���±�ţ�1004-0609(2012)10-2924-06
�����ڻ���̿�ϵ�����ģ��
������1, 2, 3���� ��4��������1, 2��������5������Ԫ1, 2�������1, 2
(1. ���ϴ�ѧ ұ���ѧ�빤��ѧԺ����ɳ 410017��
2. ���ϴ�ѧ �����ؽ�����Ⱦ���ι��̼����о����ģ���ɳ 410017��
3. ����ʡ����������ѧ�о�Ժ����ɳ 410004��4. ��ɳ����ƾ֣���ɳ 410013��
5. �л����ʹ�������о�Ժ�������ι�˾����ɳ 410007)
ժҪ�����þ�̬����ʵ���о������ڻ���̿�ϵ���������ѧ������ѧ��Ϊ������Ԥ������Ļ���̿�������ӣ������������ض�������ѧ���̣����������Ϊ0.276 1 g/g��������Ϊ-23.89 kJ/mol��������ǿ�Ƚ�С��һ�����ӷ���ͨ����������ڻ���̿�����������Ե��ϣ����ű��������������ӣ����������3 432 cm-1���ǻ������ɼ������������ɢ���ף��ɴ��Ʋ������ͨ���ǻ��ϵ���������ϡ��ۺ����������������̿�������ӵ�ģ�ͣ���Բ��������ص�ʵ������
�ؼ��ʣ����ӣ�����̿������ģ��
��ͼ����ţ�X703.1��X793 ���� ���ױ�־�룺A
Adsorption model of phenol on activated carbon
YOU Xiang-yu1, 2, 3, YANG Jie4, WANG Yun-yan1, 2, SU Yan-rong5, CHAI Li-yuan1, 2, SHU Yu-de1, 2
(1. School of Metallurgical Science and Engineering, Central South University, Changsha 410017, China;
2. National Engineering Research Center for Pollution Control of Heavy Metals, Central South University,
Changsha 410017, China;
3. Hunan Research Academy of Environmental Sciences, Changsha 410004, China;
4. Changsha Municipal Audit Bureau, Changsha 410013, China;
5. China Machinery International Engineering Design & Research Institute Co., Ltd., Changsha 410007, China)
Abstract: The dynamics and thermodynamics of activated carbon adsorbing phenol were investigated by static adsorption experiments. The adsorption process obeys the second-level dynamic equation, where the pretreatment activated carbon is used to adsorb phenol. The maximum adsorption capacity is 0.276 1 g/g. The experimental results deduced from the adsorption thermodynamics computation and the infrared spectrogram shows that the adsorption heat is -33.046 kJ/mol, the adsorption bond strength is small, and a phenol molecule adsorbs on two active points of activated carbon surface via hydrogen bond. With the increase of phenol dosage, the infrared spectrum broadens gradually on 3 432 cm-1 hydroxyl spectral line, the dissemination spectrum occurs, which indicates that phenol associates through hydrogen bond on hydroxyl. Adsorption model being built based to the obtained data could be used to explain the related experiment phenomena.
Key words: phenol; activated carbon; adsorption model
������Ŀ�����ҽܳ������ѧ����������Ŀ(50925417)��������Ȼ��ѧ�����ص���Ŀ(50830301)������ʡ�о������д�����Ŀ(CX2010B121)
�ո����ڣ�2011-10-18�������ڣ�2012-03-20
ͨ�����ߣ������࣬�����ڣ���ʿ���绰��0731-88830577��E-mail: wyy@csu.edu.cn
������һ�ֳ����ĸ߶����ѽ����л��������ҵ�ķ�չʹ���ӷ�ˮ���ŷ���Խ��Խ�ɷ�ҲԽ��Խ���ӡ����ӷ�ˮCOD�����ߣ������ṹ�ȶ������ֽ⣬�������Բ�����Ѷȴ��ǵ�ǰ���������о����ȵ����ѵ�����[1]����ˣ��о�������Ч�ĺ��ӷ�ˮ������������ʮ����Ҫ�����塣
�������ڴ������ӷ�ˮ��Ӧ�ý�Ϊ�㷺���������Է�ˮ�б��ӵ��������кܺõĿ����ԣ������Ѹ�����[2]���ܶ��ʹ�ã�����Ч�����ԣ�����������ѡ��ķ�Χ�϶ࡣ���õ������������̿[3]����ʯ[4]��������[5]�������֬[6-7]�ȶԱ��ӵ�����Ч��һ�㶼��90%���ϡ�����̿����̼Ϊ��Ҫ�ɷֵ��������ϣ��ṹ�Ƚϸ��ӡ�����̿���������ܲ��������϶�ṹ�йأ��������仯ѧ��ɺͻ�ѧ�ṹ�й�[8]����Ϊ����������Ļ���̿�������Ƿǽᾧ�����ʣ���ϸ��ʯī״���ͽ���������һ���̼�⻯���ﹹ�ɵģ����岿��֮��ļ�϶�γɿ�϶���������̿�����е��������ܡ��������[3]��ë�����̿���Ʊ������������ӷ�ˮ�������о���RADOVIC��[9]�о��˲�ͬ����̿����������ѧ���ԣ��������溬�����š���������̿���ʵȶԱ�������ȥ���ʵ�Ӱ�졣ROOSTAEI��[10]���о���ʵ������������̿�Ա��ӵ������������ڹ轺�ͻ���������VIRARAGHAVAN[11]������̿�������ӷ�ˮ��ȥ���ʴﵽ46.1%������Ч���������ڷɻ���������ղ���[12]�о�����̿�������ӷ�ˮ�Ĵ���Ч�����ڻ���̿����Ϊ14 g/L����ʼŨ��Ϊ0.505 mg/L������£�20 min�ӷ��ӵ�ȥ���ʼ��ɴﵽ96.04%����Щ�о����������û���̿�������ӷ�ˮ����ȫ���еġ�
Ŀǰ��������Ի���̿�����������ӷ�ˮ���о����ͣ���ڹ�����[13]��û�жԱ����������̽��������о���������ؽ��ͱ��ӵ�����������Ӱ��ù��յ��о���Ӧ�á����������ڲ�������̿����������Ϊ�Ĺ��̺�����������ѧ����������ѧ��������ͬŨ�ȱ��Ӻ����̿�ĺ������ͼ�Ʋ������̿�������ӵ�ģ�ͣ�Ϊ�����������չ��̵��Ż�������������̽���ṩ�������ݡ�
1 ʵ��
1.1 ���ӷ�ˮ
ģ�ⱽ�ӷ�ˮ����ȡ1.35 g��������1000 mLˮ�У����л�̼(TOC)Ũ��Ϊ950 mg/L����ѧ������(COD)Ũ��Ϊ2 850 mg/L��pHֵΪ6.5��
1.2 ʵ�����
�����õ�HA-B�ͻ���̿����ˮ��ϴ�ɾ����ٷ����ˮ����ȥ�����ʺ��ɱ��á�
��ȡһ������Ԥ���������̿������Ͷ�뵽�ѵ�pHֵ��װ��100 mLģ�ⱽ�ӷ�ˮ����ƿ�У�������(ת��170 r/min����Ӧ�¶�20 ��)����Ӧ��ɺ�ȡ������520 nm�IJ����£���722�Ϳɼ���ֹ��ȼƶ�ʣ�౽��Ũ�Ƚ��м�⡣
�������������о��У�����̿����Ϊ5 g/L����ҺpHֵΪ5.8������������ѧ�о��У�����̿����Ϊ5 g/L�����ӳ�ʼŨ��Ϊ1.35 g/L����ҺpHֵ5.8����Ӧʱ��Ϊ2 h��
ʵ���������ҺpHֵʹ��ϡH2SO4��NaOH����������ʵ�����3��ƽ�������������ݵ�ƽ��ֵ���ڽ��������
1.3 ��������
�����߷�����һ��8֧50 mL��ɫ���зֱ����0��0.5��1.0��3.0��5.0��7.0��10.0��12.5 mL���ӱ��м�Һ����ˮ��50 mL���ߣ���0.5 mL������Һ�����ȣ���ʱpHֵΪ10��0.2������4-�������������Һ1.0 mL���ȡ��ټ�1.0 mL���軯����Һ����ֻ��ȣ�����10 min����510 nm�������ù��20 mm�ı�ɫ���Դ�ˮΪ�αȣ���������ȣ����հ�У����������ȶԱ��Ӻ����ı����ߡ�

ʽ�У�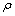 Ϊ����Ũ�ȣ�mg/L��mΪ��ˮ��У������ȣ���У�����ϲ�ñ��Ӻ�����mg��vΪ��ȡ����Һ�����L��
Ϊ����Ũ�ȣ�mg/L��mΪ��ˮ��У������ȣ���У�����ϲ�ñ��Ӻ�����mg��vΪ��ȡ����Һ�����L��
��Ʒ�ĺ�����ײ��ø���Ҷ���������(�����ȵ繫˾��Nicolet Nexus670)�ⶨ��TOC�������л�̼�ⶨ��(�ձ�����˾������TOC-VCPH)�ⶨ��
2 ���������
2.1 ��������ѧ
�ڻ���̿Ͷ����0.5 g���¶�20 �棬����Ũ��1.35 g/Lʱ���������淴Ӧʱ��ı仯��ͼ1��ʾ����ͼ1�ɿ���������ʱ������ӣ�����̿�������ӵ�����70 minʱ�ﵽƽ�⣬Ϊ�˱�֤����̿���������ܴﵽ���ͣ���ȡ������ʱ��Ϊ120 min��
�ڻ���̿�������������ӵĶ���ѧ�о��У��ֱ���һ������ѧ����(Pseudo-first-order)�Ͷ�������ѧ����(pseudo-second-order)�Ի���̿��������ѧ��Ϊ�������[14]���õ����ϵ���ֱ�Ϊ0.984 2��0996 3��������Ͻ������1��
��1 ����̿�������ӵĶ���ѧ����
Table 1 Kinetic parameters of phenol adsorption on activated carbon

Qe: equilibrium adsorption capacity with experimentally measured, mg/g; qe: equilibrium adsorption capacity calculated with kinetic equation, mg/g; t: reaction time, min; k1: adsorption constant of pseudo-first-order equation; k2: adsorption constant of pseudo-second-order equation.

ͼ1 ����̿������������ʱ��Ĺ�ϵ
Fig. 1 Relationship between adsorption capacity and reaction time
�ɱ�1����������һ��������������ѧ���̼����ƽ���������ֱ�Ϊ105.36��127.55 mg/g��ʵ���õ�ƽ��������Ϊ120.96 mg/g����������ѧ���̼������ƽ���������ӽ�ʵ��ֵ������ϵ����ϵ�����ߡ����ԣ���������ѧ���̸��ʺ���������̿�������ӵ���������ѧ����������̿�������ӵĹ���Ϊ��ѧ������
2.2 ����������
�ڲ�ͬ�ı��ӳ�ʼŨ���£�Ͷ��0.5 g����̿������ƽ��ʱ��Ϊ120 min��ʵ������ⶨƽ��Ũ�ȡ��ڲ�ͬ�¶��½�������ʵ��õ���������������ͼ2 ��ʾ��

ͼ2 ����̿��������ƽ��Ũ�ȵĹ�ϵ
Fig. 2 Relationship between adsorption capacity and equilibrium concentration
��Clausius-clapyron����ʽ�Ƶ��ɵ�
 (1)
(1)
ʽ�У�QΪ�����ȣ�kJ/mol��RΪĦ�����峣����8.314 J/(mol��K)��T1��T2�ֱ�Ϊ��Ӧ�¶ȣ�K��c1��c2�ֱ�Ϊ�¶�T1��T2ʱ��Ӧ��ƽ��Ũ�ȣ�mol/L��
����������ͬ�¶�������ƽ��Ũ�ȿɽ�����������ȡ�����ͨ��Ϊ���ȹ��̣�����ͼ2�ṩ�����ݼ�ʽ(1)���������Ϊ-23.89 kJ/mol�������Ƚ�С���������������ͻ�ѧ����֮�䣬�Ʋ����Ϊ���������
2.3 ����λ
������������ڻ���̿���ӱ��������λ�� ��[15-16]������ƽ��ʱӦ��ѭ���·���ʽ��
 (2)
(2)
ʽ�У�bΪ����ƽ�ⳣ�����ֳ�����ϵ����cΪ��ͬ��ʼŨ��ʱ��ƽ��Ũ�ȣ�mol/L�� Ϊ����λ���Ƕȣ������ò�ͬ��ʼŨ��ʱ��ƽ��������q�뱥��������qm�ı�ֵ��ʾ����
Ϊ����λ���Ƕȣ������ò�ͬ��ʼŨ��ʱ��ƽ��������q�뱥��������qm�ı�ֵ��ʾ���� ��
��
������ʽ(2)ת��Ϊ���Է���ʽ��
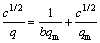 (3)
(3)
��c1/2/q��c1/2��ͼ��һֱ�ߣ������ͼ3��ʾ����ֱ��б�ʵ�qmΪ0.276 1 g/g��������ϵ��Ϊ 0.993 1��������������������ڻ���̿��ʱ���������Ե�ɼ���

ͼ3 c1/2/q��c1/2�Ĺ�ϵ
Fig.3 Relationship between c1/2/q and c1/2
2.4 �����ڻ���̿�ϵ�������̬
2.4.1 �������
Ϊ�˲��������ڻ���̿�ϵ�������̬���ⶨ��ͬ����Ũ���»���̿�������Ӻ�ĺ������ͼ����ͼ4��ʾ��ͼ4��������������1~4�ֱ�Ϊ����̿������Ũ��Ϊ0.65 g/L���ӣ�Ũ��Ϊ1.95 g/L���Ӽ�Ũ��Ϊ6.5 g/L���ӵĻ���̿������ס����ű��������������ӣ��ǻ����մ��ɻ���̿�õ��ļ�������������ɢ�Ŀ��ס�����1 000 cm-1�����մ���λ��û��ƫ�ƣ���֪�����ڻ���̿��������ã���˿�����Ϊ����̿��������ʱ�DZ��ӵ��ǻ������̿�ı��淢�����á�

ͼ4 ����̿�ĺ������
Fig.4 IR spectra of activated carbon: 1��Activated carbon; 2��0.65 g/L phenol+activated carbon; 3��1.95 g/L phenol+ activated carbon; 4��6.5 g/L phenol+activated carbon
2.4.2 ����̿�ǻ����γ�
�ɻ���̿�ĺ������ͼ�������ڲ���Ϊ3 432 cm-1ʱ���ֺ�ǿ��O��H��������������̿�����ǻ������������Ʋ����̿�����ǻ��γɡ�����̿����ʯī���ӽṹ���ɴ�����������ɣ�̼������sp2�ӻ�����γɦļ��Ǽܣ����е�һ��δ����sp2�ӻ������O��H������ԭ��һ��sp3����ɼ����γɻ���̿�����ϵ��ǻ���ʣ�µ�����sp3�ӻ�������й¶Ե��ӡ����ռۼ����ۣ����Ľṹʽ��ʾΪ
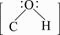 (4)
(4)
2.4.3 �����ڻ���̿�ϵ�������ѧ���ṹʽ
�ڱ��ӷ����У���ԭ�ӵļ۵�������sp2�ӻ��������ɼ������ǻ�����ԭ���ϵ�һ��δ���õ��Ӷ�����p����뱽�ӵ�6��̼ԭ�ӵ�p���ƽ���γ�����м���sp2�ӻ����������һ��sp2��������C�ɼ�����һ��sp2�����Hԭ�ӳɼ���������sp2�������δ�õŶԵ��ӣ����ӷ��ӽṹ�ɱ�ʾΪ
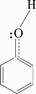 (5)
(5)
��ʽ(4)~(5)��֪������̿���ǻ���ԭ��sp3����д��ڵŶԵ��ӣ����뱽�����ǻ��е����γ���������������ڻ���̿�ϵĽṹΪ
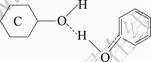 (6)
(6)
2.4.4 �����ڻ���̿�ϵ�������̬
�ɵ��·�����õ�������Ϊ-33.046 kJ/mol��Ҳ֤����������������ڻ���̿��[17]��ʽ(5)�����������ǻ�����һ��sp3�����һ��δ�õŶԵ��ӣ�ͨ�����γ������ʽ(6)��ʾ��ͼ5��ʾΪ�����ڻ���̿�ϵ�����̬ʾ��ͼ����ͼ5����������̿�е��ǻ�ʹ�����ı��ӷ����ϣ������ű���Ũ�ȵ����ӣ�ʹ�ǻ�����ԭ�������Թ¶Ե��ӣ�����һ�Կ�������һ���������ӵ��ǻ���ԭ�����γ��������ͨ���������᷽ʽʹ�����ڻ���̿�����Ϸ����ϡ���ͽ����˺�������������̿�������������������ӣ��ǻ������������������ɢ�Ŀ��ס�ʽ(5)�б����ڻ���̿��������ͨ��������������ڻ���̿�����ǻ�����ԭ���ϣ�����λΪ2����������������ʵ�ʽ��һ�¡�

ͼ5 �����ڻ���̿�ϵ�����̬ʾ��ͼ
Fig. 5 Structure of phenol adsorbed on activated carbon
2.5 ����ģ�Ͷ�ʵ������Ľ���
2.5.1 �¶ȶԻ���̿�������ӵ�Ӱ��
�ڻ���̿Ͷ����Ϊ0.5 g������ʱ��Ϊ120 min������Ũ��Ϊ1.34 g/L��pHֵΪ6ʱ�����¶ȴ�10 �����ӵ�70 ��ʱ���õ����������¶ȵĹ�ϵ��ͼ6��ʾ��

ͼ6 �¶ȶԻ���̿�������ӵ�Ӱ��
Fig.6 Effect of temperature on phenol adsorption on activated carbon
��ͼ6�ɿ��������¶�С��20 ��ʱ������̿�������ӵ������������¶ȵ����߶����ߣ����¶�Ϊ20 ��ʱ���õ�����������0.163 g/g�����¶ȴ���20��ʱ������̿�������������¶ȵ����߶����ͣ����¶ȴﵽ70 ��ʱ������̿�Ա��ӵ�������ֻ��0.1474 g/g����������Ϊ���ȷ�Ӧ���������¶ȣ�����Ч����
2.5.2 ��ʼpHֵ�Ի���̿�������ӵ�Ӱ��
���¶�20 �棬����ʱ��Ϊ120 min������Ũ��Ϊ1.34 g/L������̿Ͷ����Ϊ0.5 gʱ���ı�pHֵ��2��12���õ���ʼpHֵ�Ի���̿�������ӵ�Ӱ����ͼ7��ʾ��

ͼ7 ��ʼpHֵ�Ի���̿�������ӵ�Ӱ��
Fig.7 Effect of initial pH value on phenol adsorption on activated carbon
��ͼ7������������̿��pHֵΪ6~7ʱ������̿�Ա�������������������pHֵ���ڻ�С��6~7ʱ������̿�������ӵ����ܽ��ܵ��ܴ��Ӱ�졣���������£�����̿�������ӵ������������ͣ���Ӱ�첻���ԡ�����pHֵ����8ʱ������̿�����������½����ԣ���pHֵ����12ʱ������̿�������ܼ�����ȫɥʧ��������ֻ��0.04 g/g����ʽ(5)���������ǻ�����ԭ���ϵ�һ��δ���õ��Ӷ����ڵ�p����뱽��������̼ԭ��ƽ�У���ɹ�����ϵ��ʹ��ԭ�ӵĸ��������������ԭ���ϵĵ������ܶȣ��Ӷ�����O��H�������̿�γɵ������������OH-Ũ�ȵ����ӣ��ᷢ�����·�Ӧ��
 (7)
(7)
��OH-�ﵽһ���̶Ⱥ����ȫ�����ѣ��ʻ���̿ʧȥ�������ܡ�
3 ����
1) ����̿�Ա��ӵ��������ض�������ѧ���̣����������Ϊ0.2761 g/g��
2) ����̿�������ӷ����ʸ��Ѷ�����ʽ��������һ�����ӷ����������������Ե��ϡ�ʵ�ʲ�õ�������Ϊ-33.046 kJ/mol��������������ǿ�Ƚ�С���Ʋⱽ��ͨ����������ڻ���̿�ϡ�
3) �������ʵ����������ű���Ũ�����ӣ����������3 432 cm-1���ǻ������ɼ������������ɢ���ף��Ʋⱽ��ͨ����������ϡ�
4) ����˻���̿�������ӵ�ģ�ͣ���Բ����������ʵ����й�����
REFERENCES
[1] DOS SANTOS V L, MONTEIRO A S, BRAGA D T, SANTORO M M. Phenol degradation by Aureobasidium pullulans FE13 isolated from industrial effluents[J]. Journal of Hazardous Materials, 2009, 161(2/3): 1413-1420.
[2] LIN K L, PAN J Y, CHEN Y W, CHENG R M, XU X C. Study the adsorption of phenol from aqueous solution on hydroxyapatite nanopowders[J]. Journal of Hazardous Materials, 2009, 161(1): 231-240.
[3] ������, ������, ������, Ѧȫ��. ë�����̿�Ʊ�����Ժ����ӷ�ˮ�������о�[J]. ��У��ѧ����ѧ��, 2010, 24(4): 700-704.
WANG Gui-zhen, LI Li-xin, LI Yong-zhen, XUE Quan-min. Study on the preparation of bamboo activated carbon and its phenol adsorption properties[J]. Journal of Chemical Engineering of Chinese Universities, 2010, 24(4): 700-704.
[4] ������, �� ��, �� ǿ, ��ͮ. �Ǿ��Ǹ��Է�ʯ������ˮ�еı���[J]. ��̬����, 2008, 17(6): 2168-2172.
LI Zeng-xin, MENG Yun, LIANG Qiang, WANG Tong. Adsorption of phenol in wastewater with modified zeolite-chitosan adsorbent[J]. Ecology and Environment, 2008, 17(6): 2168-2172.
[5] MA J F, ZHU L Z. Removal of phenols from water accompanied with synthesis of organobentonite in one-step process[J]. Chemosphere, 2007, 68(10): 1883-1888.
[6] JUANG R S, SHIAN J Y. Adsorption of phenol from water onto macrovoreticularresins[J]. Journal of Hazardous Materials, 1999, B(70): 171-183.
[7] �� ܿ, ����Ƽ, ������. ������������ֵ�Ա��ӵ���������ѧ[J]. ����ʦ����ѧ��Ȼ��ѧѧ��, 2007, 30(4): 85-89.
ZHOU Yun, HU Li-ping, XU Man-cai. Adsorption thermodynamics of phenol onto polymeric adsorbents with amide groups[J]. Journal of Natural Science of Hunan Normal University, 2007, 30(4): 85-89.
[8] �� �, ��С��, ����Ԫ, ������, �� ��, �� �, Ԭ ��. ��������̿�������Ƶ�������������[J]. �й���ɫ����ѧ��, 2007, 17(6): 1008-1013.
WANG Pu, MIN Xiao-bo, CHAI Li-yuan, WANG Yun-yan, FANG Yan, CHANG Hao, YUAN Lin. Adsorption of sodium lactate on modified active carbon and its characteristics[J]. The Chinese Journal of Nonferrous Metals, 2007, 17(6): 1008-1013.
[9] RADOVIC L R, MORENO-CASTILLA C, RIVERA-UTRILLA J. Carbon materials as adsorbents in aqueous solutions[J]. Chemistry and Physics of Carbon, 2000, 27: 227.
[10] ROOSTAEI N, TEZEL F H. Removal of phenol from aqueous solutions by adsorption[J]. Journal of Environmental Management, 2004, 70(2): 157-164.
[11] VIRARAGHA V T. Removal of phenol from wastewater by adsorption on peat��fly ash and bentonite[J]. Environment Engineering, 1992, 5: 311-317.
[12] ղ ��, ��·��, �� ˧, �� ��. ����̿�Իӷ��ӵ���������[J]. �Ƽ�����, 2010, 28(24): 44-47.
ZHAN Xu, ZOU Lu-yi, SHAO Shuai, LI Jing. Absorption characteristics of activated carbon on volatile phenol[J]. Science & Technology Review, 2010, 28(24): 44-47.
[13] BUSCA G, BERARDINELLI S, RESINI C, ARRIGHI L. Technologies for the removal of phenol from fluid streams: A short review of recent developments[J]. Journal of Hazardous Materials, 2008, 160(2/3): 265-288.
[14] HAMEED B H, AHMAD A A, AZIZ N. Isotherms kinetics and thermodynamics of acid dye adsorption on activated palm ash[J]. Chemical Engineering Journal, 2007, 133: 195-203.
[15] �Ž���, ������, �� �|, ��Ⱥ��. �����ڻ���̿������ƽ��ģ�͵��о�[J]. ��ѧ��ҵ�빤��, 2001(6): 341-345.
ZHANG Jin-li, JI Xiu-ling, LI Wei, HUANG Qun-wu. Study on adsorption in activated carbon[J]. Chemical Industry & Engineering, 2001(6): 341-345.
[16] ����ΰ, ������. ��̿�Ա�������������ѧ������ѧ�������о�[J]. �����ʻ�ѧ������, 2008, 42(2): 19-22.
ZHANG Qi-wei, WANG Gui-xian. Study on the kinetic and the thermodynamic parameters for adsorption of phenol by bamboo-charcoal[J]. Biomass Chemical Engineering, 2008, 42(2): 19-22.
[17] �� ��. ��������о�����[M]. ����: ��ѧ������, 2004: 364-367.
XIN Qin. Methods for the investigation of solid catalysts[M]. Beijing: Science Press, 2004: 364-367.
(�༭ ����)