearing minerals and makes the Ti-/Si-bearing minerals diffused. In summary, the mineral phase evolution mechanism of gibbsitic bauxite during LTD, HTD and RBD process can be graphically presented in Fig. 10.
3.4 Gravity concentration of iron minerals
The magnetic hysteresis of red mud from the RBD (Bauxite A) process was determined using a vibrating sample magnetometer. The saturation magnetization of red mud is only 2.50 A・m2/kg, demonstrating that the magnetism is relatively low.
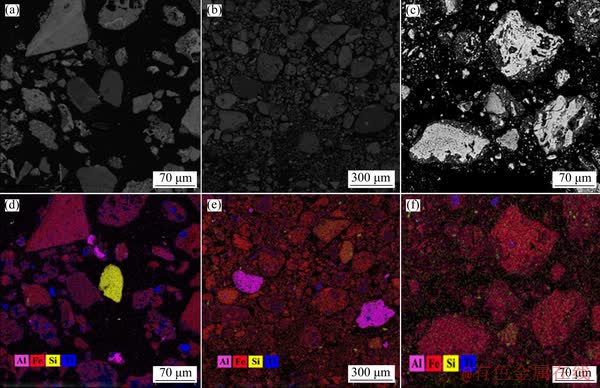
Fig. 9 BSEM and elemental map-scan images of red mud from LTD (Bauxite A) (a, d), HTD (Bauxite A) (b, e) and RBD (Bauxite A) (c, f)
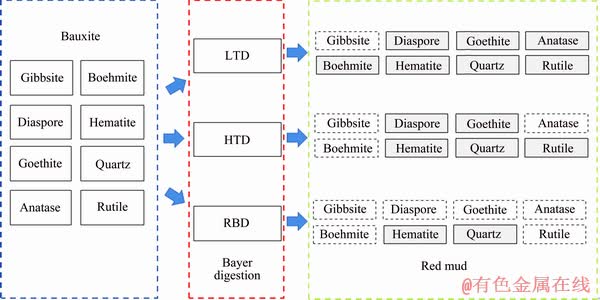
Fig. 10 Mineral phase evolution mechanism of LTD, HTD and RBD process
Considering the coarse iron mineral particles in red mud, the appropriate beneficiation method is gravity separation. To investigate the separation performance of bauxite residue obtained under different digestion processes (LTD, HTD and RBD), the gravity separation experiments on the red mud samples were conducted and the results are shown in Table 5.
Experiment 1 in Table 5 shows that the iron minerals in red mud from the LTD process cannot be separated by gravity concentration as the iron content in concentrate and tailing are close to each other. However, the iron minerals in red mud generated under the HTD conditions can be partially recovered under the same beneficiation conditions, and the output rate of concentrate is 53.56% with TFe in concentrate of 53.20 wt.% and iron recovery of 60.86% (Experiment 2). The beneficiation results for Experiment 3 reveal that the iron content in concentrate cannot be upgraded further through gravity concentration. This can be explained by the fact that the remaining impurities mainly exist in the coarse iron particles, as shown in Fig. 9. Besides, because the beneficiation will lead to the inevitable iron loss, only the red mud samples in which the iron content is lower than 50% are suggested to adopt in the separation procedure.
Experiments 4 and 5 show the results of beneficiation experiments for red muds produced by treating the medium A/S Bauxite B and low A/S Bauxite C in the RBD process. Separating the red muds can obtain iron concentrates of TFe of 52.05 wt.% with a recovery of 65.13% for Bauxite B and TFe of 50.16 wt.% with a recovery of 79.13% for Bauxite C. These results suggest that ~50% or above of red mud discharge can be decreased by the RBD process and separation.
Figures 11 and 12 show the BSEM and elemental map-scan images of the red muds, the concentrates and tailings obtained by treating the Bauxite B and Bauxite C generated from RBD process. The iron minerals in concentrates exist almost in the forms of coarse particles, while the particles of iron minerals in the tailing are relatively fine. In addition, the elemental map-scan results further reveal that Al and Si elements coexist in the fine particles with an average particle size of ~2 μm, while the distribution of Ti element is similar to that of Fe. That is to say, only the coarse iron can be recovered by separating the fine DSP particles through gravity concentration process.
Table 5 Gravity concentration results of red muds from LTD, HTD and RBD processes
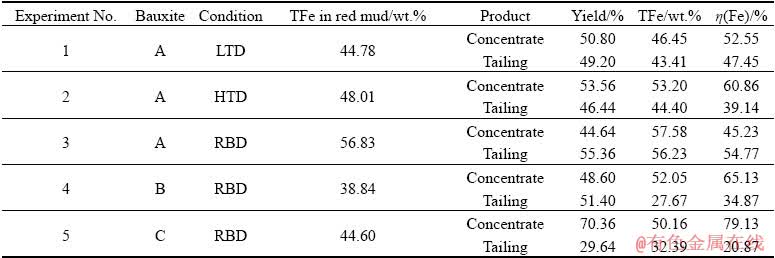
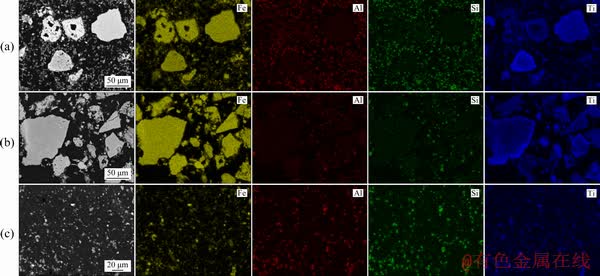
Fig. 11 BSEM images of digested red muds (a), concentrates (b) and tailings (c) of Experiment 4 (Bauxite B) in Table 5

Fig. 12 BSEM images of digested red muds (a), concentrates (b) and tailings (c) of Experiment 5 (Bauxite C) in Table 5
4 Conclusions
(1) In the reductive Bayer digestion of gibbsitic bauxite, adding appropriate organic additives can promote both the digestion of concomitant diaspore, boehmite and alumogoethite and the conversion of goethite to hematite. Meanwhile, because of the marked reduction of red mud generation, qualified iron concentrates can be directly obtained by treating high A/S Bauxite A at 260 °C for 60 min with adding 2 g/L glycerol.
(2) The beneficiation performance is mainly determined by the mineralogical characteristics of iron minerals in bauxite. The dissociated coarse iron particles can be readily recovered, while it is difficult to separate the fine iron particles. For treating the medium/low A/S gibbsitic bauxites (Bauxite B and Bauxite C), significant reductions of red mud discharges can be realized by the RBD followed by gravity separation.
(3) The application of this technique can realize the reduction of red mud discharge in alumina production and thus to decrease the adverse impact of red mud disposal. However, to effectively implement the cleaner technique, further research should be conducted focusing on the decrease of additive dosage and optimization of the beneficiation process.
References
[1] POWER G, GRAFE M, KLAUBER C. Bauxite residue issues: I. Current management, disposal and storage practices [J]. Hydrometallurgy, 2011, 108: 33-45.
[2] HUANG Yan-fang, HAN Gui-hong, LIU Jiong-tian, WANG Wen-juan. A facile disposal of Bayer red mud based on selective flocculation desliming with organic humics [J]. Journal of Hazardous Materials, 2015, 301: 46-55.
[3] LIU Wan-chao, ZHANG Xiao-shen, JIANG Wen-chen, ZHU Xin-feng, YANG Jia-kuan. Study on particle size separation pretreatment of Bayer red mud [J]. Chinese Journal of Environmental Engineering, 2011, 5(4): 921-924. (in Chinese)
[4] LI Xiao-bin, XIAO Wei, LIU Gui-hua, PENG Zhi-hong, ZHOU Qiu-sheng, QI Tian-gui. Recovery of alumina and ferric oxide from Bayer red mud rich in iron by reduction sintering [J]. Transactions of Nonferrous Metals Society of China, 2009, 19(5): 1342-1347.
[5] RAI S, NIMJE M T, CHADDHA M J, MODAK S, RAO K R, AGNIHOTRI A. Recovery of iron from bauxite residue using advanced separation techniques [J]. Minerals Engineering, 2019, 134: 222-231.
[6] ZHOU Qiu-sheng, FAN Kuang-sheng, LI Xiao-bin, PENG Zhi-hong, LIU Gui-hua. Alumina recovery from red mud with high iron by sintering process [J]. Journal of Central South University (Science and Technology), 2008, 39(1): 92-97. (in Chinese)
[7] PEPPER R A, COUPERTHWAITE S J, MILLAR G J. Comprehensive examination of acid leaching behaviour of mineral phases from red mud: Recovery of Fe, Al, Ti, and Si [J]. Minerals Engineering, 2016, 99: 8-18.
[8] TENG Chun-ying, ZHOU Kang-gen, NING Ling-feng, PENG Chang-hong, HE De-wen. Stepwise leaching of valuable metals from red mud using hydrochloric acid [J]. Chinese Journal of Environmental Engineering, 2018, 12(1): 310-315. (in Chinese)
[9] LIU Wan-chao, YANG Jia-kuan, XIAO Bo. Application of Bayer red mud for iron recovery and building material production from alumosilicate residues [J]. Journal of Hazardous Materials, 2009, 161: 474-478.
[10] WEI Hong-shan, MA Xiao-e, GUAN Xue-mao, HUANG Ang, LI Shi-hua, DONG Yun-tao, ZHAO Yue. Application research of red mud as a resource of building materials [J]. Light Metal, 2018(9): 26-29. (in Chinese)
[11] KEHAGIA F. A successful pilot project demonstrating the re-use potential of bauxite residue in embankment construction [J]. Resources, Conservation, Recycling, 2010, 54: 417-421.
[12] MUKIZA E, ZHANG Ling-ling, LIU Xiao-ming, ZHANG Na. Utilization of red mud in road base and subgrade materials: A review [J]. Resources, Conservation, Recycling, 2019, 141: 187-199.
[13] GHALEHNOVI M, ROSHAN N, HAKAK E, SHAMSABADI E A, BRITO J. Effect of red mud (bauxite residue) as cement replacement on the properties of self- compacting concrete incorporating various fillers [J]. Journal of Cleaner Production, 2019, 240: 118-213.
[14] COURTNEY R, MULLEN G, HARRINGTON T. An evaluation of revegetation success on bauxite residue [J]. Restoration Ecology, 2009, 17: 350-358.
[15] HAN Fu-song, WANG Cheng, XUE Sheng-guo, HUANG Nan, YE Yu-zhen. Plant species diversity in bauxite residue disposal areas [J]. Journal of University of Chinese Academy of Sciences, 2017, 34(5): 551-557. (in Chinese)
[16] LIU Yan-ju, NAIDU R. Hidden values in bauxite residue (red mud): Recovery of metals [J]. Waste Management, 2014, 34: 2662-2673.
[17] KHAIRUL M A, ZANGANEH J, MOGHTADERI B. The composition, recycling and utilization of Bayer red mud [J]. Resources, Conservation, Recycling, 2019, 141: 483-498.
[18] SMITH P. Reactions of lime under high temperature Bayer digestion conditions [J]. Hydrometallurgy, 2017, 170: 16-23.
[19] LI Xiao-bin, WANG Yi-lin, ZHOU Qiu-sheng, QI Tian-gui, LIU Gui-hua, PENG Zhi-hong, WANG Hong-yang. Transformation of hematite in diasporic bauxite during reductive Bayer digestion and recovery of iron [J]. Transactions of Nonferrous Metals Society of China, 2017, 27(12): 2715-2726.
[20] WANG Yi-lin, LI Xiao-bin, WANG Biao, ZHOU Qiu-sheng, QI Tian-gui, LIU Gui-hua, PENG Zhi-hong, ZHOU Ke-chao. Interactions of iron and titanium-bearing minerals under high-temperature Bayer digestion conditions [J]. Hydrometallurgy, 2019, 184: 192-198.
[21] LI Xiao-bin, WANG Yi-lin, ZHOU Qiu-sheng, QI Tian-gui, LIU Gui-hua, PENG Zhi-hong, WANG Hong-yang. Reaction behaviors of iron and hematite in sodium aluminate solution at elevated temperature [J]. Hydrometallurgy, 2018, 175: 257-265.
[22] JAMISON E, JONES A, COOLING D, STOCKTON N. Magnetic separation of red sand to produce value [J]. Minerals Engineering, 2006, 19: 1603-1605.
[23] LI Xiao-bin, YU Shun-wen, ZHANG Fang, PENG Zhi-hong, LIU Gui-hua, ZHOU Qiu-sheng, QI Tian-gui. A high pressure digestion method for diasporic bauxite: CN Patent, 102976375A [P]. 2012-12-01.
[24] LI L Y. A study of iron mineral transformation to reduce red mud tailings [J]. Waste Management, 2001, 21(6): 525-534.
[25] POST E, FIDLER B, KWIRYN D, RAPP D. Characterization of bauxite and its minerals by means of thermoanalytical methods [J]. Light Metals, 2012: 5-7.
[26] LAMERANT J M, PERRET Y. Boehmitic Reversion in a double digestion process on a bauxite containing trihydrate and monohydrate [J]. Light Metals, 2016, 377-380.
李小斌1,2,周兆宇1,2,王一霖1,3,周秋生1,2,齐天贵1,2,刘桂华1,2,彭志宏1,2
1. 中南大学 冶金与环境学院,长沙 410083;
2. 中南大学 难冶有色金属资源高效利用国家工程实验室,长沙 410083;
3. 中南大学 粉末冶金研究院,长沙 410083
摘 要:研究3种高铁三水铝石型铝土矿的矿物学特征以及不同拜耳法溶出条件对铁矿物富集与分离的影响。结果表明,添加合适的有机物还原剂,例如甘油,可以促进伴生的一水硬铝石,一水软铝石和铝针铁矿的溶出,同时还可以促进针铁矿向赤铁矿转化。利用拜尔法还原溶出处理高铝硅比三水铝石型铝土矿(A/S=25.41)可直接产出合格铁精矿(TFe>56%),实现赤泥的零排放。对于中低A/S的矿石(7.82和3.35),通过重选获得的铁精矿中相应全铁含量分别为52.05%和50.16%,铁回收率分别为65.13%和79.13%,赤泥排放量减少约50%以上。
关键词:三水铝石型铝土矿;赤泥;铁矿物;富集;矿物学特征
(Edited by Bing YANG)
Foundation item: Project (2019M652799) supported by China Postdoctoral Science Foundation; Project (51604309) supported by the National Natural Science Foundation of China
Corresponding author: Yi-lin WANG; Tel/Fax: +86-731-88830453; E-mail: wang.yi.lin@outlook.com
DOI: 10.1016/S1003-6326(20)65355-9

