J. Cent. South Univ. Technol. (2007)03-0344-04
DOI: 10.1007/s11771-007-0068-2
Synthesis and photophysical and electrochemical properties of
new cyclometalated platinum complex containing oxadiazole ligand
DENG Ji-yong(邓继勇)1,2, LIU Yu(刘 煜)1, HU Zheng-yong(胡峥勇)1,
ZHU Mei-xiang(朱美香)1, ZHU Wei-guo(朱卫国)1
(1. School of Chemistry, Xiangtan University, Xiangtan 411105,China;
2. Department of Chemistry and Engineering, Institute of Hunan Engineering, Xiangtan 411104,China)
Abstract: A new cyclometalated platinum complex containing 2, 5-bis(naphthalene-1-yl)-1,3,4-oxadiazole ligand was synthesized and characterized. The UV-Vis absorptions and photoluminescent properties of the ligand and its platinum complex were investigated. A characteristic metal-ligand charge transfer absorption peak at 439 nm in the UV spectrum and a strong emission peak at 625 nm in the photoluminescence spectrum were observed for this complex in dichloromethane. Cyclic voltammtry (CV) analysis shows that the EHOMO (energy level of the highest occupied molecular orbital) and ELUMO (energy level of the lowest unoccupied molecular orbital) of the platinum complex are about -5.69 and -3.25 eV, respectively, indicating that the oxadiazole-based platinum complex has a potential application in electrophosphorescent devices used as a red-emitting material.
Key words: cyclometalated platinum complex; oxadiazole; synthesis; photoluminescence; electrochemical property
1 Introduction
In the field of optoelectronic materials, a significant research interest has been focused on the design and photophysical properties of the third-row transition metal[1-4] and rare earth metal[5-6] complexes because of their high quantum yields of phosphorescent emission from triplet excited states. One of the most exciting applications of these metal complexes lies in the optoelectronics areas, such as light-emitting devices (LEDs)[7], chemosensors[8], and photovoltaic devices[9]. Among these complexes, cyclometalated platinum (II) complexes with a square- planar d8 electronic configuration, have displayed their potential applications in light-emitting devices[10-12]. Many cyclometalated homo- or hetero-leptic platinum (II) complexes based on different cyclometalated ligands have been developed in order to achieve their better optoelectronic properties. These cyclometalated ligands in platinum complexes reported were mostly phenylpyridine, aryl-2,2′-bipyridine, benzoquinolinate and their derivatives[2, 13-14]. However, their platinum complexes have not displayed satisfactory luminous performance in organic and polymer light-emitting devices. The platinum complexes display low luminous efficiencies compared to those of iridium complexes in the devices[15-16].
It is considered that the low luminous efficiencies of platinum complexes in the device are related to planar configuration of the complexes and unbalanced carrier transport and injection in the devices. KAVITHA et al[17] and COCCHI et al[18] exhibited their successful way to solve the issues by incorporating bulk groups into cyclometalating ligands of platinum complexes. As the bulk groups took an important role in inhibiting molecular aggregation and emission annihilation of platinum complexes, the complexes with bulk groups displayed increased luminous efficiencies in the devices.
In this paper, in order to achieve improved luminous efficiency and hole-electron balanced ability of platinum complex, a new red-emitting platinum complex with oxadiazole ligand was synthesized.
2 Experimental
2.1 Synthetic route of platinum complex
The novel platinum complex containing oxadiazole ligand is Pt[2,5-bis (naphthalene-1-yl)-1,3,4-oxadiazole (C2,N)]acetylacetonate[(DNOXD)P t(acac)]. Its synthetic route is shown in Fig.1.
2.2 Instruments and reagents
All 1H-NMR spectra were acquired on a BRUKER- AV400 NMR Instrument using CDCl3 as solvent. GC-MS data were obtained using a Trace GC- MS-2000 system (Finnigan). UV-Vis absorption spectra were recorded on a PerkinElmer Lambda 25 UV-Vis absorption spectrophotometer. The photo-luminescence spectrum was recorded on a fluorescence spectrophotometer (HITACHI-850). Elemental analysis was performed with a Perkin-Elmer 240 instrument under nitrogen atmosphere. Cyclic voltammtry was carried out on a CHI660A electrochemical workstation in a solution of tetrabutylammonium haxafluorophosphate (Bu4NPF6) (0.1 mol/L) in dichloromethane at a scan rate of 50 mV/s and room temperature under nitrogen purge. A carbon electrode was used as the working electrode, a platinum wire was used as the counter electrode, and a saturated calomel electrode was used as the reference electrode.
All raw materials were analytical reagents. POCl3, SOCl2 and pyridine were treated by distillation before use.
2.3 Synthesis of 5-bis(naphthalene-1-yl)-1,3,4-oxadiazole (DNOXD)
DNOXD was synthesized with a yield of 81.0% according to Ref.[19]. Its melting point was 182-184 ℃. 1H NMR (400 MHz, CDCl3, δ): 9.39 (d, 2H), 8.35 (d, 2H), 8.08 (d, 2H), 7.97 (d, 2H), 7.75 (t, 2H), 7.64 (t, 4H). GC-MS, m/z: 323(M+1, 20.0%), 322(M+, 76.4%), 155 (C10H7CO+, 94.5%), 127(C10H7 +, 100%).
2.4 Synthesis of chloro-bridged dimer Pt2(DNOXD)2- (μ-Cl2)
50 mL three-neck flask was charged with K2PtCl4 (0.232 g, 0.56 mmol), DNOXD (0.36 g, 1.12 mmol), 2-ethoxyethanol (18.0 mL) and water (6.0 mL). The mixture was heated to 80 ℃ and stirred for 36 h under nitrogen atmosphere, and then cooled to room temperature. The formed yellow-green precipitate was collected, washed with 2-ethoxyethanol and n-hexane in turn, and dried in vacuum to afford the (DNOXD)2Pt2- (μ-Cl2). The (DNOXD)2Pt2(μ-Cl2) was used in the next reaction without further purification.
2.5 Synthesis of (DNOXD)Pt(acac)
A mixture of (DNOXD)2Pt2(μ-Cl2) (0.364 g, 0.33 mmol), acetylacetone (82.6 mg, 0.83 mmol), Na2CO3 (0.35 g, 3.3 mmol) and 2-ethoxyethanol (22.0 mL) was heated to 100 ℃ and stirred for 36 h under nitrogen atmosphere. After cooled to room temperature, the mixture presented black precipitate. It was filtered and the precipitate was washed with 2-ethoxyethanol for several times. The yielded filtrate was mixed with water (10.0 mL) to present new yellow precipitate. The precipitate was collected, washed with anhydrous ethanol and n-hexane in turn, dried in vacuum, and then purified by column chromatography over silica using a mixed solvent (volume ratio of dichloromethane to petroleum ether was 3:1) as an eluent to obtain an orange yellow power with a yield of 32.6%. 1H NMR (400 MHz, CDCl3, δ): 9.35 (d, 2H), 8.34 (d, 2H), 8.08 (d, 1H), 8.05 (d, 1H), 7.90(d, 1H), 7.60 (t, 1H), 7.58 (d, 1H), 7.53 (t, 2H), 7.47 (t, 2H), 5.54 (s, 1H), 2.12 (s, 3H), 2.05 (s, 3H). Element analysis results are of PtC27H20N2O3 as follows: calculated (mass fraction, %): C 52.68, H 3.25, N 4.55;found (mass fraction, %): C 52.62, H 3.22, N 4.47.
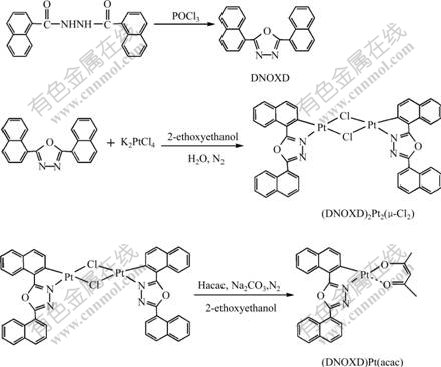
Fig.1 Synthetic route of platinum complex (DNOXD)Pt(acac)
3 Results and discussion
3.1 Synthesis of platinum complex
The platinum complex with 2, 5-bis (naphthalene- 1-yl)-1,3,4-oxadiazole ligand was synthesized with a yield of 32.6% according to Ref.[12]. The chloro-bridged reaction of 2, 5-bis(naphthalene-1-yl)-1,3,4-oxadiazole ligand with K2PtCl4 and debridged reaction of the dimer presented a low yield compared to the similar reactions reported by BROOKS et al[13]. It is considered that the electron-withdrawing ability of the oxadiazole unit in 2,5-bis(naphthalene-1-yl)- 1,3,4- oxadiazole ligand has a negative effect on the chloro-bridged reaction and debridged reaction, which finally results in a low yield of (DNOXD)Pt(acac). Hence, it is necessary to extend the reaction time to increase the yield of (DNOXD)Pt(acac).
3.2 UV-Vis absorption spectra of oxadiazole ligand and its platinum complex
The UV-Vis absorption spectra of the oxadiazole ligand DNOXD and its platinum complex (DNOXD)Pt(acac) in dichloromethane at room temperature are shown in Fig.2. The UV absorption peaks located at 327 nm for DNOXD, and 325 nm and 439 nm for (DNOXD)Pt(acac) are displayed in the UV-Vis spectral profiles. Different UV absorption bands are observed between the oxadiazole ligand and its platinum complex. Compared with the oxadiazole ligand, its platinum complex exhibits an additional absorption peak at 439 nm, which is assigned to metal-ligand charge transfer (MLCT) transition of the complex. The significant absorption of the MLCT transition from this cyclometalized platinum complex with 2, 5-bis (naphthalene-1-yl)-1,3,4-oxadiazole ligand is similar to those from the reported cyclometalated iridium complexes and platinum complexes[9-14]. This indicates that the platinum complex has a potential application in phosphorescence-based LEDs used as a new electrophosphorescent material.
3.3 Photoluminescence spectra of ligand and its platinum complex
Fig.3 shows the photoluminescence spectra of the ligand DNOXD under excitation of the light at 272 nm and its platinum complex (DNOXD)Pt(acac) under excitation of the light at 420 nm in CH2Cl2 at room temperature.
As is seen from Fig.3, the maximum emission peak of DNOXD appears at 403 nm, whereas that of (DNOXD)Pt(acac) reveals at 625 nm with a shoulder at
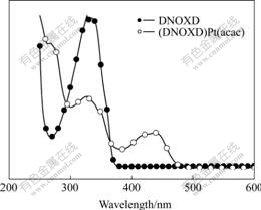
Fig.2 UV-Vis absorption spectra of ligand (DNOXD) and platinum complex (DNOXD)Pt (acac) in CH2Cl2
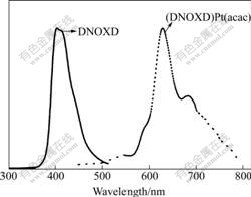
Fig.3 Photoluminescence spectra of ligand (DNOXD) and platinum complex (DNOXD)Pt(acac) in CH2Cl2 at room temperature
681 nm in the photoluminescence profiles. No emission from DNOXD is observed in the photoluminescence spectral profile of the platinum complex solution, which indicates that the emission of the complex is attributed to triplet phosphorescence emission of the platinum complex rather than the ligand. The intense red emission from (DNOXD)Pt(acac) at room temperature under excitation of the light at 420 nm indicates that the achieved oxadiazole-based platinum complex can be used as a red-emitting material in the phosphorescence- based LEDs. As this platinum complex contains an oxadiazole unit, it should be expected to have good electron-transporting and carrier-balancing abilities based on the previous researches[20- 21].
3.4 Electrochemical properties of platinum complex
In order to obtain information about the HOMO/LUMO(highest occupied molecular orbital/ lowest unoccupied molecular orbital) energy levels and band gap energy (Eg) of the platinum complex to make the complex applied on light-emitting devices based on energy level corresponding law, the electrochemical properties of the platinum complex in dichloromethane solution were studied by cyclic voltammetry method. Fig.4 shows the cyclic voltammogram of the platinum complex. As can be seen from Fig.4 that the complex shows a reversible oxidation peak at -1.37 V, and the corresponding reduction peak appears at 1.95 V. The oxidative process starts at -0.89 V. According to an empirical formula[22], the energy level EHOMO of the platinum complex is estimated from the oxidative onset potential to be -5.69 eV. The bandgap energy (Eg) is estimated from the onset of its UV-Vis absorption spectra to be -2.44 eV, as shown in Fig.2. The energy level ELUMO of the platinum complex is estimated to be -3.25 eV by subtracting Eg from EHOMO.
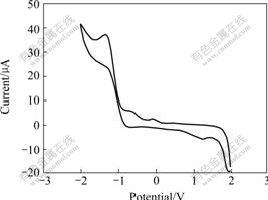
Fig.4 Cyclic voltammogram of platinum complex in 0.1 mmol/L CH2Cl2 with 0.1 mol/L Bu4NPF6 at room temperature
4 Conclusions
1) A novel cyclometalized platinum complex Pt[2,5-bis(naphthalene-1-yl)-1,3,4-oxadiazole(C2,N)]ace-tylacetonate showing strong red phosphorescence is synthesized and characterized. In the process of synthesis, it is necessary to extend the reaction time for increasing the yield of the platinum complex.
2) The fact that the platinum complex has two obvious UV-Vis absorption peaks at 327, 439 nm and a photoluminescence peak at 625 nm indicates that the achieved platinum complex can be used as a promising red-emitting material in phosphorescence-based LEDs.
References
[1] ZHANG Xiao-wei, YANG Cui-luo, LI Zhong-an, et al. Synthesis and characterization of novel orange-emitting iridium(Ⅲ) complexes with diphenylquinoline ligands containing fluorinated substituent[J]. Chin Chem Lett, 2006, 17(3): 411-414.
[2] SUN W F, ZHU H J, BARRON P M. Binuclear cyclometalated platinum(II) 4,6-diphenyl-2, 2-bipyridine complexes: Interesting photoluminescent and optical limiting materials[J]. Chem Mater, 2006, 18(10): 2602-2610.
[3] SHAVALEEV N M, ADAMS H, BEST J, et al. Deep-red luminescence and efficient singlet oxygen generation by cyclometalated platinum(Ⅱ) complexes with 8-hydroxyquinolines and quinoline-8-thiol[J]. Inorg Chem, 2006, 45(23): 9410-9415.
[4] YIN B L, NIEMEYER F, WILLIAMS J A G, et al. Snythesis, structure, and photophysical properties of luminescent platinum(Ⅱ) complexes containing cyclometalated 4-styryl-functionalized 2-phenylpyridine ligands[J]. Inorg Chem, 2006, 45(21): 8584-8596.
[5] WANG Zheng-xiang, SHU Wan-yin, ZHOU Zhong-cheng, et al. Fluorescence properties and application of doping complexes Eu1-xLx(TTA)3phen as light convesion agents[J]. Journal of Central South University of Technology, 2003, 10(4): 342-346.
[6] GUO Dong-cai, SHU Wan-yin, ZHANG Xin-qian, et al. Synthesis and luminescent properties of ternary complexes of terbium with thenoyltrifluoroacetone and reactive ligand[J]. Journal of Central South University of Technology, 2004, 11(3): 304-308.
[7] GONG X, BENMANSOUR H, BAZAN G C, et al. Red electrophosphorescence from a soluble binaphthol derivative as host and iridium complex as guest[J]. J Phys Chem B, 2006, 110(14): 7344-7348.
[8] KUNUGI Y, MANN K R, MILLER L L, et al. A vapochromic LED[J]. J Am Chem Soc, 1998, 120(3): 589-590.
[9] MCGARRAH J E, EISENBERG R. Dyes for photoinduced charge separation based on platinum diimine bis(acetylide) chromophores: Synthesis, luminescence and transient absorption studies[J]. Inorg Chem, 2003, 42(14): 4355-4365.
[10] HUA F, KINAYYIGIT S, CABLE J R, et al. Green photoluminescence from platinum(II) complexes bearing silylacetylide ligands[J]. Inorg Chem, 2005, 44(3): 471-473.
[11] GUO F Q, SUN W F. Synthesis, photophysics and optical limiting of platinum(II) 4-tolyterpyridyl arylacetylide complexes[J]. Inorg Chem, 2005, 44(11): 4055-4065.
[12] WONG W Y, HE Z, SO S K, et al. A multifunctional platinum-based triplet emitter for OLED applications[J]. Organometallics, 2005, 24(16): 4079-4082.
[13] BROOKS J, BABAYAN Y, LAMANSKY S, et al. Synthesis and characterization of phosphorescent cyclometalated platinum complexes[J]. Inorg Chem, 2002, 41(12): 3055-3066.
[14] DIEZ A, FOMIES J, GARCIA A, et al. Synthesis, structural characterization, and photophysical properties of palladium and platinum(II) complexes containing 7, 8-benzoquinolinate and various phosphine ligands[J]. Inorg Chem, 2005, 44(7): 2443-2453.
[15] LIU Jian, ZHU Mei-xiang, JIANG Chang-yun, et al. Green-yellow electrophosphorescence from di-[2,5-diphenyl-1,3,4-oxadiazole C2′ , N3] platinum (II) doped PVK devices[J]. Chin Phy Lett, 2005, 22(3): 723-727.
[16] JIANG C Y, YANG W, PENG J B, et al. High-efficiency, saturated red-phosphorescent polymer light-emitting diodes based on conjugated and non-conjugated polymer doped with an Ir complex[J]. Adv Mater, 2004, 16(6): 537-541.
[17] KAVITHA J, CHANG S Y, CHI Y, et al. In search of high-performance platinum phosphorescent materials for the fabrication of red electroluminescence devices[J]. Adv Funct Mater, 2005, 15(2): 223-229.
[18] COCCHI M, FATTORI V, VIRGILI D, et al. Highly efficient organic electrophosphorescent light-emitting diodes with a reduced quantum efficiency roll off at large current density[J]. Appl Phys Lett, 2004, 84(7): 1052-1054.
[19] WU Zhong-lian, ZHU Mei-xiang, LIU Yu, et al. Synthesis and photoluminescence of a novel iridium complex (BuPhOXD)2Ir(acac) with unit of 1,3,4-oxadiazole[J].Chin Chem Lett, 2005, 16(2): 241-244.
[20] LIANG Fu-shun, ZHOU Quan-guo, CHENG Yan-xiang, et al. Oxadiazole-functionalized europium(Ⅲ) β-diketonate complex for efficient red electroluminescence[J]. Chem Mater, 2003, 15(10): 1935-1937.
[21] LIANG Bo, JIANG Chang-yun, CHEN Zhao, et al. New iridium complex as high-efficiency red phosphorescent emitter in polymer light-emitting devices[J]. J Mater Chem, 2006, 16: 1281-1286
[22] THOMAS K R J, VELUSAMY M, LIN J T, et al. Efficient red-emitting cyclometalated iridium(III) complexes containing lepidine-based ligands[J]. Inorg Chem, 2005, 44(16): 5677-5685.
(Edited by CHEN Wei-ping)
Foundation item: Project(50473046) supported by the National Natural Science Foundation of China; Project(204097) supported by the Science Foundation of the Ministry of Education of China; Project(04JJ1002) supported by the Outstanding Youth Foundation of Hunan Province, China; Project(06JJ2008) supported by the Natural Science Foundation of Hunan Province, China
Received date: 2006-07-27; Accepted date: 2006-09-18
Corresponding author: ZHU Wei-guo, Professor; Tel: +86-732-8882658; Fax:+86-732-8292477; E-mail: zhuwg18@126.com