Hydrogen absorption and desorption in metallic glass andnanocrystalline Zr52.5Cu17.9Ni14.6Ti5Al10 alloy
来源期刊:中国有色金属学报(英文版)2009年第2期
论文作者:程晓英 王 芳
文章页码:377 - 382
Key words:metallic glasses; nanocrystals; catalysis; hydrogen storage
Abstract: To illuminate the intrinsic surface activity of Zr52.5Cu17.9Ni14.6Ti5Al10 alloy in its glass and nanocrystalline states, hydrogen absorption and desorption in both states was investigated by gas chromatographic analysis. The results show that the Zr52.5Cu17.9Ni14.6Ti5Al10 alloy in the nanocrystalline state can absorb a larger amount of hydrogen than that in glass state at room temperature after activation. According to the desorption process and surface state, the significant change in absorption induced by crystallization is proposed to result from that the glassy alloy has a higher desorption energy, which can adsorb gas physically and nonselectively, and is difficult to activate, while the nanocrystalline alloy can absorb much hydrogen due to the inter-atomic or intra-atomic electron transfer, which accelerates the kinetics of the catalytic reaction for the dissociation of molecular hydrogen into atomic hydrogen.
基金信息:the Science and Technology Committee of Shanghai, China
the National Natural Science Foundation of China
Shanghai Leading Academic Discipline, China
by the Natural Science Foundation of Shanghai, China

CHENG Xiao-ying(程晓英), WANG Fang(王 芳)
Institute of Materials Science, Shanghai University, Shanghai 200072, China
Received 5 May 2008; accepted 24 October 2008
Abstract: To illuminate the intrinsic surface activity of Zr52.5Cu17.9Ni14.6Ti5Al10 alloy in its glass and nanocrystalline states, hydrogen absorption and desorption in both states was investigated by gas chromatographic analysis. The results show that the Zr52.5Cu17.9Ni14.6Ti5Al10 alloy in the nanocrystalline state can absorb a larger amount of hydrogen than that in glass state at room temperature after activation. According to the desorption process and surface state, the significant change in absorption induced by crystallization is proposed to result from that the glassy alloy has a higher desorption energy, which can adsorb gas physically and nonselectively, and is difficult to activate, while the nanocrystalline alloy can absorb much hydrogen due to the inter-atomic or intra-atomic electron transfer, which accelerates the kinetics of the catalytic reaction for the dissociation of molecular hydrogen into atomic hydrogen.
Key words: metallic glasses; nanocrystals; catalysis; hydrogen storage
1 Introduction
The intrinsic surface activity of Zr-based metallic glasses is most important for the study of their oxidation rule[1-2] and their potential application, e.g. as penetrators[3-6], as materials for hydrogen or deuterium storage[7], or as catalysts[8-9]. The oxidation of Zr-based metallic glasses and nanocrystalline alloys shows that the nanocrystalline alloys are better in oxidation resistance, as compared with their glassy state [1-2]. The possible reason is that the glassy state possesses a higher free energy and therefore also a larger driving force for oxidation. However, in many hydrogen storage alloys, their hydrogen storage capacity in the amorphous state is lower than that in the crystalline state; and in many cases, the hydrogen absorption rate is dozens of times higher in the crystalline state than that in the amorphous state[10-11]. From the free energy consideration, it is not easy to explain the large difference between the two states. On the other hand, a key difference between the two states in hydrogen storage properties is the activation, that is, the amorphous alloy is more difficult to activate. Additional investigation is thus required to clarify the difference in surface activity through hydrogen absorption between metallic glasses and nanocrystalline alloys.
Zr-Ti-Cu-Ni alloys containing early transition metals(ETM) and later transition metals(LTM) are among the best glass-forming alloys[12-13]. For example, the metallic glass Zr52.5Cu17.9Ni14.6Ti5Al10 alloy has been investigated widely, with its glass transition temperature Tg=683 K (400 ℃), and the first crystalliza- tion temperature Tx1=713 K (440 ℃), based on the DSC study at a heating rate of 10 K/min[14]. These features make them good candidate materials for hydrogen storage. Generally speaking, the hydrogenation of hydrogen storage materials includes the following stages: 1) molecular hydrogen physisorption on the metal surface, 2) dissociation of the molecules, 3) adatom migration and chemisorption, 4) diffusion of the hydrogen through the hydride layer being formed, and 5) chemical reaction at the hydride/alloy interface. The former three stages are referable to the surface reaction and not associated with the bulk factor. Also, the former three stages are directly related to the activating process. The dynamic pulse method is a useful way to measure surface activity in situ. In this work, the intrinsic surface activity of Zr52.5Cu17.9Ni14.6Ti5Al10 alloy in its glass and nanocrystalline states was illuminated.
2 Experimental
Ingots of Zr52.5Cu17.9Ni14.6Ti5Al10 (mole fraction, %) were prepared by arc melting the constituent elements on a water-cooled copper hearth in a titanium gettered argon atmosphere and casting into a copper mold with a diameter of 7 mm. Both XRD and metallographic examination indicated that the alloy ingot was in the full glass state. For obtaining nanocrystalline alloy, the cast alloy was heat treated at 500 ℃ for 8 h. The powder samples of metallic glass and nanocrystalline alloy were filed from the corresponding bulk materials at ambient atmosphere just before testing in gas chromatograph in order to lessen surface oxidation. The morphology of the powder samples was studied by optical microscopy, and the structure of the powder samples before or after test was detected by XRD with a Cu Kα radiation.
The hydrogen absorption test was conducted by a dynamic pulse method[15] and the amount of hydrogen absorption V(H2) (mL/g) was calculated as following:
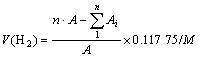 (1)
(1)
where n is the count of H2 uptake pulse, A is the area under the recorded saturation peak, Ai is the area under the recorded ith peak and M is the mass of the sample (g).
Some samples absorbed hydrogen during purging with Ar+5%H2 and heating at a programmed temperature. To study the activation process, the desorption process was recorded during purging with argon and heating at a programmed temperature.
3 Results
Figs.1(a) and (b) show the X-ray diffraction patterns of the samples just after crushing from the bulk alloys. As expected, the cast alloy is glassy, while the heat-treated alloy is in nanocrystalline structure[16-17]. Since the phase separation process is only dominant in the early stages of annealing[17], the morphology of the peaks arising from the nanocrystalline phases does not change a lot after the test of absorption hydrogen during purging with Ar+5%H2 and heating at a programmed temperature (Figs.1(c) and (d)). However, the positions of the peaks shift to lower angle due to solution hydrogen after the test.

Fig.1 XRD patterns of samples: (a) metallic glass and (b) nanocrystalline counterpart just after crush; (c) metallic glass and (d) nanocrystalline counterpart after testing in Ar+5%H2 (Solid circles indicate peaks from fcc nanocrystalline phase, open squares indicate quasicrystalline phase[17])
Fig.2 shows the morphologies of metallic glass and nanocrystalline powders after the test of hydrogen absorption during purging with Ar+5%H2 and heating at a programmed temperature. It exhibits similar in size and shape between the metallic glass and the nanocrystalline samples, indicating that both powder samples have the same specific surface. However, a slight devitrified surface layer is observed in metallic glass sample, while no obvious devitrified surface layer is revealed in nanocrystalline sample after test.

Fig.2 Morphologies of metallic glass (a) and nanocrystalline counterpart (b) after test of hydrogen absorption during purging with Ar+5%H2 and heating at programmed temperature
Fig.3 shows the thermal desorption spectra(TDS) for the activation of both samples heated at a rate of 20 K/min in purged argon. It can be clearly seen that the area under the desorption peaks of metallic glass is larger than that of nanocrystalline one. Since these samples do not take any absorption test except the exposure to air before the activation test, the amount of adhesive gas is larger on the surface of the glassy powder than that on the surface of the nanocrystalline powder. In addition, the metallic glass needs to heat to about 550 ℃ in order to desorb gas completely, while the nanocrystalline one desorbs gas completely only after heating to 450 ℃. Because Tx1 of the metallic glass Zr52.5Cu17.9Ni14.6Ti5Al10 is 440 ℃[14], the activation temperature for the metallic glass is chosen to be 400 ℃.

Fig.3 Thermal desorption spectra of Zr52.5Cu17.9Ni14.6Ti5Al10 alloy: (a) Metallic glass; (b) Nanocrystalline counterpart
After activation, the hydrogen absorption was recorded (in Fig.4) using Ar as carrier gas and a number of H2 pulse injected into the flow reactor[15]. It is obvious that in the nanocrystalline state the alloy absorbs a larger amount of hydrogen than that in the metallic glass state. The amount of the absorbed hydrogen is 0.744 mL/g for the nanocrystalline state, which is about 30 times larger than that for the metallic glass state (0.022 mL/g).
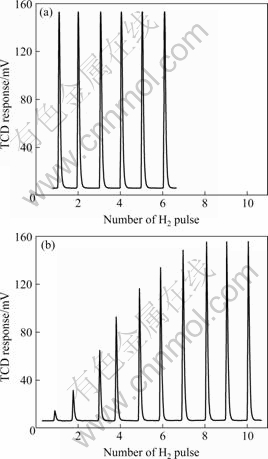
Fig.4 Progress of H2 absorption at room temperature for glassy (a) and nanocrystalline counterpart (b) after activation
Figs.5 and 6 show the hydrogen absorption curves obtained during purging with Ar+5%H2 and heating at a programmed temperature. The hydrogen absorption peaks are downwards the baseline, while the gas desorption peaks are upwards the baseline. If the samples are not activated, the curves show a desorbing gas peak besides the absorbing hydrogen peaks (see Fig.5). Fig.5(b) is the amplified morphology of Fig.5(a). The desorbed peak for the metallic glass is much higher than that for the nanocrystalline one, which is consistent with the previous results shown in Fig.3. In addition, after the gas is desorbed, the glassy alloy can first absorb hydrogen faster than the nanocrystalline alloy between 405 ℃ and 454 ℃, i.e. between Tg and Tx. The behavior in this stage is some ambiguous. More investigation is in progress. However, above Tx, the metallic glass begins to crystallize and in the mean time can absorb hydrogen as same as the nanocrystalline alloy does. If the samples are activated after heating to 400 ℃, the curves only show the peaks of absorbing hydrogen (Fig.6). The metallic glass also shows a retarded absorption until Tx. The total amount of hydrogen integrated through the absorption peaks per gram is same between metallic glass and nanocrystalline alloy.
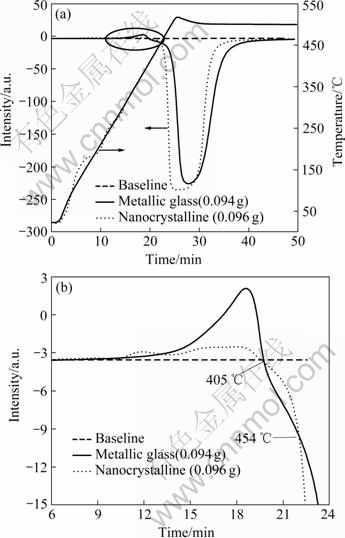
Fig.5 Absorbing hydrogen curves during purging with Ar+5%H2 and heating at programmed temperature without activation (Fig.5(b) is amplification of ellipse in Fig.5(a))

Fig.6 Absorbing hydrogen curves during purging with Ar+5%H2 and heating at programmed temperature after activation
4 Discussion
Chemisorption on solid metal surfaces involves a number of reaction steps occurring in a sequential manner, such as molecular gas physisorption on the metal surface, dissociation of the molecule, adatom migration and chemisorption, and solution. There are two factors affecting the chemisorption: structural and electronic[18]. However, the structural factor is expected to be more remarkable than the electronic factor in the first step, i.e. molecular gas physisorption on the metal surface. For example, the desorption energy of molecules adsorbed on step site of kinked surfaces, such as a Pt (557) surface in fcc, is much larger than that for the siteson a uniform surface, such as a Pt(111) surface in fcc [19]. The desorption energy of the sites on lattice imperfections of surfaces also becomes larger[19]. The step sites of kinked surfaces or the sites on lattice imperfections of surfaces are extensively reactive in adsorbing molecular gas[18]. The physisorption is nonselective and commonly weak. On the contrary, the electronic factor is thought to be the most important one in the dissociation of the molecule[20]. The dissociative adsorption is selective and commonly strong. According to theory and experimental data on single component systems, the existence of d holes (a high d type local density of states) at the Fermi energy is essential in the dissociative adsorption process of molecular hydrogen [21]. As an H2 molecule approaches a metal surface, its 1 σg orbital feels Pauli repulsion from the tails of the s electrons in the metal. If the metal has d holes, an s electron can be promoted to the d band, reducing the Pauli repulsion, and thereby reducing the subsequent energy barrier[22]. In addition, to dissociate H2 molecule, the adsorbate orbitals should be split by strong interactions with the sharper d-band. This implies that the outer s electron should be less and d-band should be nearly full. As expected, transition metals, like Ni and Pd, readily dissociate and adsorb hydrogen, while for noble or simple metals, like Cu or Al, H2 dissociation is an activated process[23].
It is well known that the difference between metallic glass and nanocrystalline states is obvious. The surface structure of metallic glass is fully disordered, while the nanocrystalline one is ordered. From the consideration of adhesive energy, the desorption energy of molecules adsorbed on the metallic glass surface is much higher than that for the nanocrystalline surface. Therefore, the amount of adhesive gas on the surface of glassy powder is much lager than that on the surface of nanocrystalline powder, and the end temperature of glassy powder desorption is higher than that of nanocrystalline powder, as shown in Fig.3 and Fig.5. Hence, the physisorption on the surface of metallic glass seems strong. Since the powders were filed from the bulk materials and inevitably exposed to air, the adhesive gas may be mainly oxygen. In addition, slight oxidization is observed after heating, from the morphologies (Fig.2). However, as shown in Fig.6, even after heating to 400 ℃, in order to desorb molecular gas, the metallic glass also shows a retarded absorption until Tx. The activity of hydrogen absorption for metallic glass is inert compared with that for nanocrystalline one.
In comparing the hydrogen properties of amorphous glass and its crystalline counterpart, many researches [24-27] did not mention the gas adsorption process prior to hydrogenation. However, a phenomenon similar as our results revealed in the hydrogen adsorption by milled C60 [28]. The more this amorphous phase exists in the sample, the more amount of hydrogen adsorption is observed.
The hydrogenation in amorphous and crystalline alloys has been widely investigated[29-31]. However, the conclusions differ greatly. For example, the hydrogenation in benzene, amorphous Ni-B has little active center due to the disorder array of Ni and B though it has larger specific surface area, while the nanocrystalline Ni3B benefits to hydrogenation due to the definite surface array and electron transfer between Ni and B[29-30]. Our results also show that the amount of absorption hydrogen for the nanocrystalline powder is 30 times larger than that for the metallic glass, as shown in Fig.4. This phenomenon is also consistent with our previous work that the atomic ordering can accelerate the catalytic activity to dissociate molecular hydrogen into atomic hydrogen and therefore promote hydrogen absorption due to the large number of electrons in d band of Ni[32-34]. The electronic structure of this alloy in both metallic glass and nanocrystalline states should be investigated further. Since the study of amorphous alloys as catalysts[35], many amorphous alloy catalysts have been developed, especially for Ni-based amorphous alloys[36]. They proposed that the amorphous alloys exhibit excellent activity due to the fine particle, high free energy and longer distance in Ni―Ni bond. But comparing the intrinsic catalytic activity between amorphous and crystalline alloys, the condition should be same. We rule out these factors such as powder size and suggest that the nanocrystalline alloy is more active than metallic glass in absorption hydrogen after activation.
5 Conclusions
1) The Zr52.5Cu17.9Ni14.6Ti5Al10 alloy in the nanocrystalline state can absorb a larger amount of hydrogen than that in glass state at room temperature after activation.
2) The glassy alloy can adsorb gas physically and nonselectively, and therefore it is difficult to activate.
3) The nanocrystalline alloy can adsorb less gas physically, but can absorb much hydrogen due to the inter-atomic or intra-atomic electron transfer, which accelerates the kinetics of catalytic reaction for the dissociation of molecular hydrogen into atomic hydrogen.
Acknowledgement
The authors thank Prof. C.T. Liu of Oak Ridge National Laboratory, USA, for providing the material.
References
[1] K?STER U, JASTROW L. Oxidation of Zr-based metallic glasses and nanocrystalline alloys [J]. Mater Sci Eng A, 2007, 449/451: 57-62.
[2] WU Y, NAGASE T, UMAKOSHI Y. Effect of crystallization behavior on the oxidation resistance of a Zr-Al-Cu metallic glass below the crystallization temperature [J]. J Non-Cryst Solids, 2006, 352: 3015-3026.
[3] ITOH N, XU W C, HARA S, KIMURA H M, MASUMOTO T. Permeability of hydrogen in amorphous Pd1-xSix alloys at elevated temperatures [J]. J Membr Sci, 1998, 139: 29-35.
[4] DOLAN M D, DAVE N C, ILYUSHECHKIN A Y, MORPETH L D, MCLENNAN K G. Composition and operation of hydrogen-selective amorphous alloy membranes [J]. J Membr Sci, 2006, 285: 30-55.
[5] KIM K B, KIM K D, LEE D Y, KIM Y C, FLEURY E, KIM D H. Hydrogen permeation properties of Pd-coated Ni60Nb30Ta10 amorphous alloy membrane [J]. Mater Sci Eng A, 2007, 449/451: 934-936.
[6] HARA S, SAKAKI K, ITOH N, KIMURA H M, ASAMI K, INOUE A. An amorphous alloy membrane without noble metals for gaseous hydrogen separation [J]. J Membr Sci, 2000, 164: 289-294.
[7] ZANDER D. Hydrogen in metastable Zr-Cu-Ni-Al alloys [D]. University of Dortmund, Berlin: Logos Verlag, 2001.
[8] ZANDER D, TAL-GUTELMACHER E, JASTROW L, K?STER U, ELIEAER D. Hydrogenation of Pd-coated Zr-Cu-Ni-Al metallic glasses and quasicrystals [J]. J Alloys Comp, 2003, 356/357: 654-657.
[9] SCHL?GL R, LOOSE G, WESEMANN M, BAIKER A. Oxidation of carbon monoxide over palladium on zirconia prepared from amorphous Pd-Zr alloy (Ⅱ): The nature of the active surface [J]. J Catal, 1992, 137: 139-157.
[10] TANAKA K, SOWA M, KITA Y, KUBOTA T, TANAKA N. Hydrogen storage properties of amorphous and nanocrystalline Zr-Ni-V alloys [J]. J Alloys Comp, 2002, 330/332: 732-737.
[11] LI X G, OTAHARA T, TAKAHASHI S, SHOJI T, KIMURA H M, INOUE A. Hydrogen absorption and corresponding changes in structure and thermal stability of Zr60Al10Ni30 amorphous alloy [J]. J Alloys Comp, 2000, 297: 303-311.
[12] PETER A, JOHNSON W L. A highly processable metallic glass Zr41.2Ti13.8Cu12.5Ni10.0Be22.5 [J]. Appl Phys Lett, 1993, 63: 2342- 2344.
[13] LIN X H, JOHNSON W L, RHIM W K. Effect of oxygen impurity on crystallization of an undercooled bulk glass forming Zr-Ti-Cu-Ni-Al alloy [J]. Mater Trans JIM, 1997,38: 473-477.
[14] PEKARSKAYA E, L?FFLER J F, JOHNSON W L. Microstructural studies of crystallization of a Zr-based bulk metallic glass [J]. Acta Mater, 2003, 51: 4045-4057.
[15] WAN X J, CHEN Y X, CHEN A P, YAN S R. The influence of atomic order on H2-induced environmental embrittlement of Ni3Fe intermetallics [J]. Intermetallics, 2005, 13: 454-459.
[16] MULAS G, SCUDINO S, COCCO G. H2 absorption in amorphous and nanostructured Zr-based alloys under milling [J]. Mater Sci Eng A, 2004, 375/377: 961-964.
[17] PEKARSKAYA E, L?FFLER J F, JOHNSON W L. Microstructural studies of crystallization of a Zr-based bulk metallic glass [J]. Acta Mater, 2003, 51: 4045-4057.
[18] NONOSE S, SONE Y, ONODERA K, SUDO S, KAYA K. Reactivity study of alloy clusters made of aluminum and some transition metals with hydrogen [J]. Chemical Physics Letters, 1989, 164: 427-432.
[19] KOEL B E, SOMORJAI G A. Surface structural chemistry [M]// ANDERSON J R, BUDART M. Catalysis: Science and Technology. Berlin: Springer, 1985.
[20] HAMMER B, N?RSKOV J K. Theretical surface science and catalysis-calculations and concepts [J]. Adv Catal, 2000, 45: 71-129.
[21] HARRIS J, ANDERSSON S. H2 dissociation of metal surface [J]. Phys Rev Lett, 1985, 55: 1583-1586.
[22] HANBICKI A T, BADDORF A P, PLUMMER E W, HAMMER B, SCHEFFLER M. Interaction of hydrogen with the (110) surface of NiAl [J]. Surf Sci, 1995, 331/333: 811-817.
[23] CHRISTMANN K. Interaction of hydrogen with solid surfaces [J]. Surf Sci Rep, 1988, 9(1/3): 1-163.
[24] TANAKA K, ARAKI T, ABE T, YOSHINARI O. Thermal desorption spectra of hydrogen from glassy Pd35Zr65 and crystalline PdZr2 [J]. J Less-Common Metals, 1991, 172: 928-935.
[25] ZALUSKA L, ZALUSKA A, TESSIER P, STR?M-OLSEN J O, SCHULZ R. Effects of relaxation on hydrogen absorption in Fe-Ti produced by ball-milling [J]. J Alloys Comp, 1995, 227: 53-57.
[26] ZALUSKA L, ZALUSKA A, STR?M-OLSEN J O. Nanocrystalline metal hydrides [J]. J Alloys Comp, 1997, 253/254: 70-79.
[27] ZALUSKA A, ZALUSKA L, STR?M-OLSEN J O. Nanocrystalline magnesium for hydrogen storage [J]. J Alloys Comp, 1999, 288: 217-225.
[28] TALYZIN A V, JACOB A. Hydrogen adsorption by ball milled C60 [J]. J Alloys Comp, 2005, 395: 154-158.
[29] JIANG Zheng, YANG Hong-wei, WEI Zheng, XIE Zhi, ZHONG Wen-jie, WEI Shi-qiang. Catalytic properties and structures of nano-amorphous Ni-B alloys affected by annealing temperatures [J]. Applied Catalysis A: General, 2005, 279: 165-171.
[30] WEI Shi-qiang, LI Zhong-rui, ZHANG Xin-yi, CHEN Chang-rong, LIU Wen-han, HU Tian-dou. The anneal crystallization and its catalytic properties of ultrafine Ni-B amorphous alloy [J]. Sci Lett, 2000, 45: 1941-1944.
[31] LI Hui, LI He-xing, DENG Jing-fa. The crystallization process of ultrafine Ni-B amorphous alloy [J]. Mater Lett, 2001, 50: 41-46.
[32] CHENG X Y, LIN G W, LI H G. Effect of atomic ordering on the hydrogen-induced environmental embrittlement of Ni4Mo intermetallics [J]. Intermetallics, 2005, 13: 289-294.
[33] CHENG X Y, LIN G W, LI H G. The effect of atomic ordering on the hydrogen absorption and desorption of Ni4Mo alloy [J]. Scripta Mater, 2004, 50: 1293-1296.
[34] CHENG X Y, FENG X Y, LIU J L, WAN X J. The electronic structure and hydrogen gas-induced environmental embrittlement in Ni4Mo alloy [J]. Scripta Mater, 2007, 56(12): 1007-1010.
[35] SMITH G V, BROWER W E, MATYJASZCZYK M S, PETIT T L. Metallic glasses: New catalyst systems [C]// SEIYAM A T, TANA E K. New Horizonsin Catalysis: Proceedings of the Seventh International Congress on Catalysis. Tokyo: Elsevier, 1980: 355-358.
[36] HU Z, HSIA Y, ZHENG J, SHEN J, YAN Q, DAI L. A study of Fe-Ni-B ultrafine alloy particles produced by reduction with borohydride [J]. J Appl Phys, 1991, 70: 436-438.
Foundation item: Project(0452NM002) supported by the Science and Technology Committee of Shanghai, China; Project(50671057) supported by the National Natural Science Foundation of China; Project(T0101) supported by Shanghai Leading Academic Discipline, China; Project(02ZE14033) supported by the Natural Science Foundation of Shanghai, China
Corresponding author: CHENG Xiao-ying; Tel: +86-21-56336532; E-mail: chengxy@staff.shu.edu.cn
DOI: 10.1016/S1003-6326(08)60281-2
(Edited by YUAN Sai-qian)

